Abstract
Introduction
There is a lack of data in the literature on surgical correction of severe neuromuscular scoliosis in patients with serious extent of cerebral palsy. The purpose of this retrospective cohort study was to analyze the radiological and clinical results after posterior-only instrumentation (group P) and combined anterior–posterior instrumentation (group AP) in severe scoliosis in patients with Gross Motor Function Classification System grades IV and V.
Materials and methods
All eligible patients who underwent surgery in one institution between 1997 and 2012 were analyzed, and charts, surgical reports, and radiographs were evaluated with a minimum follow-up period of 2 years.
Results
Fifty-seven patients were included (35 in group P, 22 in group AP), with a median follow-up period of 4.1 years. The preoperative mean Cobb angles were 84° (34 % flexibility) in group P and 109° (27 % flexibility) in group AP. In group P, the Cobb angle was 39° (54 % correction) at discharge and 43° at the final follow-up, while in group AP the figures were 54° (50 % correction) at discharge and 56° at the final follow-up. Major complications occurred in 23 vs. 46 % of the patients, respectively. Preoperative curve flexibility was an important predictor for relative curve correction, independently of the type of surgery.
Conclusion
Posterior-only surgery appears to lead to comparable radiological results, with shorter operating times and shorter intensive-care unit and hospital stays than combined surgery. The duration of surgery was a relevant predictor for complications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerebral palsy (CP) is a nonprogressive form of encephalopathy caused by damage to motor control centers in the immature brain. In a recent study, Persson–Bunke et al. reported that the risk of developing neuromuscular scoliosis in CP patients increases along with their level in the Gross Motor Function Classification system (GMFCS; Table 1) and with age [1, 2]. Patients with a GMFCS level of IV or V have a 50–75 % risk for developing moderate or severe neuromuscular scoliosis [1–3]. Spinal deformity often involves restrictions for the patients and their caregivers in everyday life. Apart from affecting ambulatory status and sitting balance, scoliosis can lead to cardiopulmonary and gastrointestinal dysfunction and pain in these patients [4–6]. Although surgical correction of neuromuscular scoliosis often leads to good radiological results, the appropriate type and extent of the surgical procedure are still unclear. Posterior-only and combined anterior–posterior spinal instrumented fusion using modern instrumentation systems are established techniques in the treatment of scoliosis in CP patients [7–10]. However, several studies have indicated high overall perioperative complication rates, ranging from 22 to 68 % [7, 11–13]. The perioperative morbidity is known to be high particularly in patients with severe cerebral palsy and nonambulatory status [12–15]. However, to the best of our knowledge there have been no published studies so far focusing exclusively on scoliosis in severe CP patients with a GMFCS of IV or V.
The purpose of this study was to analyze and present the radiological and clinical outcomes in patients with severe CP and neuromuscular scoliosis who underwent two different types of surgical procedure: posterior-only or combined anterior–posterior instrumented fusion.
Materials and methods
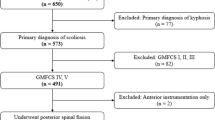
In a retrospective cohort study, patients with neuromuscular scoliosis due to CP who underwent surgery in a single institution between 1997 and 2012 were analyzed. The inclusion criteria were neuromuscular scoliosis due to severe CP with a GMFCS of IV or V, surgical treatment of scoliosis either with posterior-only or combined anterior–posterior spinal instrumented fusion, a minimum follow-up period of 2 years, and a complete set of radiographs before surgery, at discharge, 2 years after surgery, and at the final follow-up examination. Patients who received halo traction were excluded, as well as patients who underwent anterior release without anterior instrumentation. Patient records, surgical reports, anesthesia documentation, and radiographs were evaluated.
The indications for surgery were established on a very individual basis, taking into account not only the radiological spine deformity but also the patients’ overall general health parameters (pulmonary dysfunction, pain, impairment of balanced sitting, comorbidities).
