Abstract
Background
As the use of minimally invasive techniques in colorectal surgery has become increasingly prevalent, concerns remain about the oncologic effectiveness and long-term outcomes of minimally invasive low anterior resection (MI-LAR) for the treatment of rectal cancer.
Study design
The 2010–2015 National Cancer Database (NCDB) Participant Data Use File was queried for patients undergoing elective open LAR (OLAR) or MI-LAR for rectal adenocarcinoma. A 1:1 propensity match was performed on the basis of demographics, comorbidity, and tumor characteristics. Outcomes were compared between groups and Cox proportional hazard modeling was performed to identify independent predictors of mortality. A subset analysis was performed on high-volume academic centers.
Results
35,809 patients undergoing LAR were identified of whom 18,265 (51.0%) underwent MI-LAR. After propensity matching, patients receiving MI-LAR were less likely to have a positive circumferential radial margin (CRM) (5.5% vs. 6.6%, p = 0.0094) or a positive distal margin (3.6% vs. 4.6%, p = 0.0022) and had decreased 90-day all-cause mortality (2.0% vs. 2.6%, p = 0.0238). MI-LAR resulted in decreased hospital length of stay (5 vs. 6 days, p < 0.0001) but a greater rate of 30-day readmission (7.6% vs. 6.5%, p = 0.0054). Long-term overall survival was improved with MI-LAR (79% vs. 76%, p < 0.0001). Cox proportional hazard modeling demonstrated a decreased risk of mortality with MI-LAR (HR 0.859, 95% CI 0.788–0.937).
Conclusion
MI-LAR is associated with improvement in CRM clearance and long-term survival. In the hands of experienced surgeons with advanced laparoscopy skills, MI-LAR appears safe and effective technique for the management of rectal cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Colorectal cancer, the third most common cancer worldwide, remains a major cause of morbidity and mortality. It is estimated that colorectal cancer is responsible for 50,000 deaths per year in the United States [1]. Coordination of multidisciplinary care and advancements in chemotherapy and radiation therapy have significantly improved outcomes in the past 40 years, however, surgical resection of the primary tumor remains the cornerstone of curative treatment. In recent years, the use of minimally invasive surgical (MIS) approaches, including both laparoscopic and robotic techniques, has gained traction for the management of colon cancer due to accumulating evidence demonstrating improved short-term outcomes and oncologic equivalency [2, 3]. In comparison, the adoption of laparoscopic approaches in the management of rectal cancer has occurred at a slower pace due to concerns for the increased difficulty of pelvic dissection and thus the ability to perform a total mesorectal excision (TME) equivalent to open procedures [4, 5].
The ACOSOG Z6051 trial raised concerns about the oncologic efficacy of laparoscopic proctectomy for patients with stage II/III rectal cancer as laparoscopic rectal resection failed to demonstrate non-inferiority compared to open proctectomy [6]. Z6051 utilized a composite pathologic endpoint and, although these findings raised concerns about long-term survival the trial did not report survival data. Several additional studies have compared minimally invasive (defined as either laparoscopic or robotic) low anterior resection (MI-LAR) and open LAR (OLAR), however, concerns over oncologic efficacy and long-term outcomes between these two approaches remain incompletely understood [6,7,8,9,10].
As colorectal surgeons continue to fine tune their laparoscopic and robotic skillsets, it can be reasonably expected that they will become increasingly facile with more complicated operations, including MI-LAR, resulting in improved outcomes. The goal of this study was to utilize a large, contemporary national database to expand on previous analyses, conducting the largest study to date and using the most recent data to examine differences in both oncologic outcomes and long-term follow-up between MI-LAR and OLAR.
Methods
Data source
The National Cancer Data Base (NCDB) is prospectively maintained by the American College of Surgeons’ Commission on Cancer (CoC) and the American Cancer Society [11]. The NCDB gathers data from more than 1500 CoC-accredited centers and is estimated to capture approximately 70% of all newly diagnosed cases of cancer in the United States and Puerto Rico. It now contains upwards of 30 million patient records.
