Abstract
Introduction
Established treatment options of spondylodiscitis, a rare but serious infection of the spine, are immobilization and systemic antibiosis. However, the available data for specific treatment recommendations are very heterogeneous. Our intention was to develop a classification of the severity of spondylodiscitis with appropriate treatment recommendations.
Materials and methods
From 10/1/1998 until 12/31/2004, 37 cases of spondylodiscitis were examined regarding medical history, gender status, location and extent of spondylodiscitis, type and number of operations. Subsequently, a classification of six grades according to severity has been developed with specific treatment recommendations. The further evaluation of our classification and corresponding treatment modalities from 1/1/2005 to 12/31/2009 including further 132 cases, resulted in a classification of only three grades of severity (the SSC—spondylodiscitis severity code), with a follow-up until 12/31/2011. Between 01/01/2012 and 12/31/2013, a prospective study of 42 cases was carried out. Overall, 296 cases were included in the study. 26 conservatively treated cases were excluded.
Results and Conclusion
The main localization of spondylodiscitis was the lumbar spine (55 %) followed by the thoracic spine (34 %). The classification of patients into 3 grades of severity depends on clinical and laboratory parameters, the morphological vertebral destruction seen in radiological examinations and the current neurological status. Therapies are adapted according to severity and they include a specific surgical management, systemic antibiotic therapy according to culture and sensitivity tests, physiotherapy and initiation of post-hospital follow-up. 40.6 % of patients are associated with neurological deficits, classified as severity grade 3 and treated surgically with spinal stabilization and decompression. 46.9 % of patients corresponded to severity grade 2, with concomitant vertebral destruction were dorsoventrally stabilized. The 31 patients of severity Grade 1 were treated surgically with dorsal stabilization. From 1998 to 2013, the time from the onset of symptoms to the first surgical treatment was about 69.4 days and has not changed significantly. However, the time from admission to surgical treatment had been reduced to less than 2 days. Also the time of hospitalization was reduced and we see positive effects regarding the sensation of pain. 270 patients underwent surgery. We treated 89 % dorsally and 21 % dorsoventrally. With the spondylodiscitis severity code, a classification of the severity of spondylodiscitis could be established and used for a severity-based treatment. In addition, specific parameters for the treatment of individual grades of severity can be determined in a clinical pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The term osteomyelitis of the vertebral discs, also called spondylodiscitis, denotes a state of bacterial inflammation of the osteoligamentous spine, which can spread to adjacent structures of the spinal canal and the surrounding soft tissues. This is a life-threatening disease, which had a mortality rate of more than 50 % in the first half of the twentieth century. This could be reduced towards the end of the last century to less than 15 % [24]. According to previous studies, this disease has an incidence of 1:200,000–1:250,000 which is only 2–4 % of all infectious bone diseases [14, 23]. Currently, an increasing number of surgical procedures in the spine area are emerging. In addition, there are an increased number of immunosuppressed people from infectious diseases, intravenous drug addiction as well as an increasing number of people with diabetes, rheumatological diseases and malign tumors. A higher life expectancy with the result of older patients and a change of the bacterial spectrum with the appearance of multidrug-resistant bacteria are described as the causes of the increase in incidence of spondylodiscitis [2, 7, 25, 42].