Between 1997 and 2012, the surgical technique used for posterior instrumentation developed from the use of proximal hooks together with pedicle screws in the middle and distal instrumentation (hybrid instrumentation) to pedicle screw–only instrumentation. Patients who underwent both techniques were included. All of the patients were treated with a double-rod steel or titanium posterior system. Bilateral facetectomies were performed in all cases for posterior release. Additional posterior osteotomies were not performed. Following instrumentation and correction maneuvers, spinal fusion was ensured by autologous or allogeneic spondylodesis. The patients in whom the anterior–posterior surgical technique was used received anterior release prior to anterior instrumentation with a single or dual-rod system, followed immediately or in a two-stage procedure by posterior instrumentation.
Evaluation of the patients’ health status preoperatively included their ambulatory status, assessed using the GMFCS. Mental retardation was quantified using three categories: “severe,” “moderate,” and “mild/none” [7, 10]. Preoperative lung disorders (frequent recurrent respiratory infections), dislocations of the hip, and previous operations to treat spasticity (psoas or adductor tenotomies) were noted.
Perioperative data were collected, including the duration of the operation, estimated intraoperative blood loss (ESBL), early and late complications, intensive-care unit (ICU) stay, and hospital stay. Early complications comprised intraoperative and early postoperative complications occurring during the hospital stay, in comparison with late complications, which occurred after hospital discharge or after 6 weeks of a hospital stay [16]. The study focused specifically on the complications defined by Master et al. [12].
Full-spine radiographs (anterior–posterior and lateral views) were taken preoperatively, at discharge, and at the 2-year and final follow-up examinations, either in the sitting or lying position, depending on the patient’s ambulatory status. In addition, preoperative anterior–posterior radiographs under traction were taken in order to evaluate curve flexibility. Coronal Cobb angles and pelvic obliquity were measured on anterior–posterior views. Pelvic obliquity was defined as the angle between a line tangential to both iliac crests and the horizontal line [13, 17]. In addition to the assessment of pelvic obliquity in all of the patients, a subanalysis was also carried out that included only sitting patients, on the assumption that seated versus (vs.) supine postures influence pelvic obliquity. Preoperative flexibility was measured as the relative correction of the Cobb angle of the main curve achieved by traction on the preoperative anterior–posterior view (Cobb angle without traction minus Cobb angle under traction/Cobb angle without traction) [13]. Lumbar lordosis (L1–L5) and thoracic kyphosis (T4–T12) were evaluated on lateral views.
Statistical analysis
Descriptive statistical analyses were carried out using mean and standard deviation as measures of location and scale. In case of outliers or skewness, the median and interquartile ranges were computed. Statistical connections between variables were assessed using Fisher’s exact test, the Chi-squared test, the Mann–Whitney U test, and the Spearman correlation coefficient with corresponding correlation test. Multivariate regression model building was performed by backward variable selection based on the likelihood ratio test. Throughout this paper, a P value ≤0.05 is considered as statistically noticeable. The term “significant” is avoided, as the P values are interpreted in an exploratory rather than a confirmatory fashion, and adjustment for multiple testing is not applied. All statistical calculations were performed using IBM SPSS Statistics for Windows, version 22.0.
Results
A total of 57 patients were included in the study. Thirty-five patients were treated with posterior-only instrumentation (group P, Fig. 1) and 22 patients received combined anterior–posterior instrumentation (group AP, Fig. 2). The mean follow-up period was 4.1 years (range 24–141 months). No statistically noticeable differences between the two surgical groups with regard to the follow-up period were detected (mean follow-up periods 47 months in group P and 53 months in group AP). Four patients died during the follow-up (6–12 years after surgery) of causes independent of their deformities or scoliosis operations. Table 2 presents the basic preoperative data for groups P and AP. Patients with a GMFCS level of V had larger preoperative Cobb angles in the main curve (P = 0.007) than those with GMFCS level IV.
