Abstract
Introduction
Cervical spondylotic myelopathy (CSM) can lead to significant disability through a spectrum of clinical manifestations ranging from dexterity loss to more profound weakness, incontinence and paralysis.
Aim
To determine the outcome of surgical decompression for CSM and investigate pre-operative predictors of outcome.
Methods
Prospectively collected data on all patients who underwent decompressive surgery for CSM and completed 12-month follow-up were reviewed. Data on age, MRI T1 and T2 signal changes pre-operatively, surgical approach and the Nurick’s Myelopathy Grade (NMG) was analysed pre-operatively and 1 year post-surgery.
Results
Data on 93 consecutive patients who underwent surgery for CSM were reviewed. Median age was 62 (23–94) years and 59 % were male. The median follow-up was 37 (17–88) months. The approach was anterior in 38 (42 %) patients, posterior in 55 (58 %); improvement was not significantly different when the two groups were compared. The number of levels decompressed increased with age (p value <0.0001). The group with a pre-operatively high signal on T1-weighted MRI images [n = 28 (30 %)] was associated with less neurological recovery post-operatively compared to the patients with a normal T1 cord signal. None of the patients deteriorated neurologically post-operatively, while 66 % improved by at least one NMG.
Conclusion
Surgical decompressions for CSM stop the progress of symptoms at 12 months post-surgery and may result in a significant improvement of NMG in two-thirds of the patients. Changes in the T1-weighted MRI images predict worse outcomes following surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The exact prevalence of cervical spondylotic myelopathy (CSM) is not known but is considered to be a leading cause of spinal cord dysfunction in the adult population [1]. The natural history of the condition appears to favour progression in the majority of cases [2] with up to 60 % of patients deteriorating clinically without surgical intervention. The likelihood of improvement with conservative management was small [3–6] with some studies showing progressive deterioration in all patients without surgery [7]. Surgery appears to be an effective management option not only in stopping the symptom progression but serves as a catalyst for functional improvement in this group of patients [8, 9]. In this paper, we aim to explore outcome predictors in a consecutive cohort of patients managed surgically for CSM and investigate the surgical outcome comparing the anterior and posterior approaches.
Methods
Prospectively collected data on all patients surgically treated for cervical spondylotic myelopathy (CSM) in one spine surgical unit were reviewed. CSM diagnosis was based on clinical examination and confirmed by MRI scans. Patients with the primary diagnosis of CSM who were surgically managed over the period of 4 years between June 2006 and June 2010 with a minimum follow-up of 12 months were included. Data on age, gender, pre-operative MRI cord signal changes in T1- and T2-weighted images, operative data such as the surgical approach (anterior or posterior), the number of levels decompressed, type of surgical intervention (anterior cervical decompression and fusion (ACDF), corpectomy, cervical disc replacement (CDR), posterior decompression ± fusion, laminoplasty) were documented. Nurick’s classification system for myelopathy (NMG) [10] was used to grade the gait abnormality in patients pre-operatively and 1 year post-operatively (Table 1). Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS v.17); following normality studies, the data are presented as median (range). Significance was tested with non-parametric tests for continuous data, while Chi-square test was used for categorical data. A binary logistic regression analysis model was used to explore the predictors of improvement in NMG post-surgery.
Results
93 consecutive patients who underwent intervention were prospectively enroled in the study over a period of 4 years. The median age of the patients was 62 (23–94) years of which 59 % were male. The median follow-up period was 37 (17–88) months. An anterior approach was used in 38 (42 %) patients, while a posterior approach was used in the remaining 55 (58 %) patients. In the anterior approach group, 28 patients were treated with ACDF, 6 had corpectomy and 4 had CDR. In the posterior approach group, 45 patients had posterior decompression (laminectomy) and fusion, while 10 were treated with laminoplasty.
The number of levels decompressed increased with age (Pearson correlation coefficient = 0.41, p value < 0.0001), 15 patients with a mean age of 52 years had one level decompressed, 12 patients with mean age of 64 years had two levels decompressed, 14 patients with a mean age of 68 years had three levels decompressed, 13 patients with a mean age of 68 years had four levels decompressed and 15 patients with a mean age of 70 years had five levels decompressed.
Younger patients were managed with CDR (n 4) with a mean age of 37, laminoplasty (n 10) with a mean age of 51, and ACDF (n 11) with mean age of 54 while older patients underwent corpectomy (n 6) mean age of 65 and posterior decompression and fusion (n 45) with a mean age of 73 years. Posterior instrumented fusion was performed on all patients who underwent posterior laminectomy.