Study design
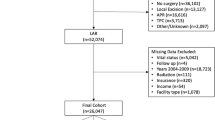
The Duke University Institutional Review Board granted approval for this retrospective analysis. The NCDB was queried for all adult patients diagnosed with rectal adenocarcinoma from 2010 to 2015 who underwent LAR. Patients undergoing LAR were selected using Surgery Procedure of the Primary Site codes 30 and 40, as designated by the Facility Oncology Registry Data Standards criteria. Any patients with non-malignant pathology or preoperatively identified metastatic disease were excluded. Patients who underwent conversion to an open procedure were included in the MI-LAR cohort. The primary endpoint was overall survival. Secondary endpoints included: adequacy of lymph node harvest (defined as greater 12 or more lymph nodes harvested in the dataset), hospital length of stay (LOS), distal margin positivity, circumferential radial margin (CRM) positivity, 30-day readmission, and 90-day mortality.
Statistical analysis
Patients were stratified by surgical approach as MI-LAR, which includes both laparoscopic and robotic approaches, or open LAR (OLAR). Baseline characteristics and unadjusted outcomes were compared using the Kruskal–Wallis test for continuous variables and Pearson χ2 test for categorical variables. After stratification by surgical approach a 1:1 propensity match was performed using a nearest neighbor algorithm. For long-term survival a multivariable Cox proportional hazards model was constructed adjusting for patient age, gender, race, insurance status, comorbidities, clinical tumor stage, hospital type and volume, use of neoadjuvant therapy, and use of adjuvant therapy.
Sensitivity analysis
MI-LAR is more likely to be performed at academic and high-volume centers. This could potentially result in overestimation of outcomes from MI-LAR. Thus, a subset analysis of matched patients treated at high-volume academic medical centers was performed. High-volume hospitals were defined as those in the top tertile of annual hospital case volume. Designation of academic hospital was provided in the dataset. Surgical and oncologic outcomes were compared between MI-LAR and OLAR. Overall survival was examined using the Kaplan Meier method and Cox Proportional Hazards modeling.
Subset analyses
The risk of several outcomes in this study, including margin positivity or nodal spread, is low in patients with T0 or Tis tumors. We performed a subset analysis excluding patients with T0 and Tis tumors (by either clinical or pathological staging) to provide a more representative sample of patients.
Although robotic and laparoscopic approaches are both considered to be minimally invasive approaches these require distinct skillsets, different advanced training, and different patient selection. In order to define the difference between these two approaches to MI-LAR a subset analysis was performed comparing individuals who underwent Robotic LAR (R-LAR) and Laparoscopic LAR (L-LAR).
Model diagnostics were assessed. A p value of less than 0.05 was considered statistically significant. Statistical analysis was performed using SAS 9.4 (SAS Institute, Cary, North Carolina).
Results
A total of 35,809 patients were included in this study, of whom 18,265 (51%) underwent MI-LAR. Baseline demographics and unadjusted outcomes are presented in Table 1. Patients who underwent MI-LAR were younger (61 vs. 63 years, p < 0.0001), less likely to be black (7.0% vs. 8.7%, p < 0.0001), and more likely to have private insurance (53.0% vs. 44.6%), and more often from a higher annual income area (63.3% vs. 55.7%, p < 0.0001) and/or higher education (60.0% vs. 53.6%, p < 0.0001). Patients undergoing MI-LAR were less comorbid (76.2% with Charlson–Deyo score of 0 vs. 73.8%, p < 0.0001), more likely to be treated in high-volume (38.9% vs. 27.4%, p < 0.0001) or academic (37.3% vs. 33.2%, p < 0.0001) medical centers, and more likely to be treated in the northeast or west compared with the south (p < 0.0001). Use of MI-LAR increased throughout the study period (10.8% in 2010 vs. 22.5% in 2015). MI-LAR less frequently resulted in an inadequate lymph node harvest (21.2% vs. 24.9%, p < 0.0001), positive distal margin (3.9% vs. 5.8%, p < 0.0001), positive CRM (5.6% vs. 7.7%, p < 0.0001), and increased rates of R0 resection (97.6% vs. 96.7%, p < 0.0001). Patients undergoing MI-LAR had a shorter duration of hospital stay (5 vs. 6 days, p < 0.0001) with no difference in 30-day readmission rates (7.0% vs. 6.8%, p = 0.3685), and decreased 90-day mortality (2.0% vs. 3.1%, p < 0.0001).
