Abstract
The alterations in serum super oxide dismutase (SOD) enzymes activities in relation to sequential haematological changes were determined to assess the impacts of oxidative stress in Wistar rats experimentally infected with Trypanosoma brucei brucei. Forty adult male rats allocated into infected and control groups, of 20 rats each based on their mean body weight were used. Rats in infected group were administered 1 ml of reconstituted infected blood containing 1 × 106 trypanosomes/ml obtained from T. b. brucei–infected donor rat. Blood sample in EDTA sample tubes was collected for haematological parameters determinations, and sera were also harvested for determination of SOD activities on day 0 post-infection (pi) (day of infection) and days 3, 5 and 7 pi. Results indicated pre-patent period of 3.75 ± 0.11 days and increase in levels of parasitaemia throughout the study. Packed cell volume (PCV) and total white blood cell count (TWBC) were significantly (p < 0.05) reduced with values of 33.00 ± 2.89% and 6.93 ± 0.48 × 109/L, respectively on day 7 pi compared with control and pre-infection values. Serum SOD activity had a significant (p < 0.05) negative correlation (г = − 0.9) with the levels of parasitaemia in the T. b. brucei–infected rats. In conclusion, infection of rats with T. b. brucei caused a decrease in the serum SOD activities proportional to the level of parasitaemia and duration of the infection, and this is suggested to be responsible for sequential development of anaemia and leucopenia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
African trypanosomosis is caused by flagellate protozoan parasites of the genus, Trypanosoma (Hoffman et al. 2013). The parasites inhabit the blood plasma, intercellular tissues and body cavity fluid of an infected animal precipitating anaemia and tissue damage (Sharma et al. 2000). African trypanosomosis is a serious threat to human and animal health in sub-Saharan Africa placing 60 million people and over 30% of cattle populations in this region at risk of the infection (WHO 2005; Sachs 2010). Human African trypanosomiasis or sleeping sickness have been reported to only occur in Sub-Saharan Africa (WHO 2005). It is caused by two subspecies of Trypanosoma brucei brucei, namely Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense (Simo et al. 2012). Trypanosoma cruzi though not found on the African continent, causes Chagas disease, which is a devastating human disease in America (WHO 2005). Animal African trypanosomiasis is caused by trypanosome species and subspecies other than those affecting humans, and these include Trypanosoma brucei, Trypanosoma congolense, Trypanosoma equiperdum, Trypanosoma simiae, Trypanosoma suis and Trypanosoma vivax (Muhanguzi et al. 2017). Trypanosoma brucei is of great importance as it causes sleeping sickness in humans and nagana in cattle (Ndondo et al. 2016). Despite extensive research efforts, trypanosomiasis has continued to be an impediment to profitable livestock production (Esievo and Saror 1991; Omotainse et al.2000).
Anaemia which is a major cardinal sign of trypanosomosis, had been reported to result from generation of reactive oxygen species (Igbokwe 1994; Ogunsami and Taiwo 2007; Akanji et al.2009), and in the development of tissue degenerative changes observed in the infected host (Ekanem and Yusuf 2008; Akanji et al.2009). The involvement of oxidative stress, alterations of endogenous antioxidant enzyme such as SOD (Punitha et al.2005) and development of free radicals have been reported to play roles in the pathophysiologic mechanism of trypanosomosis (Serem et al.2013; Kobo et al.2014). Also, the interplay between the parasite and the mononuclear phagocyte system through the release of macrophage migration inhibitory factor (MIF) and TNF-α have been associated with anaemia development (Stijlemans et al. 2018). African trypanosomosis is a global concern of biomedical scientists, necessitating the development of integrated approach towards its disease via application of all findings in natural hosts and laboratory animal models (Abenga 2014). The knowledge of alterations in the serum profiles of SOD in trypanosomal infection may prove vital to further understanding of the mechanism of development of the disease in affected animals. Hence in this study, level of parasitaemia and its sequential effect on serum SOD activities in relations to the observed haematological changes were determined following experimental trypanosomosis in Wistar rats.
Methodology
Ethical approval
Ethical approval for the use of animals in this study was obtained from the Ahmadu Bello University Committee on Animal Use and Care (ABUCAUC).