The intraoperative and perioperative data are presented in Table 3. There were statistically noticeable differences between groups P and AP with regard to the duration of surgery, length of hospital stay, length of ICU stay, and ESBL. In group AP, the median ICU stay was 5 days, with one patient receiving intensive care for 34 days due to respiratory insufficiency and wound infection. Statistically noticeable positive correlations were found between age at the time of surgery and the duration of surgery (r = 0.35, P = 0.007), as well between the duration of surgery and the length of the hospital stay (r = 0.46, P < 0.0001), length of ICU stay (r = 0.41, P < 0.002) and ESBL (r = 0.68, P < 0.0001). Positive correlations were also found between the preoperative Cobb angle of the main curve and duration of surgery (r = 0.43, P = 0.001), length of hospital stay (r = 0.57, P < 0.0001) and intensive-care unit stay (r = 0.50, P < 0.0001).
The results of the detailed radiographic analyses are shown in Table 4. The mean preoperative Cobb angle in the entire group was 94° ± 21° (range 42°–140°), corrected to 45° ± 21° after surgery and 48° ± 20° at the final follow-up (mean absolute correction at discharge: 49° ± 21°; mean relative correction: 52 ± 19 %).
No statistically noticeable effects of the preoperative Cobb angle and surgical technique on the relative correction of the Cobb angle at discharge were detected (P = 0.674 and P = 0.451, respectively), but positive correlations were found between preoperative flexibility and relative correction (r = 0.55, P < 0.0001). When groups P and AP were analyzed separately, no statistically noticeable effects of the preoperative Cobb angle on the relative correction were observed in patients in group P alone (P = 0.800), nor in patients in group AP alone (P = 0.928). On the other hand, a statistically noticeable influence of curve flexibility on relative correction was found for patients in group P (r = 0.56, P < 0.0001) and in group AP (r = 0.56, P = 0.008).
Dislocated hips, possibly due to severe spasticity, were associated with low preoperative flexibility (P = 0.007). Patients with dislocated hips had poorer correction results than those with nondislocated hips (mean absolute Cobb correction 41° ± 18° vs. 59° ± 21°; P = 0.003; mean relative correction 46 ± 19 % vs. 61 ± 15 %; P = 0.002).
Loss of correction of Cobb angles turned out to be minimal in both group P and group AP. The greatest loss of correction was observed in patients with a high degree of absolute correction (r = 0.366, P = 0.005) and patients who were treated with hybrid instrumentation (hybrid mean 8° ± 6°, pedicle screws–only mean 3° ± 4°; P = 0.011). No other statistically noticeable differences were found between hybrid constructs with hooks and pedicle screws and pedicle screw–only constructs, especially regarding duration of surgery (hybrid mean 355 min ± 182 vs. pedicle screw–only mean 419 min ± 215; P = 0.125), blood loss (hybrid mean 1760 ml ± 914 vs. pedicle screw–only mean 2430 ml ± 1424; P = 0.124), primary correction (hybrid mean 56 % ± 16 vs. pedicle screw–only mean 49 % ± 20; P = 0.117) and rate of complications (hybrid mean 26 % ± 45 vs. pedicle screw–only mean 37 % ± 49; P = 0.388).
As the overall preoperative radiological data differed between groups P and AP (Table 4), an analysis of subgroups with similar preoperative radiographic data in the two groups was performed. As shown above, preoperative curve flexibility has a statistically noticeable influence on curve correction, and in clinical practice the preoperative Cobb angle is one of the major criteria for establishing indications. We therefore focused particularly on the subgroup of patients with preoperative Cobb angles between 90° and 115° and a flexibility of ≤30 %. In these patients, both a posterior approach and a combined one might be considered in everyday clinical decision-making. In group P (n = 5), there was a mean preoperative Cobb angle of 98° with 59° at the final follow-up (a correction of 41 %), whereas in group AP (n = 7) there was a mean preoperative Cobb angle of 107° with 63° at the final follow-up (a correction of 46 %). These differences were not statistically noticeable. The median ICU stay in this subgroup was 1 day in group P vs. 5 days in group AP; hospital stay 12 vs. 29 days, mean duration of surgery 286 vs. 521 min.
