Abstract
Trypanosomosis is a serious constraint to livestock productivity in sub-Saharan Africa. Chemotherapy which is the main means of control is unsatisfactory because of drug resistance and toxicity. Thus, the search for alternative antitrypanosomal agents has become imperative. The study was aimed at evaluating the effects of astaxanthin alone or in combination with diminazene aceturate (DZ) on parasitaemia and haematological parameters in Trypanosoma brucei brucei–infected Wistar rats. Eighty Wistar rats were divided into eight groups of ten rats each and treated as follows: I, DW (uninfected and treated with distilled water); II, S/OIL (uninfected and treated with soya oil); III, T (infected with 106 trypanosomes/mL); IV, PreA [pretreated with astaxanthin (100 mg/kg)] + T; V, PreA + T + DZ (3.5 mg/kg); VI, T + A (astaxanthin); VII, T + A + DZ; VIII, T + DZ. Parasitaemia was monitored daily, and blood was collected for evaluation of haematological parameters at the end of the experiment. Parasitaemia increased progressively in the T, PreA + T and T + A groups till the termination of the experiment, but was lower (P < 0.05) in the PreA group alone, compared to the T and T + A groups at days 6 and 8. There was a resurgence of parasitaemia in 10% of rats in the T + A + DZ group at day 7 post-treatment. The red blood cell counts, PCV and haemoglobin concentration were higher (P < 0.05), but lower neutrophil and total leucocyte counts (P < 0.05) in PreA and/or DZ groups compared to the T and T + A groups. Rats in the T and T + A groups had higher (P < 0.05) erythrocytic indices. Pre-treatment with astaxanthin and/or diminazene aceturate was more effective than post-treatment in decreasing parasitaemia and ameliorating the anaemia caused by T. brucei brucei infection in Wistar rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
African trypanosomosis is one of the major factors responsible for rural underdevelopment in many sub-Saharan African countries (Franco et al. 2018). The disease affects both humans and animals with pronounced negative effects on human settlement and income generating ability of the affected communities (Ojo et al. 2021). The disease has also become a threat to food security in sub-Saharan Africa due to vast arable land that is rendered uncultivable (Gaithuma et al. 2019). Trypanosomosis is characterised by the spasmodic presence of the parasites in the patient bloodstream (which produce several changes in the blood constituents) and recurrent fever (Egbuji et al. 2023; Mohammed et al. 2023). The anaemic condition normally develops in animals infected with the parasite, and this is accompanied by weight loss, decreased productivity and high mortality (Mohammed et al. 2023). African trypanosomes modulate the mammalian host immune responses (Beschin et al. 2014) by manipulating cells of the myeloid phagocytic system, including macrophages, monocytes and granulocytes (neutrophils) (Stijlemans et al. 2015). Oxidative stress has been implicated in the pathogenesis of animal trypanosomosis (Saleh et al. 2009). Oxidative stress results in increased production of reactive oxygen species and depletion of tissue antioxidant reserves (Umar et al. 2007, 2010; Saleh et al. 2009), thus causing damage to plasma membranes, thereby contributing to the cellular degenerative changes observed in trypanosomosis.
To date, the control of animal trypanosomosis relies principally on the use of chemotherapeutic and chemoprophylactic agents (Chekwube et al. 2014; Ukwueze et al. 2022). Currently, the chemotherapy of African trypanosomosis is unsatisfactory because of the development of resistance to the drugs by the parasite, toxicity to the host and cost, especially in developing countries (Ukwueze et al. 2022; Egbuji et al. 2023). Attention is now focused on the search for non-toxic and readily available natural products that possess antioxidant properties for the treatment of trypanosomosis (Ibrahim et al. 2014; Egbuji et al. 2023).
Astaxanthin is a xanthophyll carotenoid, commercially available as a nutritional supplement in many countries, including the USA, Sweden, Japan and South Korea (Choi et al. 2011; Ambati et al. 2014). Its products are available in the form of capsules, soft gels, tablets, powders, biomass, creams, energy drinks, oils and extract (Ambati et al. 2014). Like other carotenoids, astaxanthin plays important functions in regulating immunity and disease aetiology (Park et al. 1998, 2010). Astaxanthin possesses tenfold the antioxidant activity of zeaxanthin, lutein, canthaxanthin and β-carotene and 100 times more comparable to α-tocopherol (Park et al. 2012; Ambati et al. 2014). It is conceivable that the administration of dietary astaxanthin alone or in combination with diminazene aceturate may prevent the relapse of infection that occurs following treatment with diminazene aceturate, decrease parasitaemia and anaemia in trypanosome-infected animals.
The aim of the study was to evaluate the effects of astaxanthin alone or in combination with diminazene aceturate (DZ) on survivability, parasitaemia and haematological parameters in Trypanosoma brucei brucei–infected Wistar rats.
