Abstract
Due to the toxic effects from using conventional trypanocidal drugs, there is need for development of safer alternatives to combat trypanosomosis. The aim of this study was to evaluate the effects of n-hexane and ethylacetate fractions of Terminalia catappa leaf extract in Wistar rats experimentally infected with Trypanosoma brucei brucei. Forty-five (45) adult Wistar rats were divided into 9 groups (A–I) comprising of 5 rats each after acclimatisation for 7 days. Groups A-H were infected intraperitoneally with T. brucei brucei (day 0), treatment given to groups A–G at day 3 post-infection while group I were uninfected. Groups A, B and C are treated daily with 100, 200 and 300 mg/kg n-hexane fraction (HF); D, E and F treated daily with 100, 200 and 300 mg/kg ethylacetate fraction (EAF); and G treated with diminazene aceturate once. Blood was collected for parasitaemia determination and haematology. Results revealed parasitaemia detection by day 2 pi and complete clearance by day 10 pi in groups F and G, and 100% mortality in groups A and G. There was significant decrease in packed cells volume, haemoglobin concentration, red and total white blood cells in all infected groups by day 3 pi. Following treatment at day 10 pi, these haematological parameters significantly increase towards pre-infection values and were significantly higher in groups F and G compared to the other treatment groups. The ethylacetate fraction of Terminalia catappa leaf extract at 300 mg/kg exhibited in vivo trypanocidal activity similar to diminazene aceturate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Blood parasites constitute severe problem to livestock production and these parasites include the causative agents of babesiosis and trypanosomosis (Hashem et al. 2018). Trypanosomosis is an endemic disease affecting both humans and animals (Swallow, 2000; Ezeani et al. 2008; Mbaya et al. 2012). In Africa, the disease is caused by Trypanosoma brucei gambiense and T. brucei rhodesiense referred to as human African trypanosomosis (HAT) or sleeping sickness transmitted by tsetse flies (Mbaya et al. 2012; Radwanska et al. 2018). American trypanosomosis also referred to as Chagas’ disease is caused by Trypanosoma cruzi and transmitted by reduviid bugs (Echeverria and Morillo 2019). The Trypanosoma species that commonly affect animals include T. brucei brucei, Trypanosoma congolense, Trypanosoma vivax, Trypanosoma evansi and Trypanosoma equiperdum (Radwanska et al. 2018; Büscher et al. 2019).
Trypanosomosis has continued to adversely affect economic and social well-being of sub-Saharan Africans causing great losses in livestock production (Muhanguzi et al. 2017). The disease is characterised by intermittent fever, anaemia, occasional diarrhoea, rapid loss of condition and death (Dinsa et al. 2013; Radwanska et al. 2018). Survivals usually remain infected for several months or years serving as reservoirs with low level of fluctuating parasitaemia (Mehlitz and Molyneux 2019). Despite efforts channelled towards chemotherapy and control of tsetse flies, the disease still pose danger threatening food security (Muhanguzi et al. 2017).
The treatment of trypanosomosis is faced with challenges including late stage of the disease, nervous system involvement and drug toxicity (Melaku and Birasa 2013; Giordani et al. 2016). Berenil (diminazene aceturate), being the most widely used drug for the treatment of animal trypanosomiasis, has been reported to be highly toxic, presented with brain damage, quivering and restlessness (Kellner et al. 1985; Melaku and Birasa 2013).
Plants have been reported to possess therapeutic effects as demonstrated in several studies. These include ameliorate effect of Echinacea purpurea on changes induced by Escherichia coli in broiler chickens (Abdallah et al. 2020a), protective effects of Thymus vulgaris and Curcuma longa on thermally oxidised oil-induced alterations in rabbits (Abdallah et al. 2020b, 2020c), potential benefits of Chlorella vulgaris, β-glucan, sodium alginate and Morus alba in Oreochromis niloticus and Clarias gariepinus (Mahmoud et al. 2020; Neamat-Allah et al. 2020a, 2020b, 2020c, 2020d).
The use of plants in the effective treatment of parasitic diseases such as trypanosomosis has been adopted by local herdsmen. Terminalia catappa has been reported to possess medicinal properties against bacterial, fungal and parasitic diseases (Inabo and Fathuddin 2011; Terças et al. 2017). So, in order to avoid the damages produced by conventional trypanocidal drugs, there is need to seek for safer alternatives in medicinal plants such as Terminalia catappa. Hence, the aim of this study was to evaluate the effects of n-hexane and ethylacetate fractions of Terminalia catappa leaf extract in Wistar rats experimentally infected with Trypanosoma brucei brucei.