Etiology and classification
A uniform classification of spondylodiscitis has not been established. According to etiology, spondylodiscitis can be differentiated into bacterial, fungal and parasitic spondylodiscitis. Fungal and parasitic spondylodiscitis is more likely an exotic diagnosis. Furthermore spondylodiscitis can be specific like Tbc, M. Bang, Lues or non-specific. According to origin of infection, the disease can be described as an endogenous or an exogenous infection. The progress of spondylodiscitis is either acute or chronic. It can also be described according to the localization in the spine [39, 41]. In the more common non-specific spondylodiscitis, as an endogenous infection, the main pathogenetic agents are Staphylococcus aureus, followed by Streptococci, Enterococci, Salmonella, Pseudomonas and Brucella hematogenous. It occurs as direct infection from adjacent inflamed spinal structures or as focus spread, both arterial and venous with bacteremic cell embolization of capillaries, resulting into infarction [41]. A venous backward flow in the paraspinal venous plexus (Batson plexus) caused by increased intraperitoneal pressure can lead to the spread of a current intraabdominal or pelvic infection into the spinal canal. Furthermore, spinal anesthesia, lumbar punctures, urogenital infections, and urological and gynecological operations can lead to spondylodiscitis [1, 31, 38]. An exogenous spondylodiscitis is the consequence of bacterial contamination. Almost exclusively the disease was preceded by surgical procedures such as discectomy, discography, chemonucleolysis or vertebroplasty, kyphoplasty or stabilizations. Less frequently it is the result of penetrating injuries [1, 31, 39].
Clinical signs, investigations and diagnosis
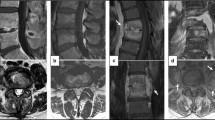
The clinical symptoms and signs are elevated temperature and back pain. Chronic disease shows low-grade fever, fatigue and weight loss. Moreover, neurological deficits of the bladder and rectum may occur. About 60–75 % of patients show pathologically elevated white blood cell count, increased erythrocyte sedimentation rate and pathological values of C-reactive protein [29, 39]. A microbiological blood sample taken during temperature rise is essential to isolate a pathogen [22].
Treatment goals
The goal of therapy is primarily the treatment of infection while avoiding further complications, such as spinal instability, neurological deficits and occurrence of septic spreading with the risk of general sepsis and death. This goal can be achieved in less severe and chronic cases by the well-known conservative treatment. It includes consequent immobilization, wearing a corset, long-term administration of antibiotics in consideration of drug resistance and control of infectious clinical signs as well as the X-ray findings. The antibiotic therapy is initially intravenous until there is a significant decrease of inflammatory markers––clinically and by means of laboratory parameters (CRP and leukocytes). Then, the antibiotic will be continued by oral intake. Regarding the duration of antibiotic therapy, different recommendations can be found [6, 32, 39, 41]. Decision making regarding the termination of antibiotic therapy also depends on radiological results (X-ray, CT and MRI). Conservative treatment includes immobilization of the spine. The timing of careful mobilization in an orthesis should be pain adapted and according to improvement of clinical symptoms and a drop of inflammatory parameters [5, 34]. Persistence, progression or spreading of infection into surrounding tissues can lead to neurological deficits, pneumonic infiltrates and even abscess formation. Other risks of conservative treatment are a possible instability in the affected spinal segment and pseudarthrosis. Surgical therapy is important in cases of advanced bone destruction of the vertebral bodies with instability, imminent neurological deficits, abscess formation and sepsis. The aim of surgery is an “internal fixation” to eliminate the instability and compression of the spinal cord, also the removal of the inflammatory focus, which can include filling of defect zones, excision and drainage of abscesses. Simultaneously there is the possibility of biopsy for histological and microbiological studies [22, 32, 39].
Due to the usually uncharacteristic symptoms, the diagnosis of spondylodiscitis is often delayed. About 30 % of the patients receive an appropriate therapy at least 3 months after appearance of first symptoms [31, 39]. That explains a very heterogeneous course of disease in different patients. This leads to the necessity of a severity orientated surgery [2]. Publications on a possible severity classification of spondylodiscitis with recommendations for treatment algorithms are rare and often with a small number of cases [32, 40].
Since 2004 we treat spondylodiscitis in answer to the following three questions:
-
Is there bone destruction with imminent instability?
-
Is there a neurological deficit?
-
Are adjacent structures involved?
The intention of this work is the presentation of spondylodiscitis therapy based on a classification of severity. Therefore, we analyze the periods 1998–2004, 2005–2009, 2010–2011 and 2012–2013 regarding various parameters such as pain level, duration of stay in hospital and inflammatory parameters.