Experimental Animals and Procedures
A total of 40 adult male rats kept in rat cages were acclimatized for a period of 14 days prior to the commencement of the experiment. On the day of commencement of the experiment, the rats were allocated, on basis of mean body weight, to two groups (infected and control) of 20 rats each. The trypanosome parasite (Federi strain) obtained from donor rat blood was reconstituted using physiological saline to obtain a concentration of 1 × 106 trypanosomes/ml. The inoculations of rats in the infected group were carried out as described by Ekanem and Yusuf (2005) using 1 ml of reconstituted infected blood via intraperitoneal administration.
Sample collection
Before infection with T. b. brucei infected blood on the day of inoculation (day 0) blood samples were collected from rats in the infected and control groups for haematologic and SOD estimations; whole blood in EDTA coated sample tube and in plain sample tube for serum respectively. Blood was also collected from both control and infected groups at day 3, 5 and 7 pi for haematologic and SOD estimations.
Determination of parasitaemia in T. b. brucei infected Rats
Beginning from day 1 post infection (pi) and throughout the course of the experiment, blood was collected from the tail directly unto a clean glass slide and covered with cover slip. The wet mount was viewed under the light microscope at × 40 magnification to monitor and score parasitaemia according to the rapid matching method described by Herbert and Lumsden (1976).
Determination of packed cell volume and total leucocyte count
Packed cell volume was determined using microhaematocrit method and total leucocyte count was determined using haemocytometer method as described by Dacie and Lewis (1991) in both control and infected group.
Determination of serum superoxide dismutase activity
The activity of superoxide dismutase (SOD) was measured using the Northwest Life Science Specialties SOD kit (NWLSS™ NWK-SOD02), based on the method of monitoring the auto-oxidation rate of haematoxylin, originally described by Martin et al. (1987) and modified to enhance reliability. Briefly, 230 μl of assay buffer was added to wells of the microplate. Then, 10 μL of assay buffer (for blank) and 10 μl of serum sample were added. The wells were properly shaken, mixed and incubated for 2 min. A multi-channel pipette was used to add 10 μl of haematoxylin reagent to begin the reaction. The content of each well was quickly mixed using instrument’s shaker function, and, immediately, the absorbance at 560 nm was recorded. The SOD activity was calculated as: SOD U/ml = 1.25× % inhibition.
Data analysis
Summarized results were expressed as mean ± standard error of mean (± SEM). Data obtained were subjected to Student’s t test and Pearson’s correlation analysis. Values of P < 0.05 were considered significant (Snedecor and Cochran 1994). Graph pad prism version 4.2 was used for the analysis.
Results
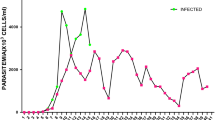
A parasitaemia was detected in the T. b. brucei-infected rats with a pre-patent period of 3.75 ± 0.11 days. The mean parasitaemia in the infected rats on days 3, 5 and 7 pi were 2.24 ± 0.63 × 106 (mean log10 = 6.30 ± 0.13), 1.08 ± 0.11 × 108 (mean log10 = 8.03 ± 0.04) and 4.04 ± 0.49 × 108 (mean log10 = 8.68 ± 0.017) parasites/ml of blood, respectively (Fig. 1).
The pre-infection mean PCV values in the T. b. brucei–infected and control groups were 52.00 ± 11.14 and 48.33 ± 5.24 %, respectively (Fig. 2). Following infection, the mean PCV values in the two groups remained fairly at the same level up to day 3 pi. Afterwards, the mean PCV values in the T. brucei–infected rats was significantly (p < 0.05) lower than the pre-infection and corresponding control group, recording a lowest value of 33.00 ± 2.89 % on day 7 pi.
The pre-infection mean TWBC in the infected and control groups were 10.85 ± 2.09 and 10.03 ± 1.16 x109/L, respectively (Fig. 3). Following infection, the mean TWBC in the infected rats showed an initial decrease to 6.33 ± 0.47 on day 3 pi, increase to 7.58 ± 0.66 on day 5 pi and later decrease to 6.93 ± 0.48 x109/L on day 7 pi; these were significantly (p < 0.05) lower than the corresponding control group.
The pre-infection mean serum SOD activities in the T. b. brucei–infected and control groups of rats were 7.23 ± 0.63 and 7.36 ± 0.53 U/ml, respectively (Fig. 4). Following infection, mean serum SOD activity in T. b. brucei–infected rats increased to 8.22 ± 0.83 U/ml on day 3 pi followed by a progressive decrease to 6.10 ± 0.37 U/ml and 5.58 ± 0.86 U/ml on day 5pi and 7 pi respectively. The mean serum SOD activities in the two groups post-infection were not significantly different (p > 0.05).
