Abstract
Introduction
Radiosurgery (RS) is an alternative to open standard stereotactic procedures (deep-brain stimulation or radiofrequency thalamotomy) for drug-resistant essential tremor (ET), aiming at the same target (ventro-intermediate nucleus, Vim). We investigated the Vim RS outcome using voxel-based morphometry by evaluating the interaction between clinical response and time.
Methods
Thirty-eight patients with right-sided ET benefited from left unilateral Vim RS. Targeting was performed using 130 Gy and a single 4-mm collimator. Neurological and neuroimaging assessment was completed at baseline and 1 year. Clinical responders were considered those with at least 50% improvement in tremor score on the treated hand (TSTH).
Results
Interaction between clinical response and time showed the left temporal pole and occipital cortex (Brodmann area 19, including V4, V5 and the parahippocampal place area) as statistically significant. A decrease in gray matter density (GMD) 1 year after Vim RS correlated with higher TSTH improvement (Spearman = 0.01) for both anatomical areas. Higher baseline GMD within the left temporal pole correlated with better TSTH improvement (Spearman = 0.004).
Conclusions
Statistically significant structural changes in the relationship to clinical response after Vim RS are present in remote areas, advocating a distant neurobiological effect. The former regions are mainly involved in locomotor monitoring toward the local and distant environment, suggesting the recruiting requirement in targeting of the specific visuomotor networks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Essential tremor (ET) is a gradually progressive movement disorder, characterized mainly by kinetic and postural tremor, affecting the head, forearms and hands [17]. Recent clinical and neuroimaging studies have suggested cerebellar involvement and mediation of tremor by the cerebello-thalamo-cortical (CTC) loop, but also including multiple other anatomical regions within the basal ganglia (basal ganglia loop) and cortex [5, 6].
Pharmacological treatment is unsatisfactory in the vast majority of cases. Drug-resistant ET can benefit from standard deep brain stimulation (DBS) or radiofrequeny thermocoagulation or alternatively from minimally invasive radiosurgery (RS) and high-focused ultrasound (HIFU), aiming at the same target (ventro-intermediate nucleus, Vim) [3, 12, 14, 30]. Radiosurgical targeting is indirect, and the clinical effect proceeds progressively, over a mean period of months, up to 1 year [30], unlike open surgery, with immediate tremor stop. It has been previously hypothesized that, besides a lesioning effect, Vim RS might have a neuromodulatory one [24].
Neuroimaging procedures have given new insight into the pathophysiology of tremor [27]. Computational approaches for analyzing high-resolution structural MRI provide a powerful and non-invasive tool for characterizing individual and/or group differences in brain anatomy [1]. With regard to structural changes, voxel-based morphometry (VBM) has been used at both 1.5 and 3 T, including in ET, with a large spectrum of controversial findings [2, 4, 7,8,9,10, 16, 21, 23]. In fact, VBM assesses subtle regional differences between subjects, in white or gray matter (GM), by a well-established and robust methodology [1].
In the present study, we used VBM to investigate whether Vim RS induces structural gray matter density (GMD) transformations 1 year after the intervention, in relationship with clinical effect. We hypothesize that changes in remote areas relate to clinical response after Vim RS, besides the changes within the targeted site (e.g. left thalamus).
Methods
Subjects
We analyzed 38 patients (mean age 71.8 years, range 52–80) with severe refractory right-dominant in right-handed ET, treated with unilateral left Vim RS, between April 2012 and March 2015. All were part of a research (including neuroimaging) protocol, aiming at the understanding of Vim RS radiobiology. All had given informed written consent. The study was approved by the local ethics committee (in compliance with the national legislation and Declaration of Helsinki).
Tremor definition and inclusion criteria
Essential tremor has been defined according to Elble et al. [11]. Parkinson’s disease cases were excluded, as well as those patients with previous stroke or epilepsy, etc., susceptible to induced structural changes. The indications for Vim RS rather than DBS were medical co-morbidities, drug-resistance, advanced age or patient’s refusal.
Gamma Knife thalamtomy
All Vim RS procedures were performed by the same neurosurgeon (JR). After application of the Leksell® coordinate G Frame (Elekta AB, Stockholm, Sweden), under local anesthesia [30], all underwent both stereotactic CT and MRI. Indirect targeting was performed in all cases using Guiot’s diagram, placed 2.5 mm above the AC-PC line, 11 mm lateral to the wall of the third ventricle, and at a mean of 7.3 mm in front of the PC (3.9–9.9; SD: 1.33). A unique 4-mm isocenter was used and a maximal prescription dose of 130 Gy.