Materials and methods
Based on a retrospective data collection from 01/01/1998 to 12/31/2004, a clinical–radiological classification of severity of spondylodiscitis, focused on surgical treatment, has been developed for planning the therapy. This classification was developed in consideration of imminent instability, neurological deficits and the involvement of adjacent structures (Table 1). Therefore, an additional subgrouping (A and B), depending on an involvement of neighboring tissues of the spine was established until 12/31/2009. The subgroup B includes abscesses in the neighborhood of spine ore muscles, which were treated with excision. The following classification of severity is the result of our spondylodiscitis cases from 10/01/1998 until 12/31/2004.
Severity grade I A/B
All cases with verified spondylodiscitis without neurological deficits, instability, destruction of the vertebral bodies, kyphosis or narrowing of the spinal canal have been included. Conservative treatment is an option. Depending on the subgroup (A or B), a thoracic or lumbar dorsal internal fixation was performed, excluding the affected vertebral bodies. Antibiotic therapy was administered for 3 months following surgery and was pathogen-specific. An X-ray follow-up was performed 2 and 6 weeks after surgery.
Severity grade II A/B
All cases with destabilizing destruction of the vertebral bodies, but without neurological deficits were included. The surgical therapy was a dorsal internal fixation in the thoracic or lumbar spine, with an attempt to correct the kyphosis. Antibiotic therapy was, whenever possible, pathogen-specific and was administered postoperatively for 3 months. We did a CT scan follow-up 3 months after surgery to check ventral fusion and to decide whether a further procedure in the mean of ventral internal fixation was necessary. When there still was an apparent destruction of the vertebral body, a ventral internal fixation with removal of the inflammatory focus was performed.
Severity grade III A/B
All cases with neurological symptoms were included. The degree of bone destruction was secondary. Surgery was performed immediately after admission including thoracic or lumbar dorsal internal fixation. Decompression of the spinal canal was achieved by laminotomy or laminectomy. Moreover, a biopsy was taken and the inflammatory focus was removed from a costotransversal or dorsolateral approach with simultaneous insertion of an antibiotic releasing material. Depending on the subgroup, an additional ventral stabilization was performed in a short interval after the initial surgery. Antibiotic therapy was pathogen-specific and was administered postoperatively for 3 months. An X-ray follow-up was performed 2 and 6 weeks after surgery for early detection of complications such as reformation of kyphosis and implant failure.
After the re-evaluation in 2009, we performed an adjustment of severity classification since 01/01/2010 (Table 2). Furthermore, we developed a scoring system for spondylodiscitis, which we term SponDT, (Spondylodiscitis Diagnosis and Treatment) based on three traits: (I) the inflammatory marker C-reactive protein (CRP) (mg/dl), (II) pain according to a numeric rating scale (NRS) and (III) magnetic resonance imaging (MRI), to monitor its progression following treatment. During this year, many paravertebral abscesses were drained CT-guided. For this reason, a subgrouping was not necessary anymore (Table 3). We performed following surgical technique: internal dorsal stabilization (grade 1), an additional second stage ventral supply of the thoracic or lumbar spine in case of instability or absence of fusion (grade 2) or spinal decompression and early ventral surgery (grade 3).
Statistics was done by the Kruskal–Wallis test as a statistical test for differences in the central tendency of rank data with more than 2 groups. Paired comparisons were performed using Tukey’s HSD. For the questions of frequency, the test was performed by Monte Carlo exact test. The correlation questions were answered by Cramer’s V as a measure of the relationship between two discrete variables.
A positive ethical approval of the University of Jena is existent.
Results
From 01/01/1998 until 12/31/2013, we were able to analyze 296 cases of spondylodiscitis. 26 were treated conservatively and found no further consideration in this analysis of surgically treated spondylodiscitis. On an average, 20–30 patients with spondylodiscitis were surgically treated per year (Fig. 1).
