Abstract
Background
Success rate of endoscopic dilation (ED) of complex benign esophageal strictures (CBES) can be as low as 65%. Since EDs are usually performed at 2–4-week intervals, the aim of this study was to evaluate the clinical outcomes of EDs done initially at weekly intervals.
Methods
A cohort of patients with CBES (luminal diameter < 10 mm) underwent ED at weekly intervals and subsequent dilation intervals adjusted based on response. Weekly EDs were also re-initiated in those requiring additional interventions (electro-cautery/stents). Group A patients: Failed prior EDs done at ≥ 2-week intervals. Group B: CBES with no prior dilations. Success was defined as achieving and maintaining a luminal diameter of ≥ 14 mm and patient remaining dysphagia-free with minimal re-interventions.
Results
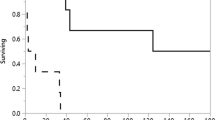
488 EDs were performed on a cohort of 57 consecutive patients with CBES. Median follow-up was 4 years. Group A: 21 patients (mean age 65 ± 13 years; mean interval between prior failed dilations 17 ± 9 days). 57% of these patients achieved long-term success with weekly dilations (mean 8 ± 4.7 dilations/patient). Group B: 36 patients (mean age 61 ± 13 years, mean 6.5 ± 5.5 dilations/patient). Long-term success was 83.3% (P = 0.033). Despite weekly dilations, unable to achieve a diameter of 14 mm in 5 patients. AE: perforation 1 (0.2%), bleeding 1 (0.2%).
Conclusion
Significant proportion of patients with CBES who failed prior dilations done at ≥ 2-week intervals achieved dysphagia-free status by initiating weekly dilations. Hence, before considering other options (electro-cautery/stents), one can consider using this approach. This approach can also be used upfront in patients with newly diagnosed CBES.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Endoscopic dilation (ED) in a step-up manner is the primary modality used to manage benign esophageal strictures [1, 2]. Majority of EDs are done initially at 2–4-week intervals. Simple strictures respond well by ≤ 3 ED sessions [3,4,5]. Complex benign esophageal strictures (CBES) that are long, tight, tortuous, or multifocal can be refractory (RBES) defined as inability to reach a luminal diameter of ≥ 14 mm despite 5 dilations at 2-week intervals, or requiring ≥ 1 dilation every 4 weeks to maintain a diameter of 14 mm [6]. Based on the etiology, the success rates of ED in these patients could be as low as 60–65% [7, 8]. Additional interventions such as electro-cautery incision or stents may be required.
Following breach in mucosal barrier (example, after dilation), fibrosis sets in by 2 weeks [9, 10]. Hence, at one tertiary-care center, a strategy of dilating patients with CBES initially every week was adopted, especially in those who were referred after failed dilations done at ≥ 2-week intervals. The objective of this pilot study was to evaluate the long-term clinical outcomes in a cohort of patients where this approach was used.
Materials and methods
Data was extracted from a prospectively maintained database on a cohort of consecutive patients with CBES who underwent endoscopic dilations done initially every week as per the protocol described below and in Fig. 1. The study was approved by the local Institutional Review Board and patients/health-care power of attorney gave informed consent.
Study subjects
All patients were ≥ 18 years and had biopsy proven CBES defined as being long (> 2 cm, except anastomotic strictures that can be short), tight (< 10 mm diameter), tortuous, or multifocal. Exclusion criteria were (a) malignant stricture, (b) extrinsic compression of the esophagus, (c) fistula/leaks, (d) eosinophilic esophagitis, (e) primary motility disorders, (f) cannot keep follow-up appointments, (g) other malignancies or co-morbidities limiting life expectancy, and (h) unsafe (laryngeal penetration/aspiration) oro-pharyngeal swallowing. Complex benign esophageal strictures resulting from radiation given for head-and-neck cancer underwent swallow study at baseline to document safe swallowing. This study was repeated if patient developed episodes of aspiration.
