Abstract
The olfactory organ of turtles consists of an upper chamber epithelium (UCE) with associated glands, and a lower chamber epithelium (LCE) devoid of glands. The UCE and LCE are referred to as the air-nose and the water-nose, respectively, because the UCE is thought to detect airborne odorants, while the LCE detects waterborne odorants. However, it is not clear how the two are used in the olfactory organ. Odorant receptors (ORs) are the major olfactory receptors in turtles; they are classified as class I and II ORs, distinguished by their primary structure. Class I ORs are suggested to be receptive to water-soluble ligands and class II ORs to volatile ligands. This study analyzed the expression of class I and II ORs in hatchlings of the green sea turtle, Chelonia mydas, through in situ hybridization, to determine the localization of OR-expressing cells in the olfactory organ. Class I OR-expressing cells were distributed mainly in the LCE, implying that the LCE is receptive to waterborne odorants. Class II OR-expressing cells were distributed in both the UCE and LCE, implying that the entire olfactory organ is receptive to airborne odorants. The widespread expression of class II ORs may increase opportunities for sea turtles to sense airborne odorants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Olfactory organs are used to collect information on diet, predators, and potential mates from the environment. The tetrapod olfactory organ consists of the olfactory epithelium and vomeronasal organ, which are separated from each other anatomically and functionally, and send axons to the main and accessory olfactory bulbs, respectively (Taniguchi and Taniguchi 2014). Mammals have two additional olfactory organs, which send axons to the main olfactory bulb: the septal organ of Masera (Rodolfo-Masera 1943) and the Grüneberg ganglion (Grüneberg 1973). The turtle olfactory organ comprises the upper chamber epithelium (UCE) and the lower chamber epithelium (LCE), which send axons to the ventral and dorsal parts of the olfactory bulb, respectively. Although the two are clearly distinguished by the presence or absence of associated glands, they are only separated anatomically by non-sensory epithelium (Parsons 1970; Schwenk 2008).
Generally, the tetrapod olfactory epithelium is associated with glands (Taniguchi and Taniguchi 2014), which play important roles in detecting odorants by dissolving airborne odorants with their secretory products (Getchell and Getchell 1992). In the olfactory organ of the African clawed frog, Xenopus laevis, the olfactory epithelium lining the principal chamber is associated with glands and detects airborne odorants, while the middle chamber epithelium, i.e., the olfactory epithelium lining the middle chamber, is devoid of associated glands and detects waterborne odorants (Altner 1962). In the turtle olfactory organ, the UCE is associated with glands, whereas the LCE is not. Therefore, the UCE and LCE are thought to detect airborne and waterborne odorants, respectively (Eisthen and Polese 2007; Schwenk 2008).
Vertebrate olfaction is mediated by several families of G protein-coupled receptors (GPCRs), collectively referred to as the olfactory receptors, and by non-GPCRs, which are membrane-spanning 4A (MS4A) proteins (Buck and Axel 1991; Dulac and Axel 1995; Fülle et al. 1995; Greer et al. 2016; Herrada and Dulac 1997; Matsunami and Buck 1997; Liberles and Buck 2006; Liberles et al. 2009; Rivière et al. 2009). Examples of the former include the odorant receptors (ORs), vomeronasal type 1 (V1Rs) and 2 (V2Rs) receptors, trace amine-associated receptors (TAARs), and formyl peptide receptors (FPRs). ORs and TAARs are coexpressed with Gαolf, V1Rs are coexpressed with Gαi2, and V2Rs are coexpressed with Gαo. FPRs are coexpressed with Gαi2 or Gαo, but they have been demonstrated only in rodents. The expression of Gαolf and Gαo in the turtle olfactory organ implies that most olfactory receptor neurons (ORNs) in the UCE and LCE express either ORs or TAARs and V2Rs (Wakabayashi and Ichikawa 2008; Nakamuta et al. 2016a). However, a recent study of the olfactory organs of freshwater turtles implied that only a small population of ORNs express vomeronasal receptors, while the majority express ORs (Abdali et al. 2020).