Clinical evaluation and outcome measures after Vim radiosurgery
The same neurologist (TW) performed all clinical assessments, at baseline and 1 year after Vim RS. The tremor score on the treated hand (TSTH) was assessed according to the Fahn-Tolosa-Marin Tremor Rating Scale [28]. It has been previously advocated that medication diminishes TSTH by approximately 50% [19] in around 50% of cases. In this sense, to be at least as effective as medication, clinical responders (R) were considered those with at least 50% amelioration of TSTH 1 year after Vim RS to take into account the delayed clinical effect [30]. At the time of the study, no patient was under medication.
MRI acquisition and evaluation after GKT
All imaging was obtained on a 3-T magnetic resonance imaging (MRI) scanner, a Siemens Skyra TrioTim Scanner (Munich, Germany), with a 32-channel receive array head coil. For all cases, the acquisition protocol included a high-resolution, contrast-enhanced, T1-weighted (T1w), TR/TE = 2300/2.03, inversion time 900 ms, isotropic voxel of 1 mm3 and 192 slices. Images artifacted by movement were excluded.
Data processing and analysis
Statistical Parametric Mapping (SPM12) (London, United Kingdom), performed with MATLAB 2014a, 2014 (MathWorks, Boston, MA, USA), was utilized for preprocessing and analyzing of the MRI data.
The anatomical MR images were normalized to the Montreal Neurological Institute (MNI) atlas, with voxel size of 2 × 2 × 2 mm. The normalization procedure allows the warped images to fit into a standard brain template. Furthermore, this establishes a voxel-to-voxel correspondence between brains of different individuals, which will enable driving the group statistics and reporting the results in a standard coordinate system (e.g. MNI, which is based upon data from many individuals and is fully 3D, with data at every voxel).
They were further divided into GM, white matter and cerebro-spinal fluid. Furthermore, they were spatially smoothed using an 8-mm full-width-at-half-maximum (FWHM) Gaussian kernel in SPM12 to blur individual variations in gyral anatomy and to increase the signal-to-noise ratio. The smoothing procedure enables the subsequent voxel-by-voxel analysis comparable to a region-of-interest approach, as the voxel will contain the average concentration of gray matter from around the voxel (where the voxel is characterized by the form of the smoothing kernel). The former is often referred to as the GMD. In a structural MRI of the brain, the intensity of each volumetric pixel, or voxel, relates to the density of the gray matter in that region. The VBM technique thus compares brain structures on a voxel-by-voxel basis. After the classical preprocessing steps, the intensity values of the voxels are compared to identify localized differences in GMD.
The between-group SPM (T) GM maps were obtained using an analysis of variance (ANOVA) full factorial model at a height threshold (voxel-level significance) of p < 0.05, corrected for multiple comparisons for the cluster (FWE, family-wise error method). Secondarily, if no change was initially found using FWE correction, with a smaller constraint voxel-threshold of p < 0.005, correction was only done for the cluster volume to avoid type II errors as recommended [15]. Age and gender were used as nuisance variables. The interaction between clinical response and time, mainly between R versus non-responders (NR) patients, was evaluated with the different time frame (baseline versus 1 year after Vim RS) to identify the eventual structural changes in GMD. The primary aim was to find differences between groups (R versus NR) in interaction with the time point (before and after Vim RS).
The SPM analysis was done by medical staff (CT, EG) not involved in the indication for treatment, Vim RS procedure or follow-up evaluation. Gray matter density was further extracted from statistically significant regions. For correlation between GMD and TSTH improvement, STATA version 11 (STATA Corp LLC, College Stations, TX, USA) was used and p values evaluated with the Spearman correlation coefficient.
Results
Thirty-one (81.6%) patients were R and 7 (18.4%) NR. Overall improvement in TSTH was 62.1% (mean 62.6%, standard deviation 32.3; range 0–100%) and in the R subgroup was 75.2%. No difference in clinical characteristics was found between R and NR (age, gender, tremor severity before Vim RS; p > 0.05). No side effect was found 1 year after Vim RS.
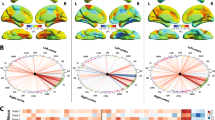
Changes in GMD between R and NR, during time (group factor X time, Fig. 1)
Changes in GMD between R and NR with time showed the left temporal pole (Brodmann area, BA 38) and a larger cluster [kc = 135 (left occipital) > kc = 124 (left temporal), indicating the number of voxels in each cluster] within the left occipital area. The former included, besides the left occipital visual association area (BA 19), the V4, V5 and parahippocampal place area (PPA). Figure 1 (1.1 and 1.2) displays the boxplots with the overall values and the median for baseline and 1 year, for R and NR, by the individual cluster.