Patients were classified into two groups. Group A included patients who failed to achieve success (as defined below) despite EDs done at ≥ 2-week intervals. Group B included patients with newly diagnosed CBES in whom the protocol was used upfront. Dysphagia was graded on a previously described scale as Grade 0: can swallow normal diet, no dysphagia; Grade 1: difficulty in swallowing some solids; Grade 2: can swallows only semi-solid/soft to liquid diet; Grade 3; can swallow only liquids, and Grade 4: cannot swallow even liquids, total dysphagia [11]
Protocol (Fig. 1)
Step-up EDs were performed starting at weekly intervals (Fig. 1) till an esophageal luminal diameter of 14 mm was reached. Attempts were made to safely go up to higher diameters so as to achieve more durable response [12]. In patients with radiation or corrosive injuries, wire-guided bougie was preferred over balloon to achieve pan-esophageal dilation as these patients may have multifocal strictures. Moreover, segments between 10 and 13 mm in diameter can be missed using a standard 9.5 mm diameter endoscope while focusing only on the tight (< 9 mm) regions if a balloon is used. Focal strictures were dilated using balloon or bougie.
All dilations were performed with anesthesia assisted sedation. Fluoroscopy was used only for guidewire placement if even the 5 mm diameter endoscope would not traverse the stricture. Based on the stricture diameter as assessed by the endoscope (< 9.5 mm standard or < 5 mm thin endoscope), and/or using open biopsy forceps, dilation was performed to a value that was considered safe (resistance-to-feel) using standard techniques and recommendations. After dilation, re-look endoscopy was performed in all patients. Patients also underwent immediate contrast fluoroscopy evaluation if perforation was suspected. All strictures were periodically biopsied (every 2–3 months).
At each weekly ED, how much the stricture has regressed (or not) in diameter was assessed. If the stricture had partially regressed (“two-steps-forwards and one-step-back”), dilation was performed to higher diameters and the weekly dilations continued till a diameter of ≥ 14 mm was achieved. Following this, the frequency of dilations required to maintain this diameter was determined by increasing (or decreasing) the intervals between dilations. Steroids were injected into the stricture if the patients continued to require frequent dilations.
Additional measures
If the patient continued to require weekly dilations and the stricture kept regressing to its original diameter (“two-steps-forward and two-steps-back”), additional measures were used. For short strictures, electrocautery-incision with needle-knife was performed (Fig. 2, Video 1) [13,14,15,16,17]. Lumen apposing metal stents (LAMS) were also used for short (< 1 cm) strictures (Fig. 3) [18, 19]. For longer strictures, fully-covered self-expanding metal stent (FC-SEMS, 18/23 mm diameter) were used [20,21,22,23,24]. Stents were sutured in place only if they migrated and required re-positioning [25, 26]. All patients where the stent bridged across the GE junction were give proton pump inhibitors and advised to follow anti-reflux measures. Stents were removed after 4 weeks. Combination techniques such as electrocautery-incision followed by FC-SEMS placement in the same session was also used in patients with tight strictures (< 5 mm) to further “tear” open the incised stricture and seal micro-perforation if any (Video 1).
One week after additional measures (electrocautery/stent removal), weekly EDs were re-initiated. This was done to assure that the gains made by the additional measures were not lost by waiting longer.
Objectives: success and failure
Primary objective was to determine the proportion of patients who achieved success defined as achieving a luminal diameter of ≥ 14 mm and patient remaining dysphagia-free (grade ≤ 1) with none to no more than 1 dilation required every 3 months to maintain this diameter/dysphagia-free status. These patients were advised to call if dysphagia recurred. Secondary objectives were to determine what proportion of patients needed additional measures besides dilation to achieve success, did not achieve 14 mm diameter, failure rates, and adverse events (AE).
Patients who did not achieve success (14 mm diameter) as per the above criteria were classified as failures. Those who achieved success and then reverted to > 1 dilation every 3 months were also considered as failures. These patients were offered training in self-dilation. [27, 28] They were trained in real-time on their first visit watching a fluoroscopy screen while performing self-dilation with a weighted dilator (Maloney®, Fig. 4) and then instructed to dilate themselves at home at least once daily. Patients were advised to call for “rescue” ED if they developed discomfort/increasing resistance while passing the dilator.
Training in self-dilation of esophageal stricture. A Initially the patient is trained to self-dilate while watching fluoroscopy monitor in real-time as biofeedback. B Two months later at one follow-up clinic visit, patient demonstrating self-dilation (no fluoroscopy). Patient preferred to stand instead of the recommendation to sit for concerns of falling. The tape on the dilator (arrow) marks the level up to which the dilator needs to be inserted and is based on prior endoscopy assessment of the stricture location
Patients who refused self-dilation and required EDs at the rate of more ≥ 2 every 3 months were also considered as failures. Similarly, those who preferred long-term stenting were counted as failures despite being dysphagia-free.