There were 56.7 % male patients and 43.3 % of female patients. 26 patients were treated conservatively. Of the surgically treated patients, 16 passed away during treatment. Thus, the mortality rate is 5.9 %. The age distribution shows in the course of the 15-year study a shift to older age and a significant age peak in the 60–80 years of age group (Fig. 2).
The etiology of spondylodiscitis stays uncertain in 34.3 % of cases. In 36.2 %, an iatrogenic cause was considered as they occurred after a medical procedure such as injection or surgery. A previous infection was causative in 14.3 %. Other origins were vertebral fractures (7.5 %), systemic diseases (6.0 %) and tumors (1.5 %). The time between the onset of symptoms and admission to hospital with diagnosed spondylodiscitis did not change significantly (64.4 days). In the prospective period 2012/2013, spondylodiscitis could receive appropriate treatment in 42 days after symptom onset (Fig. 3).
Prior to admission, 70 % of patients had not received any treatment before. 30 % of patients were in hospital before, but only 6.3 % of them had been treated surgically (Fig. 4).
Spondylodiscitis is found most frequently in the lumbar spine (55 %), then in the thoracic spine (38 %) and in the cervical spine (11 %).
Between 1998 and 2004, 10 patients of spondylodiscitis were included in consideration of the three severity levels with the subgrouping A and B. A clear predominance of cases with neurological deficits was seen (60 %). From 01/01/2005 to 12/31/2009, 132 patients were grouped into these three severity levels. Depending of subgrouping, patients were treated within six treatment regimens. We found that an only dorsal stabilization is often not sufficient. An additional ventral stabilization was necessary in approximately 50 % of cases. In 34.6 %, a neurological impairment was surgically treated by spinal decompression. In these severe cases, surgical treatment of spondylodiscitis is preferable to conservative therapy and should be carried out within an established clinical pathway. Furthermore, many paravertebral abscesses were drained CT guides. For these reasons, a subgrouping was not necessary anymore. This resulted in a reduction of the previous six treatment options to three grades of severity with specific treatment guidelines each since 01/01/2010. We treated 35.2 % patients with grade 1, 40 % 2 and 24.8 % with decompression in grade 3 per year.
The symptom-based treatment is also reflected in the distribution of neurological deficits according to the ASIA classification. Overall, about 37.4 % of the patients were neurologically impaired at admission. These patients were treated according to grade 3 of severity. Thereby, a remission in ASIA A to ASIA C classified cases could be achieved, so that 84.4 % of these patients could be discharged from hospital with ASIA D or E.
Another condition for severity assessment is the extent of vertebral bone destruction, which has been visualized in 78.1 % by CT scan and in 64.5 % by X-ray. No bone destruction could be seen in 6.7 % of cases by CT and 14.3 % by radiography, so that these cases were treated according to severity level 1 or remained conservative. The initial CT findings showed no significant change at discharge.
The average time of hospitalization was 40.8 days, with an average stay on ICU of 5.9 days. The systematic treatment depending on the grades of severity of spondylodiscitis in the period 2012/2013 could almost halve the duration of hospitalization to 23.6 days (Fig. 5). Likewise, the ICU stay was reduced to 2.3 days.
Beside the reduction of neurological deficits, vertebral stabilization and pain reduction, the decline in inflammatory markers is a crucial parameter of successful treatment. By early surgical treatment after an average of 5.2 days after admission and additional antibiotic treatment, a significant reduction of CRP was achieved. On admission, the average CRP value was 119.2 mg/dl. At the beginning of our observed time span, CRP was 95.8 mg/dl and during the last period of measuring from 2012 to 2013, 135.5 mg/dl. In the second week of treatment, the CRP value had decreased to an average of 72.9 mg/dl, in the prospective observation period even to 54.8 mg/dl (Fig. 6).