Propensity-matched analysis
Following propensity matching a total of 16,708 patients were available for analysis. Matched demographics and outcomes are presented in Table 2. Matched groups demonstrated no difference in demographics or tumor characteristics, with the exception of year of diagnosis, and the use of MI-LAR increasing over the study period. Patients who underwent MI-LAR were less likely to have positive distal margins (3.6% vs. 4.6%, p = 0.0022) or CRM (5.5% vs. 6.6%, p = 0.0094). MI-LAR was associated with decreased length of hospital stay (5 vs. 6 days, p < 0.0001) and reduced 90-day mortality (2.0% vs. 2.6%, p = 0.0238), but increased 30-day readmission (7.6% vs. 6.5%, p = 0.0054).
Matched survival analysis
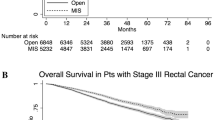
Median follow-up time was 53 months (range 1–156 months). MI-LAR was associated with improved long-term overall survival over OLAR (Fig. 1). At 5-years, overall survival was 79% among patients undergoing MI-LAR vs. 76% among patients undergoing OLAR. After adjustment, MI-LAR was associated with a decreased risk of long-term mortality (HR 0.859, 95% CI 0.788–0.937, p = 0.0006) (Table 3).
Subgroup analysis of matched patients treated at high-volume academic hospitals
The first subgroup analysis included 4028 matched patients treated in high-volume academic centers. There were no clinically significant differences in baseline demographics, with the exception being year of diagnosis (Table 4). MI-LAR was associated with shorter length of hospital stay (5 vs. 6 days, p < 0.0001) but there were no statistically significant differences in other outcomes, including rates of positive margins, adequacy of lymph node harvest, and 90-day mortality. After adjustment, use of MI-LAR was not associated with any difference in long-term survival (HR 0.849, 95% CI 0.678–1.063, p = 0.154) (Table 5).
Subgroup analysis of matched patients excluding T0 and Tis tumors
The second subgroup analysis included 17,094 matched patients excluding patients with either clinical or pathological T0 and Tis tumors. There were no clinically significant differences in baseline demographics, with the exceptions of year of diagnosis and attainment of higher education in patients undergoing MI-LAR (Table 6). MI-LAR was associated with decreased rates of positive distal margin (4.2% vs 5.3%, p = 0.0006), CRM (6.0% vs 7.0%, p = 0.0118), and shorter length of hospital stay (5 vs. 6 days, p < 0.0001). MI-LAR was also associated with improved 90-day mortality (2.0% vs 2.6%, p = 0,0376) and 5-year survival (78% vs 74%, p < 0.0001).
Subgroup analysis of matched patients treated at high-volume academic hospitals
The final subgroup analysis stratified 16.006 patients by either laparoscopic (n = 11,583) or robotic approach (n = 4.423). Patients undergoing Robotic LAR (R-LAR) were more likely to be male, younger than 65, have private insurance, fewer comorbidities, treated at academic medical centers, treated at centers with higher volumes, and diagnosed more recently. R-LAR was also associated with higher clinical but not pathological disease stage and with neoadjuvant treatment (Table 7). Fewer patients undergoing R-LAR required conversion to an open procedure (8.0% vs. 14.6%, p < 0.0001), however, there were no differences in inadequate lymph node harvest (19.0% vs. 20.4%, p = 0.0537), positive distal margin (4.0% vs. 4.4%, p = 0.2390) or positive CRM (5.8% vs. 6.1%, p = 0.4979). R-LAR was associated with decreased use of adjuvant treatment (7.1% vs. 9.7%, p < 0.0001) but improved 30-day (0.8% vs. 1.2%, p = 0.0499) and 90-day mortality (1.5% vs. 2.3%, p = 0.0055) and, however, by 5 years the mortality of each approach was 78%.