Materials and methods
Experimental animals
Eighty (80) male adult Wistar rats, weighing between 180 and 210 g, were used for the experiment. The animals were obtained from the Animal House of the Nigerian Institute of Trypanosomosis Research (NITR), Vom, Plateau State. They were transported to the animal house of the Department of Veterinary Pharmacology and Toxicology, Ahmadu Bello University, Zaria, Kaduna State, and were allowed to acclimatise to laboratory conditions for 14 days before the commencement of the experiment. The rats were housed in plastic cages with wood shavings as bedding, which was changed weekly. They were fed with a pelleted starter mash (Chikun Feed®) and water ad libitum.
Trypanosome parasites
Trypanosoma brucei brucei (Federe strain) was obtained from the Nigerian Institute for Trypanosomosis Research, Kaduna. The parasite was maintained by serial passages in other donor rats. Parasitaemia was monitored daily by preparing a wet mount and viewed under the light microscope (Olympus® CH23, Germany) at ×400 magnification.
Inoculation of rats
The infected blood from a donor rat (when the parasitaemia was 2.5 × 105) was collected and diluted with physiological saline. Each rat was inoculated with a suspension containing 3 or 4 trypanosomes per microscopic field at ×100 magnification (approximately 106 trypanosomes/mL) as described by Adeyemi et al. (2012).
Experimental design
Eighty (80) experimental rats were randomly allotted into eight groups (I, II, III, IV, V, VI, VII and VIII) of ten animals each and treated as follows:
-
Group I (DW): distilled water (2 mL/kg), per os, only.
-
Group II (S/OIL): soya oil (2 mL/kg), per os, only.
-
Group III (T): intraperitoneally infected with 106 trypanosomes/mL.
-
Group IV (PreA + T): pretreated with astaxanthin (Source Naturals, Santa Cruz, CA, USA) at 100 mg/kg (Choi et al. 2011), per os, for 3 weeks, infected with 106 trypanosomes/mL. Astaxanthin administration continued for the remaining part of the experiment.
-
Group V (PreA + T + DZ): pretreated with astaxanthin, per os, for 3 weeks, infected with 106 trypanosomes/mL and administered with diminazene aceturate (Berenil®, Hoechst, Germany) at 3.5 mg/kg, intraperitoneally. Astaxanthin administration continued for the remaining part of the experiment.
-
Group VI (T + A): infected with 106 trypanosomes/mL, intraperitoneally and treated with astaxanthin for the remaining part of the experiment.
-
Groups VII (T + A + DZ): infected with 106 trypanosomes/mL, treated with diminazene aceturate (3.5 mg/kg), intraperitoneally, followed by oral administration of astaxanthin (100 mg/kg) for the remaining part of the experiment.
-
Group VIII (T + DZ): infected with 106 trypanosomes/mL and administered with diminazene aceturate (3.5 mg/kg), intraperitoneally.
All animals were monitored daily for parasitaemia and death. The groups treated with diminazene aceturate alone or its combination with astaxanthin were monitored for relapse of infection after treatment.
Determination of survivability and parasitaemia
Survivability of the rats was determined by calculating the difference between the day of infection and the day the animal died (Aremu et al. 2018). Parasites in the blood were estimated every other day by examining wet blood smears at ×40 magnification using the rapid “matching” method of Herbert and Lumsden (1976).
Determination of haematological parameters
Blood samples were collected in labelled sample bottles containing ethylenediaminetetraacetic acid (EDTA) as anticoagulant for the determination of haematological parameters such as packed cell volume (PCV), haemoglobin (Hb) concentration, red blood cell (RBC), platelet and absolute and differential leucocyte counts using the automated haematologic analyser (Sysmex, KX-21, Japan) as described by Dacie and Lewis (1991). Erythrocytic indices of mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH) and mean corpuscular haemoglobin concentration (MCHC) were calculated from the value of PCV, Hb concentration and RBC count as described by Schalm et al. (1975).
Results
Effect of treatment on the survivability of rats
There was a significant (P < 0.05) decrease in the survivability of rats in the infected untreated and the group post-treated with astaxanthin alone when compared with all other treatment groups. The survivability of rats in the groups pre- and post-treated with astaxanthin and/or diminazene aceturate was significantly (P < 0.05) lower than those of astaxanthin pretreated alone (Fig. 1).
Effect of astaxanthin and diminazene aceturate on the survivability of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil, T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on the level and resurgence of parasitaemia
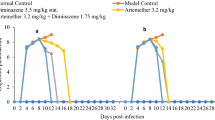
Parasitaemia was first observed on day 2 post-infection; and by day 4 post-infection, all the rats in the infected groups came down with parasitaemia. The level of parasitaemia dropped to zero at day 6 post-infection in all groups treated with diminazene aceturate alone or in combination with astaxanthin (Fig. 2). However, there was a resurgence of parasitaemia in 10% of the rats in the group post-treated with astaxanthin and administered with diminazene aceturate at day 7 post-treatment (Table 1). The level of parasitaemia in the infected untreated and the groups pre- and post-treated with astaxanthin alone progressively increased till the termination of the experiment, but was significantly (P < 0.05) lower in astaxanthin pretreated than in the infected untreated or astaxanthin post-treated groups at both days 6 and 8 post-infection (Fig. 2).