Anterior vs. posterior approach
Selection of approach was based on age, number of levels involved and cervical spine alignment (kyphotic or lordotic) [11]. When compared to the anterior approach, patients within the posterior approach group were significantly older, had a worse pre-operative gait (as indicated by their NMG), had a higher prevalence of T2-weighted cord signal changes on MRI and demonstrated more modest improvement post-operatively. With this in mind, both groups demonstrated a significant post-operative improvement in their NMG (p value <0.0001 for both groups, Wilcoxon signed-ranks test). Table 2 outlines the variances between the two surgical groups (anterior vs. posterior approach).
Cord signal changes and post-operative improvement
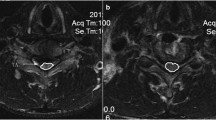
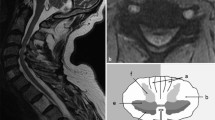
When cord signal changes on the T2-weighted images were considered, there was no significant difference in NMG improvement post-surgery when patients with a positive signal change [n = 65 (70 %)] were compared to patients with a normal cord signal [n = 28 (30 %)] in this imaging sequence (p value 0.96, Chi-square test). On the other hand, a significantly lower improvement in the NMG was noted in patients with a positive cord signal change in T1-weighted images [n = 28 (30 %)] compared to those with a normal cord signal [n = 63 (70 %)] in this phase [NMG improvement = 0 (1), 1 (1), respectively, p value 0.028, Chi-square test]. Pre-operative better NMG scores were recorded amongst patients with normal cord signal on T1- and T2-weighted MRI images (Fig. 1), while worst post-operative improvement was identified amongst patients with pre-operative cord signal changes in both T1- and T2-weighted images (Table 3).
Median pre-operative NMG scores for the three groups. The scores of a group were significantly better than group b, p value = 0.004, Mann–Whitney U test and better than group c although statistically not significant (p value = 0.37, Mann–Whitney U test). There was no significant difference between groups b and c (p value = 0.5, Mann–Whitney U test)
Predictors of post-operative NMG improvement
None of the patients showed any deterioration in gait following surgery. At the time of final assessment, 34 % of the patients did not show any improvement in the post-operative NMG score. Of the remaining group (66 %) who improved, 46 % improved by one NMG grade, 16 % improved by two NMG grades while 4 % achieved an improvement of three grades. Figure 2 illustrates that an improvement was achieved even amongst the higher NMG grades.
A binary logistic regression model investigating improvement by 1 or more NMG grades against age, gender, surgical approach, type of surgical intervention, extent of decompression (number of levels decompressed), pre-operative NMG and pre-operative cord signal changes on MRI (for both T1- and T2-weighted images) did not identify significant improvement predictors (Table 4).
Complications
We have not identified any complications related to implants; However, we encountered five complications in five patients; three superficial wound infections all in patients who were managed through a posterior approach, one wound dehiscence in a case managed with laminoplasty and one fatality due to catastrophic post-operative upper gastrointestinal bleeding.
Discussion
Irrespective of the surgical approach, the main objective of surgery in CSM is to achieve adequate spinal canal decompression while maintaining or restoring spinal stability. This ultimately results in improved cord perfusion and reduced cord oedema [12]. In our cohort, posterior decompression surgery was largely performed in older patients who had multilevel compression and significantly worse NMG scores. This selection bias, we believe, resulted in a smaller post-operative NMG improvement compared to the anterior surgery group making our findings at odds with previously published evidence that suggested no significant difference in the post-operative functional outcome between the two groups [13–17]. A recent systematic review suggested a similar outcome between the two approaches based on eight level III retrospective cohort studies; the authors concluded that there was no significant difference when the improvements in JOA (Japanese Orthopaedic Association) scores and post-operative C5 palsy rates (0–10.2 %) were compared, but the infection rates were lower with anterior surgery and dysphagia rate (7.1–30 % in anterior compared to 0 % for posterior) was lower with posterior surgery [18].
We considered additional factors when choosing the approach including the location of the compressive abnormality and the sagittal alignment of the cervical spine pre-operatively. With posterior decompression, a peak post-operative spinal cord shift >3 mm was associated with a good clinical outcome and was mostly observed in the lordotic compared to straight or kyphotic cervical spine (3.1,<3, <2 mm, respectively) [11].