No statistically noticeable correlations were found between the anchor density per level fused and the Cobb angle correction, either in relation to all patients or in groups P and AP separately.
Complications are listed in Table 5, with an overall rate of major complications as defined by Master et al. [12] of 23 % in group P and 46 % in group AP. However, the difference was not statistically noticeable (P = 0.088).
Patients with major postoperative complications had statistically noticeably longer operations (P = 0.009) and longer instrumentations (P = 0.036). No statistically noticeable effects on the rate of major complications were observed in relation to the preoperative Cobb angle and flexibility, sex, age at time of surgery, body mass index (BMI), GMFCS level, grade of mental retardation, preoperative frequent recurrent respiratory infections, preexisting dislocation of the hip, previous tenotomies, presence of a percutaneous endoscopic gastrostomy (PEG) tube, intraoperative blood loss, or number of pedicle screws used. Patients with major respiratory complications had statistically noticeably larger preoperative Cobb angles (P = 0.012) and statistically noticeably longer hospital stays (P = 0.039) and ICU stays (P < 0.0001). Frequent preoperative recurrent respiratory infections (P = 0.089) and combined anterior–posterior treatment (P = 0.086) tended to be associated with a higher rate of major respiratory complications. A stepwise multivariate linear regression analysis indicated that preoperative Cobb angle, duration of surgery, and major postoperative respiratory complications were statistically noticeable predictors for a long hospital stay (adjusted R 2 = 0.462, P < 0.0001) and for a long ICU stay (adjusted R 2 = 0.679, P < 0.0001). Patients with a BMI of less than 15 had more overall complications than those with a BMI above 15 (P = 0.032), however, no noticeably more major complications according to Master et al. (P = 0.389) [12].
In addition, the duration of surgery, estimated blood loss, and length of instrumentation had a statistically noticeable influence on the overall complication rate (P = 0.005, P = 0.039, and P = 0.015, respectively). The preoperative Cobb angle and curve flexibility, age at surgery, sex, GMFCS, mental retardation, presence of a PEG tube, hip dislocation, previous tenotomies, and the number of pedicle screws did not have any statistically noticeable effect on the overall complication rates.
In relation to the time of occurrence of postoperative complications in group P, 6 patients (17 %) had early postoperative complications, one patient (3 %) had early and late complications, and one patient (3 %) had only a late postoperative complication. In group AP, 7 patients (32 %) had early postoperative complications and 3 patients (14 %) had early and late complications.
There were no statistically noticeable differences between the two groups with regard to the revision rate (11 % in group P; 18 % in group AP). The two wound infections in group AP occurred after posterior surgery and required revision during the index hospital stay. Revision surgery was also necessary in 2 patients of group AP in whom pedicle screw dislocation occurred several years after the posterior surgery. In group P one case of pedicle screw malposition led to incomplete paraparesis requiring immediate revision and in another case surgical correction resulted in a postoperative spinal shock due to overcorrection, which had to be revised the same day. Another patient in group P developed multiple dislocations of pedicle screws and had to undergo a revision procedure 4 months after surgery. Wound infection occurred in one patient in group P, requiring revision during the primary hospital stay.
Discussion
This study presents good results of overall Cobb correction of 54 % in group P and 50 % in group AP, comparable with the literature, but in a group of patients with cerebral palsy having more severe deformities and more severe impairment than comparable studies. Despite these extreme patients, complication rates are similar to the literature. Despite the large curves the surgical goals can be achieved with posterior-only surgery and patients benefit from sparing use of the anterior approach with lower complication rates, reduced surgical trauma and lower hospitalization duration. Preoperative curve flexibility was the most relevant predictor for relative curve correction.
As only nonambulatory patients, i.e., GMFCS levels of IV or V, were included, most of the patients were in poor preoperative health, with low BMI, frequent respiratory infections, and progressive deformity. To the best of our knowledge, the present study is the first published series focusing only on patients with GMFCS IV and V. These patients often have several comorbid conditions due to severe neurological impairment, such as frequent respiratory infections, spasticity with hip dislocations, and PEG tubes. There were no statistically noticeable differences with regard to these parameters between the patients in groups P and AP, with a typically high prevalence of frequent respiratory infections in their medical histories of about 50 %.