Discussion
Despite considerably improved diagnostic possibilities, there are no standardized treatment guidelines for spondylodiscitis due to a heterogeneous patient population and multiple therapeutic options [13, 39]. Few standardized treatments are suggested, but at present there are no prospective randomized controlled trials and the level of evidence for treatment recommendations remains low [2, 3, 13]. Therefore, the aim was to develop a treatment standard, which includes the advantages of different treatment options, taking into account the latest developments in inpatient care and focused on current recommendations.
The only common recommendation for successful therapy of spondylodiscitis is the immobilization of the affected vertebral section and the targeted antibiotic therapy. Further recommendations such as debridement, decompression and operative stabilization vary a lot, depending on the extent of disease [11, 21, 26, 28, 36].
The aim of surgery is the removal of the septic focus with simultaneous pathogen detection and the stabilization of the infected spinal segment with subsequent fusion. In addition to a debridement, the surgical stabilization is the standard procedure [10, 11, 21]. However, implantation of osteosynthetic materials or interposition of bone implants into an infected area is discussed controversially at present. The use of titanium implants is increasing [27, 33, 37]. Nevertheless, there is no broad agreement on the appropriate surgical strategy [4, 9, 11, 28]. During the first period, we treated our patients by insertion of bone grafts. In the more recent years, we mainly used titanium cages. Recommendations for intravenous and oral antibiotic therapy are also not consistent, for non-specific spondylodiscitis it is usually an oral antibiotic therapy for 6–12 weeks [6, 8, 25, 39].
The key point when treating spondylodiscitis is the surgical removal of the infectious focus with simultaneous internal fixation according to recent publications where radical debridement is described as superior to conservative therapy [30]. Furthermore, current literature indicates that surgically treated patients have a better quality of life and more patient satisfaction [35]. Our analysis shows that surgically treated patients are less in pain and show better fusion of the spondylodiscitis-affected segments. Other studies report that surgically treated patients go through a significantly shorter time of immobilization and hospitalization [12, 17]. An only conservative treatment is an option for a low-grade spondylodiscitis (Grade 1) and patients with high-risk co-morbidities for surgery or yet uncertain diagnosis [23, 38].
An epidural abscess without involvement of the vertebral body and the intervertebral disc space cannot be described as spondylodiscitis. In these cases, the treatment of affected adjacent structures and the discitis could be performed non-operatively. In cases of spondylodiscitis with abscess formation, drainage is necessary in addition to debridement and internal fixation [15, 16].
In the interest of facile practicability and easy implementation of a clinical pathway, the original 6 treatment options have been reduced to a number of three, as a result of this study. We aim for the rapid improvement of the patients’ general condition, proper immobilization of the affected spinal segment and removal of the infectious through appropriate surgery, also for the prevention of secondary deformities, a low recurrence rate and shorter hospitalization [11, 21, 26, 28, 36]. Besides the long-preferred ventral approach with removal of the affected segments and their reconstruction, now the dorsoventral or the solely dorsal approach has been established [4, 10, 17, 23, 28, 35]. Therefore, the solely dorsal stabilization is recommended in grade 1 severity of spondylodiscitis without bone destruction or kyphosis caused by disc loss and distraction of the vertebral bodies. In case of infection of the intervertebral disc and the vertebral body, too, an internal fixation with taking a sample for lab analysis should be performed. The reasons for that decision are:
-
Increasing the chance to identify the pathogenic agent.
-
Patients are instantly mobile without an obstructive and uncomfortable corset.
-
The region of inflammation is immobilized.
There is nothing to be said against a percutaneous approach. If the patient was not eligible for surgery or did not consent to it, we treated conservatively.
In cases of grade 2 severity, the formation of a segmental kyphosis with loss of alignment can be effectively prevented by posterior stabilization and an additional ventral fixation [21].