Follow-up data was collected each time during scheduled endoscopies. Those who responded were followed at approximately 3 months, 6 months, and then yearly (some via telephone).
Statistical analysis
Statistical analysis was done using StatDirect 3.3.5. Descriptive statistics were used to summarize demographic and clinical characteristics of the patients. Measures of central tendency (mean and median for normally distributed variables and for skewed distribution respectively) and measures of spread (standard deviation and range) was used for continuous data while frequency listing was used for categorical data summarized using proportions. Continuous variables were compared using t-test for normal data and Mann–Whitney test for skewed data while the Chi-square or Fisher’s exact test were used to compare categorical variables. P value of < 0.05 was used as a determinant of statistical significance.
Results
Study subjects
A cohort of 57 consecutive patients with CBES were enrolled and underwent a total of 488 dilations (not counting dilations done daily by those performing self-dilations).
Group A: Cohort of 21 patients (mean age 65 ± 13 SD years; male 9, female 12) with CBES were enrolled. At baseline, 10 (48%) patients had esophageal lumen of < 5 mm and dysphagia grade of 3–4. In 11 patients (48%), the baseline diameter was between 5 and 10 mm, and dysphagia grade was between 2 and 3. These patients had failed a median of 6 (range of 4–12) prior dilations done at a mean interval of 17 ± 9 SD days between each dilation. Six of these patients who failed prior EDs also had steroid injected into the stricture. Weekly ED as per the protocol was initiated and patients were followed for a median of 4 years (range of 1–9 years). One patient was lost to follow-up for 5 months and then re-established care after recurrent dysphagia.
Group B: This group included 36 patients (mean age 61 ± 13 SD years; male 21, female 15) with newly diagnosed CBES [Baseline luminal diameter < 5 mm in 19 (53%) with dysphagia grade 3–4; luminal diameter between 5 and 10 mm in 17 (47%) with dysphagia grade 2–3]. Majority of these referrals were from within the institute referred by oncology, surgery, gastroenterology, and internists. Following interventions as per protocol, these patients were also followed up for a median of 4 years (range of 1–9 years).
The etiology of the strictures between the two groups were comparable (Table 1). One patient (Group A) had lichen planus induced stricture and another was NG-tube related following post-ERCP pancreatitis. Two patients in Group B had esophageal leaks managed with FC-SEMS. After the stent removal, the leaks resolved but they developed CBES (< 5 mm).
Clinical outcomes
Success: In 52 of the 57 patients (92%), a luminal diameter of ≥ 14 mm was achieved. On follow-up, the stricture recurred in 10 that required more than one ED every 3 months. Hence the overall success rate of 74%. In Group A, 57% (12 of 21) of patients who failed dilations done at the traditional 2-week intervals achieved long-term success with weekly EDs (mean 8 ± 4.7 dilation/patient). The success rate was even higher in those where this protocol was applied upfront (Group B: 83.3%; P 0.033; mean 6.5 ± 5.5 dilations/patient). Of the 12 patients who achieved success in Group A, 7 patients (58%, ITT 33%) achieved long-term success by just dilating them initially once every week. Remaining 5 patients (42%) did not respond to weekly dilations and required temporary FC-SEMS. Following stent removal, they started responding to weekly dilations and eventually achieved long-term success (Table 2). In Group B, 25 of 30 patients (83.3%, ITT 69%, mean 6.5 ± 5.5 dilations/patient) achieved long-term success by weekly dilations. In 5 (16.7%) patients additional interventions (FC-SEMS: 4, electro-cautery-incision plus SEMS: 1) followed by re-initiating weekly EDs were needed to achieve long-term success (P = 0.057 compared to Group A).
