Abstract
Purpose
Most morphometric studies on lumbar degenerative spondylolisthesis (DS) have focused solely on the L4–L5 slipped level, neglecting the shape of the entire lumbar segments. The purpose of this study was to present a morphometric analysis of the entire lumbar IVDs and VBs in DS.
Methods
Out of 500 lumbar CTs, the first 100 CTs, 50 with DS at L4 and 50 age- and sex-matched control CTs, were randomly selected. All lumbar IVD and VB heights, widths, lengths and sagittal wedging as well as lumbar lordosis (LL) and sacral inclination (SI) were measured and relevant ratios calculated. The prevalence of lumbar vertebral osteophyte was also measured.
Results
A total of 6700 measurements were taken. Age, height, weight and BMI had no effect on all parameters. Compared with controls, in females with DS, the majority of IVDs were flatter, with increased kyphotic wedging at L1–L2 (Δ1.3°) and L2–L3 (Δ1.8°), turning to lordotic wedging at L3–L4 (Δ5.9°), and decreased lordotic wedging at L4–L5 (Δ2.7°) and L5–S1 (Δ5.3°). The posterior IVD/VB ratio of all lumbar levels, middle IVD/VB ratio of L3–S1 and anterior IVD/VB ratio of L4–S1 were smaller. In males with DS, the L2–L3 IVD manifested more kyphotic wedging (Δ3.8°), the L4 VB wedging was more lordotic (Δ2.4°) and all L4–L5 IVD/VB ratios and L3–L4 middle and posterior IVD/VB ratios were smaller.
Conclusions
Individuals with DS have a more generalized degenerative disc disease on all lumbar vertebral levels, characterized by decreased disc space heights and kyphotic posture of the upper lumbar segments, occurring more predominantly in females than in males with DS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Degenerative lumbar spondylolisthesis (DS) is defined as an anterior slip of one vertebra in relation to the next caudal vertebra occurring in a segment with degenerative changes, but without an associated defect of the neural arch. The prevalence of DS increases with age and is rarely found under the age of 50. DS is the most common cause of radiculopathy and intermittent neurogenic claudication in the elderly and can be found in patients suffering from spinal stenosis [1, 2], degenerative scoliosis and or segmental instability with various clinical presentations [3, 4]. The prevalence of DS is 8.4 % in women and 2.7 % in men [5]. In approximately one-third of the cases, a double-level slip can be found [6]. The most common level where DS occurs is at L4–5 followed by L3–4 and L5–S1. In women, DS is associated with increased body mass index (BMI) [5] and decreased levels of estrogen production [7].
The shape of the lumbar spine and pelvis and the presence of degenerative changes are considered relevant factors in the pathomechanism of DS evolvement [3, 8–10]. Pelvic incidence (PI) is usually found to be greater in patients with DS [10], yet no definite conclusions have been drawn as to the possible correlation between DS and lumbar lordosis (LL), since there are several patterns of normal lumbar lordosis (Roussouly type 4, normal lordosis sub-types) [11] leading to a complex relationship of LL and DS.
Some studies have indicated that LL is greater in DS than in spines without a degenerative slip [3, 9, 12], whereas others have indicated that there are no differences in LL between DS and normal spines [13, 14]. Barrey et al. [9] found that DS was associated with greater PI leading to compensatory hyperlordosis of the lower lumbar spine. Indeed, DS is found mostly in Roussouly type 3 and 4 lumbar profiles [15]. Degenerative changes at the level of the slip are the hallmark of DS; however, a controversy still exists as to which changes appear first: IVD degeneration or facet joints arthrosis. Kirkaldy-Willis in 1978 [16] described the degenerative cascade and emphasized the strong link between IVD and facet joints. Kalichman [17] found that almost all cases of DS showed signs of facet arthritis, while only 50 % exhibited evidence of IVD degeneration. Sanderson et al. [18] concluded that the vertebral slippage in DS occurs before the appearance of arthritic changes. Farafan [19] introduced the concept of rotatory deformity in DS and not just forward slippage.
Some studies have endeavored to explore the morphometric characteristics of DS at the slip level; however, the rest of the lumbar spine has not been properly investigated. To the best of our knowledge, no previous study has examined the morphological characteristics and variation of the entire lumbar IVDs (L1–S1) and their adjacent vertebral bodies (VB) in cases of DS.