Vertebrate OR genes have been categorized into seven groups, designated group α to group η. Genes belonging to groups α and γ are found exclusively in tetrapods, those belonging to group β are found in both tetrapods and teleosts, and those belonging to groups δ, ε, ζ, and η are found only in teleosts and amphibians (Niimura and Nei 2005; Niimura 2012). These observations indicate that ORs belonging to groups α and γ detect airborne odorants, those belonging to groups δ, ε, ζ, and η detect water-soluble odorants, and those belonging to group β may detect odorants that are both water-soluble and volatile. Meanwhile, the tetrapod OR genes are also grouped into class I ORs corresponding to groups α and β, and class II ORs corresponding to group γ (Glusman et al. 2000; Zhang and Firestein 2002; Niimura and Nei 2005). In the X. laevis olfactory organ, class I ORs are expressed in the middle chamber epithelium, while class II ORs are expressed in the olfactory epithelium lining the principal chamber. As the middle chamber epithelium detects waterborne odorants and the olfactory epithelium lining the principal chamber detects airborne odorants, it has been suggested that class I ORs detect water-soluble odorants while class II ORs detect volatile odorants (Freitag et al. 1995, 1998). It has also been shown that ligands for class I and class II ORs in mammals tend to be hydrophilic and hydrophobic, respectively (Mezler et al. 2001). In all, 254 OR genes have been identified in the green sea turtle, and they consist of 158 genes belonging to group α, 1 gene belonging to group β, and 95 genes belonging to group γ (Wang et al. 2013). Therefore, green sea turtle ORs can be classified into 159 class I ORs and 95 class II ORs, but their sites of expression are not yet known.
In this study, we clarified the spatial distribution of cells expressing olfactory receptors in the olfactory organ of the green sea turtle, Chelonia mydas, through in situ hybridization. Hybridization signals showed a scattered distribution characteristic of olfactory receptor genes, suggesting that individual genes are expressed by sparsely distributed ORNs. Notably, genes encoding class II ORs, which have been suggested to detect volatile odorants, were found within both the UCE and LCE. There are nearly 300 turtle species worldwide. Although most are semi-aquatic, they inhabit environments ranging from terrestrial to fully aquatic. Sea turtles, which spend most of their life under water and do not go onto land except to lay eggs, are known to detect both waterborne odorants (Constantino and Salmon 2003; Piovano et al. 2004) and airborne odorants (Endres et al. 2009; Endres and Lohmann 2012). The broad expression of class II ORs demonstrated in this study implies the detection of airborne odorants by the entire olfactory organ and may increase opportunities for sea turtles to sense airborne odorants.
Materials and methods
The following experiments were approved by the Animal Ethics Committee of Iwate University and performed in accordance with the Guide for the Care and Use of Experimental Animals of Iwate University (approval no. A202143). Eggs were provided by the Port of Nagoya Public Aquarium (Aichi, Japan) and incubated at 29 °C, at which temperature both female and male hatchlings were expected to hatch (Morreale et al. 1982). Hatchlings (n = 10) were anesthetized with 0.2% tricaine methanesulfonate (MS-222) and decapitated. Subsequently, the olfactory organs were dissected out, fixed with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4), and decalcified in 10% ethylenediamine tetraacetic acid (EDTA) in 0.1 M phosphate buffer. After washing, the specimens were cryoprotected by passage through a sucrose gradient (10%, 20%, and 30%), embedded in OCT compound (Sakura Finetek, Tokyo, Japan), and frozen at −20 °C. Several sections were stained with hematoxylin and eosin for histological observation.