Failures: Etiology of the strictures in those who failed to achieve success are shown in Table 3. Majority of these were post-radiation (40%) and anastomotic stricture (33%). In 5 patients, a diameter of 14 mm could not be achieved and in others, the strictures recurred requiring > 1 dilation every month after achieving a 14 mm diameter. Table 4 shows the outcomes of those who failed the protocol. Four patients initially achieved success but within 12-month returned with recurrence of dysphagia requiring more than 1 dilation every 12 weeks. Although 3 of them again achieved success, their need for dilations kept varying from one dilation every 2 to 4 months. Only 2 accepted to train in self-dilation (in one with radiation stricture, the husband dilated the patient every day). One patient with anastomotic stricture initially had a LAMS placed with good benefit. Following stent removal, the stricture recurred within 4 weeks. He developed a pseudo-diverticulum proximal to the stricture that required partial trans-esophageal diverticulotomy. As the patient continued to require frequent dilations and was not a candidate for self-dilations (diverticulum) he opted for long-term LAMS.
Mortality
Eleven patients deceased of whom 5 were in the failure group and 6 had already achieved success. Except for one patient as described below (1; failure group), all other causes for mortality were unrelated to EDs:
Mortality in failure group: (1) Radiation stricture being dilated every 4–6 weeks. Patient did not want SEMS or self-dilation. At 28 months perforation occurred during a dilation that resulted in a trachea-esophageal fistula. Covered SEMS was placed. Family opted for hospice care as the patient had severe dementia, (2) 12-month follow-up developed metastatic head-and-neck cancer, (3) 42-month follow-up, respiratory failure from smoking and worsening emphysema. (4) 24-month follow-up, recurrent lung cancer, (5) 18-month follow-up, recurrent laryngeal cancer.
Mortality in success (dysphagia-free) group: (1) 36-month follow-up, recurrent adenocarcinoma at anastomotic site with brain metastasis, (2) 20-month follow-up, myocardial infarction, (3) 78-month follow-up, metastatic colon cancer, (4) 18-month follow-up, metastatic head-and-neck cancer, (5) 36-month follow-up, cirrhosis with hepatic decompensation, and (6) 42-month follow-up, progressive multiple sclerosis with urinary sepsis, hospice care.
Other adverse events
Chest pain: following FC-SEMS in one patient with post-RFA/EMR stricture, Stent had to be removed within a week. Another patient required pain medications for 5 days following FC-SEMS.
Continuous oozing of blood following dilation in one patient with post-radiation stricture who also had significant neovascularization of the esophageal mucosa. Although this patient did not require hospitalization or blood transfusion, the continuous oozing required tamponade with a FC- SEMS that was removed after one week. No further significant bleeding was observed with subsequent dilations.
Esophago-pharyngeal reflux with nocturnal cough in one patient with gastric pull-up and anastomotic stricture following LAMS placement. Following life-style changes and raising the head-end of the bed, symptoms resolved. Options like injection botulinum toxin into the pyloric sphincter of G-POEM were not required.
Unrelated to the dilations, one patient with surgery for recurrence of cancer developed a retropharyngeal abscess that resolved with drainage and antibiotics.
Discussion
Endoscopic dilation is the primary approach in managing patients with benign esophageal strictures [1, 2]. The goal is to achieve a luminal diameter at ≥ 14 mm [6], although more durable response has been reported with larger diameters [12]. Simple strictures such as webs, rings, or peptic respond well to ≤ 3 ED sessions [3,4,5]. CBES such as those from radiation, corrosives, ESD, pills, or surgery can be refractory despite EDs done at 2-week intervals as defined by Kochman et al. [6] These patients are then characterized as “failure” to dilations and referred to tertiary-care centers for additional measures such as electrocautery-incisions or FC-SEMS that are not without risks and generally not performed in the community. In this study, instead of considering these patients as “failure” to dilations, the authors investigated the clinical outcomes of a protocol where EDs were initially done every week instead of the traditional every 2-weeks. Compared to success being defined as achieving a luminal diameter of ≥ 14 mm, this study also incorporated maintaining this diameter over long-term follow-up in the definition of success.
Using this approach, over 55% of patients who failed prior dilations done at ≥ 2-week intervals achieved long-term success and many of these patients just responded to weekly dilations without the need of stents or electro-cautery-incision. Over a median of 4-year follow-up, these patients continue to remain dysphagia-free with minimal interventions. When this protocol was applied upfront in those with newly diagnosed CBES, the success rate was even higher. The exact reason for this difference is not known. Based on the principles of wound healing as described below, it could be those who were referred after several prior dilation sessions done at ≥ 2-week intervals progressively developed worsening fibrosis as compared to the group where the protocol was applied soon after the diagnosis. Higher success rates were also reported in another study where dilations were performed every week [4]. However, they included some patients with simple strictures and the follow-up was less than 2 years.