Effect of treatment with astaxanthin and diminazene aceturate on the level of parasitaemia of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means on the same day with different superscript letters (a, b) are significantly different (P < 0.05). Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on total red blood cell count
The RBC count of rats in the group pretreated with astaxanthin alone or in combination with diminazene aceturate was significantly (P < 0.05) higher when compared to other treatment groups. There was a significant (P < 0.05) increase in the RBC count of rats in the group post-treated with astaxanthin and administered with diminazene aceturate in comparison to infected untreated, astaxanthin post-treated and the group administered with diminazene aceturate alone (Fig. 3).
Effect of treatments with astaxanthin and diminazene aceturate on erythrocyte count of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on packed cell volume
There was a significant (P < 0.05) increase in the PCV of rats in the group pretreated with astaxanthin and administered with diminazene aceturate in comparison to all other treatment groups (Fig. 4). The astaxanthin pretreated, astaxanthin post-treated and administered with diminazene aceturate and the group administered with diminazene aceturate alone had significantly (P < 0.05) higher PCV when compared to the values obtained in infected untreated and astaxanthin post-treated groups.
Effect of treatments with astaxanthin and diminazene aceturate on packed cell volume of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on haemoglobin concentration
Figure 5 shows the effect of treatment on Hb concentration. The Hb concentration of rats in the group pretreated with astaxanthin and administered with diminazene aceturate was significantly (P < 0.05) higher when compared to other treatment groups. The astaxanthin pretreated and the group post-treated with astaxanthin and administered with diminazene aceturate had higher (P < 0.05) Hb concentration in comparison to the concentration obtained in infected untreated, astaxanthin post-treated and the group administered with diminazene aceturate alone.
Effect of treatments with astaxanthin and diminazene aceturate on haemoglobin concentration of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on erythrocytic indices
There were significant (P < 0.05) increases in MCV, MCH and MCHC of rats in infected untreated and astaxanthin post-treated groups when compared to other treatment groups. The MCV and MCH of rats in the group pretreated with astaxanthin alone or in combination with diminazene aceturate were significantly (P < 0.05) lower compared to those of astaxanthin post-treated and/or diminazene aceturate groups. A significant (P < 0.05) decrease was recorded in the MCV of rats pretreated with astaxanthin and administered with diminazene aceturate when compared with pretreated with astaxanthin alone (Fig. 6).
Effect of treatments with astaxanthin and diminazene aceturate on erythrocytic indices of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d, e) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; MCV, mean corpuscular volume; MCH, mean corpuscular haemoglobin; MCHC, mean corpuscular haemoglobin concentration
Effect of treatments on platelet count
The platelet count of rats in the infected untreated group was significantly (P < 0.05) lower than other treatment groups (Fig. 7). There was a significant (P < 0.05) decrease in platelet count of rats in the group pretreated with astaxanthin alone when compared to those pretreated and administered with diminazene aceturate. The group pretreated with astaxanthin and administered with diminazene aceturate had higher (P < 0.05) platelet count, in comparison to those post-treated with astaxanthin and/or diminazene groups.
Effect of treatments with astaxanthin and diminazene aceturate on platelet count of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d, e, f) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments on total and differential leucocyte counts
There was no difference in lymphocyte counts of rats in all the treatment groups.
The WBC and neutrophil counts in the infected untreated and astaxanthin post treated groups were higher (P < 0.05) than the counts recorded in other groups (Figs. 8 and 9). The group pretreated with astaxanthin and administered with diminazene aceturate had significantly (P < 0.05) lower WBC and neutrophil counts when compared to astaxanthin pretreated, astaxanthin post-treated and/or diminazene aceturate groups.