Additional factors which may contribute to the development of DS are lumbar lordosis as well as formation of osteophytes, osteoporosis, ligamentum flavum hypertrophy and ossifications [9, 20, 21]. Computerized tomography (CT) is considered an accurate imaging modality for identifying DS [22] and the delineation of the soft tissue changes and bony encroachment accompanying this pathology [22–25].
The hypothesis of this study was that in individuals with DS at L4–5, the shape of the IVDs and VBs along the entire lumbar spine was unique. The aim of the current CT study was to present a comprehensive morphometric analysis and shape variation of the IVDs and their adjacent VBs along the lumbar spine (L1–S1) in individuals with a single-level L4–5 DS and compare these parameters in subjects with no degenerative slip.
Materials and methods
Following the approval of the relevant institutional Helsinki and ethics committees, 100 CT scans of the lumbar spine were retrospectively randomly selected from a database of approximately 500 new CTs in 2010 from a leading private hospital conducting more than 500 lumbar spine surgeries per year. The CTs were divided equally into two groups, a study group (N = 50) and controls (N = 50). The study group included the first 50 CT images of individuals with single-level DS (grades 1–2) at L4–5 (25 males and 25 females ranging in age from 50 to 80). The diagnosis was carried out separately by two senior spine surgeons.
The study group was compared to controls consisting of the first 50 individuals without DS, matched according to gender and age (25 males, 25 females) (Table 1). All CT scans with other than L4–5 slips, double-level slips, evidence of previous surgery, vertebral fractures, spondyloarthropathies, scoliosis and isthmic spondylolisthesis were excluded.
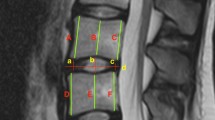
Lumbar lordosis (the angle between the superior endplate lines of L1 and S1), sacral inclination (the angle between the superior endplate line of S1 and a horizontal line) and the following IVDs and VBs parameters were measured from L1 to S1 (Fig. 1): anterior, posterior and middle IVD heights and sagittal IVD wedging; anterior, posterior and middle VB heights, superior and inferior VB widths and lengths and sagittal VB wedging; anterior, middle and posterior IVD/VB and sagittal height ratios. The prevalence of vertebral osteophytes (anterior, anterolateral and posterior) along the lumbar spine was also examined from different axial views. All CT measurements were taken by the first author (SA) from 2D projections using a computer software program (K-Pacs Workstation Version 1.0.1).
CT measurements: left image (mid-sagittal view)—lumbar lordosis (between lines A and F), sacral inclination (between lines F and G), vertebral body sagittal wedging (between lines D and E), intervertebral disc sagittal wedging (between lines B–C); middle image (mid-sagittal view)—vertebral body heights (A anterior, B middle, C posterior), intervertebral disc heights (D anterior, E middle, F posterior); right image (transverse view)—vertebral body length (A–B) and width (C–D)
Prior to the study procedure, intra and inter-reliability trials were conducted on ten CT images. The intra-reliability trials were based on repeating twice the same measurements from the same 10 CTs by SA with an interval of 1 week in between. The inter-reliability trials involved another tester (YM) who separately carried out the same measurements using the same protocol and under the same conditions.
Statistical analysis
Descriptive statistics were performed for all measurements. The Kolmogorov–Smirnov test was used to check whether the data were normally distributed. Multivariate regression analysis was used to examine the effects of age, weight, height, and BMI on the measured spinal parameters. Analysis of variance was used to examine the differences between the DS and controls. The intraclass correlation coefficient (ICC) was used to determine the intra-tester and inter-tester reliability of the measurements. An ICC of >0.75 was considered to be good reproducibility, whereas an ICC <0.75 was considered to be poor reproducibility of the measurements taken.
Results
A total of 6700 measurements from 100 lumbar CTs (67 × 100 CTs) were taken. All p values for the Kolmogorov–Smirnov test were >0.05, indicating normal distribution of all variables. Both intra-tester and inter-tester reliability for all measurements were good (0.85 < ICC < 1 and 0.85 < ICC < 0.92, respectively). Males were significantly taller and heavier than females in both the DS and controls (p < 0.05). Multivariate regression analysis indicated that age, height, weight and BMI had no effect on all measured spinal parameters (0.994 > p > 0.053). Mean values (±standard deviations) of all measured spinal parameters are shown in Tables 2 and 3.