Materials and methods
Ethics statement
This study involved the use of animals in accordance with internationally accepted guidelines. Ethical approval was granted by the Ahmadu Bello University Committee on Animal Use and Care (ABUCAUC).
Plant collection, extract preparation and fractionation
The leaves of Terminalia catappa used in this study were collected within Zaria, identified and deposited with 1556 as voucher number. Extract of the leaves was prepared by drying at room temperature for 3 weeks, grinding, soaking in ethanol and washing with same solvent. After soaking and washing, solvent was evaporated using rotary evaporator. Fractions of the extract were prepared using n-hexane and ethylacetate as solvents. Lethal dose-50 (LD50) of the fractions was determined using the method described by Lorke (1983) to be over 1000 mg/kg.
Trypanosome parasite
The Trypanosoma brucei brucei used in this study was obtained from the Nigerian Institute for Trypanosomiasis Research (NITR), Kaduna. The parasite was continually passaged in mice to be maintained in the laboratory. Animals were infected by intraperitoneal administration of T. brucei brucei-infected blood containing 106 trypanosomes/ml, parasitaemia-monitored and treatment-administered at day 3 post-infection (pi).
Experimental design
Forty-five (45) adult Wistar rats were acclimatised for 7 days and divided into 9 groups (A–I) comprising 5 rats each. Rats in groups A, B and C were treated daily with 100 mg/kg, 200 mg/kg and 300 mg/kg n-hexane fraction (HF), respectively; D, E and F were treated daily with 100 mg/kg, 200 mg/kg and 300 mg/kg ethylacetate fraction (EAF), respectively; G treated once with diminazene aceturate; H were infected and untreated, while rats in group I were uninfected.
Blood collection
Blood was collected at pre-infection (day 0) and post-infection (days 2, 3, 5, 7, 9 and 10) from all rats into EDTA-coated sample tubes and processed for analysis (Neamat-Allah 2015; Neamat-Allah and El-Damaty 2016; Neamat-Allah and Mahmoud 2019). The haematological parameters determined were packed cells volume, haemoglobin concentration, red and total white blood cells counts as described by Schalm et al. (1975) and Dacie and Lewis (1991).
Data analysis
Values obtained were expressed as mean ± SEM and presented using charts. Statistical analysis of date was achieved by two-way analysis of variance (ANOVA) with Bonferroni multiple comparison test, using Graph Pad Prism version 5.0 for windows (Graph Pad Software, San Diego, CA, USA). Values of P ≤ 0.05 were considered significant.
Results
Parasitaemia
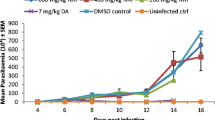
Parasitaemia became patent from day 2 post-infection (pi) in all infected groups (Fig. 1), and 100% mortality recorded at day 7 pi (infected untreated) and day 9 pi (100 mg/kg HF). Following treatment, there was complete parasitaemia clearance from day 7 pi (diminazene aceturate) and day 9 pi (300 mg/kg EAF) but with persistence of parasitaemia up to day 10 pi in other treatment groups (Fig. 1).
Packed cells volume
No significant (P > 0.05) difference existed for packed cells volume (PCV) in all groups of rats pre-infection (Fig. 2). There was significant (P < 0.05) decrease in PCV in all infected groups at day 3 pi. Following treatment, PCV was significantly (P < 0.05) increased in groups VI (51.33 ± 0.88%) and VII (50.00 ± 1.50%), and showed no significant (P > 0.05) differences with those of group IX (51.50 ± 1.50%), but with significant (P < 0.05) differences compared to other treatment groups at day 10 pi (Fig. 2).
Haemoglobin concentration
Haemoglobin concentration (HGB) showed no significant (P > 0.05) difference in all groups of rats pre-infection but decreased significantly (P < 0.05) in all infected groups at day 3 pi (Fig. 3). At day 10 pi following treatment, HGB was significantly (P < 0.05) increased in groups VI (15.00 ± 0.06 g/dl), VII (14.85 ± 0.05 g/dl) and IX (15.20 ± 0.20 g/dl), with no significant (P > 0.05) differences between them, but significantly (P < 0.05) higher compared to the other treatment groups (Fig. 3).