Effect of treatments with astaxanthin and diminazene aceturate on absolute leucocyte count of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Effect of treatments with astaxanthin and diminazene aceturate on lymphocyte and neutrophil counts of Wistar rats experimentally infected with Trypanosoma brucei brucei. Means with different superscript letters (a, b, c, d) are significantly (P < 0.05) different. Values are mean ± SEM of N = 5. Keys: DW, distilled water; S/OIL, soya oil; T, Trypanosoma brucei brucei; PreA + T, pretreated with astaxanthin and infected with Trypanosoma brucei brucei; PreA + T + DZ, pretreated with astaxanthin, infected with Trypanosoma brucei brucei and treated with diminazene aceturate; T + A, infected with Trypanosoma brucei brucei and treated with astaxanthin; T + A + DZ, infected with Trypanosoma brucei brucei, treated with astaxanthin and diminazene aceturate; T + DZ, infected with Trypanosoma brucei brucei and treated with diminazene aceturate
Discussion
In this study, pre-treatment with astaxanthin and in combination with diminazene aceturate led to higher survivability of the rats compared to other treatment groups. This observation agrees with the results of Eze et al. (2008), who reported a higher survival rate in rats treated with a diminazene/selenium combination than those groups that had selenium and vitamin E supplements alone. The pathogenesis of trypanosomosis is associated with severe inflammation and production of radicals such as nitric oxide, which affect adversely the survival of the host (Mbuthia et al. 2011). The prolonged survivability of the rats pretreated with astaxanthin and/or diminazene aceturate may be due to the antioxidant activity of the astaxanthin, which ameliorated the inflammatory reactions and the damages caused by the generation of ROS during the course of the infection. The survivability of rats in the astaxanthin post-treated group was not different from that of the infected untreated. This may be attributed to an increase in the number of circulating trypanosomes and their by-products, resulting in enormous generation of ROS, thereby damaging cell membranes and consequently death (Yakubu et al. 2014).
Parasites were observed in the blood of infected rats as early as day 2 post-infection. By day 4 post-infection, all the infected groups developed parasitaemia. The pre-patent period of 2–4 days observed in this study disagrees with the findings of Chekwube et al. (2014) in rats and Kobo et al. (2015) in mice, who reported a pre-patent period of 5 and 4 days, respectively. The pre-patent period of 2–4 days observed in this study may be related to the level of virulence and the number of parasites inoculated. The number of parasites inoculated has been reported to influence the pre-patent period (Egbe-Nwiyi et al. 2017). Pre-treatment with astaxanthin did not affect the onset of parasitaemia and was unable to clear the parasites from blood, but led to a decrease in the level of parasitaemia. This finding agrees with the earlier reports of Kobo et al. (2015) in rats infected with Trypanosoma brucei brucei and treated with Daflon® 500 mg (an antioxidant) and Eze et al. (2015) in Trypanosoma brucei–infected rats supplemented with dietary zinc that exhibits antioxidant activity. Astaxanthin has been reported to influence the host immune system and disease aetiology (Park et al. 1998, 2010; Kuan-Hung et al. 2016), thus altering the susceptibility of the host to infectious diseases. All the groups treated with diminazene aceturate alone or in combination with astaxanthin became aparasitaemic 48 h post-treatment; an exception was the group post-treated with astaxanthin and diminazene aceturate, where there was a resurgence of parasitaemia in 10% (that is one out of three) of the rats at day 7 post-treatment and the rat died a day after the resurgence of parasitaemia was observed. This corroborates the report of Ezeh et al. (2009, 2016), who observed relapse infection after treatment with diminazene aceturate. Reports of relapse of T. b. brucei infection have been attributed to the presence of T. b. brucei in drug-inaccessible sites such as the brain, and this often occurs when there is a prolonged period between infection and treatment (Eke et al. 2017) such that the parasite migrates to brain, and after treatment recedes from the brain to the bloodstream (Eze et al. 2019). However, in the present study, the treatment was started on day 4 post-infection, when parasites were 3 or 4 per microscopic field, suggesting that the parasites may not have invaded the brain as treatment was started immediately. Thus, the reemergence of parasitaemia may not be as a result of the parasites reappearing into the bloodstream from the brain but may be attributed to other factors such as mutation, amplification or deletion, altered drug uptake, drug metabolism, drug target interaction or efflux (Eze et al. 2019). The prevalence of fake and adulterated drugs may also lead to the emergence of drug-resistant trypanosomes (Ezeokonkwo et al. 2007).
Acute haemolysis has been demonstrated as a cardinal feature in African trypanosomosis (Kobo et al. 2015; Ojo et al. 2021). The significant decrease in the levels of RBC, PCV and Hb concentration observed in the infected untreated rats agrees with the findings of Karaye et al. (2017) and Oparah et al. (2017), which may be attributed to the trypanosome-induced disruption of RBC membrane (Akanji et al. 2009). This could have resulted in subsequent haemolysis as reflected in the low RBC count. The decrease in PCV may also be attributed to reduced glutathione concentration on the membrane surface of the RBC induced by the parasites, thus rendering the membrane liable to oxidative lysis (Akanji et al. 2009; Jolayemi et al. 2020; Ojo et al. 2021). Oxidative cell damage is a prominent feature in T. b. brucei infections (Omer et al. 2007; Saleh et al. 2009; Kobo et al. 2015). The RBC, PCV and Hb concentration of astaxanthin-pretreated group alone and astaxanthin pretreated and administered with diminazene aceturate remained significantly higher than the infected untreated group. This result is consistent with the report of Kobo et al. (2015) who observed that the pre-treatment of rats with a flavonoid mixture (Daflon® 500 mg) kept the RBC, PCV and Hb concentration higher than in the infected untreated rats. This finding could be attributed to the beneficial effect of astaxanthin as an antioxidant and immunostimulant that resulted in the protection of the integrity of RBCs, thereby preventing the cells from oxidative haemolysis. The group pretreated with astaxanthin and administered with diminazene aceturate had the highest RBC, PCV and Hb concentration, when compared to other treatment groups. This result may be attributed to the antioxidant effect of astaxanthin and the trypanocidal effect of diminazene aceturate that eliminated the parasites from peripheral circulation, thereby protecting the RBCs from destruction by the lashing action of the trypanosome flagella (Oparah et al. 2017). The RBC, PCV and Hb values in the astaxanthin post-treated group alone were not different from that of the infected untreated group, indicating that astaxanthin was better as a prophylactic than curative agent.