Gender had no effect on LL and SI and no significant differences were indicated in these parameters between DS and the controls (LL—females: control = 44.3 ± 8.0, DS = 42.2 ± 6.4; males: control 41.2 ± 5.8, DS = 45.1 ± 7.9; SI -females: control = 53.7 ± 9.4, DS = 54.5 ± 9.5; males: control 50.3 ± 7.9, DS = 54.2 ± 10.8). The prevalence of osteophytes along the lumbar spine (L1–S1) was significantly greater in individuals with DS (both in females and males; 29 % vs 17 % in females (p = 0.000) and 32 vs 22 % in males (p = 0.03) (Fig. 2) The pattern of shape variation of all measured IVD and VB parameters along the lumbar spine are depicted in Figs. 3, 4, 5, 6 and 7.
Sagittal wedging variations of the vertebral body and intervertebral disc along the lumbar spine in degenerative spondylolisthesis and controls/normal groups. MN normal males, MS DS males, FN normal females, FS DS females, All measurement mean. Positive angle kyphotic angle, negative angle lordotic angle
Intervertebral disc (IVD)
IVD wedging (Fig. 7): in general, the IVD kyphotic wedging increased from L1–L2 to L2–3 followed by a lordotic wedging up to L5–S1. IVD heights (Fig. 6): the anterior disc height increases along the lumbar spine. The middle disc height increases from L1–L2 to L3–L4, remains almost constant until L4–L5 and then tends to decrease toward L5–S1. The posterior disc height rises moderately from L1–L2 to L4–L5 (approximately 1 mm), and decreases toward L5–S1 (approximately, 1 mm). In DS females, the anterior disc height tends to remain unchanged from L4–L5 to L5–S1 compared with a tendency to increase in the other groups (Fig. 6).
In females with DS, most IVD heights (9 out of 15) in most lumbar segments were significantly smaller (i.e., thinner) compared with the control females (p < 0.05) (Table 3). In males with DS, however, only some IVD heights (5 out of 15) of L3–L4, and L4–L5 were significantly smaller compared to control males (p < 0.05) (Table 3). In females with DS, the IVD manifested more kyphotic wedging at L1–L2 (Δ1.3°) and L2–L3 (Δ1.8°) and more lordotic wedging at L3–L4 (Δ5.8°); however, less lordotic wedging was manifested at the slip L4–5 level (Δ2.7°) compared to control females (p < 0.05). In males with DS, the IVD of L2–L3 manifested more kyphotic wedging than in control males (Δ3.8°) (p < 0.05) (Table 3).
Vertebral body (VB)
VB wedging (Fig. 7): in general, the VB sagittal wedging continuously decreased from the kyphotic wedging at L1 to the lordotic wedging at L5. VB heights (Fig. 3): the anterior VB height increases from L1 to L2 (about 2 mm), remains almost constant until L4 and then tends to decline toward L5 (approximately 1 mm). The middle VB height increases from L1 to L3 (approximately, 1 mm) and remains almost constant until L5. The posterior VB height increases from L1 to L2 (approximately, 1 mm), remains almost constant until L3 and then decreases toward L5 (about 4 mm). VB lengths (Fig. 4): the inferior VB length increases from L1 to L2 (approximately 2 mm), remains almost constant until L4 and increases toward L5 (approximately, 1 mm). The superior VB length increases sharply from L1 to L3 (approximately, 4 mm), and continues to rise moderately toward L5 (approximately, 1 mm). VB widths (Fig. 5): the superior VB width increases sharply from L1 to L3 (approximately 7 mm) and then descends moderately toward L5. The inferior VB width increases from L1 to L3, remains almost constant until L4 and then tends to decrease toward L5.
All VB dimensions (except for middle VB heights) at all lumbar levels (L1–S1) were significantly smaller in females than in males in both groups (p < 0.05) (Table 2). The L5 anterior VB height was significantly lower in females with DS than the control females (28.57 versus 30.01 mm) (p < 0.05). The L4 VB wedging was significantly more lordotic (−2.4°) in males with DS than control males (p < 0.05) (Table 2). In females, no significant differences in VB sagittal wedging were found between DS and controls in all lumbar levels.
