Abstract
Introduction
Drug-resistant essential tremor (ET) can benefit from open standard stereotactic procedures, such as deep-brain stimulation or radiofrequency thalamotomy. Non-surgical candidates can be offered either high-focused ultrasound (HIFU) or radiosurgery (RS). All procedures aim to target the same thalamic site, the ventro-intermediate nucleus (e.g., Vim). The mechanisms by which tremor stops after Vim RS or HIFU remain unknown. We used voxel-based morphometry (VBM) on pretherapeutic neuroimaging data and assessed which anatomical site would best correlate with tremor arrest 1 year after Vim RS.
Methods
Fifty-two patients (30 male, 22 female; mean age 71.6 years, range 49–82) with right-sided ET benefited from left unilateral Vim RS in Marseille, France. Targeting was performed in a uniform manner, using 130 Gy and a single 4-mm collimator. Neurological (pretherapeutic and 1 year after) and neuroimaging (baseline) assessments were completed. Tremor score on the treated hand (TSTH) at 1 year after Vim RS was included in a statistical parametric mapping analysis of variance (ANOVA) model as a continuous variable with pretherapeutic neuroimaging data. Pretherapeutic gray matter density (GMD) was further correlated with TSTH improvement. No a priori hypothesis was used in the statistical model.
Results
The only statistically significant region was right Brodmann area (BA) 18 (visual association area V2, p = 0.05, cluster size Kc = 71). Higher baseline GMD correlated with better TSTH improvement at 1 year after Vim RS (Spearman’s rank correlation coefficient = 0.002).
Conclusions
Routine baseline structural neuroimaging predicts TSTH improvement 1 year after Vim RS. The relevant anatomical area is the right visual association cortex (BA 18, V2). The question whether visual areas should be included in the targeting remains open.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Tremor characterizes one of the clearest consequences of unusual synchronization within the motor network. Although many studies have attempted to understand the pathophysiology of tremor [17, 18, 44], its underlying mechanisms remain largely undiscovered. Essential tremor (ET) is the most common movement disorder [27]. It has previously been reported that, in the context of ET, thalamic neurons display discharging patterns correlated with tremor, predominantly in the cerebellar input-receiving area (mainly the dentate nucleus) and further in the contralateral red nucleus, thalamic ventro-intermediate nucleus (Vim) and precentral gyrus [21]. The “tremor network” thus encompasses the cerebello-thalamo-cortical loop [18]. It has also been reported that the inferior olivary nucleus might produce an abnormal rhythmic output, which further affects synchronization of Purkinje cell firing [19], passively propagated through the cerebello-thalamic tract [5, 30] and lastly regulating motor activity [31].
Drug-resistant ET can benefit from standard stereotactic procedures, including deep-brain stimulation (DBS) [3], by means of an electrode positioned in the ventral thalamus (the ventro-intermediate nucleus, i.e., Vim) or radiofrequency thalamotomy [28]. Both have the advantage of intraoperative confirmation of targeting, with the possibility of adjusting the placement of the electrode if necessary, which can be performed on the basis of the intraoperative electrophysiology and clinical response; furthermore, they provide an immediate postoperative clinical effect regarding alleviation of the tremor [6, 20]. Non-surgical candidates can be advised to undergo radiosurgery (RS; e.g., Vim RS, also called Gamma Knife stereotactic thalamotomy) [8, 23, 41] or high-focused ultrasound (HIFU) [14, 15, 26]. The former aim at the same target (e.g., the Vim). The mechanisms by which RS and HIFU produce tremor arrest remain unknown. Radiosurgical targeting is indirect, and the clinical effect develops gradually, up to 1 year after the procedure [41], unlike open surgery. It has been previously hypothesized that, besides a purely lesioning effect, Vim RS might have a neuromodulatory one [33].
The advances in neuroimaging procedures have provided a new understanding of the pathophysiology of tremor [34]. Computational approaches to analyzing high-resolution structural MRI provide a powerful and non-invasive tool for characterizing individual and/or group differences in brain anatomy [1]. Regarding structural MRI, voxel-based morphometry (VBM) is a well-established and robust methodology, allowing insight into depicting the structural brain features. It uses routinely acquired T1-weighted imaging (T1w) to study volume subparts, gray or white matter density [1]. For ET, VBM has been used at both 1.5 and 3 T, with a large spectrum of controversial findings [2, 4, 7, 10,11,12, 25, 29, 32]. However, all these studies have compared ET cases versus a group of healthy controls.