Cryosections 20 μm thick were thaw-mounted onto glass slides and subjected to in situ hybridization using digoxigenin (DIG)-labeled RNA probes. DNA fragments of olfactory marker protein (OMP), alpha subunit of olfactory neuron-specific G protein (Gαolf), cyclic nucleotide gated channel alpha 2 (CNGA2), transient receptor potential channel C2 (TRPC2), and V1Rs were amplified from olfactory organ cDNA with the primer pairs listed in Table 1. OMP encodes a cytoplasmic protein that marks mature ORNs in the olfactory organ of many vertebrate species, and has been suggested to have multiple functions, including roles in signal transduction, axon targeting, and behavior (Dibattista et al. 2021). CNGA2 encodes a cation channel subunit and is a marker of OR-expressing cells (Brunet et al. 1996). TRPC2 encodes an element of the signal transduction cascade and is a marker of vomeronasal receptor-expressing cells (Liman et al. 1999). Five class I ORs, namely CMY16, CMY17, CMY18, CMY19, and CMY20, and five class II ORs, namely CMY11, CMY12, CMY14, CMY15, and CMY22, were chosen as representatives of the 159 class I and 95 class II ORs, respectively, according to their positions in the phylogenetic tree (Fig. 1, Table 2). Protein sequences used for phylogenetic analyses were obtained from Wang et al. (2013). Sequence alignments were performed using Clustal W in MEGA 7 (Kumar et al. 2016). Then detailed phylogenetic analyses were conducted based on the multiple sequence alignments using the neighbor-joining method in MEGA 7 with 500 bootstrap replicates. Vomeronasal type 1 receptor was also added as an outgroup. All DNA fragments were cloned into the pCRII TOPO vector (ThermoFisher Scientific, Waltham, MA, USA), and confirmed by sequencing. DIG-labeled probes were synthesized using SP6/T7 DIG RNA labeling kits (Sigma-Aldrich, St. Louis, MO, USA). Section pretreatment, probe hybridization, post-hybridization washing, and signal detection were performed using in situ hybridization reagents (ISHR; Nippon Gene, Tokyo, Japan) in accordance with the manufacturer’s protocol, as described previously (Nakamuta et al. 2023). Briefly, hybridization was performed overnight at 55 °C using ISHR7 hybridization buffer (Nippon Gene). Anti-DIG antibody coupled to alkaline phosphatase, 4-nitroblue tetrazolium chloride, and 5-bromo-4-chloro-3-indolyl-phosphate were used for signal detection (all from Roche Diagnostics, Basel, Switzerland). Negative control experiments using sense probes yielded no specific signals.
Phylogenetic tree of odorant receptors (ORs) in green sea turtle constructed using the Neighbor-Joining method. Vomeronasal type 1 receptors are also added as outgroup. The ORs are classified into class I (blue) and II (red) ORs according to their primary structure. Five class I and five class II ORs were chosen as representatives of 159 class I and 95 class II ORs, respectively
Results
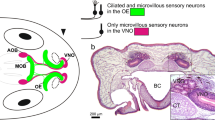
In general, the turtle nasal cavity is divided into upper and lower chambers. However, since the lower chamber is further subdivided into anterodorsal (AD), anteroventral (AV), and posteroventral (PV) regions, the upper chamber is referred to as the posterodorsal (PD) region in sea turtles (Fig. 2a-c). As reported by Saito et al. (2000) and Kondoh et al. (2019), the AD, AV, and PV regions of the nasal cavity were lined by the LCE, a sensory epithelium devoid of associated glands, while the PD region was lined by the UCE, a sensory epithelium associated with glands (Figs. 2d-g, S1). Nuclei of the supporting cells, ORNs, and basal cells were situated in the apical, middle, and basal layers of each epithelium, and the UCE and LCE could be readily distinguished from non-sensory epithelium.