A posterior decompression was therefore considered contraindicated in patients with a neutral or a kyphotic cervical spine [12] but the findings at follow-up for an average of 5.6 years of 136 patients (mixed cohort of disc herniation and spondylosis) comparing the two approaches identified no significant difference in the neurological recovery as long as the normal anatomic sagittal alignment of the cervical spine is maintained or restored with posterior surgery [19]. Furthermore, Chiba et al. reviewed 70 patients who underwent posterior decompression for CSM with post-operative cervical malalignment and hypothesised that the “slack in the cord” due to the spinal loss of height with degeneration allowed for an acceptable recovery even in a kyphotic or straight spine [20].
Cord signal changes
Although an MRI signal change is not uniformly present in all patients with CSM, signal changes in T1- and in T2-weighted sequences were suggested to indicate a worse clinical picture and predicted limited neurologic improvement after decompression surgery [21–25]. Three patterns of MRI cord signal change in T1/T2 sequences were described in CSM; normal/normal, normal/high and low/high [22]. Such changes were suggested to reflect various stages of intra-spinal oedema, neuronal death, gliosis, microcavitation, and/or demyelination [12] and described to indicate chronicity and severity of the compression [24]. When the patterns were compared in a number of studies, the neurologic recovery was better in patients who had normal cord signal in the T1-weighted sequence [21, 22] as was multi-segmental high-intensity changes on T2-weighted sequences [23] and well-defined signal change borders on T2-weighted sequences [26]. In our cohort, the absence of cord signal change was associated with a better NMG score, but significant neurological improvement was recorded in patients irrespective of the cord signal change pattern. The maximum improvement was identified in patients with a normal/high signal, which might be explained by the severe pre-operative NMG in a cord that has not lost the capacity to recover (cord oedema but no fatty infiltration/irreversible grey matter necrosis [27]). The least improvement was observed in those with a low/high cord signal pattern.
Predictors of outcome
It is recognised that the patient’s over all condition such as an associated neurogenic or vascular claudication, hip/knee advanced osteoarthritis and cardiopulmonary reserve adversely affect the NMG [10]. We have not adjusted our analysis for such factors but we found age not to have a significant predictive effect on post-operative improvement in our cohort. Kawaguchi et al. also found the post-operative improvement amongst 20 patients older than 70 years of age not to be significantly different from 69 younger patients [28].
The time from onset of symptoms is a very elusive parameter. What might be recalled by one patient as abnormal might escape another as relevant. A reasonable proportion of CSM are elderly and suffer with degenerative changes in their upper and lower limbs which might affect their walking, writing and even interfere with fine movements in the hands. Authors who considered this factor in their analysis suggested that earlier intervention results in a better prognosis [29–31]. Other factors implicated for poor post-operative outcome include pre-operative bladder dysfunction [29] and post-operative kyphosis [32]. Our experience with posterior decompression has been to add a postero-lateral instrumented fusion in the area of decompression with the aim to restore pre-operative cervical anatomic alignment and prevent the post-operative destabilising effect of an isolated laminectomy. This has been implicated in iatrogenic kyphosis post-laminectomy and was shown not to be influenced by the number of decompressed levels, resulting in a recurrence of the myelopathic symptoms through progression of compression at adjacent levels [33, 34]. We were unable to identify factors predictive of post-operative outcome in this study; the cord signal changes in the T1-weighted images did not reach statistical significance but had a reasonably good odds ratio for predicting a one or more NMG improvement post-operatively [OR = 1.13 (0.29–4.32)].
We appreciate there was a selection bias in the study because of age difference but we were unable to address that as the type of surgery was related to the age and number of levels affected.
Our study demonstrates that a favourable outcome is expected with the majority of patients managed with surgical decompression for CSM regardless of the approach or the type of surgical intervention. An adequate decompression is expected to halt the progression of the condition and improve function in a good proportion of the patients irrespective of the pre-operative severity of symptoms or MRI scan changes although a T1 signal change might suggest a more reserved post-operative outcome.