In the present study, we use pretherapeutic VBM assessment and correlate pretherapeutic gray matter density (GMD) with tremor score on the treated hand (TSTH) improvement at 1 year after Vim RS. We used no a priori assumption. We hypothesized (based on previous work from our group) that visual areas might play a role in tremor alleviation and/or arrest after Vim RS.
Methods
Subjects
We analyzed 52 patients (30 male, 22 female; mean age 71.6 years, range 49–82, standard deviation 6.9). All had severe refractory right dominant in right-handed ET, treated with unilateral left Vim RS, in Marseille, France. All had given informed written consent. The study was approved by the local ethics committee (in compliance with the national legislation and the Declaration of Helsinki). All were part of a research protocol aiming at understanding Vim RS radiobiology. Inclusion and exclusion criteria are shown in Table 1. Essential tremor was defined according to Elble et al. [13]. The indications for Vim RS rather than DBS were medical comorbidities, drug resistance, advanced age or patient’s refusal.
Ventro-intermediate nucleus radiosurgery procedure
All Vim RSs were performed by the same neurosurgeon (JR) in Marseille, France, using a uniform procedure: Leksell® coordinate G Frame (Elekta AB, Stockholm, Sweden) application under local anesthesia [41], stereotactic CT and MRI and indirect targeting using Guiot’s diagram [2.5 mm above the AC-PC line, 11 mm lateral to the wall of the third ventricle and at a mean of 7.3 mm in front of the PC (3.8–9.8; SD: 1.3)]. A unique 4-mm isocenter was used with a uniform maximal prescription dose of 130 Gy.
Clinical evaluation and outcome measures after Vim radiosurgery
The same neurologist (TW) performed all clinical assessments at baseline and 1 year after Vim RS to account for delayed clinical effect [41]. The tremor score on the treated hand (TSTH) was assessed according to the Fahn-Tolosa-Marin Tremor Rating Scale [36]. At the time of study, no patient was under medication.
MRI pretherapeutic acquisition
All imaging was obtained on a 3-T magnetic resonance imaging (MRI) scanner (Siemens Skyra TrioTim Scanner, Munich, Germany) with a 32-channel receive array head coil. The acquisition protocol included a high-resolution T1-weighted (T1w) image, TR/TE = 2300/2.03, inversion time 900 ms, isotropic voxel of 1 mm3 and 192 slices. Images artifacted by movement were excluded.
Data processing and analysis
Statistical parametric mapping (SPM12) (Department of Neurology, London), performed with MATLAB 2014a, 2014 (MathWorks, Boston, MA, USA), was utilized for pre-processing and analyzing of the MRI data. The anatomical MR images were normalized to the Montreal Neurological Institute (MNI) atlas, with voxel size of 2 × 2 × 2 mm. The normalization procedure allows the warped images to fit into a standard template brain. Furthermore, this establishes a voxel-to-voxel correspondence between brains of different individuals. This enables driving the group statistics and reporting the results in a standard coordinate system (e.g., MNI, which is based on data from many individuals and is fully 3D, with data at every voxel). They were further segmented into the GM, white matter and cerebrospinal fluid. Furthermore, they were spatially smoothed using an 8-mm full-width-at-half-maximum (FWHM) Gaussian kernel in SPM12 to blur individual variations in the gyral anatomy and to increase the signal-to-noise ratio. The smoothing procedure enables subsequent voxel-by-voxel analysis comparable to a region-of-interest approach as the voxel will contain the average concentration of gray matter from around the voxel (where the voxel is characterized by the form of the smoothing kernel). The former is often referred to as the GMD. In a structural MRI of the brain, the intensity of each volumetric pixel or voxel relates to the density of the gray matter in that region. The VBM technique thus compares brain structures on a voxel-by-voxel basis. After the classical preprocessing steps, the intensity values of the voxels are compared to identify localized differences in the GMD.
SPM 12 was used to create a flexible factorial analysis of variance (ANOVA) model. Pretherapeutic T1w imaging was used, and TSTH improvement at 1 year after Vim RS was added as a covariate using the corresponding continuous values. The aim was to depict the anatomical area that correlates best with tremor stop after Vim RS. The uncorrected p value at the cluster level was set at ≤ 0.05 and for the peak level < 0.001.