Considering the higher invasiveness of ventral procedures, except in the cervical spine, the anterior internal fixation is not recommended as a stand-alone therapy. There are no recommendations regarding the lack of tension band in isolated posterior instrumentation in the case of existing anterior stability. Furthermore, loosening of the ventrally placed material can occur in cases of osteoporosis [10, 28]. Spinal decompression was not much discussed in the treatment of spondylodiscitis in the case of intraspinal space-occupying lesion. However, an isolated intraspinal abscess is not attributable to the clinical picture of spinal osteomyelitis. In the treatment of cases with severity 3, in addition to spinal decompression is also the causative pronounced spondylodiscitis, usually of several segments, a focus of therapy. In addition to the immediate surgical treatment, in the neurologically impaired patients is paralysis suitable training and treatment essential. This allows about half of the paralyzed patients to expect an improvement of the initial symptoms [20]. The therapy is according to standardized treatment approach followed a clinical pathway is recommended [18, 19].
The classification of patients for spondylodiscitis therapy results from three criteria. In case of neurological symptoms, severity grade 3 is used and includes the immediate spinal decompression, internal fixation followed by an aftercare according to the degree of paralysis. If there is neither a neurological impairment nor a radiological evidence of bone destruction of the affected segment, the patient is treated according to grade 1. In these cases, an antibiotic therapy according to peripheral and local pathogen determination and an internal fixation is appropriate. Thus, the possibility of contamination of the implanted foreign material is limited. Cases with bone destruction are considered as severity grade 2 and are attended to by two-step dorsoventral surgery for thoracic or lumbar spine.
Magnetic resonance imaging and computer tomography are the standard radiological methods to diagnose spondylodiscitis [3, 31, 43]. Our results also show that in case of spondylodiscitis an osteolytic lesion of the vertebral bodies is a frequent finding in radiological examinations. Hence, dorsoventral internal fixation is the most prevalent form of treatment.
During the early years of our study with the initial six grades of severity (1998–2009), patients with neurological deficits were predominant, which explains the prolonged inpatient stay due to the more complex surgical, antibiotic and rehabilitative therapy.
The reduction of initially 6 to finally three grades of severity of spondylodiscitis was due to a more practicable handling in clinical practice and easier distribution to electronic-based clinical pathways. It had no influence on the proportion of the severity grades. However, during the course of the analyzed years, an initial preponderance of grade 3 stood out. We see an explanation in the still small number of cases and a possible unintentional selection of rather severe cases, due to our level-one facility. In the later years of our observed period, there were a relatively constant number of 20–30 patients per year. In our opinion, older age of the patient is the cause of the observed increasing ASA classification, worse general condition on admission to hospital and also significant higher inflammatory markers over the time of our study.
In summary, the standardized treatment of spondylodiscitis according to three grades of severity is highly recommended. It results in a significant shortening of the therapy-free interval and an early reduction of inflammatory markers, compared to non-standardized treatment. The time of hospitalization and intensive care is shortened. Furthermore, our classification of severity of spondylodiscitis can be translated into electronic-based clinical pathways leading to a significant increase of the quality of treatment.