Some patients in both groups did not respond to weekly dilations. Hence additional intervention such as FC-SEMS or electro-cautery-incision were needed. There was a trend toward less need for additional interventions in those where the protocol was applied upfront (P = 0.057). Interestingly, when weekly dilation protocol was re-started in a week after additional interventions, a significant proportion of patients in both groups started responding to ED when initially they were not.
The rationale behind the approach of initially starting with weekly dilations and using this approach again immediately after FC-SEMS/electro-cautery was to not lose out on the gains made by waiting too long. Although the exact duration of “waiting too long” has not been defined, we opted for the weekly intervals based on studies looking at wound healing. After the mucosal barrier is broken (such as after dilation), fibrosis with luminal distortion (stricture) sets in by 2 weeks while re-epithelization can take over 5 weeks [9, 10]. Dilating the stricture in a step-up manner every week may be one option to address this as was done in this study. Although delaying fibrosis with steroid injection into the stricture has been tried (including several patients in this study), results have been variable [29,30,31,32]. Since re-epithelization to restore the mucosal barrier can take over 5 weeks [10], another option could have been to rapidly restore the epithelial barrier as has been tried using principles of regenerative medicine such as injecting autologous pluripotent cells into the site of the mucosal break, or using autologous cell-sheets to immediately cover the breached epithelium [33,34,35,36,37,38,39,40,41,42]. Besides animal studies, these techniques have also shown promising results in human beings [38, 42]. As these techniques are expertise driven, expensive, and have regulatory limitations, the authors adopted the frequent dilation approach.
Since patients with CBES can have a variable course over time, studies looking at the response to interventions with short term follow-ups of less than 1–2 years may not represent the long-term outcomes. One of the strengths of this study was the median follow-up of 4 years. There are however several limitations such as there was no control arm. However, as several patients had failed prior dilations done at ≥ 2-weekly intervals, they acted as their own controls. Over 55% of these patients started responding to weekly dilations when previously they had failed to do so. Secondly, no cost analysis was done on frequent EDs. Although the initial cost of weekly dilations was much higher, 74% of patients were dysphagia-free with a mean of 8 ± 4.7 dilations per patient in the group who had failed previous dilations done at ≥ 2-week intervals. This was 6.5 ± 5.5 dilations per patient in the group where this protocol was applied upfront. Hence it is anticipated that over a long-term period (median follow-up of 4 years in this study), this led to significant cost savings, not to also forget a significant improvement of quality of life. Accessibility to GI endoscopy centers for weekly dilations may be an issue based on the area/country of practice. Experience from a very busy tertiary-care center like ours showed that, once patients were accepted for weekly dilations, appointment for the next dilation in a week was already successfully factored into the schedule and the intervals between dilations accordingly adjusted if the patient starts responding.
Whether the frequent dilation approach exposed the patients to higher risks of adverse events was not shown to be the case in this study. There was one dilation related perforation and one bleeding episodes among the 488 EDs performed. Although all EDs were performed by advanced endoscopy faculty, most of the gastroenterologists are trained in ED (core training) and can easily adopt the weekly ED protocol in the community setting hence obviating the need for measure such as electrocautery-incision or FC-SEMS. Lastly, since the number of patients in each group was small, sub-group analysis on the response rates based on etiology was not possible.
In summary, the protocol of initially dilating every week and then increasing the interval between dilation sessions based on response was shown to be safe and effective in the management of patients with CBES who failed dilations done at the traditional 2-week or more intervals. If these results are validated in large prospective randomized studies, it will have great implications on how we manage these patients since weekly EDs can safely be performed in the community as against electro-cautery incision or placing FC-SEMS. Better outcomes were observed in those where this approach was used sooner than later. Those who initially did not respond to weekly dilation and hence needed additional interventions such as FC-SEMS/electro-cautery, re-initiating weekly dilations led to further success. Prospective randomized studies on a larger cohort of patients will be required to do subgroup analysis on the influence of the etiology of the stricture on outcomes.