Increased MCV (macrocytosis) in the infected untreated group corroborates the findings of Abenga et al. (2005) in Trypanosoma congolense infection of Nigerian puppies but contrasts with the report of Kobo et al. (2015) in rats infected with T. brucei brucei. An increase in MCV and MCH is typically seen in cases of haemolytic anaemias. Macrocytosis in African trypanosomosis usually arises from increased erythropoiesis in the bone marrow, with the resultant release of macrocytic immature red cells into the circulation, known to occur during the acute phase of the anaemia (Abenga et al. 2005; Oparah et al. 2017). The increase in the value of MCH recorded in the infected untreated group was probably due to free circulating haemoglobin arising from the haemolysis (Igbokwe and Anosa 1989). The MCHC is a more reliable predictor in the classification of anaemia. The increase in MCHC suggests a qualitative bone marrow output during the acute stage of infection. The significant decreases in the MCV and MCH recorded in the group pretreated with astaxanthin alone and its combination with diminazene aceturate suggest that astaxanthin reduced the anaemic effect of T. brucei brucei in the infected rats.
The decrease observed in platelet count in the infected untreated group corroborates the findings of Ezebuiro et al. (2012) in rabbits, Kobo et al. (2015) in rats and Oparah et al. (2017) in donkeys. Thrombocytopaenia could be the result of damage to platelets due to haemorrhage, vasoconstriction or tissue damage observed in trypanosomosis (Davis 1982). Low platelet count may also be due to increased splenic sequestration of platelets or an increased destruction of the platelets due to disseminated intravascular coagulation reaction (Kobo et al. 2015; Oparah et al. 2017). In addition, the thrombocytopaenia recorded in the infected untreated group may also be due to oxidative damage to the platelet membranes, provoked by the trypanosome parasite. This may result in the formation of reactive oxygen species within the platelet membranes, thereby causing cellular lysis (Kobo et al. 2015). Pre-treatment with astaxanthin and diminazene aceturate significantly increased the platelet count better than other treatment groups. This result may be attributed to the immune-stimulatory and antioxidant effects of astaxanthin and the trypanocidal effect of diminazene aceturate.
The significant increase in total leucocyte counts observed in this study agrees with the reports of Ukpai and Nwabuko (2014) and Abenga et al. (2017) in T. b. brucei–infected rats and pigs, respectively, but disagrees with the findings of Kobo et al. (2015) in T. b. brucei–infected rats and El-Ashmawy et al. (2016) in T. evansi infected rats. The increase in WBC count in the infected untreated rats was an indication of infection and may be attributed to the ability of the body to employ its immune arsenals to combat the invading parasites and in the process of immune response to enhance more WBC production (Ukpai and Nwabuko 2014; Ukwueze et al. 2022). Leucocytosis which may be due to an increase in lymphocyte, neutrophil, monocytes or eosinophil counts has been implicated in trypanosomosis (Ezebuiro et al. 2012). It is believed to be part of an immunological response influenced by the ever-changing variable surface glycoprotein of the infecting trypanosomes (Abenga et al. 2017; Oparah et al. 2017). Lymphocytosis could also result from the host’s inflammatory response due to the presence of the infecting organisms (Satue et al. 2014). This report supports the findings in this study where there was an increase in lymphocyte and neutrophil counts in the infected groups, when compared to the uninfected control group. Treatment of infected rats with astaxanthin and/or diminazene aceturate caused a decrease in WBC, lymphocyte and neutrophil count, when compared to the infected untreated group. This may be due to the finding that astaxanthin, being a potent antioxidant with anti-inflammatory activity (Liu and Lee 2003; Ambati et al. 2010), was able to counter the host’s inflammatory response induced by the parasite, thereby maintaining the WBC, lymphocyte and neutrophil counts at lower levels. Astaxanthin pretreated and/or diminazene aceturate groups had lower WBC, lymphocyte and neutrophil counts when compared to astaxanthin post-treated group alone, demonstrating that pre-treatment with astaxanthin was better at stimulating the immune system of the host than post-treatment.