Discussion
This national analysis of more than 35,000 patients is the largest study to date to examine the differences between surgical approaches in the management of rectal cancer. We found that MI-LAR was associated with clinically and statistically significant improvements in short-term oncologic outcomes including adequacy of lymph node harvest and rates of negative circumferential radial and distal margins even when excluding patients with low-stage tumors. This study also provides long-term follow-up which demonstrates that MI-LAR was associated with improved survival. Importantly, we were able to demonstrate these improved outcomes despite including patients who required conversion to open procedures, which would be expected to blunt the size of these effects.
Although the role of MIS approaches to proctectomy has been an area of investigation for two decades, the existing evidence is limited and the influence of approach on long-term mortality remains unclear. The United Kingdom Medical Research Council’s CLASICC trial (1996–2002) was the first prospective multi-center randomized control trial to evaluate the use of laparoscopy in the treatment of rectal cancer. Of the 794 patients with either colon or rectal cancer randomized to receive laparoscopic or open surgery, only 246 patients underwent LAR for rectal cancer and 31% of MI-LAR cases required conversion to open leaving only 112 cases in the MI-LAR cohort. Within this limited cohort, a twofold increase in the incidence of a positive CRM (12% vs. 6%) was observed in patients undergoing MI-LAR, this was not statistically significant and, on long-term follow-up there has been no difference in disease-free or overall survival between treatment groups for patients with rectal cancer [12,13,14]. Subsequently the COREAN (2006–2009) and COLOR II (2004–2010) trials detected no significant differences between surgical approaches in the rates of CRM positivity, locoregional recurrence, disease-free survival, or overall survival at 3 years [8, 9, 15].
The similar outcomes between approaches seen in these early trials left the decision of MI-LAR or OLAR to the discretion of individual surgeons. However, two subsequent non-inferiority trials failed to demonstrate non-inferiority of laparoscopic proctectomy. In the ALaCaRT trial, a total of 475 patients underwent either laparoscopic or open rectal resection [10]. The rate of positive CRM was 7% in the laparoscopic group compared to 3% in the open surgical group. Similarly, the ACOSOG Z6051, a North American trial of 462 individuals undergoing either laparoscopic or open rectal resection between 2008 and 2013, failed to demonstrate non-inferiority utilizing a composite endpoint of distal margin positivity, circumferential margin positivity, and completeness of TME [6]. Although these trials found similar short-term outcomes between surgical approaches, these pathological findings raised concerns about the oncologic equivalency between groups; however, neither trial examined long-term outcomes which substantially limits the clinical significance of their findings. The use of a composite endpoint for oncologic adequacy in Z6051 further limits the interpretation and application of these findings.
