Abstract
Opinions differ as to the exact mechanism responsible for spondylolysis (SP) and whether individuals with specific morphological characteristics of the lumbar vertebral neural arch are predisposed to SP. The aim of our study was to reveal the association between SP and the architecture of lumbar articular facets and the inter-facet region. Methods: Using a Microscribe three-dimensional apparatus (Immersion Co., San Jose, CA, USA), length, width and depth of all articular facets and all inter-facet distances in the lumbar spine (L1–L5) were measured. From the Hamann-Todd Human Osteological Collection (Cleveland Museum of Natural History, OH, USA) 120 normal male skeletons with lumbar spines in the control group and 115 with bilateral SP at L5 were selected. Analysis of variance was employed to examine the differences between spondylolytic and normal spines. Results: Three profound differences between SP and the norm appeared: (1) in individuals with SP, the size and shape of L4’s neural arch had significantly greater inter-facet widths, significantly shorter inter-facet heights and significantly shorter and narrower articular facets; (2) only in the L4 vertebra in individuals with SP was the inferior inter-facet width greater in size than the superior inter-facet width of the vertebra below (L5) (38.7 mm versus 40 mm); (3) in all lumbar vertebrae, the right inferior articular facets in individuals with SP were flatter compared to the control group. Conclusions: Individuals with L4 “SP” characteristics are at a greater risk of developing fatigue fractures in the form of spondylolysis at L5.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Isthmic spondylolysis (SP) is a separation of the neural arch of a lumbar vertebra occurring at the pars interarticularis between the superior and inferior articular facets, most commonly at L5 [2, 12, 14]. Opinions differ as to the exact mechanism responsible for SP. Heredity, repeated stress and lumbar hyperlordosis have been suggested as possible causative agents [1, 3–6, 8, 10, 12]. Several studies have shown that, whatever the causative agent, individuals with specific morphometric characteristics of the neural arch are more likely to develop SP [7, 13].
We sought to test the hypothesis that spondylolytic defects at L5 are indeed associated with a distinctive shape of articular facets and inter-facet region of the lumbar vertebrae.
Methods
One hundred and fifteen adult male skeletons with SP at L5 and 120 male age–race-matched control skeletons (individuals with spondyloarthropathies and compression fractures excluded) were studied. The mean age (SD) for the SP group was 47.9 (13.8) and for the control, 49.1 (17.1) (p = 0.20). Additionally, no differences between the two groups were found in regard to stature and body weight. The skeletons were part of the Hamann-Todd Human (HTH) Osteological Collection housed at the Cleveland Museum of Natural History, Cleveland, OH, USA. This collection comprised 3,000 white Caucasians and African-American human skeletons of individuals born between 1825 and 1910 (mostly of low socio-economic status). Data on age, sex, race, stature and weight were obtained from the medical records of these individuals. All lumbar spines with bilateral spondylolysis at L5 were visually identified and isolated. Adhesive plasticine was used to attach the separated part of the neural arch at L5 to its original position. Direct measurements were taken by one of the authors (MY) using a Microscribe 3D apparatus (Immersion Co., San Jose, CA, USA). A special metal device properly stabilized the vertebrae.
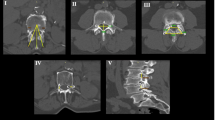
The parameters included in the study were facet length, facet width, superior facet concavity (depth), inferior facet convexity (height), left and right inter-facet heights and superior and inferior inter-facet widths at all lumbar vertebrae (Table 1).
Descriptive statistics were carried out for all the measurements in all the vertebrae. Analysis of variance (ANOVA) examined the differences between spondylolytic and normal spines. Prior to this test, we applied the Kolmogorov–Smirnov test to check whether the data was normally distributed. The intraclass correlation coefficient (ICC) was used to determine the intra-tester and inter-tester reliability of the measurement. An ICC of greater than 0.75 is considered good reproducibility and less than 0.75 indicates poor reproducibility [11]. An ICC above 0.90 is considered as an excellent reproducibility. Intra-tester reliability of the measurement taken was assessed by one of the authors (YM). Measurements were taken twice with 3.5 days separation. Inter-tester reliability involved two testers (YM and IH), who carried out the measurements in the same method within 20 min. Both testers were blinded to the results of the measurements. To estimate the reliability of the adhesion procedure, attachment process was carried twice (a week interval) by the same author (YM), measurements taken after each time, and reassembled and measured again for the third time by a second author (IH).