Comparable studies on neuromuscular scoliosis have usually included only Cobb angles of up to 79° [7–10, 13, 18, 19], with only one study reporting a mean preoperative Cobb angle of 88° [16]. The present study with a mean Cobb angle of 94° included patients with greater deformities than those described in any available studies in the literature. It was therefore only possible to correct the scoliosis to an average Cobb angle of 48° at the final follow-up (in comparison with other results of around 30°), but with a comparable mean relative correction rate of 52 %. In the literature (Table 6), correction rates of between 50 and 68 % have been reported in neuromuscular scoliosis.
In patients with severe deformities, surgeons may need to choose between different types and extents of surgery. One key question prior to surgery is whether a posterior-only approach is sufficient or whether anterior release and instrumentation followed by posterior instrumentation will produce relevantly better results. In this retrospective series, patients were therefore grouped according to these two types of surgery—group P and group AP. Due to the retrospective character of the study, the preoperative radiological parameters differ to a certain extent between the two groups (Table 1). However, analyzing and comparing the two groups provides valuable data that may help improve the establishment of indications in the future. The main difference between group P and group AP is the different mean preoperative Cobb angles of 84° vs. 109°. The relative flexibilities of the main curve showed similar results (34 vs. 27 %). Pelvic obliquity was statistically noticeably poorer in group AP (15° vs. 26°). However, the sagittal balance parameters (thoracic kyphosis, lumbar lordosis) were similar.
Due to the two surgical approaches and greater invasiveness of the procedure, patients in Group AP had statistically noticeably longer operations with greater blood loss and longer hospital stays and ICU stays. The length of posterior instrumentation did not differ between the two groups, nor did the numbers of anchors used.
Correction of the main curve was very similar in the two groups at discharge (group P 54 % vs. group AP 50 %), with only minor loss of correction in both groups at the final follow-up, corresponding well with the literature [7, 8, 13]. Interestingly, the surgical technique did not have a statistically noticeable effect on the relative Cobb correction. These results correspond to the findings reported by Teli et al., who compared patients who underwent posterior-only instrumentation (74° Cobb angle before surgery, 58 % correction) with patients undergoing combined anterior–posterior surgery (76° Cobb angle before surgery, 59 % correction) [9]. Low preoperative flexibility and preoperative hip dislocations had a statistically noticeable negative impact on the absolute and relative curve corrections. Although the preoperative Cobb angle is used in everyday practice as a key parameter for establishing the indications, the present data do not show a statistically noticeable influence of preoperative Cobb angles on relative corrections, either for the overall study cohort or for groups P and AP separately.
Patients with proximal hooks had slightly more loss of correction than those with proximal pedicle screws, a finding that supports the results of earlier studies [20]. No other statistically noticeable differences were found between hybrid constructs and pedicle screw–only constructs.
The correction rates of Cobb angles in this series were not statistically noticeably affected by anchor density. However, the anchor densities in the two groups were very similar. The available studies on neuromuscular scoliosis have not investigated anchor density. In patients with idiopathic scoliosis, Larson et al. described improved relative corrections of major coronal curves in patients with more than 1.54 anchors per level fused in comparison with patients who had fewer anchors [21].