Despite the evidence provided by these RCTs, the impact of surgical approach on long-term survival remains extremely limited. Moreover, concerns persist about the external validity of these highly selective studies which may not reflect contemporary practice, particularly during a two decade period of significant change in the use of minimally invasive approaches. Unfortunately beyond these international RCTs, the majority of the literature comparing surgical approaches to proctectomy consists of single institution studies which demonstrate similar outcomes, with shorter hospital length of stay and reduced infection rates in patients receiving laparoscopic procedures [16, 17]. Small studies investigating oncologic outcomes specifically, including CRM negativity, have found similar or improved outcomes with MI-LAR with other studies suggesting a specific role for MI-LAR in facilitating simultaneous resection of liver metastases and management of bulky T4 tumors [18,19,20,21,22]. Previous studies utilizing administrative databases to compare surgical approaches to proctectomy have consistently demonstrated improvements in hospital length of stay and perioperative morbidity with laparoscopic approaches [23, 24]. Comparisons between laparoscopic and robotic approaches, including work by Speicher et al., in 2015, have demonstrated similar operative and oncologic outcomes between approaches [25, 26]. These findings are redemonstrated here, with robotic approaches demonstrating improvements in operative and short-term outcomes, although long-term survival does not differ between approaches and these cohorts differed significantly in several important demographics and comorbidities. Although some studies have demonstrated improved CRM clearance, lymph node harvest, and rates of R0 resection with minimally invasive approaches, the impact of surgical procedure on long-term mortality has been inconsistent [24, 27, 28]. Sujatha-Bhaskar et al., in a similar NCDB national analysis, demonstrate superior rates of CRM negativity and improved survival in patients receiving laparoscopic proctectomy although the inclusion criteria of this analysis selected for locally advanced disease, with all patients receiving neoadjuvant chemoradiation, surgical resection, and adjuvant therapy. The result of this is a limited population of 6,313 patients and heterogeneity of the specific surgical resection received.
When considering only patients treated at high-volume academic centers, which we considered to be a proxy for high-volume surgeons with skills in advanced laparoscopy or fellowship training, we found that surgical approach did not impact oncologic outcomes or perioperative morbidity and mortality in these centers (Tables 4, 5) [29, 30]. This suggests that improved outcomes may be due to a combination of surgeon volume, patient selection, and the multidisciplinary approach offered at high-volume centers. Taken together, these findings suggest that although MI-LAR is a safe and effective choice of treatment for rectal cancer, these procedures should be done by experienced surgeons on appropriately selected patients and approached in a multidisciplinary fashion at high-volume centers.
Although our findings suggest that MI-LAR is associated with improved oncologic outcomes and survival overall, laparoscopic approaches to advanced cases could be considered a proxy to surgical care in high-volume academic centers with highly trained experts [30]. Our subgroup analysis, considering only operations performed in academic medical centers in the highest tertile of operative volume, found no clinically or statistically significant differences in oncologic outcomes or survival between MI-LAR and OLAR. This highlights that although MI-LAR is safe and effective, with similar outcomes to OLAR, it should be performed on appropriately selected patients by surgeons experienced with advanced laparoscopy in high-volume centers with a robust multidisciplinary approach to cancer care.
Although the NCDB is the single largest clinical cancer database in the nation, and is widely accepted to capture contemporary practice patterns, it is susceptible to miscoding as are all administrative datasets. Additionally, although the NCDB provides several tumor-specific variables not included in other databases its granularity is ultimately more limited than institutional data and as such our ability to assess clinically important variables is limited. These include the specific agents used for neoadjuvant and adjuvant therapy and the completion of that therapy. Additionally, we were unable to determine disease-specific survival using the available data, limiting our ability to discriminate differences in this important outcome. Lastly, it is difficult to directly compare this analysis to RCTs as this is a retrospective and non-randomized analysis and as such is vulnerable to selection bias and unmeasured confounders influencing the surgical approach chosen for a given patient. Despite these limitations our study is the largest to date comparing MI-LAR and OLAR for rectal cancer in terms of pathologic oncologic outcomes and long-term survival. Our findings of improved CRM clearance with MI-LAR strengthen the conclusions drawn from prior RCTs, single institution studies, and smaller database studies and support the continued expansion of MI-LAR.
Conclusions
This study is the largest to date comparing the oncologic efficacy of MI-LAR and OLAR for the treatment of rectal cancer and provides important data on long-term survival with these approaches. MI-LAR is associated with improved short-term outcomes, oncologic outcomes including circumferential radial margin, and long-term survival compared to OLAR. Importantly, until long-term follow-up from ongoing RCTs is available this provides important evidence for the use of MI-LAR due to both pathologic surrogates for oncologic efficacy and improvements in long-term survival. Nonetheless, the decision to perform MI-LAR is dependent upon the skillset of individual surgeons and the characteristics of individual medical centers and should be performed by experienced surgeons in high-volume academic centers.