Gross anatomical and histological characterization in the olfactory organ of sea turtle. a Lateral, b dorsal, and c ventral views of the olfactory organ. The nasal cavity of sea turtle is divided into the anterodorsal (AD), anteroventral (AV), posteroventral (PV), and posterodorsal (PD) regions. d–g Hematoxylin and eosin-stained sections. The PD region of the nasal cavity is lined by the upper chamber epithelium (UCE), whereas the AD, AV, and PV regions of the nasal cavity are lined by the lower chamber epithelium (LCE). The UCE and LCE are distinguished from each other by the presence of associated glands (arrows) in the UCE and absence of them in the LCE. Asterisks indicate non-sensory epithelium on the medial and lateral walls of nasal cavity. Scale bars: a–c 2 mm, d–g 500 µm
OMP was expressed by cells broadly distributed in the middle layers of both the UCE and LCE (Fig. 3), indicating the presence of ORNs in each of the four nasal cavity regions. In both the UCE and LCE, probes for Gαolf and CNGA2 showed ubiquitous labeling, extending throughout the sensory epithelia defined by OMP expression (Figs. 4 and 5). These observations imply the expression of ORs by most ORNs in the UCE and LCE. In contrast, TRPC2 was expressed by a small subset of ORNs within both the UCE and LCE (Fig. 6, Supplementary Fig. S2), implying the expression of vomeronasal receptors by a small population of ORNs. These observations also imply that cells expressing vomeronasal receptors are intermingled with those expressing ORs in the sea turtle olfactory organ.
In situ hybridization analysis for the expression of the gene encoding OMP, a marker for olfactory receptor neurons, in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See extensive expression of OMP mRNA. AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a”, b” 250 µm
In situ hybridization analysis for the expression of the gene encoding Gαolf, an alpha subunit of G-protein co-expressed with odorant receptors, in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See extensive expression of Gαolf mRNA. AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a”, b” 100 µm
In situ hybridization analysis for the expression of the gene encoding CNGA2, a marker for cells expressing odorant receptors, in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See extensive expression of CNGA2 mRNA. AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a”, b” 100 µm
In situ hybridization analysis for the expression of the gene encoding TRPC2, a marker for cells expressing vomeronasal receptors, in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See sparse expression of TRPC2 mRNA. Signals are shown as blue circles in (a, b), or pointed by arrows in (a’, b’, a”, b”). AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a”, b” 250 µm
Cells expressing class I ORs were abundant in the LCE and rare in the UCE. For instance, among 551 cells labeled for CMY16 in 14 sections, 6 were found in the UCE and 545 were found in the LCE (Figs. 7, S3). Similar results were obtained for CMY17, CMY18, CMY19, and CMY20 (Supplementary Figs. S4–S7). Conversely, class II ORs were predominantly expressed in the UCE. For instance, among 297 cells labeled for CMY11 in 14 sections, 293 were found in the UCE and 4 in the LCE (Figs. 8, S8). Similar results were obtained for CMY15 and CMY22 (Supplementary Figs. S9, S10). However, signals for some class II ORs were detected in both the UCE and LCE. For instance, among 382 cells labeled for CMY12 in 14 sections, 128 were found in the UCE and 254 in the LCE (Fig. 9, Supplementary Fig. S11). Similarly, among 112 cells labeled for CMY14 in 13 sections, 95 were found in the UCE and 17 in the LCE (Supplementary Fig. S12). Cells expressing V1R were distributed in both the UCE and LCE (Figs. 10, S13), as expected from TRPC2 expression.
In situ hybridization analysis for the expression of the gene encoding class I OR (CMY16) in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See expression of class I OR mRNA in the LCE. Signals are shown as blue circles in (a, b), or pointed by arrows in (a’, b’, a”, b”). AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a”, b” 100 µm
Spatial distribution of cells expressing class II OR (CMY11) in the olfactory organ of sea turtle. See expression of class II OR mRNA in the UCE. Fourteen sections were picked up at intervals of 200 µm and arranged from a anterior to n posterior end of nasal cavity. Signals (293 in UCE, 4 in LCE) are shown as blue circles. AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bar: 1 mm
Spatial distribution of cells expressing class II OR (CMY12) in the olfactory organ of sea turtle. See expression of class II OR mRNA in the UCE and LCE. Fourteen sections were picked up at intervals of 200 µm and arranged from a anterior to n posterior end of nasal cavity. Signals (128 in UCE, 254 in LCE) are shown as blue circles. AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bar: 1 mm
In situ hybridization analysis for the expression of the gene encoding V1R (CMY09) in the olfactory organ of sea turtle. a anterior and b posterior regions of the olfactory organ. a’, a” and b’, b” are higher magnification views of (a, b). See sparse expression of V1R mRNA. Signals are shown as blue circles in (a, b), or pointed by arrows in (a’, b’, a”). AD, anterodorsal; AV, anteroventral; PD, posterodorsal; and PV, posteroventral regions of the nasal cavity. Scale bars: a, b 1 mm, a’, b’, a” 100 µm, b” 200 µm
Discussion
Previous studies distinguished two types of sensory epithelia in the nasal cavity of the sea turtle: the UCE, associated with glands and lining the dorsally situated “olfactory region” of Parsons; and the LCE, devoid of glands and lining the ventrally situated “intermediate region” of Parsons (Parsons 1970; Saito et al. 2000). The UCE contains ciliated ORNs, while the LCE contains microvillous ORNs. However, the presence of a third type of epithelium lining the posteroventral region of the nasal cavity, lacking associated glands, and containing microvillous ORNs has been proposed (Kondoh et al. 2019). The present study was performed to determine possible functional differences among these sensory epithelia by examining the expression of olfactory receptor genes.