IVD/VB sagittal height ratios
In females with DS, the posterior IVD/VB ratios of all lumbar levels (L1–S1), middle IVD/VB ratios of L3–S1 and anterior IVD/VB ratios of L4–S1 were significantly smaller than in the female controls (p < 0.05, Table 4). In males with DS, all ratios of L4–L5 and the middle and posterior IVD/VB ratios of L3–L4 were significantly smaller than in the male controls (p < 0.05) (Table 4).
Discussion
To the best of our knowledge, this is the first study to investigate the shape of all IVDs and their adjacent VBs along the entire lumbar spine of patients with DS. The morphometric data and degenerative changes observed in the current study indicate that individuals with DS experience a more generalized degenerative process in all lumbar vertebral levels studied, which is characterized by decreased disc heights, kyphotic posture of the upper lumbar segments and greater osteophyte prevalence along the lumbar vertebrae. A similar trend of decreased IVD height was previously described in another study, although the measurements taken from the lateral spine radiographs were only at the L4–L5 levels, thus indicating lower values of IVD heights (approximately, 1 mm) [26]. This trend is more pronounced in females than in males with DS, corroborating that prevalence of DS is greater in women than in men [5]. Although degenerative changes were also present in gender- and age-matched controls without DS, kyphotic disc spaces and generalized degenerative changes of the entire lumbar spine in the form of thinner IVDs and greater prevalence of vertebral osteophytes were found more predominantly in individuals with DS. This in turn may affect the segmental mobility and/or stability of the lumbar slipped level in DS [27].
Our results have clearly shown that both in controls and in DS, the IVDs in the two upper lumbar segments (L1–2, L2–3) are kyphotic in shape and not lordotic (see positive values in Table 3). However, in individuals with DS, the positive values were significantly more kyphotic than in the controls. Furthermore, the IVDs appearing at the lower segments of L4–5 and L5–S1 are significantly less lordotic (i.e., relatively more kyphotic) in females with DS than in the controls. Based on this, a clear image is presented wherein DS in general, and in females with DS in particular (another reason why DS is more prevalent in females than in males), the IVD configuration along the upper lumbar spine contributes more to a kyphotic alignment when compared with controls (Fig. 8). As the vertebral bodies at these levels (L1–L3 in females and L1–L2 in males) are also kyphotic in shape, it is reasonable to suggest that DS is characterized by an upper kyphotic lumbar sagittal shape. This unique upper lumbar kyphotic configuration fits well with the Roussouly sagittal lumbar pelvic classification of type I and type II, where a thoracolumbar kyphosis and a short caudal lumbar lordosis exist [28]. Accordingly, individuals in general and females in particular with a ‘kyphotic’ upper lumbar spine are at greater risk of developing DS. However, Roussouly has indicated that DS is more common in types III and IV of the lumbopelvic alignment probably due to the fact that their classification is based on standing X-rays compared to a supine CT in the current study. The upper lumbar kyphotic configuration in DS, as presented in the current study, may cause a relatively anterior displacement of the center of gravity in the upper lumbar spine, thereby overstressing s the lower lumbar levels leading eventually into DS at L4–L5 (Fig. 8). Previous studies have suggested a possible correlation between an anterior displacement in the sagittal vertical axis and the development of low back pain and degenerative changes in the spine [28].
The upper lumbar kyphotic configuration in degenerative spondylolisthesis. This configuration may cause a relatively anterior displacement of the center of gravity in the upper lumbar spine which overstresses the lower lumbar levels (L4–L5) leading eventually to DS at L4–L5. Lumbar lordosis (between lines a–b and m), sacral inclination (between lines a–b and c), lordotic discs at L5–S1 (between lines c and d) and L4–L5 (between lines e and f), kyphotic discs at L1–L2 (between lines k and l) and L2–L3 (between lines i and j), and kyphotic vertebral body at L1 (between lines m and l) and L2 (between lines j and k)
As discussed above, compared to controls, the IVDs in females with DS were thinner, more kyphotic at the upper lumbar segments and less lordotic at the lower segments, whereas in males with DS, only the VB wedging of L4 was more lordotic than in the control males. This information could also explain why the value of lumbar lordosis (LL) in the current study was similar in all examined groups, as more kyphotic upper lumbar segments probably neutralized the more lordotic lower lumbar segments. Another explanation could be related to the fact that a CT examination is obtained in the supine position (i.e., non-weight bearing); thus, any differences in the IVD sagittal wedging and/or flattening as currently observed were probably neutralized when measuring the total LL from L1 to S1. Nevertheless, there is contradictory information in the literature concerning LL in individuals with DS. Some studies have indicated higher LL values in DS [3, 9, 12], while others reported no difference in the LL between DS and controls [13, 14]. It is therefore reasonable to suggest that LL when measured during a routine supine CT should not be considered as unique morphometric characteristics in DS.