References
American Cancer Society (2017) Colorectal Cancer Facts & Figures 2017-2019. Atlanta, pp 1–40
Esemuede IO, Gabre-Kidan A, Fowler DL, Kiran RP (2015) Risk of readmission after laparoscopic vs. open colorectal surgery. Int J Colorectal Dis 30(11):1489–1494
Pasam RT, Esemuede IO, Lee-Kong SA, Kiran RP (2015) The minimally invasive approach is associated with reduced surgical site infections in obese patients undergoing proctectomy. Tech Coloproctol 19(12):733–743
Read TE (2007) Laparoscopic proctectomy for rectal adenocarcinoma. J Surg Oncol 96:660–664
Turner M, Adam MA, Sun Z, Kim J, Ezekian B, Yerokun B et al (2017) Insurance status, not race, is associated with use of minimally invasive surgical approach for rectal cancer. Ann Surg 265:774–781
Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M et al (2015) Effect of laparoscopic-assisted resection vs open resection of stage II or III rectal cancer on pathologic outcomes the ACOSOG Z6051 randomized clinical trial. JAMA-J Am Med Assoc 314(13):1346–1355
Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AMH et al (2005) Short-term endpoints of conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (MRC CLASICC trial): Multicentre, randomised controlled trial. Lancet 365(9472):1718–1726
Jeong SY, Park JW, Nam BH, Kim S, Kang SB, Lim SB et al (2014) Open versus laparoscopic surgery for mid-rectal or low-rectal cancer after neoadjuvant chemoradiotherapy (COREAN trial): survival outcomes of an open-label, non-inferiority, randomised controlled trial. Lancet Oncol 15(7):767–774
Bemelman WA, Ph D, Andersson J, Angenete E, Ph D, Rosenberg J et al (2015) A randomized trial of laparoscopic versus open surgery for rectal cancer. N Engl J Med 372:1324–1332
Stevenson ARL, Solomon MJ, Lumley JW, Hewett P, Clouston AD, Gebski VJ et al (2015) Effect of laparoscopic-assisted resection vs open resection on pathological outcomes in rectal cancer: the ALaCaRT randomized clinical trial. JAMA-J Am Med Assoc 314(13):1356–1363
Boffa DJ, Rosen JE, Mallin K, Loomis A, Gay G, Palis B et al (2017) Using the national cancer database for outcomes research. JAMA Oncol 3:1722–1728
Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AMH et al (2007) Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-Year results of the UK MRC CLASICC trial group. J Clin Oncol 25(21):3061–3068
Jayne DG, Thorpe HC, Copeland J, Quirke P, Brown JM, Guillou PJ (2010) Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer. Br J Surg 97(11):1638–1645
Green BL, Marshall HC, Collinson F, Quirke P, Guillou P, Jayne DG et al (2013) Long-term follow-up of the Medical Research Council CLASICC trial of conventional versus laparoscopically assisted resection in colorectal cancer. Br J Surg 100(1):75–82
van der Pas MH, Haglind E, Cuesta MA, Fürst A, Lacy AM, Hop WC et al (2013) Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol 14(3):210–218
Baik SH, Kwon HY, Kim JS, Hur H, Sohn SK, Cho CH et al (2009) Robotic versus laparoscopic low anterior resection of rectal cancer: short-term outcome of a prospective comparative study. Ann Surg Oncol 16(6):1480–1487
Zhou T, Zhang G, Tian H, Liu Z, Xia S (2014) Laparoscopic rectal resection versus open rectal resection with minilaparotomy for invasive rectal cancer. J Gastrointest Oncol 5(1):36–45
Sammour T, Malakorn S, Bednarski BK, Kaur H, Shin US, Messick C et al (2018) Oncological outcomes after robotic proctectomy for rectal cancer: analysis of a prospective database. Ann Surg 267(3):521–526