The SPM analysis was done by the medical staff (CT, DVDV, EG) not involved in the treatment indication, Vim RS procedure or follow-up evaluation. Gray matter density (GMD) was further extracted from statistically significant regions. For correlation between the GMD and TSTH improvement, STATA version 11 (StataCorp LLC, College Station, TX, USA) was used, and p values were evaluated with the Spearman correlation coefficient.
Results
One-year overall improvement in TSTH in the present population was 67.8% (range 0–100%, standard deviation 32.9). No side effect was encountered. The vast majority of the patients presented a left thalamic “cocade” lesion on follow-up MRI, which is classically considered associated with the clinical response [37, 41]. However, four patients with severe pretherapeutic tremor were considered clinical non-responders but had a visible thalamic lesion on follow-up MRI, while eight patients were considered clinical responders and had a smaller lesion on follow-up MRI.
Right visual association area (Brodmann area 18, V2) predicts tremor alleviation
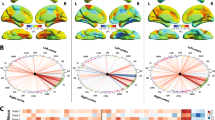
The only statistically significant cluster was the right Brodmann area (BA) 18 (visual association area V2; MNI 12, −74, −4; puncor = 0.05, Kc = 71; Fig. 1). Higher pretherapeutic GMD correlated with better TSTH improvement (Spearman = 0.002; Fig. 1).
The only statistically significant cluster was right Brodmann area (BA) 18 (visual association area V2). The upper part shows an illustration of the cluster in the axial, coronal and sagittal plane. Below, a scatterplot correlates the baseline GMD with TSTH improvement (Spearman = 0.002). The bottom part shows the SPM result at the cluster and peak level
Discussion
To the best of our knowledge, we performed the first VBM analysis endeavoring to predict which structural changes and/or adaptations in patients with drug-resistant ET would best correlate with 1-year outcome (e.g., tremor score on the treated hand) after Vim RS. We report that right Brodmann area 18 best correlates with TSTH improvement. Furthermore, higher baseline gray matter density within this region associates with better TSTH improvement.
Visual association area V2, also called the prestriate cortex, is the second major area in the visual cortex. This region receives connections from the V1 and sends strong connections to the V3, V4 and V5 areas. Most of the neurons are regulated to simple visual features such as orientation, spatial frequency, size, color and shape. It is mainly involved in visuo-spatial information processing [39]. Other roles include detection of light intensity, detection of patterns, discrimination of finger gestures or orientation-selective attention [24]. In fact, according to some functional studies, it additionally participates in visual priming and visual attention [9].
The previous studies related to VBM and ET patients have mainly compared the ET with healthy controls and revealed inconsistent results (at 1.5 and 3 T [2, 4, 7, 10, 11, 25, 32], showing atrophy, no change or volume increase in different brain areas). In the present study, it was beyond our purpose to analyze a healthy control group, as we were interested in correlating an eventual statistically significant structural feature, present pre-therapeutically, to the clinical response at 1 year after Vim RS.
In line with our present findings, using a VBM methodology, Daniels et al. [11] revealed a relative expansion of gray matter bilaterally in the temporo-parietal junction and the right middle occipital cortex in patients with ET as compared with healthy controls. The authors concluded that these areas, involved in higher order visuospatial processing and control, might exhibit an adaptive reorganization and compensate skilled movements in case of tremor. In a recent functional resting-state MRI study, Jang et al. [22] evaluated the effect of Vim HIFU on brain networks in ten patients using graph analysis. Interestingly, the authors observed changes in functional connectivity in the right lateral inferior occipital cortex. They hypothesized that a loss of strong interactions might appear or a reduced functional interaction on the way to other brain regions. Concerning the eventual distant effects of a thalamotomy procedure, structural diffusion tensor imaging (DTI) data after HIFU thalamotomy for ET [40] showed that fractional anisotropy changes included the pre- and post-central subcortical white matter, thalamus, red nucleus, inferior olive, bilateral parts of the superior vermis or central tegmental tract. After correction for multiple comparisons, only distant DTI changes were correlated with clinical improvement [40].