References
Bartels RH, Fehlings MG, Abumi K (2013) Cervical spondylotic myelopathy: an update: editorial. Eur Spine J. doi:10.1007/s00586-013-3105-x
Clarke E, Robinson PK (1956) Cervical myelopathy: a complication of cervical spondylosis. Brain 79:483–510
Karadimas SK, Erwin WM, Ely CG, Dettori JR, Fehlings MG (2013) Pathophysiology and natural history of cervical spondylotic myelopathy. Spine (Phila Pa 1976) 38:S21–S36. doi:10.1097/BRS.0b013e3182a7f2c3
Matz PG, Anderson PA, Holly LT, Groff MW, Heary RF, Kaiser MG, Mummaneni PV, Ryken TC, Choudhri TF, Vresilovic EJ, Resnick DK (2009) The natural history of cervical spondylotic myelopathy. J Neurosurg Spine 11:104–111. doi:10.3171/2009.1.SPINE08716
Kalsi-Ryan S, Karadimas SK, Fehlings MG (2013) Cervical spondylotic myelopathy: the clinical phenomenon and the current pathobiology of an increasingly prevalent and devastating disorder. Neurosci Rev J Bringing Neurobiol Neurol Psychiatry 19:409–421. doi:10.1177/1073858412467377
Al-Tamimi YZ, Guilfoyle M, Seeley H, Laing RJ (2013) Measurement of long-term outcome in patients with cervical spondylotic myelopathy treated surgically. Eur Spine J Off Pub Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc 22:2552–2557. doi:10.1007/s00586-013-2965-4
Sadasivan KK, Reddy RP, Albright JA (1993) The natural history of cervical spondylotic myelopathy. Yale J Biol Med 66:235–242
Furlan JC, Kalsi-Ryan S, Kailaya-Vasan A, Massicotte EM, Fehlings MG (2011) Functional and clinical outcomes following surgical treatment in patients with cervical spondylotic myelopathy: a prospective study of 81 cases. J Neurosurg Spine 14:348–355. doi:10.3171/2010.10.SPINE091029
Sampath P, Bendebba M, Davis JD, Ducker TB (2000) Outcome of patients treated for cervical myelopathy. A prospective, multicenter study with independent clinical review. Spine (Phila Pa 1976) 25:670–676
Nurick S (1972) The pathogenesis of the spinal cord disorder associated with cervical spondylosis. Brain 95:87–100
Sodeyama T, Goto S, Mochizuki M, Takahashi J, Moriya H (1999) Effect of decompression enlargement laminoplasty for posterior shifting of the spinal cord. Spine (Phila Pa 1976) 24:1527–1531 (discussion 1531–1522)
Rao R, Gourab K, David KS (2006) Operative treatment of cervical spondylotic myelopathy. J Bone Joint Surg Am 88:1619–1640. doi:10.2106/JBJS.F.00014
Sakaura H, Hosono N, Mukai Y, Ishii T, Iwasaki M, Yoshikawa H (2005) Long-term outcome of laminoplasty for cervical myelopathy due to disc herniation: a comparative study of laminoplasty and anterior spinal fusion. Spine (Phila Pa 1976) 30:756–759. doi:10.1097/01.brs.0000157415.79713.7e
Wada E, Suzuki S, Kanazawa A, Matsuoka T, Miyamoto S, Yonenobu K (2001) Subtotal corpectomy versus laminoplasty for multilevel cervical spondylotic myelopathy: a long-term follow-up study over 10 years. Spine (Phila Pa 1976) 26:1443–1447 (discussion 1448)
Fehlings MG, Barry S, Kopjar B, Yoon ST, Arnold P, Massicotte EM, Vaccaro A, Brodke DS, Shaffrey C, Smith JS, Woodard E, Banco RJ, Chapman J, Janssen M, Bono C, Sasso R, Dekutoski M, Gokaslan ZL (2013) Anterior versus posterior surgical approaches to treat cervical spondylotic myelopathy: outcomes of the prospective multicenter AOSpine North America CSM study in 264 patients. Spine (Phila Pa 1976) 38:2247–2252. doi:10.1097/BRS.0000000000000047
Liu T, Xu W, Cheng T, Yang HL (2011) Anterior versus posterior surgery for multilevel cervical myelopathy, which one is better? A systematic review. Eur Spine J Off Pub Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc 20:224–235. doi:10.1007/s00586-010-1486-7
Zhu B, Xu Y, Liu X, Liu Z, Dang G (2013) Anterior approach versus posterior approach for the treatment of multilevel cervical spondylotic myelopathy: a systemic review and meta-analysis. Eur Spine J Off Pub Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc 22:1583–1593. doi:10.1007/s00586-013-2817-2
Lawrence BD, Jacobs WB, Norvell DC, Hermsmeyer JT, Chapman JR, Brodke DS (2013) Anterior versus posterior approach for treatment of cervical spondylotic myelopathy: a systematic review. Spine (Phila Pa 1976) 38:S173–S182. doi:10.1097/BRS.0b013e3182a7eaaf