Boutros M, Hippalgaonkar N, Silva E, Allende D, Wexner SD, Berho M (2013) Laparoscopic resection of rectal cancer results in higher lymph node yield and better short-term outcomes than open surgery: a large single-center comparative study. Dis Colon Rectum 56(6):679–688
Barnajian M, Pettet D, Kazi E, Foppa C, Bergamaschi R (2014) Quality of total mesorectal excision and depth of circumferential resection margin in rectal cancer: a matched comparison of the first 20 robotic cases. Color Dis 16(8):603–609
Hatwell C, Bretagnol F, Farges O, Belghiti J, Panis Y (2013) Laparoscopic resection of colorectal cancer facilitates simultaneous surgery of synchronous liver metastases. Color Dis 15(1):e21–e28
de’Angelis N, Landi F, Vitali GC, Memeo R, Martínez-Pérez A, Solis A et al (2017) Multicentre propensity score-matched analysis of laparoscopic versus open surgery for T4 rectal cancer. Surg Endosc Other Interv Tech 31(8):3106–3121
Greenblatt DY, Rajamanickam V, Pugely AJ, Heise CP, Foley EF, Kennedy GD (2011) Short-term outcomes after laparoscopic-assisted proctectomy for rectal cancer: results from the ACS NSQIP. J Am Coll Surg 212(5):844–854
Sun Z, Kim J, Adam MA, Nussbaum DP, Speicher PJ, Mantyh CR et al (2016) Minimally invasive versus open low anterior resection equivalent survival in a national analysis of 14,033 patients with rectal cancer. Ann Surg 263(6):1152–1158
Speicher PJ, Englum BR, Ganapathi AM, Nussbaum DP, Mantyh CR, Migaly J (2015) Robotic low anterior resection for rectal cancer: a national perspective on short-term oncologic outcomes. Ann Surg 262(6):1040–1045
Jayne D, Pigazzi A, Marshall H, Croft J, Corrigan N, Copeland J et al (2017) Effect of robotic-assisted vs conventional laparoscopic surgery on risk of conversion to open laparotomy among patients undergoing resection for rectal cancer. JAMA 318(16):1569
Nussbaum DP, Speicher PJ, Ganapathi AM, Englum BR, Keenan JE, Mantyh CR et al (2014) Laparoscopic versus open low anterior resection for rectal cancer: results from the national cancer data base. J Gastrointest Surg 19:124–132
Sujatha-Bhaskar S, Jafari MD, Gahagan JV, Inaba CS, Koh CY, Mills SD et al (2017) Defining the role of minimally invasive proctectomy for locally advanced rectal adenocarcinoma. Ann Surg 266(4):574–581
Kayano H, Okuda J, Tanaka K, Kondo K, Tanigawa N (2011) Evaluation of the learning curve in laparoscopic low anterior resection for rectal cancer. Surg Endosc 25(9):2972–2979
Cagino K, Altieri MS, Yang J, Nie L, Talamini M, Spaniolas K et al (2017) Effect of academic status on outcomes of surgery for rectal cancer. Surg Endosc 32:2774–2780
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Brian Gilmore, Mohamed A. Adam, Kristen Rhodin, Megan C. Turner, Brian Ezekian, Christopher R. Mantyh, John Migaly have no financial ties or conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
NCDB Provision Disclosure: The National Cancer Data Base (NCDB) is a joint project of the Commission on Cancer (CoC) of the American College of Surgeons and the American Cancer Society. The CoC's NCDB and the hospitals participating in the CoC NCDB are the source of the de-identified data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the author.
Rights and permissions
About this article
Cite this article
Gilmore, B., Adam, M.A., Rhodin, K. et al. Evolution of minimally invasive surgery for rectal cancer: update from the national cancer database. Surg Endosc 35, 275–290 (2021). https://doi.org/10.1007/s00464-020-07393-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07393-y