Abstract
Background
In the field of rectal cancer surgery, there remains ongoing debate on the merits of high ligation (HL) and low ligation (LL) of the inferior mesenteric artery (IMA) in terms of perfusion and anastomosis leakage. Recently, infrared fluorescence of indocyanine green (ICG) imaging has been used to evaluate perfusion status during colorectal surgery.
Objective
The purpose of this study is to compare the changes in perfusion status between HL and LL through quantitative evaluation of ICG.
Methods
Patients with rectosigmoid or rectal cancer were randomized into a high or LL group. ICG was injected before and after IMA ligation, and region of interest (ROI) values were measured by an image analysis program (HSL video©).
Results
From February to July 2020, 22 patients were enrolled, and 11 patients were assigned to each group. Basic demographics were similar between the two groups, except for albumin level and cardiac ejection fraction. There were no significant differences in F_max between the two groups, but T_max was significantly higher and Slope_max was significantly lower in the HL group than in the LL group. Anastomosis leakage was significantly associated with neoadjuvant chemoradiation and F_max.
Conclusion
After IMA ligation, T_max increased and Slope_max decreased significantly in the HL group. However, the intensity of perfusion status (F_max) did not change according to the level of IMA ligation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
There is controversy over the appropriate level of ligation on the inferior mesenteric artery (IMA) in rectal cancer surgery. From the oncological view, high ligation (HL) could remove lymph nodes (LN) around the IMA and is considered a more oncologically favorable surgery, but there have been very few studies that actually support favorable oncological outcomes with HL [1,2,3,4].
Aside from the oncological view, surgeons who prefer low ligation (LL) claim that HL could reduce perfusion at the distal colon and increase the risk of anastomosis leakage (AL) [2, 5, 6]. Although several studies reported no association between HL and anastomosis leakage, the risk of anastomosis leakage depending on the ligation level of IMA remains controversial [1, 3, 4, 6].
AL is one of the worst complications after colorectal cancer surgery. AL causes high morbidity and mortality after surgery and increases the duration of hospital stay and medical cost [7]. Several studies have reported that checking colonic perfusion status using fluorescent angiography with indocyanine green (ICG) could lower AL [8,9,10,11]. Recently, several studies performed a quantitative analysis of ICG fluorescent imaging and reported its association with the risk of anastomosis leakage [12, 13]. However, no study has evaluated perfusion status for HL and LL using quantitative analysis of ICG. In the present study, the authors compare the colonic perfusion status between the HL and LL groups by quantitative analysis of the near infrared (NIR)-ICG fluorescence perfusion test.
Materials and method
Patients and data collection
Patients who underwent surgery for rectosigmoid or rectal cancer from February to July 2020 at Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, were enrolled in this study. Patients with metastasis, local excision, abdominoperineal operation, Hartmann's operation, or intersphincteric resection were excluded. The patients were randomized into two groups, HL and LL, using a web random number generator. The patients’ demographics, clinical characteristics, and hemodynamic data were collected. This study (XC19MEDT0088K) was approved by the Institutional Review Board of the Ethics Committee of the College of Medicine, The Catholic University of Korea. All patient records were anonymized and de-identified prior to analysis.
Surgical technique
High ligation (HL)
The aorto-mesenteric window was opened wide, and the inferior mesenteric vessels were exposed. After peeling to the point where the origin of the IMA was visible, the ligation was performed at about 1 cm from the root, while the left colic artery and the inferior mesenteric vein were ligated near the lower border of the pancreas (Fig. 1_A).
Drawing of the surgical procedure of inferior mesenteric artery (IMA) ligation and perfusion check of the proximal end of the colon compared to the small bowel using indocyanine green (ICG). A High ligation, ligation of the inferior mesenteric artery at its origin with left colic artery ligation; B Low ligation, ligation of the superior rectal artery just distal to the left colic artery; ★, Region of interest (ROI) for quantitative analysis of colon and small bowel perfusion
Low ligation (LL)
The aorto-mesenteric window was opened wide, and the inferior mesenteric vessels were exposed. Apical lymph node dissection was performed around the IMA origin, and the left colic artery was identified and preserved, while the superior rectal artery was ligated (Fig. 1_B).