This study clarified the distribution of cells expressing olfactory receptors in the sea turtle olfactory organ, and the results implied functional diversification of the UCE lining the PD region of the nasal cavity and LCE lining the AD, AV, and PV regions. Class I ORs, which have been suggested to detect water-soluble ligands, were expressed mainly in the LCE, implying that the LCE is receptive to waterborne odorants. Our results are consistent with the findings of other studies suggesting that the turtle olfactory organ detects waterborne odorants with the LCE, which does not have associated glands (Eisthen and Polese 2007; Schwenk 2008). They are also consistent with a simulation using a silicon-mold model, which showed that water flows readily into the AD, AV, and PV regions of the nasal cavity, while little enters the PD region, which remains filled with air while underwater (Kondoh et al. 2019). Therefore, the sea turtle olfactory organ likely detects waterborne odorants via the LCE lining the AD, AV, and PV regions of the nasal cavity.
Unlike class I ORs, class II ORs were expressed chiefly in the UCE. More importantly, some class II ORs were expressed in both the UCE and LCE. These observations indicate that the UCE and LCE are not completely separated from each other with respect to their functions. The expression of class II ORs, which have been suggested to detect volatile ligands, implies the detection of airborne odorants. Therefore, the LCE, as well as the UCE, may participate in the detection of airborne odorants. Indeed, sea turtles can expel water from the entire nasal cavity before respiration. Consequently, the entire olfactory organ of sea-turtle is supposed to contact with air when the nasal cavity serves as a pathway for air. In addition, the angle of the olfactory organ relative to the water surface changes as the sea turtle swims upward and downward. Therefore, given that class II ORs are widely expressed, the olfactory organs can detect airborne odorants at any angle, which would increase the chances that sea turtles could detect airborne odorants.
Signals were detected for one of the two V1Rs, but not for the other, in the sea turtle olfactory organ (Supplementary Fig. S14). Of course, we cannot exclude the possibility of the expression of the V1R that was not detected in this study. Meanwhile, V2R expression was not examined in the present study, as the only known V2R in the sea turtle genome is a pseudogene (Wang et al. 2013). However, the V1R was expressed by only a small population of ORNs, and TRPC2 showed more numerous signals than V1R. Therefore, unknown olfactory receptors, including other V1Rs or V2Rs, may be coexpressed with TRPC2. The population of cells expressing unknown V1Rs/V2Rs in the sea turtle olfactory organ is assumed to be small, considering the small population of ORNs expressing TRPC2 in the present study. Gαo is a marker of cells expressing V2Rs (Berghard and Buck 1996) and is expressed by most ORNs in both the UCE and LCE of the turtle olfactory organ (Wakabayashi and Ichikawa 2008; Nakamuta et al. 2016b). This implies that V2Rs are expressed by most ORNs constituting the organ. However, we recently demonstrated that only a small population of ORNs express vomeronasal receptor genes and TRPC2 in the olfactory organ of freshwater turtles, including red-eared sliders and soft-shelled turtles (Abdali et al. 2020). Therefore, it is unlikely that Gαo is always coexpressed with V2Rs, although we observed Gαo expression in most ORNs (data not shown).