Several recent publications have described various aspects of disc degeneration and spinal stenosis as related to degenerative spondylolisthesis or low back pain [29–33]. Most studies that have examined IVD morphology in DS have focused only on the L4–5 vertebrae and linked the flattening of the IVD with the degenerative changes, facet tropism and regional instability, leading in turn to vertebral slippage [25, 34, 35]. Others have indicated that the mechanism works in reverse, i.e., following vertebral slippage, a load increase occurs on the IVDs thus leading to degenerative changes. This possible mechanism may explain why the IVD’s heights in other studies remained unchanged in DS compared with normal [9].
The question raised is which of the degenerative lumbar changes plays a primary role in the genesis of DS—the one occurring in the anterior portion of the vertebrae or in its posterior elements? This question is beyond the scope of this paper, but will be presented in a future study where unique facet orientation was observed in DS. Nevertheless, it is clear from our data that DS is not only a one-level problem, but rather a more generalized multilevel disc degeneration with a greater prevalence of vertebral osteophytes in the lumbar spine.
Although a possible biomechanical explanation related to the anterior displacement of the center of gravity in the lumbar spine due to its upper kyphotic configuration was suggested above, the issue of “cause-and-effect” contributing factors remains debatable. One may adopt the interactive cycling model to deal with this issue as previously suggested for isthmic spondylolysis [36, 37]. According to the model, the key point for understanding isthmic spondylolisthesis is not “which comes first”, but rather the relationships between the various factors associated with the pathology (anatomical features, spinal posture related to pelvic incidence, daily activities, etc.). As DS is partially associated with similar factors, such as posture and anatomical features, it is possible to use the same model to explain its pathomechanism. Here too, each contributing factor can initiate DS, i.e., degenerated IVDs [13, 38, 39], greater VB lordotic wedging [12], articular facets orientation and asymmetric facet arthritis [8, 21, 25, 34, 40–43] as well as spino-pelvic sagittal posture [9, 10, 25, 43]. It has been suggested that it is the type and intensity of the interaction between the various factors which will eventually determine if DS will develop or not. This would explain why DS is commonly seen in the elderly rather than in young individuals [20]. With aging, the intensity of all the above-mentioned factors may increase, leading to vertebral slippage [20, 34, 38, 40, 45].
Although DS is easily diagnosed by most spinal imaging modalities, it is important to develop specific tools that may predict either the occurrence of the slippage or the potential of progression. One such tool may be the use of calculated ratios as suggested in this study. The benefit of using morphometric ratios rather than absolute dimensions in characterizing the lumbar shape in DS is obvious, as ratios are not affected by the individual’s height, weight and BMI. Future studies can use these simple ratios to examine whether they may act as predictors for the development and deterioration of DS.
Clinical and surgical implications
Measuring the various lumbar geometric parameters as indicated in the current study using CT imaging may be beneficial for spinal clinicians in the early diagnostic and treatment stages of DS. One of the most common indications for lumbar spine surgery in the elderly is DS. Awareness of the actual size and shape of the IVD and VB may assist the surgeon in choosing the most adequate surgical procedure with appropriate fixation plates or screws to the different vertebral levels. In particular, segmental hypermobility related to lumbar shape variation at the slipped level, may require rigid internal fixation with pedicular screw fixation and interbody cages. As most lumbar IVDs show a clear tendency to disc degeneration in DS patients, a stabilizing surgical fixation with facet replacement to prevent slip progression rather than a formal fusion [46] should be considered.
Conclusions
Individuals with DS have more generalized degenerative lumbar discs emanating from all lumbar vertebral levels, characterized by decreased disc space heights and kyphotic posture of the upper lumbar segments. This trend is more pronounced in females than in males. This information may constitute an important diagnostic component and a better understanding of the pathomechanism of DS.