Previous studies have postulated that visual areas must be linked to the motor ones as a need for sensory guidance of movement of the hands and fingers [16] and further in motor regulation. Several possibilities could explain this: one is a modulation by the corpus callosum (mediating tasks in which the visual cortex on one part of the body is linked to the contralateral motor cortex); a second is a visual input to motor areas (by the caudate nucleus and claustrum); third, the cerebellum might play an essential role in calibrating the relationship between visual and somatosensory/motor information.
In a recently published paper [38], we evaluated longitudinal GMD changes in time between the pretherapeutic state and 1 year after Vim RS. We assessed whether these structural MR changes related to TSTH improvement by conventionally separating two different groups, clinical responders (≥ 50% improvement) and non-responders (< 50% improvement). Furthermore, we also appraised whether pretherapeutic GMD within the statistically significant clusters correlated with 1-year TSTH improvement, in the same study. We found that GMD was lower at baseline in the non-responder group for left BA 19 and left temporal pole (BA 38), V4, V5 and the parahippocampal place area. Moreover, for the left temporal pole, higher baseline GMD predicted better TSTH improvement.
In the present study , we exclusively used pretherapeutic structural T1w imaging. The VBM preprocessing steps were identical. However, the research question and statistical analysis model pertinent to this question were different. Here, we constructed a general linear model allowing the prediction of whether an eventual pretherapeutic GMD in a specific anatomical area would relate to TSTH improvement. Of particular importance, we used TSTH as a continuous variable, without the splitting of any group, as in the previous study [38]. We found that higher pretherapeutic GMD within right BA 18 predicted better TSTH improvement.
In our opinion, these findings should be seen as complementary, not exclusive. Both studies point to the involvement of the visual association areas in tremor arrest after Vim RS. Furthermore, together, they show that lower baseline GMD in these areas (independently if right or left) and in the left temporal pole was associated with lower TSTH improvement. Some other additional aspects should be taken into account because of their relevance. First, from a methodological point of view, as previously explained, the research questions were different. Second, it is well known that several visual functions (e.g., detection of light intensity, feature extraction or detection of patterns, etc.) simultaneously activate both BA 18 and 19, which are in close anatomical location, suggesting a common brain network. Third, regarding visual perception, it is now well acknowledged that it is not just carried out in one area, but involves many different regions of the cerebral cortex [42, 43], receiving input from V1. Typically, the corresponding processing of different information streams continues beyond V1. Many outputs go to V2 (e.g., BA 18) and relate to motion analysis, and this information is further processed in V3 (e.g., BA 19) and then V5, the medial temporal (MT) cortex (part of the study already published) and medial superior temporal (MST) cortex. All these steps happen before the information moves to other visual association areas, such as the posterior parietal cortex. Fourth, regarding functions related to the visual association cortex, electrophysiology data suggest sensitivity to motion-delineated forms [35] and its role as a differentiation point between the ‘what’ and ‘where’ visual pathways. Additional involvement is in visuospatial information processing, horizontal saccadic eye movements, orientation-selective attention and tracking visual motion patterns [42]. These functions involve both hemispheres and are not limited to a certain unilateral and individual area (whether V2 or V3, V4, etc.). Ultimately, a possibility that has been raised over time and that was already mentioned here is that the cerebellum might play the crucial role in adjusting the link between visual and somatosensory/motor information [16]. This former feature might be of particular interest in ET.
We believe that these findings, from two different studies in which two different methodologies were used, provide rich and complementary information about what structural change relates to a better clinical response after Vim RS. Our research question was not conducted to support existing pathophysiological theories, and thus our findings might provide new insight. The depicted changes in relationship to tremor response might have several possible explanations. One would be that adaptive structural MR changes appear during the disease course and, depending on these changes, the “reset” of the “tremor network” might be either easier or more difficult after Vim RS. A second hypothesis sees ET as a family of diseases rather than a uniform entity. Regarding this, some of the patients, depending on their phenotype, might develop structural MRI changes making them more “sensitive” or “resistant” to Vim RS as a trait of ET subclinical phenotype.
The advantages of the VBM analysis are related to the unbiased and objective procedure, being fully automated, not being based on regions of interest and being more exploratory. Furthermore, it depicts differences and/or changes (in our study) on a global and local scale. The limitations of our study are mainly related to the absence of a blinded neurological examination and to the VBM method (preprocessing steps, statistical challenges, etc.).