The results of the present study indicate that ORs are expressed by most ORNs in the sea turtle olfactory organ. The expression of ORs by most ORNs has also been suggested in other turtles based on the expression of Gαolf (Wakabayashi and Ichikawa 2008; Nakamuta et al. 2016a; Nakamuta et al. 2016b) and CNGA2 (Abdali et al. 2020). Such olfactory receptor expression resembles that in the olfactory epithelium of mammals, in which most ORNs express ORs and only a small population express vomeronasal receptors (Wakabayashi et al. 2007). Therefore, both the UCE and LCE of the turtle olfactory organ can be considered olfactory epithelia based on the expression of olfactory receptors. This is in marked contrast to the situation in other reptiles, such as snakes, which have many V2Rs and most ORNs in the vomeronasal organ express V2Rs (Brykezynska et al. 2013). The UCE and LCE are referred to as the olfactory epithelium and vomeronasal organ in turtles, based on the fine structure of the ORNs. In many turtles, the UCE contains ciliated ORNs and the LCE contains microvillous ORNs (Graziadei and Tucker 1970; Hatanaka et al. 1982; Saito et al. 2000; Wakabayashi and Ichikawa 2008; Nakamuta et al. 2016a; Nakamuta et al. 2016b), whereas the olfactory epithelium contains ciliated ORNs and the vomeronasal organ contains microvillous ORNs in mammals. Therefore, the turtle olfactory organ is unique from other vertebrates, in that it contains microvillous ORNs expressing ORs. So far, there is no evidence that ORs are expressed in microvillous ORNs in fish or amphibians (Eisthen 2004; Hansen et al. 2003, 2004; Nakada et al. 2014; Sato et al. 2005). Even in the olfactory epithelium of fish, where both ciliated and microvillous ORNs are distributed, it has been suggested that ORs are expressed in the ciliated ORNs and vomeronasal receptors in the microvillous ORNs. Meanwhile, not only in the olfactory organ of soft-shelled turtles, where microvillous ORNs are not found, but also in the olfactory organ of red-eared sliders, where microvillous ORNs are contained in the LCE, it has been suggested that most ORNs express ORs and limited number of ORNs express the genes encoding vomeronasal receptors (Abdali et al. 2020). Thus, the olfactory organ of turtle is of interest for studies of the relations between the expression of olfactory receptors and the fine structure of ORNs.
Data availability
The data that support the findings of this article are available from the corresponding author upon reasonable request.
References
Abdali SS, Nakamuta S, Yamamoto Y, Nakamuta N (2020) Distribution of cells expressing vomeronasal receptors in the olfactory organ of turtles. J Vet Med Sci 82:1068–1079. https://doi.org/10.1292/jvms.20-0207
Altner H (1962) Untersuchungen uber Leistungen und Bau der Nase des sudafrikanischen Krallenfrosches Xenopus laevis (Daudin, 1803). Z Vergl Physiol 45:272–306. https://doi.org/10.1007/BF00302326
Berghard A, Buck LB (1996) Sensory transduction in vomeronasal neurons: evidence for Gαo, Gαi2, and adenylyl cyclase II as major components of a pheromone signaling cascade. J Neurosci 16:909–918. https://doi.org/10.1523/JNEUROSCI.16-03-00909.1996
Brunet LJ, Gold GH, Ngai J (1996) General anosmia caused by a targeted disruption of the mouse olfactory cyclic nucleotide-gated cation channel. Neuron 17:681–693. https://doi.org/10.1016/S0896-6273(00)80200-7
Brykczynska U, Tzika AC, Rodriguez I, Milinkovitch MC (2013) Contrasted evolution of the vomeronasal receptor repertoires in mammals and squamate reptiles. Genome Biol Evol 5:389–401. https://doi.org/10.1093/gbe/evt013