Both PCV (r = − 0.97) and serum SOD (r = − 0.91) had negative correlation with parasitaemia and were statistically significant (p < 0.05) at days 5 and 7 pi (Table 1).
Discussion
In this study, the level of parasitaemia and its sequential effect on serum SOD activities in relations to the observed haematological changes were determined following experimental T. b. brucei infection in Wistar rats. The mean prepatent period for the infection with T. brucei of 3.75 ± 0.11 days recorded in this study, with a progressive parasitaemia up to day 7 pi was similar to previous reports by Umar et al. (2007) and Kobo et al. (2014). The significant reduction in packed cell volume in the T. b. brucei–infected rats, as observed in this study could result in anaemia (Esievo 2017), which is consistent with the established clinical manifestations of animal trypanosomosis as reported by Anosa (1988) and Esievo and Saror (1991). The reduction in packed cell volume coincided with parasitaemia. This finding is in conformity with previous reports that correlated development of anaemia with onset and the degree of parasitaemia (Dargie et al. 1979; Umar et al.2009).
The significant reduction in total white blood cell count in the T. b. brucei–infected rats was in agreement with earlier reports by Fialkow et al. (2007) and Kobo et al. (2014). The decrease in total leucocyte count observed in the infected rats were attributed to factors such as variable surface antigen of the trypanosomes and bone marrow depression of their production (Kagira et al.2006; Kobo et al. 2014); lipoperoxidation can also predispose them to direct cellular lysis or phagocytosis just as with the case of erythrocytes because of the polyunsaturated lipids in their membranes (Itou et al. 1996; Fialkow et al. 2007).
The higher activity of serum SOD in the infected rats at the onset of the infection (day 3pi) is probably due to an upregulation of SOD being a first line of antioxidant defence involved with the conversion of superoxide anion (O2−) into hydrogen peroxide and other peroxides which are degraded by either glutathione peroxidase (GPx) or catalase (Valko et al.2006). Increased SOD synthesis and activity following the upregulation would concomitantly increase the formation of less toxic radicals. In a parallel study by Souza et al. (2014) with a different tissue parasite (canine visceral leishmaniasis), they found an increased SOD activity in the blood of infected dogs corroborated by the work of Dimri et al. (2012) on dogs infected with Dirofilaria immitis with similar findings. The progressive lower serum levels of SOD in the infected rats on days 5 and 7pi compared with the control could be attributed to possible exhaustion of the endogenous antioxidant enzymes embattled with the increasing generations of ROS during the infection (Igbokwe et al. 1996; Omer et al. 2007). SOD is involved in the direct elimination of ROS, through dismutation of superoxide radicals (Punitha et al.2005), but in excess generation of ROS, there is depletion of the endogenous antioxidant defence system (Igbokwe et al. 1996; Baydas et al.2002; Yusuf et al.2012), and this could be the possible reason for the reduction in serum SOD activities observed in this study.
The negative correlation of the levels of parasitaemia with PCV in the T. b. brucei–infected rats indicated that the erythrocytes are either more sensitive or more affected by the trypanosomes induced blood cell damage. The findings in this study on SOD activities were contrary to previous reports by Ataley et al. (2000), Ogunsami and Taiwo (2007) and Yusuf et al. (2012), who reported increased antioxidant enzymes activities. This might be due to differences in the concentration of released reactive oxygen or nitrogen species in the trypanosomal infected rats resulting from parasitaemia. This is because the more the reactive species released, the higher the tendency to overwhelm the antioxidant capacity of the host animal. The reduction, though not significant statistically, in superoxide dismutase (SOD) activities following infection supports the fact that reactive oxygen species were released during the infection, and the reduction may have been a consequence of SOD in the mopping up of the oxidant substance (Igbokwe et al.1996; Omer et al.2007; Yusuf et al.2012).
Conclusion
The infection of rats with T. b. brucei resulted in decreased serum SOD activities proportional to level of parasitaemia and duration of infection. The level of parasitaemia also has an impact on both PCV and TLC. This is therefore suggested to be responsible for the sequential development of anaemia and leucopenia observed.