The role of the right visual association areas depicted on routine structural pretherapeutic neuroimaging is reported for the first time as a predictor of tremor improvement after Vim RS. How the “tremor network” (e.g., the cerebello-thalamo-cortical pacemaker) modulates these structural adaptations in patients with ET remains unknown.
Conclusion
Right visual association (BA18, V2) predicts tremor arrest after Vim RS. The mechanism by which this visual area modulates the clinical response is currently unknown. The question of whether visual areas should be included in the targeting is still open.
References
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. NeuroImage 11:805–821
Bagepally BS, Bhatt MD, Chandran V, Saini J, Bharath RD, Vasudev MK, Prasad C, Yadav R, Pal PK (2012) Decrease in cerebral and cerebellar gray matter in essential tremor: A voxel-based morphometric analysis under 3T MRI. J Neuroimaging Off J Am Soc Neuroimaging 22:275–278
Benabid AL, Pollak P, Gao D, Hoffmann D, Limousin P, Gay E, Payen I, Benazzouz A (1996) Chronic electrical stimulation of the ventralis intermedius nucleus of the thalamus as a treatment of movement disorders. J Neurosurg 84:203–214
Benito-Leon J, Alvarez-Linera J, Hernandez-Tamames JA, Alonso-Navarro H, Jimenez-Jimenez FJ, Louis ED (2009) Brain structural changes in essential tremor: Voxel-based morphometry at 3-Tesla. J Neurol Sci 287:138–142
Bhalsing KS, Saini J, Pal PK (2013) Understanding the pathophysiology of essential tremor through advanced neuroimaging: A review. J Neurol Sci 335:9–13
Blomstedt P, Hariz GM, Hariz MI, Koskinen LO (2007) Thalamic deep brain stimulation in the treatment of essential tremor: A long-term follow-up. Br J Neurosurg 21:504–509
Buijink AW, Broersma M, van der Stouwe AM, Sharifi S, Tijssen MA, Speelman JD, Maurits NM, van Rootselaar AF (2016) Cerebellar atrophy in cortical myoclonic tremor and not in hereditary essential tremor—a voxel-based morphometry study. Cerebellum 15:696–704
Campbell AM, Glover J, Chiang VL, Gerrard J, Yu JB (2015) Gamma knife stereotactic radiosurgical thalamotomy for intractable tremor: A systematic review of the literature. Radiother Oncol J Eur Soc Ther Radiol Oncol 114:296–301
Carrasco M (2011) Visual attention: The past 25 years. Vis Res 51:1484–1525
Cerasa A, Messina D, Nicoletti G, Novellino F, Lanza P, Condino F, Arabia G, Salsone M, Quattrone A (2009) Cerebellar atrophy in essential tremor using an automated segmentation method. AJNR Am J Neuroradiol 30:1240–1243
Daniels C, Peller M, Wolff S, Alfke K, Witt K, Gaser C, Jansen O, Siebner HR, Deuschl G (2006) Voxel-based morphometry shows no decreases in cerebellar gray matter volume in essential tremor. Neurology 67:1452–1456
Daniels JK, Gaebler M, Lamke JP, Walter H (2015) Grey matter alterations in patients with depersonalization disorder: A voxel-based morphometry study. J Psychiatry Neurosci JPN 40:19–27
Elble RJ (2000) Diagnostic criteria for essential tremor and differential diagnosis. Neurology 54:S2–S6
Elias WJ, Huss D, Voss T, Loomba J, Khaled M, Zadicario E, Frysinger RC, Sperling SA, Wylie S, Monteith SJ, Druzgal J, Shah BB, Harrison M, Wintermark M (2013) A pilot study of focused ultrasound thalamotomy for essential tremor. N Engl J Med 369:640–648
Elias WJ, Lipsman N, Ondo WG, Ghanouni P, Kim YG, Lee W, Schwartz M, Hynynen K, Lozano AM, Shah BB, Huss D, Dallapiazza RF, Gwinn R, Witt J, Ro S, Eisenberg HM, Fishman PS, Gandhi D, Halpern CH, Chuang R, Butts Pauly K, Tierney TS, Hayes MT, Cosgrove GR, Yamaguchi T, Abe K, Taira T, Chang JW (2016) A randomized trial of focused ultrasound Thalamotomy for essential tremor. N Engl J Med 375:730–739
Glickstein M (2000) How are visual areas of the brain connected to motor areas for the sensory guidance of movement? Trends Neurosci 23:613–617
Hallett M (2012) Parkinson’s disease tremor: Pathophysiology. Parkinsonism Relat Disord 18(Suppl 1):S85–S86