The main effect of the time point, interacting with clinical response. Upper part: 1.1 and 1.2, global overview of GMD at baseline and 1 year for the left temporal pole and visual association area, depending on clinical response (≤ 50% TSTH improvement versus > 50% TSTH improvement); the boxplots reveal the range of values for GMD, as well as the median for each one of them. It is interesting that for both clusters (temporal and occipital), for a TSTH improvement ≤ 50%, the baseline values are lower as compared with the ones for TSTH improvement > 50%. At 1 year, the NR patients achieve a median value comparable to that of R at baseline, while the former remain stable; 1.3 (for the left temporal cluster) and 1.4 (for the left occipital cluster), correlations between TSTH improvement and differences in GMD (Spearman = 0.01 for both clusters); 1.5, correlation between baseline GMD within the left temporal cluster and TSTH improvement (Spearman = 0.004); 1.6, illustration of GMD increase (for the NR) and respectively no change (for R), as structural differences between R and NR during time, before and after Vim RS, presented by clusters (1 = left temporal and 2 = left occipital, with a sagittal illustration for each one of them)
Lower part, table with the main SPM results: it corresponds to the statistically significant results at the cluster level and peak level, respectively; the overall p value is 0.049. For the cluster and peak level are reported th p family-wise error correction (FEW), false-discovery rate (FDR), Kc = size of the cluster, meaning the number of the included voxels in each one of them, and the p unc = uncorrected p value, as opposed to the previous (FEW and FDR). The uncorrected p values show statistical significance (<0.05); for the peak level, the MNI signifies the Montreal Neurological Institute, which is the common place where the brains have been analyzed so as to be able to compare them. Lower part: an artistic drawing illustrating the main functions of these statistically significant clusters
Interestingly, the GMD was lower at baseline in the NR group for both regions and at 1 year had a median value similar to the R groups (which presented a slight decrease in GMD).
The table at the bottom of the figure shows the respective p values at the cluster and peak level (for a detailed explanation, please see the figure legend).
Furthermore, a higher GMD decrease between 1 year and baseline correlated with better TSTH improvement (Spearman = 0.01 for clusters 1 and 2, respectively; Figs. 1, 1.3 and 1.4). For cluster 1 (left temporal pole), higher baseline GMD predicted better improvement (Spearman = 0.004; Figs. 1, 1.5).
The GMD increase at 1 year after Vim RS was fairly similar for both clusters in NR, while it tended to be zero in R (Figs. 1, 1.6).
Discussion
To the best of our knowledge, we performed the first VBM analysis after Vim RS for drug-resistant ET. We report changes in GMD between R and NR with time within the left temporal pole (BA 38) and a larger cluster including the left visual association area (BA 19) and parts of V4, V5 and PPA as statistically significant. Gray matter density was lower at baseline in the NR group for both regions and at 1 year had a median value similar to the R groups (which presented a slight decrease in GMD). A higher GMD decrease between 1 year and baseline correlated with better TSTH improvement for both anatomical regions. For the left temporal pole, higher baseline GMD predicted better TSTH improvement.
VBM methodology has been previously used in ET (at 1.5 and 3 T), with studies reporting inconsistent results [2, 4, 7,8,9, 16, 23], while showing no change, atrophy or volume increase in different regions. In the present study, it was beyond our purpose to analyze a healthy control group, as we were interested in how the brain structurally adapts to Vim RS in relationship to clinical response.
The involvement of the left temporal pole and occipital BA 19 is a very interesting and novel finding, suggesting a distant effect of Vim RS. The left temporal pole is mainly involved in complex functions, including visual (“what” and “where” visual pathway distinction), emotional (visual processing of emotional images, emotional attachment), processing phonological properties of written words, sign language or complex auditory processing [20]. The visual cluster actually extends beyond the BA 19 and includes parts of V4 and V5 and the PPA (posterior subdivision). Visual area V4 is part of the extrastriate visual cortex [25] and is tuned for orientation, spatial frequency and color, but also long-term plasticity and stimulus salience, and is gated by signals coming from the frontal eye fields (FEF) [31]. The former connections would be of special interest in patients with ET: in a crowded visual scene, all factors that could help distinguish the target include color, shape and eye movements.
Vim RS is classically performed with the aim of inducing a small area of necrosis after several months up to 1 year after the procedure [24]. By the expected radiobiological effect, it mimics a histologically destructive one compared to that produced by thermocoagulation [26]. However, Ohye [22] was the first to advocate that solely a necrotic lesion was not sufficient to produce tremor alleviation. Moreover, he advocated that the magnitude of the lesion induced by Vim RS was too small to account for the benefic clinical effect. The proposed “cockade theory” advocates for four distinct zones that might appear after Vim RS, including necrotic, subnecrotic, neuromodulatory and showing no effect [24].