Abbreviations
- ED:
-
Endoscopic dilation
- CBES:
-
Complex benign esophageal stricture
- RBES:
-
Refractory benign esophageal stricture
- FC-SEMS:
-
Fully covered self-expanding metal esophageal stent
- LAMS:
-
Lumen apposing metal stent
- AE:
-
Adverse events
References
Sami SS et al (2018) UK guidelines on oesophageal dilatation in clinical practice. Gut 67(6):1000–1023
Poincloux L, Rouquette O, Abergel A (2017) Endoscopic treatment of benign esophageal strictures: a literature review. Expert Rev Gastroenterol Hepatol 11(1):53–64
Robles-Medranda C et al (2019) Treating simple benign esophageal strictures with Savary-Gilliard dilators: is the rule of three still necessary? Arq Gastroenterol 56(1):95–98
Pereira-Lima JC et al (1999) Endoscopic dilation of benign esophageal strictures: report on 1043 procedures. Am J Gastroenterol 94(6):1497–1501
Persson ST, Fraser AG, Lane MR (1999) Long-term follow-up of the management of benign oesophageal strictures at Auckland Hospital 1990–1994. N Z Med J 112(1081):28–30
Kochman ML, McClave SA, Boyce HW (2005) The refractory and the recurrent esophageal stricture: a definition. Gastrointest Endosc 62(3):474–475
Vermeulen BD et al (2020) Risk factors and clinical outcomes of endoscopic dilation in benign esophageal strictures: a long-term follow-up study. Gastrointest Endosc 91(5):1058–1066
Badir M et al (2021) Endoscopic dilation of benign post-esophagectomy anastomotic strictures: long-term outcomes and risk of recurrence. Ann Gastroenterol 34(3):337–343
Honda M et al (2010) Process of healing of mucosal defects in the esophagus after endoscopic mucosal resection: histological evaluation in a dog model. Endoscopy 42(12):1092–1095
Kakushima N et al (2006) Histopathologic characteristics of gastric ulcers created by endoscopic submucosal dissection. Endoscopy 38(4):412–415
Mellow MH, Pinkas H (1985) Endoscopic laser therapy for malignancies affecting the esophagus and gastroesophageal junction. Analysis of technical and functional efficacy. Arch Intern Med 145(8):1443–1446
van Halsema EE et al (2017) Endoscopic dilation of benign esophageal anastomotic strictures over 16 mm has a longer lasting effect. Surg Endosc 31(4):1871–1881
Beilstein MC, Kochman ML (2005) Endoscopic incision of a refractory esophageal stricture: novel management with an endoscopic scissors. Gastrointest Endosc 61(4):623–625
Choi J, Choi SI (2021) A new simple endoscopic incision therapy for refractory benign oesophageal anastomotic stricture. BMJ Case Rep 14(3).
Hordijk ML et al (2006) Electrocautery therapy for refractory anastomotic strictures of the esophagus. Gastrointest Endosc 63(1):157–163
Schubert D et al (2003) Endoscopic treatment of benign gastrointestinal anastomotic strictures using argon plasma coagulation in combination with diathermy. Surg Endosc 17(10):1579–1582
Li J et al (2020) Endoscopic incision and selective cutting for primary treatment of benign esophageal anastomotic stricture: outcomes of 5 cases with a minimum follow-up of 12 months. Ann Palliat Med 9(3):1206–1210
Adler DG (2017) Esophageal placement of a lumen-apposing metal stent in a patient with a chronic anastomotic stricture. Gastrointest Endosc 85(6):1291–1293
Irani S et al (2017) Use of a lumen-apposing metal stent to treat GI strictures (with videos). Gastrointest Endosc 85(6):1285–1289
Tandon S et al (2019) Self-expanding esophageal stents for the management of benign refractory esophageal strictures in children: a systematic review and review of outcomes at a single center. J Pediatr Surg 54(12):2479–2486
Lange B, et al (2018) Experience with fully covered self-expandable metal stents for anastomotic stricture following esophageal atresia repair. Dis Esophagus 31(11).