Pelvic obliquity is important for balanced sitting. In the literature, authors’ views regarding the lower instrumented vertebra (LIV) differ between using pedicle screws only to L4 or L5 [7, 22] and application of iliac anchors [18, 23]. Several authors recommend instrumentation down to the pelvis in patients with preoperative pelvic obliquity of more than 15° [7, 22]. In the present series, the preoperative pelvic obliquities differed between the two groups (group P 15°, group AP 26°). However, the LIV was L5 in the majority of patients. Modi et al. reported on a cohort with an average preoperative pelvic obliquity of 23°, which was corrected to 8° using pelvic fixation, in comparison with a cohort with 6° before surgery, which was corrected to 3° without pelvic fixation [7]. The results of pelvic obliquity correction were poorer in the present study, but at discharge 8° (43 % correction) was nevertheless achieved in group P and 12° (51 % correction) in group AP. The greater improvement in group AP in comparison with group P is consistent with the results reported by Teli et al. [9]. This might be attributed to better intraoperative flexibility through anterior release in patients with combined instrumentation. As these overall results include patients who were not able to sit and also sitting patients, and as it might be speculated that seated vs. supine positions may affect pelvic obliquity measurements, a subanalysis was performed including sitting patients alone. This subgroup also only showed a trend toward better correction in group AP. As the majority of the patients had L5 as the LIV, the data are not able to show whether instrumentation to the pelvis leads to better results with regard to pelvic obliquity correction than instrumentation to L5.
One limitation of this study is the fact that there were statistically noticeable differences between the patients in group P and those in group AP with regard to the preoperative Cobb angle, although they had similar degrees of flexibility. For better comparison of the two surgical techniques, a subanalysis was performed including patients with Cobb angles ≥90° and ≤115° and a flexibility of ≤30 %—i.e., patients in whom both posterior-only or combined surgery might be considered in everyday practice. This subanalysis only contains small numbers, but it presents valuable results that show that there is no relevant advantage for a combined approach over posterior-only surgery. This finding supports the results of earlier studies in patients with adolescent idiopathic scoliosis [24, 25].
Master et al. reported a 28 % rate of major complications in their cohort of 131 patients with neuromuscular scoliosis who underwent posterior or combined surgical correction, but did not identify any statistically noticeable differences between posterior-only and combined surgery [12]. In the present study, the complication rate was markedly higher in group AP at 46 % than in group P (23 %). Respiratory complications occurred more often in group AP (major 32 %) than in group P (major 11 %). In a recent meta-analysis of patients who had undergone surgical treatment for neuromuscular scoliosis, Sharma et al. reported that respiratory complications were the most frequent (at 23 %) among all postoperative complications [26]. It is not possible to determine from the present data whether the respiratory complications in the AP group were due to greater respiratory impairment resulting from greater deformity, or due to the additional anterior approach. However, longer operations correlated statistically noticeably with higher complication rates. Interestingly, preoperative Cobb angle and flexibility, sex, age at surgery, BMI, GMFCS, mental retardation, preoperative frequent recurrent respiratory infections, hip dislocation, former tenotomies, PEG tube, blood loss, and number of anchors did not have any statistically noticeable effects on the rates of major complications. However, patients with major respiratory complications had larger preoperative Cobb angles. The results show that patients with a history of frequent respiratory infections, as well as patients in whom a combined approach was used, had higher rates of major respiratory complications, leading to statistically noticeably longer ICU and hospital stays—a finding that corresponds well to the literature data [27]. In a large trial, Tsirikos et al. reported an average ICU stay of 5 days for patients with CP scoliosis who received posterior-only treatment in comparison with 7 days for those with combined operations; the two groups also had hospital stays of 19 and 25 days, respectively [10]. The present results, with median ICU stays of 1 day in group P vs. 5 days in group AP and median hospital stays of 17 vs. 29 days, appear to be similar. In addition to higher complication rates, one major reason for a prolonged ICU stay in group AP was the fact that at that time in our hospital patients were only transferred to the normal ward after removal of the chest drains. In a multivariate linear regression model of the results, the preoperative Cobb angle, duration of surgery, and postoperative respiratory complications were clear predictors for a long ICU stay and hospital stay. This needs to be taken into consideration during consultations with patients and families prior to surgery.
A BMI lower than 15 also increased the risk of overall complications, however not that of major complications. This may encourage surgeons to improve the nutritional status in these patients prior to scoliosis surgery [28].
Revision rates in CP scoliosis surgery are higher than with idiopathic deformities, ranging between 8 and 13 % in the literature [12, 13, 18, 26]. The revision rates in the present study were similar. Interestingly, revision procedures in both groups were always associated with problems in the posterior approach.