References
Markwalder M (1993) Surgical management of neurogenic claudication in 100 patients with lumbar spinal stenosis due to degenerative spondylolisthesis. Acta Neurochir (Wien) 120:136–142
Drury T, Ames SE, Costi K et al (2009) Degenerative spondylolisthesis in patients with neurogenic claudication effects functional performance and self-reported quality of life. Spine 34:2812–2817
Hasegewa K, Kitahara K, Hara T et al (2009) Biomechanical evaluation of segmental instability in degenerative lumbar spondylolisthesis. Eur Spine J 18:465–470
Hasegawa K, Shimoda H, Kitahara K et al (2011) What are the reliable radiological indicators of lumbar segmental instability? J Bone Joint Surg Br 93:650–657
Jacobsen S, Sonne-Holm S, Rovsing H et al (2007) Degenerative lumbar spondylolisthesis: an epidemiological perspective: the Copenhagen Osteoarthritis Study. Spine 32:120–125
Iguchi T, Wakami T, Kurihara A, et al 9 (2002) Lumbar multilevel degenerative spondylolisthesis: radiological evaluation and factors related to anterolisthesis and retrolisthesis. J Spinal Disord Tech 15:93–99
Imada K, Matsui H, Tsuji H (1995) Oophorectomy predisposes to degenerative spondylolisthesis. J Bone Joint Surg Br 77:126–130
Hosoe H, Ohmori K (2008) Degenerative lumbosacral spondylolisthesis: possible factors which predispose the fifth lumbar vertebra to slip. J Bone Joint Surg Br 90:356–359
Barrey C, Jund J, Perrin G et al (2007) Spinopelvic alignment of patients with degenerative spondylolisthesis. Neurosurgery 61:981–986
Legaye J, Duval-Beaupère G, Hecquet J et al (1998) Pelvic incidence: a fundamental pelvic parameter for three-dimensional regulation of spinal sagittal curves. Eur Spine J 7:99–103
Roussouly P, Pinheiro-Franco J (2011) Sagittal parameters of the spine: biomechanical approach. Eur Spine J 20:578–585
Been E, Ling L, Hunter D et al (2011) Geometry of the vertebral bodies and the intervertebral discs in lumbar segments adjacent to spondylolysis and spondylolisthesis: pilot study. Eur Spine J 20:1159–1165
Chen I, Wei T (1976) Disc height and lumbar index as independent predictors of degenerative spondylolisthesis in middle-aged women with low back pain. Spine 34:1402–1409
Berlemann U, Jeszenszky DJ, Buhler DW et al (1999) The role of lumbar lordosis, vertebral end-plate inclination, disc height, and facet orientation in degenerative spondylolisthesis. J Spinal Disord 12:68–73
Funao H, Tsuji T, Hosogane N et al (2012) Comparative study of spinopelvic sagittal alignment between patients with and without degenerative spondylolisthesis. Eur Spine J 21:2181–2187
Kirkaldy-Willis WH, Wedge JH, Yong-Hing K et al (1978) Pathology and pathogenesis of lumbar spondylosis and stenosis. Spine 3:319–328
Kalichman L, Kim DH, Li L et al (2009) Spondylolysis and spondylolisthesis: prevalence and association with low back pain in the adult community-based population. Spine 34:199–205
Sanderson P, Fraser R (1996) The influence of pregnancy on the development of degenerative spondylolisthesis. J Bone Joint Surg Br 78:951–954
Farfan HF (1980) The pathological anatomy of degenerative spondylolisthesis: a cadaver study. Spine 5:412–418
Rosenberg N (1975) Degenerative spondylolisthesis: Predisposing factors. J Bone Joint Surg Am 57:467–474
Cinotti G, Postacchini F, Fassari F et al (1997) Predisposing factors in degenerative spondylolisthesis. A radiographic and CT study. Int Orthop 21:337–342
Belfi L, Ortiz A, Katz D (2006) Computed tomography evaluation of spondylolysis and spondylolisthesis in asymptomatic patients. Spine 15:E907–E910
Saint-Louis L (2001) Lumbar spinal stenosis assessment with computed tomography, magnetic resonance imaging and myelography. Clin Orthop Relat Res 384:122–136
Paul C, Hansen A (1982) computed tomography in spondylolisthesis. Clin Orthop Relat Res 6:62–71
Schuller S, Charles Y, Steib J (2011) Sagittal spinopelvic alignment and body mass index in patients with degenerative spondylolisthesis. Eur Spine J 20:713–719