Red blood cells count
There was no significant (P > 0.05) difference in red blood cells (RBC) count in all groups of rats at day 0 but this decreased (P < 0.05) significantly in all infected groups up to day 3 pi (Fig. 4). Following treatment, RBC count increased significantly (P < 0.05) up to day 10 pi but significantly (P < 0.05) higher in groups VI (7.87 ± 0.19 × 106/μl) and VII (8.25 ± 0.15 × 106/μl) compared to the other treatment groups (Fig. 4).
Total white blood cells
Total white blood cells (TWBC) showed no significant (P > 0.05) difference pre-infection in all groups of rat, but decreased (P < 0.05) significantly in infected groups from day 2 pi till day 5 pi. This was followed by significant (P < 0.05) increase up to day 10 pi with no significant (P > 0.05) difference in groups VI (14.63 ± 0.07 × 103/μl) and VII (15.05 ± 0.15 × 103/μl), but significantly (P < 0.05) higher compared to other treatment groups (Fig. 5).
Discussion
In this study, parasitaemia due to T. brucei brucei was detected by day 2 post-infection (pi) in infected groups (A–H). However, T. brucei brucei parasitaemia has been detected by day 4 pi in the reports of studies in Wistar rats (Umar et al. 2000, 2007; Kobo et al. 2014; Ibrahim et al. 2016; Erin et al. 2019). The detection of parasitaemia suggests potential parasite replication following infection. The differences in day of parasitaemia detection could have resulted from variation in ages and weights of rats, feed, season and virulence of the parasite. Also, there was anaemia and leukopaenia in infected groups depicted by the significant decrease in haematological parameters post-infection. Anaemia has been reported to be a consistent finding in trypanosomosis resulting from trypanosome flagella lashing action, undulating pyrexia, immune complexes, trypanosome toxins and metabolites, lipid peroxidation and malnutrition (Murray and Morrison 1978; Morrison et al. 1981; Saror 1982; Igbokwe 1994; Mbaya et al. 2012). The apoptosis of leucocytes and bone marrow hypoplasia has been suggested to be responsible for the leukopaenia observed in trypanosomosis (Igbokwe and Anosa 1989; Marcondes et al. 2000; Kagira et al. 2006). The LD50 of Terminalia catappa fractions being greater than 1000 mg/kg suggests safety of the plant for use hence, necessitated the dose levels (100, 200 and 300 mg/kg) used in this study.
Following treatment, there was complete clearance of parasitaemia in groups treated with diminazene aceturate (group G) and 300 mg/kg ethylacetate fraction (group F) of Terminalia catappa leaves. Packed cells volume, haemoglobin concentration, red and total white blood cells were significantly (P < 0.05) increased in groups B–G but the increase was significantly (P < 0.05) higher in groups F and G compared to the other treatment groups at day 10 pi. Also, mortality was observed in group A despite receiving treatment at 100 mg/kg HF. The mortality could be due to low amount of trypanocidal components at this dose. However, in groups B, C, D and E, no mortality was recorded but parasitaemia was persistent in the face of increasing haematological parameters. This suggests that at these doses, trypanocidal level of the fractions was not achieved to completely clear the parasite from the blood. Rapid metabolism of the active components could possibly be responsible for the decreased trypanocidal activity observed at these lower doses. The ability of EAF at 300 mg/kg to completely clear the parasitaemia accompanied by higher haematological parameters at day 10 pi compared to HF at same dose suggests that EAF had more trypanocidal principles of importance in trypanosomosis. Antitrypanosomal activity by EAF of Punica granatum against T. b. brucei has also been reported (Inabo and Fathuddin 2011) suggesting presence of more trypanocidal constituents in EAF.
The antitrypanosomal activity of Terminalia catappa leaf fractions in this study evidenced by clearance of parasitaemia and restoration of haematological changes could be due to the presence of substances that act directly on the parasite, interfere with parasite replication, protect blood cell components and/or enhance the immune system of the host. These substances include flavonoids, terpenes and tannins (Shuaibu et al. 2008). Possible mechanisms involved erythrocytes protection from haemolysis via scavenging of reactive oxygen species (Umar et al. 2007; Ajakaiye et al. 2013), neutralisation of trypanosomes toxic metabolites and bone marrow stimulation (Ogoti et al. 2009; Mergia et al. 2014).