References
Adler CP (1985) Spondylitis–Spondylodiscitis. Radiologe 25:291–298
Akbar M, Lehner B, Doustdar S, Fürstenberg CH, Hemmer S, Bruckner T, Carstens C, Wiedenhöfer B (2011) Pyogene Spondylodiszitis der Brust- und Lendenwirbelsäule. Eine neue Klassifikation zur Entscheidungsfindung bei der Wahl der operativen therapie. Orthopade 40:614–623
Amini MH, Salzman GA (2013) Infectious spondylodiscitis: diagnosis and treatment. Mo Med 110(1):80–4. Erratum in: Mo Med. 110(2):121
Arnold PM, Baek PN, Bernardi RJ (1997) Surgical management of nontuberculous thoracic and lumbar vertebral osteomyelitis: report of 33 cases. Surg Neurol 47:551–561
Bettini N, Girardo M, Dema E, Cervellati S (2009) Evaluation of conservative treatment of non specific spondylodiscitis. Eur Spine J 18:143–150
Colmenero JD, Jiménez-Mejías ME, Sánchez-Lora FJ, Reguera JM, Palomino-Nicás J, Martos F, de García las Heras J, Pachón J (1997) Pyogenic, tuberculous, and brucellar vertebral osteomyelitis: a descriptive and comparative study of 219 cases. Ann Rheum Dis 56:709–715
D’Agostino C, Scorzolini L, Massetti AP, Carnevalini M, d’Ettorre G, Venditti M, Vullo V, Orsi GB (2010) A seven-year prospective study on spondylodiscitis: epidemiological and microbiological features. Infection 38(2):102–107
Dünkel J, Oestmann A, Jonczy M (2013) Sterile spondylodiscitis. Praxis 18;102(19):1195–1198
Eysel P, Hopf C, Meurer A (1994) Korrektur und Stabilisierung der infektbedingten Wirbelsäulendeformität. Orthopädische Praxis 11:696–703
Eysel P, Hopf C, Vogel I (1997) Primary stable anterior instrumentation or dorsoventral spondylodesis in spondylodiscitis? Results of a comparative study. Eur Spine J 6:152–157
Frangen TM, Kalicke T, Gottwald M (2006) Surgical management of spondylodiscitis. An analysis of 78 cases. Unfallchirurg 109:743–753
Gasbarrini AL, Bertoldi E, Mazzetti M (2005) Clinical features, diagnostic and therapeutic approaches to haematogenous vertebral osteomyelitis. Eur Rev Med Pharmacol Sci 9:53–66
Grados F, Lescure FX, Senneville E, Flipo RM, Schmit JL, Fardellone P (2007) Suggestion for managing pyogenic (non-tuberculous) discitis in adults. Joint Bone Spine 74:133–139
Grammatico L, Baron S, Rusch E (2008) Epidemiology of vertebral osteomyelitis (VO) in France: analysis of hospital-discharge data 2002–2003. Epidemiol Infect 136:653–660
Haaker RG, Senkal M, Kielich T et al (1997) Percutaneous lumbar discectomy in the treatment of lumbar discitis. Eur Spine J 6:98–101
Hadjipavlou AG, Mader JT, Necessary JT et al (2000) Hematogenous pyogenic spinal infections and their surgical management. Spine 25:1668–1679
Hee HT, Majd ME, Holt RT et al (2002) Better treatment of vertebral osteomyelitis using posterior stabilization and titanium mesh cages. J Spinal Disord Tech 15:149–156
Homagk L, Deml O, Hofmann GO (2012) Der IT-gestützte Behandlungspfad als Routinearbeitsinstrument in der Unfallchirurgie. Unfallchirurg 115(12):1076–1084
Homagk L, Wiesner I, Hofmann GO et al (2013) Are IT-based clinical pathways superior to hard-copy form? Zentralbl Chir 138:64–69
Keil M, Szczerba L, Kraus G, Abel R (2012) Therapy of septic paraplegia. Orthopade 41(9):742–748
Klöckner C, Valencia R, Weber U (2009) Alignment of the sagittal profile after surgical therapy of nonspecific destructive spondylodiscitis: ventral or ventrodorsal method—a comparison of outcomes. Orthopade 30:965–976
Kraft CN, Fell M (2011) Algorithm for treatment of deep spinal infections and spondylodiscitis with in situ instrumentation. Orthopade 41(1):51–57