Conclusion
This study has demonstrated that pre-treatment with astaxanthin alone or in combination with diminazene aceturate increased the survivability of rats, decreased parasitaemia, prevented the resurgence of parasitaemia and ameliorated trypanosome-induced alterations in haematological parameters of T. brucei brucei–infected rats.
Data availability
The data for this research will be made available upon request.
References
Abenga JN, Adamu NM, Nok AJ, Ibrahim NDG, Sackey AKB, Esievo KAN (2017) Early stage leucocytosis in Nigerian pigs experimentally infected with Trypanosoma brucei. Int J Biol Chem Sci 11(1):67–78. https://doi.org/10.4314/ijbcs.v11i1.6
Abenga JN, Ezebuiro CO, David K, Fajinmi AO, Samdi S (2005) Studies on anaemia in Nigerian local puppies infected with Trypanosoma congolense. Vet Arh 75(2):165–174
Adeyemi OS, Akanji MA, Ekanem JT (2012) Ethanolic extract of Psidium guajava influences protein and bilirubin levels in T. brucei brucei-infected rats. J Biol Sci 12(2):111–110. https://doi.org/10.3923/jbs.2012.111.116
Akanji MA, Adeyemi OS, Oguntoye SO, Sulyman F (2009) Psidium guajava extract reduces trypanosomosis associated lipid peroxidation and raises glutathione concentrations in infected animals. EXCLI J 8:148–154
Ambati R, Moi Phang S, Ravi S, Aswathanarayana RG (2014) Astaxanthin:sources, extraction, stability, biological activities and its commercial applications - a review. Mar Drugs 12:128–152. https://doi.org/10.3390/md12010128
Ambati R, Raghunath Reddy RL, Baskaran V, Sarada R, Ravishankar GA (2010) Characterization of microalgal carotenoids by mass spectrometry and their bioavailability and antioxidant properties elucidated in rat model. J Agric Food Chem 58:8553–8559. https://doi.org/10.1021/jf101187k
Aremu A, Kingsley EI, Talha BK, Akeem AO, Ibrahim RA, Jimoh AG, Yusuf SK (2018) Methanolic leaf extract of Moringa oleifera improves the survivability rate, weight gain and histopathological changes of Wistar rats infected with T. brucei. Int J Vet Sci Med 6:39–44. https://doi.org/10.1016/j.ijvsm.2018.04.006
Beschin A, Van Den Abbeele J, De Baetselier P, Pays E (2014) African trypanosome control in the insect, vector, and mammalian host. Trends Parasitol 30(11):538–547. https://doi.org/10.1016/j.pt.2014.08.006
Chekwube AI, Onyema EI, Ikenna UE, Ezeokonkwo RC (2014) Effect of diminazene aceturate, levamisole and vitamin C combination therapy in rats experimentally infected with Trypanosoma brucei brucei. Asian Pac J Trop Med 7(6):438–445. https://doi.org/10.1016/S1995-7645(14)60071-7
Choi HD, Kang HE, Yang SH, Lee MG, Shin WG (2011) Pharmacokinetics and first-pass metabolism of astaxanthin in rats. Br J Nutr 105:220–227
Dacie JV, Lewis SM (1991) Practical haematology, 7th edn. Churchill Livingston, London, pp 659–661
Davis CE (1982) Thrombocytopenia: a uniform complication of African trypanosomosis. Acta Trop 39(2):123–133
Egbe-Nwiyi TN, Igwenagu E, Nwaosu AT, Maina MM (2017) Comparison of pathogenicity of relapsed, field and mixed isolates of Trypanosoma brucei brucei infections in rats. J Adv Vet Ani Res 4(1):97–103. https://doi.org/10.5455/javar.2017.d197
Egbuji JV, Ejere VC, Ugwu GC, Ugwu CL (2023) Effects of aqueous leaf extracts of Loranthus micranthus Linn on biochemical profile of Wistar rats infected with Trypanosoma brucei brucei. Trop J Nat Prod Res 7(6):3209–3214. https://doi.org/10.26538/tjnpr/v7i6.22
Eke IG, Ezeh IO, Ezeudu TA, Eze UU, Anega AO, Onyeyili PA (2017) Chemotherapeutic efficacy of secnidazole-diminazene aceturate combination therapy in experimental T. b. brucei-infection in rats. Afr J Pharm Pharmacol 11:349–353. https://doi.org/10.5897/AJPP2017.4779
El-Ashmawy I, Al-Wabel NA, Bayed AE (2016) Achillea fragrantissima rich in flavonoids and tannins potentiates the activity of diminazene aceturate against T. evansi in rats. Asian Pac J Trop Med 9(3):228–234. https://doi.org/10.1016/j.apjtm.2016.01.032
Eze JI, Anene BM, Chukwu CC (2008) Determination of serum and organ malondialdehyde concentration, a lipid peroxidation index in Trypanosoma brucei infected rats. Comp Clin Path 17(2):67–72. https://doi.org/10.1007/s00580-008-0722-6