References
Abenga JN (2014) Review: a comparative pathology of Trypanosoma brucei infections. Glob Adv Res J Med Med Sci 3(12):390–399
Akanji MA, Adeyemi OS, Oguntoye SO, Suleiman F (2009) Psidium guavaja extract reduses trypanosomiasis associated lipid peroxdation and raised glutathione concentrations in infected animals. Exp Clin Sci Int Online J Adv Sci 8:148–154
Anosa VO (1988) Haematological and biochemical changes in human and animal trypanosomiasis. Rev Elev Med Vet Pays Trop 41(2):151–164
Ataley M, Laksonem DE, Khanna S, Kaliste KE, Hanninem O, Sen CK (2000) Vitamin E regulates changes in tissues antioxidants induced by fish oil and exercise. Med Sci Sports Exerc 32:601–607
Baydas G, Canatan H, Turkoglu A (2002) Comparative analysis of the protective effects of melatonin and vitamin E on streptozotocin-induced diabetes mellitus. J Pineal Res 32:225–230
Dacie JV, Lewis SM (1991) Practical haematology, 7th edn. Churchill Livingston, London
Dargie JD, Murray PK, Murray M, Grimshaw WRT, McIntyre WIM (1979) Bovine trypanosomiasis: the cell kinetics of N’dama and Zebu cattle infected with T. Congolense. Parasitology 78:271–286
Dimri U, Singh SK, Sharma MC, Behera SK, Kumar D, Tiwari P (2012) Oxidant/antioxidant balance, minerals status and apoptosis in peripheral blood of dogs naturally infected with Dirofilaria immitis. Res Vet Sci 93(1):296–299
Ekanem JT, Yusuf OK (2005) Activities of alkaline phosphatase, glutamate oxaloacetate transaminase and glutamate pyruvate transaminase in liver and serum of Trypanosoma brucei - infected rats treated with honey. Biokemistri 17:185–191
Ekanem JT, Yusuf OK (2008) Some biochemical and haematological effects of black seed (Nigella sativa) oil on T. brucei-infected rats. Afr J Biomed Res 11:79–85
Esievo KAN (2017) Veterinary clinical pathology. Spectrum Books Limited Ibadan. First Editions
Esievo KAN, Saror DI (1991) Immunochemistry and immunopathology of animal trypanosomiasis. Vet Bull 61:765–777
Fialkow L, Wang Y, Downey GP (2007) Reactive oxygen and nitrogen species as signaling molecules regulating neutrophil function. Free Radic Biol Med 42:153–164
Herbert WJ, Lumsden WHR (1976) Trypanosoma brucei: a rapid “matching” method for estimating the host’s parasitemia. Exp Parasitol 40(3):427–431
Hoffman R, Benz JEJ, Silberstein LE, Heslop H, Weitz J, Anastasi J (2013) Hematology: basic principles and practice. Elsevier Health Sciences, London, pp 23–43
Igbokwe IO (1994) Mechanism of cellular injury in African trypanosomiasis. Vet Bull 64:611–620
Igbokwe IO, Umar IA, Omage JJ, Ibrahim NDG, Kadima KB, Obagaiye OK, Saror DI, Esievo KAN (1996) Effect of acute Trypanosoma vivax infection on cattle erythrocyte glutathione and susceptibility to in vitro peroxidation. Vet Parasitol 63:215–224
Itou T, Iida T, Kawatsu H (1996) Kinetics of oxygen metabolism during respiratory burst in Japanese eel neutrophils. Dev Comp Immunol 20:323–330
Kagira JM, Thuita JK, Ngotho M, Mdachi R, Mwangangi DM et al (2006) Haematology of experimental Trypanosomabruceirhodesiense infection in vervet monkeys. Afr J Health Sci 13:59–65
Kobo PI, Erin PJ, Suleiman MM, Aliyu H, Tauheed M, Muftau S, Mamman M (2014) Antitrypanosomal effect of methanolic extract of Zingiber officinale (ginger) on Trypanosoma brucei brucei-infected Wistar rats. Vet World 7(10):770–775
Martin JP Jr, Dailey M, Sugarman E (1987) Negative and positive assays of superoxide dismutase based on haematoxylin autoxidation. Arch Biochem Biophys 225:329–336
Muhanguzi D, Mugenyi A, Bigirwa G, Kamusiime M, Kitibwa A, Akurut GG, Ochwo S, Amanyire W, Okech SG, Hattendorf J, Tweyongyere R (2017) African animal trypanosomiasis as a constraint to livestock health and production in Karamoja region: a detailed qualitative and quantitative assessment. BMC Vet Res 13:355
Ndondo AM, Munganga JM, Mwambakana JN, Saad-Roy CM, van den Driessche P, Walo RO (2016) Analysis of a model of gambiense sleeping sickness in humans and cattle. J Biol Dyn 10:347–365