The significant clearance of parasitaemia and restoration of trypanosome-induced haematological changes by 300 mg/kg EAF in this study could be due to presence of optimum amount of constituents with antitrypanosomal activities at this dose. It could also results from the constituents acting individually or synergistically to produce these effects. The in vivo antitrypanosomal effect exhibited by Terminalia catappa is not uncommon as other reports have clearly demonstrated potent antitrypanosomal activity by different families of plants (Nok et al. 1993; Agbedahunsi et al. 2006; Bala et al. 2009; Inabo and Fathuddin 2011). The specific mechanisms by which antitrypanosomal activity is achieved by Terminalia catappa are therefore unknown and require further investigation. Hence, studies are recommended to isolate the active antitrypanosomal principles from ethylacetate fractions of Terminalia catappa leaves as an impetus for safer trypanocidal drugs development.
References
Abdallah AAM, Nasr El-Deen NAM, Neamat-Allah ANF, Hammza HEE, Abou-Elnaga HM (2020a) Impact of dietary supplementation with Echinacea purpurea on growth performance, immunological, biochemical, and pathological findings in broiler chickens infected by pathogenic E. coli. Trop Anim Health Prod 52:1599–1607
Abdallah AAM, Nasr El-Deen NAM, Neamat-Allah ANF, Abd El-Aziz HIA (2020b) Evaluation of the hematoprotective and hepato-renal protective effects of Thymus vulgaris aqueous extract on thermally oxidized oil-induced hematotoxicity and hepato-renal toxicity. Comp Clin Pathol 29:451–461
Abdallah AAM, Nasr El-Deen NAM, Abd El-Aziz HI, Neamat-Allah ANF (2020c) Effect of the aqueous root extract of Curcuma longa L. (turmeric) against thermally oxidized oil-induced hematological, biochemical and histopathological alterations. Comp Clin Pathol 29:837–845
Agbedahunsi JM, Anao I, Adewunmi CO, Croft SL (2006) Trypanocidal properties of Terminalia ivorensis A. Chev. (Combretaceae). Afr J Tradit Complement Altern Med 3(2):51–56
Ajakaiye JJ, Mazadu RM, Benjamin MS, Bizi LR, Shuaibu Y et al (2013) Effects of dietary vitamins C and E oral administration on body temperature, body weight and haematological parameters in Wistar rats infected with Trypanosoma brucei brucei (Federi strain) during the hot rainy season. Int Res J Pharm Pharmacol 3:105–111
Bala AY, Adamu T, Abubakar U, Ladan MJ, Abubakar MG (2009) Studies on the in vitro trypanocidal effect of the extracts of some selected medicinal plants in Sokoto State, Nigeria. Nigerian J Basic Appl Sci 17(2):257–264
Büscher P, Gonzatti MI, Hébert L, Inoue N, Pascucci I, Schnaufer A, Suganuma K, Touratier L, Van Reet N (2019) Equine trypanosomosis: enigmas and diagnostic challenges. Parasites Vector 12(234):234. https://doi.org/10.1186/s13071-019-3484-x
Dacie JV, Lewis SM (1991) Practical Haematology, 7th edn. Churchill Livingston, London, UK
Dinsa B, Yohannes M, Degefu H, Woyesa M (2013) Cross-sectional study of bovine trypanosomosis and major clinical signs observed in Diga District, Western Ethiopia. Afr J Agric Res 8(6):500–506
Echeverria LE, Morillo CA (2019) American trypanosomiasis (Chagas disease). Infect Dis Clin N Am 33(1):119–134
Erin PJ, Orakpoghenor O, Sani NA, Adeke JT, Atata JA, Adamu S, Esievo KAN (2019) Effects of acute experimental Trypanosoma brucei brucei infections on serum super oxide dismutase activities in relations to development of parasitaemia, anaemia and leucopenia in Wistar rats. Comp Clin Pathol 29(2):311–316
Ezeani MC, Okoro H, Anosa VO, Onyenekwe CC, Meludu SC, Dioka CE, Azikiwe CC (2008) Immunodiagnosis of bovine trypanosomiasis in Anambra and Imo states, Nigeria, using enzyme-linked immunosorbent assay: zoonotic implications to human health. J Vector Borne Dis 45:292–300
Giordani F, Morrison LJ, Rowan TG, de Koning HP, Barrett MP (2016) The animal trypanosomiases and their chemotherapy: a review. Parasitology 143:1862–1889