Krödel A, Stürz H (1989) Differenzierte operative und konservative therapie der spondylitis und spondylodiszitis. Z Orthop 127:587–596
Krogsgaard MR, Wagn P, Bengtsson J (1998) Epidemiology of acute vertebral osteomyelitis in Denmark: 137 cases in Denmark 1978–1982, compared to cases reported to the National Patient Register 1991–1993. Acta Orthop Scand 69:513–517
Lehner B, Akbar M, Rehnitz C, Omlor GW, Dapunt U, Burckhardt I (2012) Standards der mikrobiologische Diagnostik der Spondylodiszitis. Orthopade 41(9):702–710
Lerner T, Hackenberg L, Rosler S et al (2005) Surgical therapy of unspecific and specific spondylodiscitis. Z Orthop 143:204–212
Liljenqvist U, Lerner T, Bullmann V et al (2003) Titanium cages in the surgical treatment of severe vertebral osteomyelitis. Eur Spine J 12:606–612
Linhardt O, Matussek J, Refior HJ et al (2007) Longterm results of ventro-dorsal versus ventral instrumentation fusion in the treatment of spondylitis. Int Orthop 31:113–119
Maus U, Andereya S, Gravius S, Ohnsorge JA, Miltner O, Niedhart C (2009) Procalcitonin (PCT) as diagnostic tool for the monitoring of spondylodiscitis. Z Orthop Unfall 147(1):59–64
Moon MS, Woo YK, Lee KS et al (1995) Posterior instrumentation and anterior interbody fusion for tuberculous kyphosis of dorsal and lumbar spines. Spine 20:1910–1916
Mörk S, Kothe R, Ulrich C (2011) Infektionen der Wirbelsäule. Orthopädie Unfallchirurgie up2date 6:423–444
Mückley T, Kirschner M, Hierholzer C, Hofmann GO (2003) Spondylitis–Spondylodiszitis: neue Therapiekonzepte. Trauma Berufskrankh 5(2):296–304
Oga M, Arizono T, Takasita M, Sugioka Y (1993) Evaluation of the risk of instrumentation as a foreign body in spinal tuberculosis. Spine 18:1890–1894
Peters KM, Schwanitz B, Zilkens KW (1992) Spondylodiszitis-eine häufig spät gestellte diagnose. Orthop. Praxis 2:108–112
Rajasekaran S, Soundarapandian S (1998) Progression of kyphosis in tuberculosis of the spine treated by anterior arthrodesis. J Bone Joint Surg 71:1314–1323
Schinkel C, Gottwald M, Andress HJ (2003) Surgical treatment of Spondylodiscitis. Surg Infect 4:387–391
Shoakazemi A, Amit A, Nooralam N, Abouharb A, Gormley M, McKinstry S (2013) Panspinal epidural and psoas abscess with secondary cervical disc space infection. Ulster Med J 82(1):23–25
Siewe J, Oppermann J, Eysel P, Zarghooni K, Sobottke R (2013) Diagnosis and treatment of spondylodiscitis in HIV-positive patients. Acta Orthop Belg 79(5):475–482
Sobottke R, Seifert H, Fätkenheuer G, Schmidt M, Gossmann A, Eysel P (2008) Current diagnosis and treatment of spondylodiscitis. Dtsch Arztebl Int 105(10):181–187
Stüer C, Stoffel M, Hecker J, Ringel F, Meyer B (2013) A staged treatment algorithm for spinal infections. J Neurol Surg A Cent Eur Neurosurg 74(2):87–95
Vorbeck F, Morscher M, Ba-Ssalamah A, Imhof H (1996) Infektiöse Spondylitis beim Erwachsenen. Radiologe 36:795–804
Vos FJ, Kullberg BJ, Sturm PD, Krabbe PF, van Dijk AP, Wanten GJ, Oyen WJ, Bleeker-Rovers CP (2012) Metastatic infectious disease and clinical outcome in Staphylococcus aureus and Streptococcus species bacteremia. Medicine 91:86–94
Yilmaz U (2011) Spondylodiscitis. Radiologe 51(9):772–778
Conflict of interest
The authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Homagk, L., Homagk, N., Klauss, J.R. et al. Spondylodiscitis severity code: scoring system for the classification and treatment of non-specific spondylodiscitis. Eur Spine J 25, 1012–1020 (2016). https://doi.org/10.1007/s00586-015-3936-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3936-8