Eze JI, Ayogu LC, Abonyi FO, Eze UU (2015) The beneficial effect of dietary zinc supplementation on anaemia and immunosuppression in Trypanosoma brucei-infected rats. Exp Parasitol 154:87–92. https://doi.org/10.1016/j.exppara.2015.04.015
Eze UU, Eke IG, Ezeh IO, Nzeakor TA, Owube C, Dede A, Anene BM (2019) Comparative evaluation of the chemotherapeutic efficacies of two salts of diminazene aceturate in T. b. brucei infected dogs. Trop J Pharm Res 18(9):1961–1967. https://doi.org/10.4314/tjpr.v18i9.26
Ezebuiro OGC, Yohanna JA, Abdulya A, Osue HO, Abenga JN, Yakasai MA, Attahir A (2012) Haematological changes following acute Trypanosoma brucei brucei infection in rabbits. Curr Res J Biol Sci 4(4):414–416
Ezeh IO, Agbo LI, Emehelu CO, Nweze EN, Ezeokonkwo RC, Onah DN (2009) Berenil-resistant Trypanosoma brucei brucei infection in a hunting dog in Nsukka are, Enugu state, Nigeria. Niger Vet J 29:34–42
Ezeh IO, Ugwu EN, Enemuo OV, Obi CF, Iheagwam CN, Ezeokonkwo RC, Onah DN (2016) Efficacy of repeated doses of diminazene aceturate in the treatment of experimental Trypanosoma brucei infection in albino rats. Iran J Vet Res 17(2):124–129
Ezeokonkwo RC, Okoro FC, Ezeh IO (2007) The efficacy of increasing doses of Samorenil® in the treatment of Trypanosoma brucei infected albino rats. Niger Vet J 28:24–32
Franco JR, Cecchi G, Priotto G, Paone M, Diarra A, Grout L, Simarro PP, Zhao W, Argaw D (2018) Monitoring the elimination of human African trypanosomosis: update to 2016. PLOS Neglect Trop Dis 12(12):e0006890. https://doi.org/10.1371/journal.pntd.0006890
Gaithuma AK, Yamagishi J, Martinelli A, Hayashida K, Kawai N, Marsela M, Sugimoto C (2019) A single test approach for accurate and sensitive detection and taxonomic characterization of trypanosomes by comprehensive analysis of internal transcribed spacer 1 amplicons. PLOS Neglect Trop Dis 13(2):e0006842. https://doi.org/10.1371/journal.pntd.0006841
Herbert WJ, Lumsden WHR (1976) Trypanosoma brucei: a rapid “matching” method for estimating the host’s parasitaemia. Exp Parasitol 40:427–431
Ibrahim MA, Mohammed A, Isah MB, Aliyu AB (2014) Anti-trypanosomal activity of African medicinal plants: a review update. J Ethnopharmacol 153(1):26–54. https://doi.org/10.1016/j.jep.2014.04.012
Igbokwe IO, Anosa VO (1989) Leucopenia in T. vivax infection of sheep. Rev Elev Med Vet Pays Trop 42:219–221. https://doi.org/10.1982/remvt.8837
Jolayemi KO, Mamman M, Sani D, Okoronkwo MO, Amaje J (2020) In vitro and in vivo changes observed in Trypanosoma brucei brucei-infected rats treated with artesunate and/or diminazene aceturate. Sokoto J Vet Sci 18(4):211–220
Karaye GP, Tekdek LB, Dogo GI (2017) The relapse of single and mixed infections of Trypanosoma congolense and Trypanosoma brucei in Red Sokoto bucks after treatment with isometamidium chloride. Saudi J Biomed Res 2(4):79–85. https://doi.org/10.21276/sjbr
Kobo PI, Ayo JO, Aluwong A, Zezi AU, Maikai VA (2015) Haematological changes in Trypanosoma brucei brucei-infected Wistar rats treated with a flavonoid mixtureand/or diminazene aceturate. Biol Med 6:213. https://doi.org/10.4172/0974-8369.1000213
Kuan-Hung L, Kao-Chang L, Wan-Jung L, Philip-Aloysius T, Thanasekaran J, Joen-Rong S (2016) Astaxanthin, a carotenoid, stimulates immune responses by enhancing IFN- and IL-2 secretion in primary cultured lymphocytes in vitro and ex vivo. Int J Mol Sci 17:44. https://doi.org/10.3390/ijms/17010044
Liu BH, Lee YK (2003) Effect of total secondary carotenoids extracts from Chlorococcum sp. on Helicobacter pylori-infected BALB/c mice. Int Immunopharmacol 3:979–986. https://doi.org/10.1016/S1567-5769(03)00069-1
Mbuthia SK, Wachira NW, Ngure RM, Ouma J, Kagira JM (2011) Effects of tea on survival rates and liver pathology of Trypanosoma brucei brucei infected mice. J Protozool Res 21:1–7