Perfusion test protocol using indocyanine green
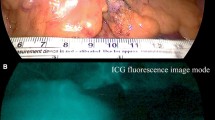
During the surgery, ICG angiography was performed three times with a fluorescence imaging system in the laparoscopic surgery (Viscera 4 K UHD system, Olympus®, Japan) and the robotic surgery (da Vinci Xi, Intuitive®, USA). ICG (1 vial, 25 mg, Dongindang®, Indocyanine Green Injection, Korea) was diluted in 10 ml of distilled normal saline, and 7.5 mg of ICG was injected into the peripheral blood vessels followed by 20 cc normal saline pushing at each injection. The first ICG dose (7.5 mg) was injected just after the creation of a pneumoperitoneum to check the baseline perfusion status of the colon and small bowel. After the ICG injection, a fluorescent image was taken. The camera was fixed for 2 min until the perfused ICG disappeared. Blood pressure and pulse rate were checked at the time of ICG injection. After checking baseline perfusion status, surgery was performed, followed by IMA ligation (low or high), mesocolon dissection, and rectal resection. After rectal resection, the surgeon placed the distal part of the resected rectum and the small bowel close to each other (Figs. 1 and 2), and the second ICG (7.5 mg) was then injected. The ICG image of both the colon and small bowel was taken with similar fashion. Then, the specimen was extracted and resected after checking perfusion status by ICG again (the third ICG injection, 7.5 mg). All surgical procedures were recorded for quantitative video analysis.
Difference in perfusion time and intensity between the small bowel and proximal limb of the colon by quantitative analysis using ICG, after ligation of the IMA and transection of the distal rectum. A–D Fluorescent intestinal perfusion images taken in chronological order. E Quantitative fluorescent analysis. The purple and green lines are the fluorescent perfusion graphs of the colon and small bowel in the above fluorescent image
Quantitative analysis of colonic perfusion
Recorded colonic fluorescence videos were changed into fluorescence intensity and measured sequentially to produce colonic perfusion graphs using a video analysis and modeling tool (HSL Video Analyzer®, Dr. Park’s software lab, Korea). To assess the colonic perfusion quantitatively, fluorescence intensity factors and perfusion time factors were calculated from each ICG fluorescence graph (Fig. 3). The fluorescence intensity factors related to arterial perfusion were fluorescence intensity at baseline (F_min), fluorescence difference between maximum and baseline intensity (F_max), and the maximal fluorescence slope (Slope_max = F_max/T_max). The fluorescence intensity factors related to venous perfusion were fluorescence difference between maximum and intensity at a sharp drop and then a gentle decrease (F_plateau) and the decreased fluorescence slope (Slope_plateau = F_plateau/T_plateau). The perfusion time factors were the time from first fluorescence increase to maximum (T_max = ΔT), time from first fluorescence increase to half of maximum (T_1/2max), and the time ratio (TR = T_1/2max/T_max). The ratio of the slope at the start of perfusion and the slope until reaching the maximum state was defined as the slope ratio (SR = Slope_1/2max / Slope_max).
Statistical analysis
The categorical variables of the groups were compared using the chi-square or Fisher’s exact test. Chi-square and Pearson correlation tests were used to evaluate the correlation between clinical factors and anastomotic complications. We used the two-sided independent samples Mann–Whitney test or T test for continuous variables of perfusion factors to identify risk factors of anastomotic complications. Wilcoxon’s signed rank test was used to compare between the baseline and the after-ligation imaging analysis. All statistical analyses were performed using SPSS for Windows software (ver. 24.0; IBM Corp., Armonk, NY, USA). P values < 0.05 were considered significant.
Results
Baseline characteristics between high or low ligation
A total of 22 patients were enrolled in this pilot study, 11 each in the HL and LL groups. The average patient age was 69.7 years old, and there were 15 males (68.2%). There were 9 cases (40.9%) of rectal cancer and 13 cases (59.1%) of recto-sigmoid (RS) colon cancer. Table 1 shows the clinical characteristics and hemodynamic parameters of the two groups. There were no significant differences in clinical features between the two groups except for albumin and cardiac ejection fraction (EF). In each group, 1 case of anastomosis leakage occurred within 30 days.