Manfredi MA et al (2014) Externally removable stents in the treatment of benign recalcitrant strictures and esophageal perforations in pediatric patients with esophageal atresia. Gastrointest Endosc 80(2):246–252
Liu J et al (2012) Removable, fully covered, self-expandable metal stents for the treatment of refractory benign esophagogastric anastomotic strictures. Dysphagia 27(2):260–264
Dua KS et al (2008) Removable self-expanding plastic esophageal stent as a continuous, non-permanent dilator in treating refractory benign esophageal strictures: a prospective two-center study. Am J Gastroenterol 103(12):2988–2994
Granata A et al (2020) Endoscopic management of post-surgical GI wall defects with the overstitch endosuturing system: a single-center experience. Surg Endosc 34(9):3805–3817
Dolezel R et al (2018) Fixation of biomaterial to metallic stent and fixation of stents after circular endoscopic dissection in the esophagus on an animal model. Rozhl Chir 97(5):208–213
Dzeletovic I et al (2013) Self-dilation as a treatment for resistant, benign esophageal strictures. Dig Dis Sci 58(11):3218–3223
van Halsema EE et al (2018) Self-dilation for therapy-resistant benign esophageal strictures: towards a systematic approach. Surg Endosc 32(7):3200–3207
Abdelhay S, Mousa M, Elsherbeny MS (2020) Corticosteroid injection of impassable caustic esophageal strictures without dilatation: does it pave the way to interval endoscopic dilatation? J Pediatr Surg 55(11):2348–2351
Dasari CS et al (2020) Intralesional steroids and endoscopic dilation for anastomotic strictures after esophagectomy: systematic review and meta-analysis. Endoscopy 52(9):721–726
Henskens N, Wauters L, Vanuytsel T (2020) Intralesional steroid injections in addition to endoscopic dilation in benign refractory esophageal strictures : a systematic review. Acta Gastroenterol Belg 83(3):432–440
Nie D, Yan X, Huang Y (2020) Efficacy of hydrocortisone sodium succinate and aluminum phosphate gel for stricture prevention after >/=3/4 circumferential endoscopic submucosal dissection. J Int Med Res 48(4):300060519894122
Honda M et al (2011) Use of adipose tissue-derived stromal cells for prevention of esophageal stricture after circumferential EMR in a canine model. Gastrointest Endosc 73(4):777–784
Sakurai T et al (2007) Autologous buccal keratinocyte implantation for the prevention of stenosis after EMR of the esophagus. Gastrointest Endosc 66(1):167–173
Ohki T et al (2006) Treatment of oesophageal ulcerations using endoscopic transplantation of tissue-engineered autologous oral mucosal epithelial cell sheets in a canine model. Gut 55(12):1704–1710
Kanai N et al (2012) Fabricated autologous epidermal cell sheets for the prevention of esophageal stricture after circumferential ESD in a porcine model. Gastrointest Endosc 76(4):873–881
Takagi R et al (2012) Cell sheet technology for regeneration of esophageal mucosa. World J Gastroenterol 18(37):5145–5150
Ohki T, et al (2012) Prevention of esophageal stricture after endoscopic submucosal dissection using tissue-engineered cell sheets. Gastroenterology 143(3):582–588 e2.
Sasaki R et al (2012) Punch and spindle-shaped biopsies for collecting oral mucosal tissue for the fabrication of transplantable autologous epithelial cell sheets. J Biomed Mater Res A 100(10):2849–2854
Liu Y et al (2021) Autologous esophageal mucosa with polyglycolic acid transplantation and temporary stent implantation can prevent stenosis after circumferential endoscopic submucosal dissection. Ann Transl Med 9(7):546
Ohki T, Yamamoto M (2020) Esophageal regenerative therapy using cell sheet technology. Regen Ther 13:8–17
Dua KS, Sasikala M (2018) Repairing the human esophagus with tissue engineering. Gastrointest Endosc 88(4):579–588
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Sowmya Palam, Mathew Mohorek, Syed Rizvi and Kulwinder Dua have declare no conflict of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (WMV 23823 kb)
Rights and permissions
About this article
Cite this article
Palam, S., Mohorek, M., Rizvi, S. et al. Clinical outcomes on weekly endoscopic dilations as the initial approach to manage patients with complex benign esophageal strictures: report on 488 dilations. Surg Endosc 36, 7056–7065 (2022). https://doi.org/10.1007/s00464-022-09248-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-022-09248-0