Hashem M, Neamat-Allah ANF, Gheith MA (2018) A study on bovine babesiosis and treatment with reference to hematobiochemical and molecular diagnosis. Slov Vet Res 55:165–173
Ibrahim A, Mbaya AW, Boniface Anene MB, Luka J (2016) Comparative parasitaemia and haematology of mice, rats and rabbits experimentally infected with Trypanosoma brucei brucei and their responses to diminazene diaceturate (Veriben®) therapy. Asian Pacific J Tropical Dis 6(7):527–532
Igbokwe IO (1994) Mechanisms of cellular injury in African trypanosomiasis. Vet Bull 64(7):611–620
Inabo HI, Fathuddin MM (2011) In vivo antitrypanosomal potentials of ethyl acetate leaf extracts of Punica granatum against T. b. brucei. Adv Agric Biotechnol 1:82–86
Kagira JM, Thuita JK, Ngotho M, Mdachi R, Mwangangi DM et al (2006) Haematology of experimental Trypanosoma brucei rhodesiense infection in vervet monkeys. African J Health Sci 13:59–65
Kellner HM, Eckert HG, Volz MH (1985) Studies in cattle of the anti-T.l drug Diminazene aceturate (Berenil). Trop Med Parasitol 36:199–204
Kobo PI, Erin PJ, Suleiman MM, Aliyu H, Tauheed M, Muftau S, Mamman M (2014) Antitrypanosomal effect of methanolic extract of Zingiber officinale (ginger) on Trypanosoma brucei brucei-infected Wistar rats. Vet World 7(10):770–775
Lorke D (1983) A new approach to practical acute toxicity testing. Arch Toxicol 54:275–287
Mahmoud EA, El-Sayed BM, Mahsoub YH, El-Murr AI, Neamat-Allah ANF (2020) Effect of Chlorella vulgaris enriched diet on growth performance, hemato-immunological responses, antioxidant and transcriptomics profile disorders caused by deltamethrin toxicity in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol 102:422–429
Marcondes MC, Borelli P, Yoshida N, Russo M (2000) Acute Trypanosoma cruzi infection is associated with anemia, thrombocytopaenia, leukopaenia and bone marrow hypoplasia: reversal by nitrofurtimox treatment. Microbes Infect 2(4):347–352
Mbaya A, Kumshe H, Nwosu CO (2012) The mechanims of anaemia in trypanosomosis: a review. (Silverberg, D. S., Ed). Croatia, In Tech Publishers, Pp. 240-282
Mehlitz D, Molyneux DH (2019) The elimination of Trypanosoma brucei gambiense? Challenges of reservoir hosts and transmission cycles: expect the unexpected. Parasite Epidemiol Control 6:e00113
Melaku A, Birasa B (2013) Drugs and drug resistance in African animal trypanosomosis: a review. Eur J Appl Sci 5(3):84–91
Mergia E, Terefe G, Teklehaymanot T, Shibeshi W (2014) Evaluation of in vivo Antitrypanosomal activity of aqueous and methanol leaf extracts of Clutia abyssinica (Euphorbiaceae) against Trypanosoma congolense. Austin J Pharmacol Ther 2(5):9–14
Morrison WI, Murray M, Sayer PD, Preston JM (1981) The pathogenesis of experimentally induced Trypanosoma brucei infection in the dog, tissue and organ damage. Am J Pathol 102:168–181
Muhanguzi D, Mugenyi A, Bigirwa G, Kamusiime M, Kitibwa A, Akurut GG, Ochwo S, Amanyire W, Okech SG, Hattendorf J, Tweyongyere R (2017) African animal trypanosomiasis as a constraint to livestock health and production in Karamoja region: a detailed qualitative and quantitative assessment. BMC Vet Res 13(355):355. https://doi.org/10.1186/s12917-017-1285-z
Murray M, Morrison WI (1978) Parasitaemia and host susceptibility in African trypanosomiasis. Proceedings of a workshop Nairobi, Kenya, pp 71–81
Neamat-Allah ANF (2015) Immunological, hematological, biochemical, and histopathological studies on cows naturally infected with lumpy skin disease. Vet World 8(9):1131–1136
Neamat-Allah ANF, El-Damaty HM (2016) Strangles in Arabian horses in Egypt: clinical, epidemiological, hematological, and biochemical aspects. Vet World 9(8):820–826
Neamat-Allah ANF, Mahmoud EA (2019) Assessing the possible causes of hemolytic anemia associated with lumpy skin disease naturally infected buffaloes. Comp Clin Pathol 28(3):747–753