Quantitative analysis of ICG perfusion before and after IMA ligation
Table 2 shows changes in the ICG perfusion intensity of the colon and small bowel after IMA ligation through quantitative analysis. Latency in the colon was increased after ligation but not significantly different (13.8 ± 11.2 s vs. 15.7 ± 8.2 s, P = 0.122). The F_max, F_plateau, and ratio of time (TR) of the small bowel showed significant differences at baseline and after IMA ligation, but this finding demonstrated the possibility of biases such as distance between target and camera during the measurement of ICG perfusion at baseline and after IMA ligation. Therefore, when comparing the ratio of colon to small bowel, significant increase of T_max (1.15 vs. 2.34, P = 0.002) and decrease of Slope_max (0.77 vs. 0.49, P = 0.002) showed after IMA ligation, but F_plateau showed no significant difference.
Table 3 shows the changes in ratio of colon to small bowel perfusion depending on the level of IMA ligation. F_max of the ratio of colon to small bowel also decreased after IMA ligation in both groups, but neither decrease was significantly different (HL: 0.81 vs. 0.76, P = 0.859; LL: 0.85 vs. 0.80, P = 0.722). The ratio of colon perfusion after ligation showed significant differences in T_max and Slope_max in the HL group but no significant differences in the LL group.
Table 4 shows a comparison of the perfusion ratio of colon to small bowel after IMA ligation between HL and LL. After LL, Slope_max was faster and T_max was shorter compared to the HL group but showed no significant differences. The trend for F_max was similar (0.76 ± 0.27 vs. 0.80 ± 0.26, P = 0.768).
Quantitative analysis of ICG perfusion depending on the presence of anastomosis leakage
Table 5 shows the relationship between anastomosis leakage and quantitative parameters. Two patients had anastomosis leakage (AL), while 20 patients had no event on anastomosis. Statistical difference was only shown in neoadjuvant chemoradiotherapy in terms of anastomosis leakage (P = 0.013). There was no association between level of ligation and anastomosis leakage. Among the quantitative ICG perfusion parameters, F_max had the only significant association with anastomosis leakage (0.82 ± 0.23 vs. 0.36 ± 0.22, P = 0.012).
Discussion
There have been over one hundred years of controversy regarding the appropriate level of ligation of the IMA since reports from Miles and Moynihan in 1908 [14]. Practically, there are three debating points in terms of the level of IMA ligation. The first point is an oncological issue. There were no differences in long-term oncological outcomes [1,2,3,4, 6]. The second point is functional outcome. Mary GM et al. reported that 103 patients with LL had fewer symptoms of urinary and sexual dysfunction than 111 patients with HL [15]. The last issue, AL, remains debated. There have been several reports of lower rate of anastomosis leakage in LL due to preservation of the left colic artery [2, 3, 6]. On the other hand, several recent studies reported that there was no statistically significant difference in AL rates between the LL and HL groups [1, 4, 15].
Since the 2000s, several studies have reported that the evaluation of intestinal perfusion using ICG could reduce the rate of anastomosis leakage in colon surgery [8]. Recently, quantitative analysis methods objectify fluorescent images analyzing several parameters such as F_max, T_max, and Slope_max [12, 13, 16]. The interpretation of various parameters can be broadly divided into two categories. It can be largely divided into inflow parameter and intensity parameter [17]. The inflow parameter has factors that indicate how well and how quickly it arrives in the colon. For example, there are factors such as T_max, T_1/2max, Slope_max, and TR. And the representative of the intensity parameter is F_max, which is a factor that intuitively measures how much colonic perfusion is. Wada et al. [13] reported that both F_max and Slope_max were significantly lower in the anastomosis leakage group. In the analysis by Son et al., an additional time ratio (TR) was defined and used for analysis.12 In Son’s study, a slow TR of 0.6 or more was reported as a significant anastomosis leakage factor. However, in this study, lower F_max and CRT were observed as significant AL factors. There are not many studies that have performed quantitative analysis yet, and existing studies are also heterogenous, so it is difficult to find a consensus on which parameter can be used as a representative perfusion factor. A randomized trial is required to raise the level of evidence and achieve consensus.