Ogunsami AO, Taiwo VO (2007) Pathobiochemical mechanisms involved in the control of diseases caused by Trypanosoma congolense in African grey duiker (Sylvicapra grimmia). Vet Parasitol 96:51–63
Omer OH, Mousa HM, Al-Wabel N (2007) Study on the antioxidant status of rats experimentally infected with Trypanosoma evansi. Vet Parasitol 145:142–145
Omotainse SO, Edegbhere H, Omoogum GA, Thompson G, Igweh CA, Ukah JAC, Ikenga MA, Halid I (2000) The prevalence of animal trypanosomosis in Konshisha Local Government Area of Benue State Nigeria. Israel J Vet Med 55(4):75–79
Punitha ISR, Rajendran K, Shirwaikar A, Shirwaikar A (2005) Alcoholic stem extract of Coscinium fenestratum regulates carbohydrate metabolism and improves antioxidant status in streptozotocin-nicotinamide induced diabetic rats. ECAM 2:375–381
Sachs J (2010) The current situation. In: Linking sustainable human and animal African trypaosomiasis control with rural development. PAAT Tech Sci Ser 10:3–8
Serem EK, Mburu J, Mdachi R, Korir S, Kibugu J, Kagira J, Ngure R (2013) Effects of crude extracts of Solanum nigrum on the liver pathology and survival time in Trypanosoma brucei rhodesiense-infected rats. Sci J Microbiol 1–7
Sharma DK, Chauhan PPS, Saxena VK, Agrawal RD (2000) Haematological changes in experimental trypanosomiasis in Barbari goats. Small Rumin Res 38:145–149
Simo G, Silatsa B, Flobert N, Lutumba P, Mansinsa P, Madinga J, Manzambi E, De Deken R, Asonganyi T (2012) Identification of different trypanosome species in the mid-guts of tsetse flies of the Malanga (Kimpese) sleeping sickness focus of the Democratic Republic of Congo. Parasit Vector 5(201):2–9
Snedecor GW, Cochran WG (1994) Statistical methods. Oxford and IBH Publishing Corporation, Calcutta 509 pp
Souza CC, Barreto TDO, Silva SM, Pinto AW, Figueiredo MM, Ferreira Rocha OG, Tafuri WL (2014) A potential link among antioxidant enzymes, histopathology and trace elements in canine visceral leishmaniasis. Int J Exp Pathol 95(4):260–270
Stijlemans B, De Baetselier P, Magez S, Van Ginderachter JA, De Trez C (2018) African trypanosomiasis-associated anaemia: the contribution of the interplay between parasites and the mononuclear phagocyte system. Front Immunol 9:218–233
Umar IA, Ogenyi E, Okodaso D, Kimeng E, Stancheva GI, Omage JJ, Isah S, Ibrahim MA (2007) Amelioration of anaemia and organ damage by combined intraperitoneal administration of vitamins A and C to Trypanosoma brucei brucei-infected rats. African J Biotechnol 6:2083–2086
Umar I, Maryoms N, Daikwo E, Gidado A, Buratai L, Igbokwe I, Ibrahim M (2009) The effect of aqueous extracts of Hibiscus sabdariffa (Sorrel) calyces on heamatological profile and organ pathological changes in Trypanasoma congolense-infected rat. African J Trad Compl Alt Med 6(4):87–94
Valko M, Rhodes CJ, Moncol J, Izakovic MM, Mazura M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160:1–40
WHO (2005) Control and surveillance of African trypanosomiasis. Tech Rep Ser 881:1–113
Yusuf AB, Umar IA, Musa UB, Nok AJ (2012) Screening of Vernonia amygdalina and Hymenocardia acida extracts and 1-3-diaminopropane for antitrypanosomal activity: in vitro model. J Med Plant Res 6(19):3573–3578
Acknowledgement
Department of Veterinary Pathology, Ahmadu Bello University, Zaria Nigeria
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article contains studies with animal subjects performed by the author and ethical approval for this study was provided by the Ahmadu Bello University Committee on Animal Use and Care (ABUCAUC).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Erin, P.J., Orakpoghenor, O., Sani, N.A. et al. Effects of acute experimental Trypanosoma brucei brucei infections on serum super oxide dismutase activities in relations to development of parasitaemia, anaemia and leucopenia in Wistar rats. Comp Clin Pathol 29, 311–316 (2020). https://doi.org/10.1007/s00580-019-03066-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-019-03066-y