The advantages of the VBM analysis are related to an unbiased and objective procedure, fully automated, not based on regions of interest and that is more exploratory. Furthermore, it depicts differences and/or changes (in our study) on a global and local scale. The limitations of our study are mainly related to those of the VBM method (preprocessing steps, statistical challenges, etc.), but also to the studied population of patients, the former including the limited sample size or the absence of a blinded neurological assessment.
With regard to the main reasons for incomplete tremor alleviation, our experience and/or our hypotheses are: the size of the lesion (small size classically associated with more failures [29]), an eventual different phenotype of ET [18] (more resistant to Vim RS?), possible adaptive changes (including structural, as depicted by VBM), which makes the “reset” of the “tremor network” more difficult, and the targeting, although we used a uniform and well-established methodology.
The role of visual association areas is reported for the first time as statistically significant and thus relevant to tremor improvement after Vim RS. How the “tremor network” (cerebello-thalamo-cortical pacemaker) modulates these structural changes in remote areas remains unknown and to be elucidated by further studies. The anatomical connections between the primary motor cortex and visual areas might be of relevance, as previously advocated [13], for sensory guidance of movements.
Conclusion
Responders present changes in areas involved in motion, mainly locomotor monitoring of the local and distant environment, suggesting the requirement of recruiting in the targeting these specific visuomotor networks.
References
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. NeuroImage 11:805–821
Bagepally BS, Bhatt MD, Chandran V, Saini J, Bharath RD, Vasudev MK, Prasad C, Yadav R, Pal PK (2012) Decrease in cerebral and cerebellar gray matter in essential tremor: a voxel-based morphometric analysis under 3T MRI. J Neuroimag: Off J Am Soc Neuroimag 22:275–278
Benabid AL, Pollak P, Gao D, Hoffmann D, Limousin P, Gay E, Payen I, Benazzouz A (1996) Chronic electrical stimulation of the ventralis intermedius nucleus of the thalamus as a treatment of movement disorders. J Neurosurg 84:203–214
Benito-Leon J, Alvarez-Linera J, Hernandez-Tamames JA, Alonso-Navarro H, Jimenez-Jimenez FJ, Louis ED (2009) Brain structural changes in essential tremor: voxel-based morphometry at 3-Tesla. J Neurol Sci 287:138–142
Benito-Leon J, Louis ED (2006) Essential tremor: emerging views of a common disorder. Nature clinical practice. Neurology 2:666–678 quiz 662p following 691
Benito-Leon J, Louis ED (2007) Clinical update: diagnosis and treatment of essential tremor. Lancet 369:1152–1154
Buijink AW, Broersma M, van der Stouwe AM, Sharifi S, Tijssen MA, Speelman JD, Maurits NM, van Rootselaar AF (2016) Cerebellar atrophy in cortical myoclonic tremor and not in hereditary essential tremor—a voxel-based morphometry study. Cerebellum 15:696–704
Cerasa A, Messina D, Nicoletti G, Novellino F, Lanza P, Condino F, Arabia G, Salsone M, Quattrone A (2009) Cerebellar atrophy in essential tremor using an automated segmentation method. AJNR Am J Neuroradiol 30:1240–1243
Daniels C, Peller M, Wolff S, Alfke K, Witt K, Gaser C, Jansen O, Siebner HR, Deuschl G (2006) Voxel-based morphometry shows no decreases in cerebellar gray matter volume in essential tremor. Neurology 67:1452–1456
Daniels JK, Gaebler M, Lamke JP, Walter H (2015) Grey matter alterations in patients with depersonalization disorder: a voxel-based morphometry study. J Psychiatr Neurosci: JPN 40:19–27
Elble RJ (2000) Diagnostic criteria for essential tremor and differential diagnosis. Neurology 54:S2–S6
Elias WJ, Huss D, Voss T, Loomba J, Khaled M, Zadicario E, Frysinger RC, Sperling SA, Wylie S, Monteith SJ, Druzgal J, Shah BB, Harrison M, Wintermark M (2013) A pilot study of focused ultrasound thalamotomy for essential tremor. N Engl J Med 369:640–648
Glickstein M (2000) How are visual areas of the brain connected to motor areas for the sensory guidance of movement? Trends Neurosci 23:613–617