In our study, we introduced several unique parameters compared to previous studies. First, we introduced the Slope_plateau, which was related to venous return of the colon. Second, we introduced the ratio of colon to small bowel as a standardization. When measuring ICG, there will always be bias caused by the environment at the time of measurement. For example, there will be a difference in the brightness of the ICG depending on the distance between the specimen and the camera, the type of camera, and the type of light emission. This kind of bias has been pointed out as a limitation of ICG perfusion measurement so far. However, by checking the ICG of the colon together with the adjacent small bowel, we could account for this bias. Furthermore, since the maximal perfusion of each patient is different, the perfusion of the small bowel can be considered as a control perfusion status for the patient, and the ratio of colon to small bowel could get rid of such bias and could compare the standardized colon perfusion values of each patient because small bowel perfusion is not affected by IMA ligation.
In the present study, after IMA ligation, T_max and Slope_max showed significant differences in the HL group but not in the LL group, while F_max showed no difference between pre- and post-IMA ligation in either group. After HL, the time for perfusion could be delayed because blood was supplied from the right side, but the total intensity and amount of perfusion were not different depending on the level of IMA ligation.
Many factors have been reported to be associated with AL [18]. In the present study, 2 cases of anastomosis leakage occurred, and preoperative radiation and F_max were observed to be significant factors associated with AL. Other ICG quantitative parameters including latency, T-max, Slope_max, and TR, which were reported to be risk factors of AL by previous studies, were not associated with AL in this study.
Several meta-analyses using recent randomized control trials reported that there was no difference in the rate of anastomosis leakage between the HL and LL groups [1], but there had been no clear explanations to support those results. This study evaluated the perfusion status between the HL and LL groups using quantitative ICG analysis and proved that perfusion intensity (F_max) between HL and LL was not different. As far as we know, this is the first study to evaluate quantitative ICG between HL and LL in rectal cancer. In addition, we introduced unique parameters that could overcome various biases pointed out as limitations of quantitative ICG analysis. The standardized parameter suggested in this study can be used as a new quantitative parameter that allows for more objective perfusion evaluation and AL predicting models by removing the limitations of previous quantification studies, thereby enabling multi-center studies and large-scale studies. Although this study had a very small sample size as a pilot study and did not suggest specific cut-off values that can predict anastomosis leakage, we think that results of this research will be able to suggest values that can predict AL and more object colonic perfusion results according to ligation by conducting additional multi-center studies on a large number of subjects.
Conclusion
T_max and Slope_max decreased after ligation of the IMA, especially in the HL group, but F_max, which indicates the intensity of perfusion, showed no significant difference between the HL and LL groups. Based on this study, the speed of blood perfusion could be more delayed after HL than LL, but the intensity of perfusion was similar between HL and LL.
References
Hajibandeh S, Hajibandeh S, Maw A (2020) Meta-analysis and trial sequential analysis of randomized controlled trials comparing high and low ligation of the inferior mesenteric artery in rectal cancer surgery. Dis Colon Rectum 63:988–999
Si MB, Yan PJ, Du ZY, Li LY, Tian HW, Jiang WJ, Jing WT, Yang J, Han CW, Shi XE, Yang KH, Guo TK (2019) Lymph node yield, survival benefit, and safety of high and low ligation of the inferior mesenteric artery in colorectal cancer surgery: a systematic review and meta-analysis. Int J Colorectal Dis 34:947–962
Yang Y, Wang G, He J, Zhang J, Xi J, Wang F (2018) High tie versus low tie of the inferior mesenteric artery in colorectal cancer: a meta-analysis. Int J Surg 52:20–24