Neamat-Allah ANF, Abd El-Hakim Y, Mahmoud EA (2020a) Alleviating effects of β-glucan in Oreochromis niloticus on growth performance, immune reactions, antioxidant, transcriptomics disorders and resistance to Aeromonas sobria caused by atrazine. Aquac Res 51:1801–1812. https://doi.org/10.1111/are.14529
Neamat-Allah ANF, Mahsoub YH, Mahmoud EA (2020b) The potential benefits of dietary β-glucan against growth retardation, immunosuppression, oxidative stress and expression of related genes and susceptibility to Aeromonas hydrophila challenge in Oreochromis niloticus induced by herbicide pendimethalin. Aquac Res. https://doi.org/10.1111/are.14910
Neamat-Allah ANF, El-Murr AI, Abd El-Hakim Y (2020c) Dietary supplementation with low molecular weight sodium alginate improves growth, haematology, immune reactions and resistance against Aeromonas hydrophila in Clarias gariepinus. Aquac Res 50:1547–1556. https://doi.org/10.1111/are.14031
Neamat-Allah ANF, Mahmmud EA, Mahboubb Y (2020d) Effects of dietary mulberry (Morus alba) on hemato-biochemical alterations, immunosuppression and oxidative stress induced by Aeromonas hydrophila in Oreochromis niloticus. Fish and Shellfish Immunology In Press, https://doi.org/10.1016/j.fsi.2020.11.028Nok AJ, Esievo KAN, Arowosafe S, Onyenekwe PC, Gimba CE, Kagbu JA (1993) In vitro activity of leaf extracts against T. brucei brucei. J Clin Biochem Nutr 15: 113–118
Ogoti P, Magiri E, Auma J, Magoma G, Imbuga M, Murilla G (2009) Evaluation of in vivo antitrypanosomal activity of selected medicinal plant extracts. J Med Plant Res 3:849–854
Radwanska M, Vereecke N, Deleeuw V, Pinto J, Magez S (2018) Salivarian trypanosomosis: a review of parasites involved, their global distribution and their interaction with the innate and adaptive mammalian host immune system. Front Immunol 9:2253
Saror DI (1982) Aspects of the anaemia of acute bovine trypanosomiasis. Proceedings of the first National Conference on Tsetse and Trypanosomiasis Research, Kaduna, Nigeria 12-14
Schalm OW, Jain NC, Carroll EJ (1975) Veterinary Haematology, 3rd Edn, Philadelphia. Lea and Febige, USA, Pa
Shuaibu MN, Wuyep PTA, Yanagi T, Hirayama K, Ichinose A, Tanaka T, Kouno I (2008) Trypanocidal activity of extracts and compounds from the stem bark of Anogeissus leiocarpus and Terminalia avicennioides. Parasitol Res 102(4):697–703
Swallow BM (2000) Impacts of trypanosomosis in African agriculture, Programme against African Trypanosomosis Technical and Scientific series. Food Agric Organ 2:45–46
Terças AG, Monteiro AS, Moffa EB, dos Santos JRA, de Sousa EM, Pinto ARB, Costa PC, Borges ACR, Torres LMB, Filho AKD, Fernandes ES, Monteiro CA (2017) Phytochemical characterization of Terminalia catappa Linn. extracts and their antifungal activities against Candida spp. Front Microbiol 8(595):1–13
Umar IA, Toh ZA, Igbalajobi FI, Gidado A, Buratai LB (2000) The role of vitamin C administration in alleviation of organ damage in rats infected with T. brucei. J Clin Biochem Nutr 28:1–7
Umar IA, Ogenyi E, Okodaso D, Kimeng E, Stancheva GI, Omage JJ, Isah S, Ibrahim MA (2007) Amelioration of anaemia and organ damage by combined intraperitoneal administration of vitamins A and C to Trypanosoma brucei brucei-infected rats. Afr J Biotechnol 6:2083–2086
Acknowledgements
Nigeria Institute of Leather and Science Technology, Zaria, Nigeria; Veterinary Parasitology and Entomology; Veterinary Pathology, Ahmadu Bello University, Zaria, Nigeria.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The use of animals in this study was approved by the Ahmadu Bello University Committee on Animal Use and Care (ABUCAUC).
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ojeleye, F.S., Inabo, H.I., Whong, C.M.Z. et al. Effects of n-hexane and ethylacetate fractions of Terminalia catappa leaf extract in experimental Trypanosoma brucei brucei infection in Wistar rats. Comp Clin Pathol 30, 67–74 (2021). https://doi.org/10.1007/s00580-020-03192-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-020-03192-y