Buck L, Axel R (1991) A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell 65:175–187. https://doi.org/10.1016/0092-8674(91)90418-X
Constantino MA, Salmon M (2003) Role of chemical and visual cues in food recognition by leatherback posthatchlings (Dermochelys coriacea L). Zoology (Jena) 106:173–181. https://doi.org/10.1078/0944-2006-00114
Dibattista M, Koborssy DA, Genovese F, Reisert J (2021) The functional relevance of olfactory marker protein in the vertebrate olfactory system: a never-ending story. Cell Tissue Res 383:409–427. https://doi.org/10.1007/s00441-020-03349-9
Dulac C, Axel R (1995) A novel family of genes encoding putative pheromone receptors in mammals. Cell 83:195–206. https://doi.org/10.1016/0092-8674(95)90161-2
Eisthen HL (2004) The goldfish knows: olfactory receptor cell morphology predicts receptor gene expression. J Comp Neurol 477(4):341–346. https://doi.org/10.1002/cne.20258
Eisthen HL, Polese G (2007) Evolution of vertebrate olfactory subsystems. In: Kaas JH (ed) Evolution of nervous system, Vol 2. Non-mammalian vertebrates, Academic Press, Oxford, pp 355–406. https://doi.org/10.1016/B0-12-370878-8/00142-7
Endres CS, Lohmann KJ (2012) Perception of dimethyl sulfide (DMS) by loggerhead sea turtles: a possible mechanism for locating high-productivity areas for foraging. J Exp Biol 215:3535–3538. https://doi.org/10.1242/jeb.073221
Endres CS, Putman NF, Lohmann KJ (2009) Perception of airborne odors by loggerhead sea turtles. J Exp Biol 212:3823–3827. https://doi.org/10.1242/jeb.033068
Freitag J, Krieger J, Strotmann J, Breer H (1995) Two classes of olfactory receptors in Xenopus laevis. Neuron 15:1383–1392. https://doi.org/10.1016/0896-6273(95)90016-0
Freitag J, Ludwig G, Andreini I, Rössler P, Breer H (1998) Olfactory receptors in aquatic and terrestrial vertebrates. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 183:635–650. https://doi.org/10.1007/s003590050287
Fülle HJ, Vassar R, Foster DC, Yang RB, Axel R, Garbers DL (1995) A receptor guanylyl cyclase expressed specifically in olfactory sensory neurons. Proc Natl Acad Sci USA 92:3571–3575. https://doi.org/10.1073/pnas.92.8.3571
Getchell ML, Getchell TV (1992) Fine structural aspects of secretion and extrinsic innervation in the olfactory mucosa. Microsc Res Tech 23:111–127. https://doi.org/10.1002/jemt.1070230203
Glusman G, Bahar A, Sharon D, Pilpel Y, White J, Lancet D (2000) The olfactory receptor gene superfamily: data mining, classification, and nomenclature. Mamm Genome 11:1016–1023. https://doi.org/10.1007/s003350010196
Graziadei PPC, Tucker D (1970) Vomeronasal receptors in turtles. Z Zellforsch Mikrosk Anat 105:498–514. https://doi.org/10.1007/BF00335424
Greer PL, Bear DM, Lassance JM, Bloom ML, Tsukahara T, Pashkovski SL, Masuda FK, Nowlan AC, Kirchner R, Hoekstra HE, Datta SR (2016) A family of non-GPCR chemosensors defines an alternative logic for mammalian olfaction. Cell 165(7):1734–1748. https://doi.org/10.1016/j.cell.2016.05.001
Grüneberg H (1973) A ganglion probably belonging to the N. terminalis system in the nasal mucosa of the mouse. Z Anat Entwicklungsgesch 140:39–52. https://doi.org/10.1007/BF00520716
Hansen A, Anderson KT, Finger TE (2004) Differential distribution of olfactory receptor neurons in goldfish: structural and molecular correlates. J Comp Neurol 477(4):347–359. https://doi.org/10.1002/cne.20202