Hallett M (2014) Tremor: Pathophysiology. Parkinsonism Relat Disord 20(Suppl 1):S118–S122
Hansel C (2009) Reading the clock: How Purkinje cells decode the phase of olivary oscillations. Neuron 62:308–309
Hariz GM, Lindberg M, Bergenheim AT (2002) Impact of thalamic deep brain stimulation on disability and health-related quality of life in patients with essential tremor. J Neurol Neurosurg Psychiatry 72:47–52
Hua SE, Lenz FA (2005) Posture-related oscillations in human cerebellar thalamus in essential tremor are enabled by voluntary motor circuits. J Neurophysiol 93:117–127
Jang C, Park HJ, Chang WS, Pae C, Chang JW (2016) Immediate and longitudinal alterations of functional networks after thalamotomy in essential tremor. Front Neurol 7:184
Kondziolka D, Ong JG, Lee JY, Moore RY, Flickinger JC, Lunsford LD (2008) Gamma Knife thalamotomy for essential tremor. J Neurosurg 108:111–117
Larsson J, Landy MS, Heeger DJ (2006) Orientation-selective adaptation to first- and second-order patterns in human visual cortex. J Neurophysiol 95:862–881
Lin CH, Chen CM, Lu MK, Tsai CH, Chiou JC, Liao JR, Duann JR (2013) VBM reveals brain volume differences between Parkinson’s disease and essential tremor patients. Front Hum Neurosci 7:247
Lipsman N, Schwartz ML, Huang Y, Lee L, Sankar T, Chapman M, Hynynen K, Lozano AM (2013) MR-guided focused ultrasound thalamotomy for essential tremor: A proof-of-concept study. Lancet Neurol 12:462–468
Louis ED (2005) Essential tremor. Lancet Neurol 4:100–110
Nagaseki Y, Shibazaki T, Hirai T, Kawashima Y, Hirato M, Wada H, Miyazaki M, Ohye C (1986) Long-term follow-up results of selective VIM-thalamotomy. J Neurosurg 65:296–302
Nicoletti V, Cecchi P, Frosini D, Pesaresi I, Fabbri S, Diciotti S, Bonuccelli U, Cosottini M, Ceravolo R (2015) Morphometric and functional MRI changes in essential tremor with and without resting tremor. J Neurol 262:719–728
Passamonti L, Novellino F, Cerasa A, Chiriaco C, Rocca F, Matina MS, Fera F, Quattrone A (2011) Altered cortical-cerebellar circuits during verbal working memory in essential tremor. Brain 134:2274–2286
Popa T, Russo M, Vidailhet M, Roze E, Lehericy S, Bonnet C, Apartis E, Legrand AP, Marais L, Meunier S, Gallea C (2013) Cerebellar rTMS stimulation may induce prolonged clinical benefits in essential tremor, and subjacent changes in functional connectivity: An open label trial. Brain Stimul 6:175–179
Quattrone A, Cerasa A, Messina D, Nicoletti G, Hagberg GE, Lemieux L, Novellino F, Lanza P, Arabia G, Salsone M (2008) Essential head tremor is associated with cerebellar vermis atrophy: A volumetric and voxel-based morphometry MR imaging study. AJNR Am J Neuroradiol 29:1692–1697
Regis J, Carron R, Park M (2010) Is radiosurgery a neuromodulation therapy?: A 2009 Fabrikant award lecture. J Neuro-Oncol 98:155–162
Sharifi S, Nederveen AJ, Booij J, van Rootselaar AF (2014) Neuroimaging essentials in essential tremor: A systematic review. NeuroImage Clin 5:217–231
Tardif E, Richer L, Bergeron A, Lepore F, Guillemot JP (1997) Spatial resolution and contrast sensitivity of single neurons in area 19 of split-chiasm cats: A comparison with primary visual cortex. Eur J Neurosci 9:1929–1939
Tolosa E, Pena J (1988) Involuntary vocalizations in movement disorders. Adv Neurol 49:343–363
Tuleasca C, Pralong E, Najdenovska E, Cuadra MB, Marques JRF, Vingerhoets F, Regis J, Bloch J, Levivier M (2017) Deep brain stimulation after previous gamma knife thalamotomy of the vim for essential tremor is feasible! Clinical, electrophysiological and radiological findings. Acta Neurochir 159:1371–1373
Tuleasca C, Witjas T, Najdenovska E, Verger A, Girard N, Champoudry J, Thiran JP, Van de Ville D, Bach Cuadra M, Levivier M, Guedj E, Régis J (2017) Assessing the clinical outcome of vim radiosurgery with voxel-based morphometry: Visual areas are linked with tremor arrest! Acta Neurochir. https://doi.org/10.1007/s00701-017-3317-7