Mohammed YA, Kabiru AY, Hamzah RU, Lukman HY, Aliu TB, Alqarni M, Batiha GE, Waard M, Lawal B (2023) Rutin-rich flavonoid subfraction of Annona senegalensis mitigates Trypanosoma brucei brucei infection and haematobiochemical changes in infected mice. Biomed Res Int 2023:6820338. https://doi.org/10.1155/2023/6820338
Ojo RJ, Enoch GA, Adeh FS, Fompun LN, Bitrus BY, Kugama MA (2021) Comprehensive analysis of oral administration of vitamin E on the early stage of Trypanosoma brucei brucei infection. J Parasit Dis 45(2):512–523. https://doi.org/10.1007/s12639-020-01322-5
Omer OH, Mousa HM, Al-Wabel N (2007) Study on antioxidant status of rats experimentally infected with Trypanosoma evansi. Vet Parasitol 145:142–145. https://doi.org/10.1016/j.vetpar.2006.11.007
Oparah QN, Sackey ABK, Lawal IA, Abdullahi US (2017) Haematological indices in Trypanosoma brucei brucei (Federe isolate) infected Nigerian donkeys (Equus asinus) treated with homidium and isometadium chloride. Maced Vet Rev 40(1):73–82. https://doi.org/10.1515/macvetrev-2017-0014
Park JS, Chew BP, Wong TS (1998) Dietary lutein from marigold extract inhibits mammary tumor development in BALB/c mice. J Nutr 128:1650–1656. https://doi.org/10.1093/jn/128.10.1650
Park JS, Chyun JH, Kim YK, Line LL, Chew BP (2010) Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr Metab 7:18–28
Park JS, Mathison BD, Hayek MG, Zhang J, Reinhart GA, Chew BP (2012) Astaxanthin modulates age-associated mitochondrial dysfunction in healthy dogs. J Ani Sci 91:268–275. https://doi.org/10.2527/jas.2012-5341
Saleh MA, Al-Salahy MB, Sanousi SA (2009) Oxidative stress in the blood of camels (Camelus dromedarius) naturally infected with Trypanosoma evansi. Vet Parasitol 162:192–199. https://doi.org/10.1016/j.vetpar.2009.03.035
Satue K, Muñoz A, Gardón JC (2014) Interpretation of the equine leukogram. J Hem Res 1(1):27–35
Schalm OW, Jain NC, Carroll EJ (1975) Veterinary haematology, 3rd edn, Philadelphia. Lea and Febiger, Pa, pp 15–18
Stijlemans B, Beschin A, Magez S, Ginderachter JAV, De Baetseliar P (2015) Iron homeostasis and Trypanosoma brucei associated immunopathogenicity development: a battle/quest for iron. Biomed Res Int 2015:819389. https://doi.org/10.1155/2015/819389
Ukpai OM, Nwabuko OP (2014) Effects on haematological parameters and pathology of internal organs of Trypanosoma brucei brucei-infected albino rats. Niger J Biotechnol 27:8–13
Ukwueze CS, Akpa PO, Odo RI, Ezema C, Anene BM, Idika K (2022) The effect of resveratrol supplementation on haematological parameters and trypanocidal effect of diminazene aceturate in dogs. Sokoto J Vet Sci 29(4):240–247
Umar IA, Ogenyi E, Okodaso D, Kimeng E, Stancheva GI, Omage JJ, Isah S, Ibrahim MA (2007) Amelioration of anaemia and organ damage by combined intraperitoneal administration of vitamins A and C to Typanosoma brucei brucei-infected rats. Afr J Biotechnol 6(18):2083–2086
Umar IA, Toma I, Akombum CA, Nnaji CJ, Mahdi MA, Gidado A, Igbokwe IO, Buratai LB (2010) The role of intraperitoneally administered vitamin during Trypanosoma congolense infection of rabbits. Afr J Biotechnol 9(32):5224–5228
Yakubu DP, Dawet A, Olaleye NA (2014) Effects of vitamin C and selenium on some blood parameters of Trypanosoma brucei brucei infected rats. Br J Appl Sci Technol 4(7):1100–1108
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not supported by any funding.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethical approval for the study was sought from the Ahmadu Bello University Committee on Animal Use and Care (ABUCAUC), Ahmadu Bello University, Zaria, and was obtained with approval number ABUCAUC/2021/029.
Informed consent
For this type of study, informed consent is not required.
Consent for publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kobo, P.I., Suleiman, M.M., Ayo, J.O. et al. Effects of astaxanthin alone or its combination with diminazene aceturate on the survivability and haematological parameters of Trypanosoma brucei brucei–infected Wistar rats. Comp Clin Pathol (2024). https://doi.org/10.1007/s00580-024-03590-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00580-024-03590-6