Results
The prevalence of SP in the HTH adult (20> years) male sample (total 2,374) was 4.8%. No significant differences in SP prevalence were found between the age cohorts (10 years interval), varying from 2.2 to 7.6%.
Metric data for all lumbar facets and inter-facet region for the SP and control groups appear in Tables 2, 3, 4 and Figs. 1 and 2. Females were excluded from the current analysis due to small sample size (only 6 females out of 700 females in the HTH collection manifested SP). Both the intra-tester and inter-tester reliability for the measurements taken were good (intra-tester ICC = 0.96–0.98; inter-tester ICC = 0.82–0.89). The adhesive procedure did not contribute to the variation of the traits measured (intra and interclass correlation coefficients of 0.89–0.94). All p-values for Kolmogorov–Smirnov test were greater than 0.05, indicating normal distribution of all variables.
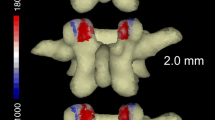
In lumbar spines with SP, the neural arch of L4 showed the greatest number of significantly different variables (p < 0.05) from the control group, including a significantly greater distance between the articular facets (i.e. inter-facet width), both in the superior (34.3 mm versus 33.0 mm) and inferior facets (39.9 mm versus 35.5 mm); a considerably lower ratio between the above distances (0.87 in SP versus 0.94 in the norm), rendering the inter-facet region a pronounced trapezoidal shape; a significantly shorter inter-facet height; and significantly shorter and narrower articular facets (Fig. 3). Two other important results are worth noting: only in individuals with SP, a reduced superior inter-facet width is indicated at L5 relative to L4 inferior inter-facet width (38.7 mm versus 40 mm), contrary to that found for the control (36.7 mm versus 35.5 mm), and in all lumbar vertebrae, the convexity of the right inferior facets was between 1.5 and 2.0 times greater in the normal group compared to SP.
Discussion
The current study describes the shape of the facet and inter-facet region in the lumbar vertebrae (from L1 to L5), in both SP and control groups. We found that the facets and inter-facet region of L4 and L5 are distinctly organized in individuals with SP.
During flexion/extension of the spine, the load on the neural arch in normal spines increases considerably from L1 to L5 when the highest mechanical stress is at the pars interarticularis of L5 [6]. In the normal lumbar spine, the zygoapophyseal joints usually absorb up to 25% of the total load applied to the vertebra [15]. The fact that in SP individuals the total area of the articular facets of L4 and L5 was found to be significantly smaller and shallower compared to the control (by ca. 16–26%) ultimately indicate that the stress on these facets in SP individuals is much greater compared to the control. Gobler et al. [9] also found a reduced transverse articular dimension in SP compared to the norm.
The two most significant morphological features of the inter-facet region in SP individuals are: (a) the wider inferior inter-facet region of L4 in SP compared to the control (40 mm versus 35 mm), lending the L4 inter-facet region a pronounced trapezoidal shape, and (b) the reduced superior inter-facet width of L5 relative to L4 inferior inter-facet width (38.7 mm versus 40 mm), contrary to that found for the control (36.7 mm versus 35.5 mm). These features result in the following: (a) L4 inferior facets are placed over the pars interarticularis of L5. This finding is in line with Ward and Latimer [13], who recently proposed that chronic spondylolytic defects at L5 are influenced by insufficient differential mediolateral distances between the inferior articular facets of L4 and the superior facets of S1, resulting in the structures impinging on the par interarticularis; (b) the moment applied to the inferior facets of L4 and L5 is considerably greater in SP due to the longer leverage (i.e. greater inter-facet distances in L4 and L5). This implies that under similar torsional motion, the load applied to the facets will be greater in SP individuals compared to the norm; (c) the reduced convexity and surface area of the inferior facets in SP individuals decrease the torsional range of motion during spinal movements. This may result in an earlier locking of the zygoapophyseal joints. The above findings indicate that under certain morphological configuration of the inter-facet region, enhanced moment created during spinal torsional motions may increase the risk for SP. This risk becomes significantly greater in extremely demanding spinal functions (eg., gymnastics). The fact that L5 movement is strongly counterbalanced by the attached ligaments to the sacrum explains why this fracture is more commonly seen in L5 than in L4. Finally, the transverse orientation of the pars fractures seems to indicate a moment applied contra-laterally (i.e. by the opposite facet) rather than ipsi-laterally. Accordingly, a fracture on the right pars interarticularis is probably caused by the left leverage (i.e. the left facet) and vice versa.