Waberski TD, Gobbele R, Lamberty K, Buchner H, Marshall JC, Fink GR (2008) Timing of visuo-spatial information processing: Electrical source imaging related to line bisection judgements. Neuropsychologia 46:1201–1210
Wintermark M, Huss DS, Shah BB, Tustison N, Druzgal TJ, Kassell N, Elias WJ (2014) Thalamic connectivity in patients with essential tremor treated with MR imaging-guided focused ultrasound: In vivo fiber tracking by using diffusion-tensor MR imaging. Radiology 272:202–209
Witjas T, Carron R, Krack P, Eusebio A, Vaugoyeau M, Hariz M, Azulay JP, Regis J (2015) A prospective single-blind study of gamma knife thalamotomy for tremor. Neurology 85:1562–1568
Zeki S (1993) The visual association cortex. Curr Opin Neurobiol 3:155–159
Zeki S, Watson JD, Lueck CJ, Friston KJ, Kennard C, Frackowiak RS (1991) A direct demonstration of functional specialization in human visual cortex. J Neurosci Off J Soc Neurosci 11:641–649
Zesiewicz TA, Elble R, Louis ED, Hauser RA, Sullivan KL, Dewey RB Jr, Ondo WG, Gronseth GS, Weiner WJ, Quality Standards Subcommittee of the American Academy of N (2005) Practice parameter: Therapies for essential tremor: Report of the quality standards Subcommittee of the American Academy of Neurology. Neurology 64:2008–2020
Funding
This work was supported by Timone University Hospital, the Swiss National Science Foundation SNSF-205321-157,040, the Centre d’Imagerie BioMédicale (CIBM) of the University of Lausanne (UNIL), the University of Geneva (UniGe), the Centre Hospitalier Universitaire Vaudois (CHUV) and the Leenaards and Jeantet Foundations.This work was carried out thanks to the support of the A*MIDEX project (no. ANR-11-IDEX-0001-02) funded by the "Investissements d’Avenir" French Government program, managed by the French National Research Agency (ANR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
The study was approved by the local ethics committee of Timone University Hospital, Marseille, France. Patient consent was acquired in all cases.
Additional information
Comments
Fifty-two patients with right-sided ET were treated with left unilateral Vim Gamma Knife (Vim) thalamotomy. Targeting was performed using 130 Gy and a single 4-mm collimator. Pre-therapeutic gray matter density in T1-weighted MRI (GMD) was correlated with tremor score improvement of the treated hand (TSTH) at 1 year after Vim RS as a continuous variable. As the only statistically significant anatomical region, higher baseline GMD in right Brodmann area 18 (visual association area V2) correlated with better TSTH improvement.
This article shows a very interesting relation between unsuspected anatomical visual association areas (BA18, V2) and tremor arrest in general by demonstrating a correlation between pre-therapeutic T1w imaging in visual association areas and outcome of left unilateral radiosurgical Vim lesions in patients with right-sided essential tremor.
Bodo Lippitz
Hamburg, Germany
This study is from a leading group in the field of radiosurgery for functional disorders. The authors were able to recruit a remarkable number of patients treated with an innovative method for a rare disease. No a priori hypothesis was used in the statistical model, which is a very important strength of the study. Also, the hypothesis that the visual areas are linked to the motor ones, as sensory guidance of the movement of the hand and fingers, appears sound and sheds new light on the understanding of the visuospatial network.
Alfredo Conti
Messina, Italy
Rights and permissions
About this article
Cite this article
Tuleasca, C., Witjas, T., Van de Ville, D. et al. Right Brodmann area 18 predicts tremor arrest after Vim radiosurgery: a voxel-based morphometry study. Acta Neurochir 160, 603–609 (2018). https://doi.org/10.1007/s00701-017-3391-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3391-x