Kawakami M, Tamaki T, Iwasaki H, Yoshida M, Ando M, Yamada H (2000) A comparative study of surgical approaches for cervical compressive myelopathy. Clin Orthop Relat Res 381:129–136
Chiba K, Toyama Y, Watanabe M, Maruiwa H, Matsumoto M, Hirabayashi K (2000) Impact of longitudinal distance of the cervical spine on the results of expansive open-door laminoplasty. Spine (Phila Pa 1976) 25:2893–2898
Suri A, Chabbra RP, Mehta VS, Gaikwad S, Pandey RM (2003) Effect of intramedullary signal changes on the surgical outcome of patients with cervical spondylotic myelopathy. Spine J Off J North Am Spine Soc 3:33–45
Morio Y, Teshima R, Nagashima H, Nawata K, Yamasaki D, Nanjo Y (2001) Correlation between operative outcomes of cervical compression myelopathy and MRI of the spinal cord. Spine (Phila Pa 1976) 26:1238–1245
Fernandez de Rota JJ, Meschian S, Fernandez de Rota A, Urbano V, Baron M (2007) Cervical spondylotic myelopathy due to chronic compression: the role of signal intensity changes in magnetic resonance images. J Neurosurg Spine 6:17–22. doi:10.3171/spi.2007.6.1.4
Yagi M, Ninomiya K, Kihara M, Horiuchi Y (2010) Long-term surgical outcome and risk factors in patients with cervical myelopathy and a change in signal intensity of intramedullary spinal cord on magnetic resonance imaging. J Neurosurg Spine 12:59–65. doi:10.3171/2009.5.SPINE08940
Cho YE, Shin JJ, Kim KS, Chin DK, Kuh SU, Lee JH, Cho WH (2011) The relevance of intramedullary high signal intensity and gadolinium (Gd-DTPA) enhancement to the clinical outcome in cervical compressive myelopathy. Eur Spine J Off Pub Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc 20:2267–2274. doi:10.1007/s00586-011-1878-3
Avadhani A, Rajasekaran S, Shetty AP (2010) Comparison of prognostic value of different MRI classifications of signal intensity change in cervical spondylotic myelopathy. Spine J Off J North Am Spine Soc 10:475–485. doi:10.1016/j.spinee.2010.03.024
Mizuno J, Nakagawa H, Inoue T, Hashizume Y (2003) Clinicopathological study of “snake-eye appearance” in compressive myelopathy of the cervical spinal cord. J Neurosurg 99:162–168
Kawaguchi Y, Kanamori M, Ishihara H, Ohmori K, Abe Y, Kimura T (2003) Pathomechanism of myelopathy and surgical results of laminoplasty in elderly patients with cervical spondylosis. Spine (Phila Pa 1976) 28:2209–2214. doi:10.1097/01.BRS.0000085029.65713.B0
Lee TT, Manzano GR, Green BA (1997) Modified open-door cervical expansive laminoplasty for spondylotic myelopathy: operative technique, outcome, and predictors for gait improvement. J Neurosurg 86:64–68. doi:10.3171/jns.1997.86.1.0064
Chiles BW, 3rd, Leonard MA, Choudhri HF, Cooper PR (1999) Cervical spondylotic myelopathy: patterns of neurological deficit and recovery after anterior cervical decompression. Neurosurgery 44:762–769 (discussion 769–770)
Tetreault LA, Karpova A, Fehlings MG (2013) Predictors of outcome in patients with degenerative cervical spondylotic myelopathy undergoing surgical treatment: results of a systematic review. Eur Spine J Off Pub Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc. doi:10.1007/s00586-013-2658-z
Kohno K, Kumon Y, Oka Y, Matsui S, Ohue S, Sakaki S (1997) Evaluation of prognostic factors following expansive laminoplasty for cervical spinal stenotic myelopathy. Surg Neurol 48:237–245
Kaptain GJ, Simmons NE, Replogle RE, Pobereskin L (2000) Incidence and outcome of kyphotic deformity following laminectomy for cervical spondylotic myelopathy. J Neurosurg 93:199–204
Kato Y, Iwasaki M, Fuji T, Yonenobu K, Ochi T (1998) Long-term follow-up results of laminectomy for cervical myelopathy caused by ossification of the posterior longitudinal ligament. J Neurosurg 89:217–223. doi:10.3171/jns.1998.89.2.0217
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salem, H.M.I., Salem, K.M.I., Burget, F. et al. Cervical spondylotic myelopathy: the prediction of outcome following surgical intervention in 93 patients using T1- and T2-weighted MRI scans. Eur Spine J 24, 2930–2935 (2015). https://doi.org/10.1007/s00586-015-4028-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-4028-5