Grobler et al. [9] who studied the association between anatomical features at the posterior vertebrae and SP could not determine whether “this anatomical feature could be the expression of an altered posterior element morphology predisposing to SP... or a result of the formation of the lesion itself” (p. 88). Although the issue of “cause-and-effect” is a central one in SP, considering the nature of the phenomenon at its early stages, it is doubted if even in the case of a comprehensive study carried out on ideal adolescent sample, an unequivocal answer can be reached. In an attempt to deal with this issue, we have developed a feedback cycling model (Fig. 4) that describes the possible mechanism by which SP may be produced. According to this model, the key point for understanding spondylolysis is not “which comes first,” but rather the relationships between the various factors associated with SP (Fig. 4). Although it is possible that each of the three elements in the model (anatomical features, microfracture and posture/activity) may cause SP independently, it is the nature and intensity of the interaction between the three during growth (as implied from the prevalence of SP in the age cohorts), which will determine who will develop SP and who will not.
The feedback cycling model of spondylolysis (SP). The model expresses the notion that SP is multifactorial in nature. The various factors are grouped into three clusters (external circle). Although each may enhance stress (middle circle) on the lumbar vertebrae, which can lead to the development of SP, it is the nature and intensity of the interaction between the elements in the three clusters during growth (inner circle), which will determine if SP will be developed
Conclusion
The configuration of the L4 and L5 inter-facet region is associated with SP.
References
Boachie-Adjei O, Lonner B (1996) Spinal deformity. Pediatr Clin North Am 43:883–897
Bridges P (1989) Spondylolysis and its relationship to degenerative joint disease in the prehistoric Southeastern United States. Am J Phys Anthropol 79:321–329
Cyron BM, Hutton WC (1978) The fatigue strength of the lumbar neural arch in spondylolysis. J Bone Joint Surg Br 60:234–238
Cyron BM, Hutton WC, Stott JR (1979) Spondylolysis: the shearing stiffness of the lumbar intervertebral joint. Acta Orthop Belg 45:459–469
Cyron BM, Hutton WC, Troup JD (1976) Spondylolytic fractures. J Bone Joint Surg Br 58:462–466
Dietrich M, Kurowski P (1985) The importance of mechanical factors in the etiology of spondylolysis: a model analysis of loads and stresses in human lumbar spine. Spine 10:532–542
Eisenstein S (1978) Spondylolysis: a skeletal investigation of two population groups. J Bone Joint Surg 60:488–494
Farfan HF, Osteris V, Lamy C (1976) The mechanical etiology of spondylolysis and spondylolisthesis. Clin Orthop Relat Res 17:40–55
Grobler L, Robertson P, Novotny J, Pope M (1993) Etiology of spondylolisthesis. Assessment of the role played by lumbar facet joint morphology. Spine 18:80–91
Nathan H (1959) Spondylolysis: its anatomy and mechanism of development. J Bone Joint Surg Am 41:303–310
Portney LG, Watkins MP (2000) Foundations of clinical research: application to practice, 2nd Edn. Appleton Lange, East Norwalk Conn pp 53–67
Troup JD (1976) Mechanical factors in spondylolisthesis and spondylolysis. Clin Orthop 117:59–67
Ward C, Latimer B (2005) Human evolution and the development of spondylolysis. Spine 30:1808–1814
Wiltse LL, Widell, Jackson DW (1975) Fatigue fracture: the basic lesion in isthmic spondylolisthesis. J Bone Joint Surg Am 57:17–22
Yang K, King A (1984) Mechanism of facet load transmission as a hypothesis for low-back pain. Spine 9:557–565
Acknowledgments
The authors would like to thank Prof. Bruce Latimer and Mr. Lyman Jellema of the Cleveland Museum of Natural History, Cleveland, OH, for their support and assistance in using the invaluable Hamman-Todd Osteological Collection; Mrs. Ana Bachar, Department of Anatomy and Anthropology, Sackler Faculty of Medicine, Tel-Aviv University, for her tremendous assistance in preparing the figures; and Mrs. Phyllis Curchack Kornspan for her editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Masharawi, Y., Dar, G., Peleg, S. et al. Lumbar facet anatomy changes in spondylolysis: a comparative skeletal study. Eur Spine J 16, 993–999 (2007). https://doi.org/10.1007/s00586-007-0328-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-007-0328-8