Limitations of this study mainly involve in its retrospective nature, the fairly small absolute number of analyzed patients and baseline differences between the two groups, e.g. different Cobb angles before surgery. However, this study is the first available one and therefore largest one focusing on severe deformities in patients with GMFCS levels IV and V. Results—including subgroup analyses—provide valuable data to improve the perioperative guidance that can be offered to patients and families. They may serve as additional help and information in everyday clinical decision-making without claiming broad general application to all other patients with cerebral palsy. Further prospective trials comparing posterior-only and combined techniques would be desirable. Additionally, patients in the presented study had very severe preoperative Cobb angles. One might surmise that much better results and with less complications might be achieved if patients undergo surgery at an earlier stage. Future studies should also focus on patients with less severe curves and compare their results with those of severe curves.
Conclusions
This study shows good results, with an overall Cobb correction of 52 % (54 % in group P, 50 % in group AP), comparable with the literature data, but in a cohort of cerebral palsy patients with more severe deformities (mean preoperative Cobb angle 94°) and severe impairment (GMFCS levels IV and V). Patients with these extreme deformities and GMFCS scores are therefore also clearly able to benefit from surgery. The complication rate is relatively high, due to a strict definition of complications, but the revision rates are similar to those in comparable studies in patients with less severe deformities and lower GMFCS grades. Posterior instrumentation appears to lead to comparable radiological results with shorter operations and shorter ICU and hospital stays than with combined surgery. Preoperative curve flexibility was an important predictor for relative curve correction. The duration of surgery was a relevant predictor for complication rates.
References
Persson-Bunke M, Hägglund G, Lauge-Pedersen H et al (2012) Scoliosis in a total population of children with cerebral palsy. Spine 37:E708–E713. doi:10.1097/BRS.0b013e318246a962
Sandström K, Alinder J, Oberg B (2004) Descriptions of functioning and health and relations to a gross motor classification in adults with cerebral palsy. Disabil Rehabil 26:1023–1031. doi:10.1080/09638280410001703503
Madigan RR, Wallace SL (1981) Scoliosis in the institutionalized cerebral palsy population. Spine 6:583–590
Majd ME, Muldowny DS, Holt RT (1997) Natural history of scoliosis in the institutionalized adult cerebral palsy population. Spine 22:1461–1466
Xue X, Shen J, Zhang J et al (2014) An analysis of thoracic cage deformities and pulmonary function tests in congenital scoliosis. Eur Spine J Off Publ Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc. doi:10.1007/s00586-014-3327-6
Kalen V, Conklin MM, Sherman FC (1992) Untreated scoliosis in severe cerebral palsy. J Pediatr Orthop 12:337–340
Modi HN, Hong J-Y, Mehta SS et al (2009) Surgical correction and fusion using posterior-only pedicle screw construct for neuropathic scoliosis in patients with cerebral palsy: a three-year follow-up study. Spine 34:1167–1175
Piazzolla A, Solarino G, De Giorgi S et al (2011) Cotrel-Dubousset instrumentation in neuromuscular scoliosis. Eur Spine J 20:75–84. doi:10.1007/s00586-011-1758-x
Teli MG, Cinnella P, Vincitorio F et al (2006) Spinal fusion with Cotrel-Dubousset instrumentation for neuropathic scoliosis in patients with cerebral palsy. Spine 31:E441–E447
Tsirikos AI, Lipton G, Chang W-N et al (2008) Surgical correction of scoliosis in pediatric patients with cerebral palsy using the unit rod instrumentation. Spine 33:1133–1140