Chen IR, Wei TS (2009) Disc height and lumbar index as independent predictors of degenerative spondylolisthesis in middle-aged women with low back pain. Spine 34:1402–1409
Galbsura F, Schmidt H, Neidliniger-Wilke C et al (2011) The effect of degenerative morphological changes of the intervertebral disc on the lumbar spine biomechanics: a poroelastic finite element investigation. Comput Methods Biomech Biomed Eng 14:729–739
Roussouly P, Gollogly S, Berthonnaud E et al (2005) Classification of the normal variation in the sagittal alignment of the human lumbar spine and pelvis in the standing position. Spine 30:346–353
Korez R, Likar B, Pernus F et al (2014) Parametric modeling of the intervertebral disc space in 3D: application to CT images of the lumbar spine. Comput Med Imaging Graph 38:596–605
Merckaert S, Pierzchala K, Kulik G et al (2015) Influence of anatomical variations on lumbar foraminal stenosis pathogenesis. Eur Spine J 24:313–318
Smorgick Y, Mirovsky Y, Fischgrund JS et al (2014) Radiographic predisposing factors for degenerative spondylolisthesis. Orthopedics 37:260–264
Maatta JH, Kraatari M, Wolber L et al (2014) Vertebral endplate change as a feature of intervertebral disc degeneration: a heritability study. Eur Spine J 23:1856–1862
Miao J, Wang S, Park WM et al (2013) Segmental spinal canal volume in patients with degenerative spondylolisthesis. Spine J 13:706–712
Dai L (2001) Orientation and tropism of lumbar facet joints in degenerative spondylolisthesis. Int Orthop 25:40–42
Toyone T, Moriya H, Kitahara H, Takahashi K, Yamagata M, Murakami M, Takahashi Y (1993) Assessment of segmental spinal instability using magnetic resonance imaging. In: Yonenobu K, Ono Y, Takemitsu Y (eds) Lumbar fusion and stabilization. Springer- Verlag, Tokyo, pp 35–44
Masharawi Y, Alperovitch-Najenson D, Steinberg N et al (2007) Lumbar facet orientation in spondylolysis: a skeletal study. Spine 6:176–180
Masharawi Y, Peleg S, Nili S et al (2007) Lumbar facet anatomy changes in spondylolysis: a comparative skeletal study. Eur Spine J 16:993–997
Matsunaga S, Sakou T, Morizono Y et al (1990) Natural history of degenerative spondylolisthesis. Pathogenesis and natural course of the slippage. Spine 15:1204–1210
Newman P (1955) Spondylolisthesis, its cause and effect. Ann R Coll Surg Engl 16:305–323
Love T, Fagan A, Fraser R (1999) Degenerative spondylolisthesis developmental or acquired. J Bone Joint Surg Br 81:670–674
Boden S, Riew K, Yamaguchi K et al (1996) Orientation of the lumbar facet joints: association with degenerative disc disease. J Bone Joint Surg Am 78:403–411
Sato K, Wakamatsu E, Yoshizumi A et al (1989) The configuration of the laminas and facet joints in degenerative spondylolisthesis: a clinicoradiologic study. Spine 14:1265–1271
Kalichman L, Suri P, Guermazi A et al (2009) Facet orientation and tropism: associations with facet joint osteoarthritis and degeneratives. Spine 34:E579–E585
Haruki F, Takashi T, Naobumi H et al (2012) Comparative study of spinopelvic sagittal alignment between patients with and without degenerative spondylolisthesis. Eur Spine J 21:2181–2187
Grobler L, Robertson P, Novotny J et al (1993) Etiology of spondylolisthesis. Assessment of the role played by lumbar facet joint morphology. Spine 18:80–91
Anekstein Y, Floman Y, Smorgick Y et al (2015) Seven years follow-up for total lumbar facet joint replacement (TOPS) in the management of lumbar spinal stenosis and degenerative spondylolisthesis. Eur Spine J 24:2306–2314
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
The authors thank Mrs. Phyllis Curchack Kornspan for her editorial assistance.
Rights and permissions
About this article
Cite this article
Abu-Leil, S., Floman, Y., Bronstein, Y. et al. A morphometric analysis of all lumbar intervertebral discs and vertebral bodies in degenerative spondylolisthesis. Eur Spine J 25, 2535–2545 (2016). https://doi.org/10.1007/s00586-016-4673-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4673-3