Kondziolka D, Ong JG, Lee JY, Moore RY, Flickinger JC, Lunsford LD (2008) Gamma knife thalamotomy for essential tremor. J Neurosurg 108:111–117
Lieberman MD, Cunningham WA (2009) Type I and type II error concerns in fMRI research: re-balancing the scale. Soc Cogn Affect Neurosci 4:423–428
Lin CH, Chen CM, Lu MK, Tsai CH, Chiou JC, Liao JR, Duann JR (2013) VBM reveals brain volume differences between Parkinson’s disease and essential tremor patients. Front Hum Neurosci 7:247
Louis ED (2005) Essential tremor. Lancet Neurol 4:100–110
Louis ED (2009) Essential tremors: a family of neurodegenerative disorders? Arch Neurol 66:1202–1208
Lyons KE, Pahwa R (2008) Pharmacotherapy of essential tremor: an overview of existing and upcoming agents. CNS Drugs 22:1037–1045
Nakamura K, Kawashima R, Sugiura M, Kato T, Nakamura A, Hatano K, Nagumo S, Kubota K, Fukuda H, Ito K, Kojima S (2001) Neural substrates for recognition of familiar voices: a PET study. Neuropsychologia 39:1047–1054
Nicoletti V, Cecchi P, Frosini D, Pesaresi I, Fabbri S, Diciotti S, Bonuccelli U, Cosottini M, Ceravolo R (2015) Morphometric and functional MRI changes in essential tremor with and without resting tremor. J Neurol 262:719–728
Ohye C, Shibazaki T, Ishihara J, Zhang J (2000) Evaluation of gamma thalamotomy for parkinsonian and other tremors: survival of neurons adjacent to the thalamic lesion after gamma thalamotomy. J Neurosurg 93(Suppl 3):120–127
Quattrone A, Cerasa A, Messina D, Nicoletti G, Hagberg GE, Lemieux L, Novellino F, Lanza P, Arabia G, Salsone M (2008) Essential head tremor is associated with cerebellar vermis atrophy: a volumetric and voxel-based morphometry MR imaging study. AJNR Am J Neuroradiol 29:1692–1697
Regis J, Carron R, Park M (2010) Is radiosurgery a neuromodulation therapy?: a 2009 Fabrikant award lecture. J Neuro-Oncol 98:155–162
Roe AW, Chelazzi L, Connor CE, Conway BR, Fujita I, Gallant JL, Lu H, Vanduffel W (2012) Toward a unified theory of visual area V4. Neuron 74:12–29
Schuurman PR, Bosch DA, Bossuyt PM, Bonsel GJ, van Someren EJ, de Bie RM, Merkus MP, Speelman JD (2000) A comparison of continuous thalamic stimulation and thalamotomy for suppression of severe tremor. N Engl J Med 342:461–468
Sharifi S, Nederveen AJ, Booij J, van Rootselaar AF (2014) Neuroimaging essentials in essential tremor: a systematic review. NeuroImage Clin 5:217–231
Tolosa E, Pena J (1988) Involuntary vocalizations in movement disorders. Adv Neurol 49:343–363
Witjas T, Carron R, Azulay JP, Regis J (2013) Gammaknife Thamamotomy for intractable tremors: clinical outcome and correlations with Neuroimaging features. MDS 17th international congress of Parkinson’s disease and movement disorders, vol volume 28, p 1247
Witjas T, Carron R, Krack P, Eusebio A, Vaugoyeau M, Hariz M, Azulay JP, Regis J (2015) A prospective single-blind study of gamma knife thalamotomy for tremor. Neurology 85:1562–1568
Zhou H, Desimone R (2011) Feature-based attention in the frontal eye field and area V4 during visual search. Neuron 70:1205–1217
Funding
This work has been supported by the Timone University Hospital, the Swiss National Science Foundation SNSF-205321-157,040, the Centre d’Imagerie BioMédicale (CIBM) of the University of Lausanne (UNIL), the University of Geneva (UniGe), the Centre Hospitalier Universitaire Vaudois (CHUV) and the Leenaards and Jeantet Foundations. This work was carried out thanks to the support of the A*MIDEX project (no. ANR-11-IDEX-0001-02) funded by the “Investissements d’Avenir” French Government program, managed by the French National Research Agency (ANR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
The study was approved by the local ethics committee of Timone University Hospital, Marseille, France.
Rights and permissions
About this article
Cite this article
Tuleasca, C., Witjas, T., Najdenovska, E. et al. Assessing the clinical outcome of Vim radiosurgery with voxel-based morphometry: visual areas are linked with tremor arrest!. Acta Neurochir 159, 2139–2144 (2017). https://doi.org/10.1007/s00701-017-3317-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3317-7