Comstock CP, Leach J, Wenger DR (1998) Scoliosis in total-body-involvement cerebral palsy. Analysis of surgical treatment and patient and caregiver satisfaction. Spine 23:1412–1424 discussion 1424–1425
Master DL, Son-Hing JP, Poe-Kochert C et al (2011) Risk factors for major complications after surgery for neuromuscular scoliosis. Spine 36:564–571. doi:10.1097/BRS.0b013e3181e193e9
Lonstein JE, Koop SE, Novachek TF, Perra JH (2012) Results and complications after spinal fusion for neuromuscular scoliosis in cerebral palsy and static encephalopathy using luque galveston instrumentation: experience in 93 patients. Spine 37:583–591. doi:10.1097/BRS.0b013e318225ebd5
Tsirikos AI, Chang W-N, Dabney KW, Miller F (2003) Comparison of one-stage versus two-stage anteroposterior spinal fusion in pediatric patients with cerebral palsy and neuromuscular scoliosis. Spine 28:1300–1305. doi:10.1097/01.BRS.0000065572.10824.AB
Lipton GE, Miller F, Dabney KW et al (1999) Factors predicting postoperative complications following spinal fusions in children with cerebral palsy. J Spinal Disord 12:197–205
Watanabe K, Lenke LG, Daubs MD et al (2009) Is spine deformity surgery in patients with spastic cerebral palsy truly beneficial?: a patient/parent evaluation. Spine 34:2222–2232
Peelle MW, Lenke LG, Bridwell KH, Sides B (2006) Comparison of pelvic fixation techniques in neuromuscular spinal deformity correction: galveston rod versus iliac and lumbosacral screws. Spine 31:2392–2398
Sponseller PD, Shah SA, Abel MF et al (2009) Scoliosis surgery in cerebral palsy: differences between unit rod and custom rods. Spine 34:840–844
Modi HN, Suh S-W, Hong J-Y et al (2010) Treatment and complications in flaccid neuromuscular scoliosis (Duchenne muscular dystrophy and spinal muscular atrophy) with posterior-only pedicle screw instrumentation. Eur Spine J 19:384–393. doi:10.1007/s00586-009-1198-z
Kim YJ, Lenke LG, Cho SK et al (2004) Comparative analysis of pedicle screw versus hook instrumentation in posterior spinal fusion of adolescent idiopathic scoliosis. Spine 29:2040–2048
Larson AN, Polly DW, Diamond B et al (2014) Does higher anchor density result in increased curve correction and improved clinical outcomes in adolescent idiopathic scoliosis? Spine 39:571–578. doi:10.1097/BRS.0000000000000204
Whitaker C, Burton DC, Asher M (2000) Treatment of selected neuromuscular patients with posterior instrumentation and arthrodesis ending with lumbar pedicle screw anchorage. Spine 25:2312–2318
Phillips JH, Gutheil JP, Knapp DR Jr (2007) Iliac screw fixation in neuromuscular scoliosis. Spine 32:1566–1570
Dobbs MB, Lenke LG, Kim YJ et al (2006) Anterior/posterior spinal instrumentation versus posterior instrumentation alone for the treatment of adolescent idiopathic scoliotic curves more than 90 degrees. Spine 31:2386–2391. doi:10.1097/01.brs.0000238965.81013.c5
Luhmann SJ, Lenke LG, Kim YJ et al (2005) Thoracic adolescent idiopathic scoliosis curves between 70 degrees and 100 degrees: is anterior release necessary? Spine 30:2061–2067
Sharma S, Wu C, Andersen T et al (2013) Prevalence of complications in neuromuscular scoliosis surgery: a literature meta-analysis from the past 15 years. Eur Spine J 22:1230–1249. doi:10.1007/s00586-012-2542-2
Hod-Feins R, Abu-Kishk I, Eshel G et al (2007) Risk factors affecting the immediate postoperative course in pediatric scoliosis surgery. Spine 32:2355–2360. doi:10.1097/BRS.0b013e3181558393
Jevsevar DS, Karlin LI (1993) The relationship between preoperative nutritional status and complications after an operation for scoliosis in patients who have cerebral palsy. J Bone Joint Surg Am 75:880–884
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beckmann, K., Lange, T., Gosheger, G. et al. Surgical correction of scoliosis in patients with severe cerebral palsy. Eur Spine J 25, 506–516 (2016). https://doi.org/10.1007/s00586-015-4107-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-4107-7