Fujii S, Ishibe A, Ota M, Suwa H, Watanabe J, Kunisaki C, Endo I (2019) Short-term and long-term results of a randomized study comparing high tie and low tie inferior mesenteric artery ligation in laparoscopic rectal anterior resection: subanalysis of the HTLT (High tie vs. low tie) study. Surg Endosc 33:1100–1110
Nayeri M, Iskander O, Tabchouri N, Artus A, Michot N, Muller O, Giger-Pabst U, Bourlier P, Kraemer-Bucur A, Lecomte T, Salame E, Ouaissi M (2019) Low tie compared to high tie vascular ligation of the inferior mesenteric artery in rectal cancer surgery decreases postoperative complications without affecting overall survival. Anticancer Res 39:4363–4370
Zeng J, Su G (2018) High ligation of the inferior mesenteric artery during sigmoid colon and rectal cancer surgery increases the risk of anastomotic leakage: a meta-analysis. World J Surg Oncol. https://doi.org/10.1186/s12957-018-1458-7
Kingham TP, Pachter HL (2009) Colonic anastomotic leak: risk factors, diagnosis, and treatment. J Am Coll Surg 208:269–278
Blanco-Colino R, Espin-Basany E (2018) Intraoperative use of ICG fluorescence imaging to reduce the risk of anastomotic leakage in colorectal surgery: a systematic review and meta-analysis. Tech Coloproctol 22:15–23
Degett TH, Andersen HS, Gogenur I (2016) Indocyanine green fluorescence angiography for intraoperative assessment of gastrointestinal anastomotic perfusion: a systematic review of clinical trials. Langenbecks Arch Surg 401:767–775
Jafari MD, Wexner SD, Martz JE, McLemore EC, Margolin DA, Sherwinter DA, Lee SW, Senagore AJ, Phelan MJ, Stamos MJ (2015) Perfusion assessment in laparoscopic left-sided/anterior resection (PILLAR II): a multi-institutional study. J Am Coll Surg 220:82–92
Ris F, Liot E, Buchs NC, Kraus R, Ismael G, Belfontali V, Douissard J, Cunningham C, Lindsey I, Guy R, Jones O, George B, Morel P, Mortensen NJ, Hompes R, Cahill RA, Near-Infrared Anastomotic Perfusion Assessment Network V (2018) Multicentre phase II trial of near-infrared imaging in elective colorectal surgery. Br J Surg 105:1359–1367
Son GM, Kwon MS, Kim Y, Kim J, Kim SH, Lee JW (2019) Quantitative analysis of colon perfusion pattern using indocyanine green (ICG) angiography in laparoscopic colorectal surgery. Surg Endosc 33:1640–1649
Wada T, Kawada K, Takahashi R, Yoshitomi M, Hida K, Hasegawa S, Sakai Y (2017) ICG fluorescence imaging for quantitative evaluation of colonic perfusion in laparoscopic colorectal surgery. Surg Endosc 31:4184–4193
Miles WE (1971) A method of performing abdomino-perineal excision for carcinoma of the rectum and of the terminal portion of the pelvic colon (1908). CA Cancer J Clin 21:361–364
Mari GM, Crippa J, Cocozza E, Berselli M, Livraghi L, Carzaniga P, Valenti F, Roscio F, Ferrari G, Mazzola M, Magistro C, Origi M, Forgione A, Zuliani W, Scandroglio I, Pugliese R, Costanzi ATM, Maggioni D (2019) Low ligation of inferior mesenteric artery in laparoscopic anterior resection for rectal cancer reduces genitourinary dysfunction: results from a randomized controlled trial (HIGHLOW Trial). Ann Surg 269:1018–1024
Diana M, Agnus V, Halvax P, Liu YY, Dallemagne B, Schlagowski AI, Geny B, Diemunsch P, Lindner V, Marescaux J (2015) Intraoperative fluorescence-based enhanced reality laparoscopic real-time imaging to assess bowel perfusion at the anastomotic site in an experimental model. Br J Surg 102:e169-176
Lutken CD, Achiam MP, Svendsen MB, Boni L, Nerup N (2020) Optimizing quantitative fluorescence angiography for visceral perfusion assessment. Surg Endosc 34:5223–5233
Park JS, Choi GS, Kim SH, Kim HR, Kim NK, Lee KY, Kang SB, Kim JY, Lee KY, Kim BC, Bae BN, Son GM, Lee SI, Kang H (2013) Multicenter analysis of risk factors for anastomotic leakage after laparoscopic rectal cancer excision: the Korean laparoscopic colorectal surgery study group. Ann Surg 257:665–671
Acknowledgements
I would like to express my gratitude to Hae-Won Shin who helped illustration and Keunheung Park who helped with the ICG quantification program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Drs. Seung-Rim Han, Chul Seung Lee, Jung Hoon Bae, Hyo Jin Lee, Mi Ran Yoon, Abdullah Al-Sawat, Do Sang Lee, In Kyu Lee, and Yoon Suk Lee have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Han, SR., Lee, C.S., Bae, J.H. et al. Quantitative evaluation of colon perfusion after high versus low ligation in rectal surgery by indocyanine green: a pilot study. Surg Endosc 36, 3511–3519 (2022). https://doi.org/10.1007/s00464-021-08673-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-021-08673-x