Hansen A, Rolen SH, Anderson K, Morita Y, Caprio J, Finger TE (2003) Correlation between olfactory receptor cell type and function in the channel catfish. J Neurosci 23(28):9328–9339. https://doi.org/10.1523/JNEUROSCI.23-28-09328.2003
Hatanaka T, Matsuzaki O, Shibuya T (1982) Fine structure of vomeronasal receptor cells in the Reeve’s turtle, Geoclemys reevesii. Zool Mag 91:190–193. https://doi.org/10.34435/zm004925
Herrada G, Dulac C (1997) A novel family of putative pheromone receptors in mammals with a topographically organized and sexually dimorphic distribution. Cell 90:763–773. https://doi.org/10.1016/S0092-8674(00)80536-X
Kondoh D, Kitayama C, Yamaguchi Y, Yamagawa M, Kawai YK, Suzuki C, Itakura R, Fujimoto A, Satow T, Kondo S, Sato T (2019) Nasal cavity of green sea turtles contains 3 independent sensory epithelia. Chem Senses 44:427–434. https://doi.org/10.1093/chemse/bjz033
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Liberles SD, Buck LB (2006) A second class of chemosensory receptors in the olfactory epithelium. Nature 442:645–650. https://doi.org/10.1038/nature05066
Liberles SD, Horowitz LF, Kuang D, Contos JJ, Wilson KL, Siltberg-Liberles J, Liberles DA, Buck LB (2009) Formyl peptide receptors are candidate chemosensory receptors in the vomeronasal organ. Proc Natl Acad Sci USA 106:9842–9847. https://doi.org/10.1073/pnas.0904464106
Liman ER, Corey DP, Dulac C (1999) TRP2: a candidate transduction channel for mammalian pheromone sensory signaling. Proc Natl Acad Sci USA 96:5791–5796. https://doi.org/10.1073/pnas.96.10.5791
Matsunami H, Buck LB (1997) A multigene family encoding a diverse array of putative pheromone receptors in mammals. Cell 90:775–784. https://doi.org/10.1016/S0092-8674(00)80537-1
Mezler M, Fleischer J, Breer H (2001) Characteristic features and ligand specificity of the two olfactory receptor classes from Xenopus laevis. J Exp Biol 204:2987–2997. https://doi.org/10.1242/jeb.204.17.2987
Morreale SJ, Ruiz GJ, Spotila JR, Standora EA (1982) Temperature-dependent sex determination: Current practices threaten conservation of sea turtles. Science 216:1245–1247. https://doi.org/10.1126/science.7079758
Nakada T, Hagino-Yamagishi K, Nakanishi K, Yokosuka M, Saito TR, Toyoda F, Hasunuma I, Nakakura T, Kikuyama S (2014) Expression of G proteins in the olfactory receptor neurons of the newt Cynops pyrrhogaster: their unique projection into the olfactory bulbs. J Comp Neurol 522(15):3501–3519. https://doi.org/10.1002/cne.23619
Nakamuta N, Nakamuta S, Kato H, Yamamoto Y (2016) Morphological study on the olfactory systems of the snapping turtle, Chelydra serpentina. Tissue Cell 48:145–151. https://doi.org/10.1016/j.tice.2016.03.011
Nakamuta S, Yamamoto Y, Miyazaki M, Sakuma A, Nikaido M, Nakamuta N (2023) Type 1 vomeronasal receptors expressed in the olfactory organs of two African lungfish, Protopterus annectens and Protopterus amphibius. J Comp Neurol 531:116–131. https://doi.org/10.1002/cne.25416
Nakamuta S, Yokosuka M, Taniguchi K, Yamamoto Y, Nakamuta N (2016) Immunohistochemical analysis for G protein in the olfactory organs of soft-shelled turtle, Pelodiscus sinensis. J Vet Med Sci 78:245–250. https://doi.org/10.1292/jvms.15-0359
Niimura Y (2012) Olfactory receptor multigene family in vertebrates: from the viewpoint of evolutionary genomics. Curr Genomics 13:103–114. https://doi.org/10.2174/138920212799860706
Niimura Y, Nei M (2005) Evolutionary dynamics of olfactory receptor genes in fishes and tetrapods. Proc Natl Acad Sci USA 102:6039–6044. https://doi.org/10.1073/pnas.0501922102
Parsons TS (1970) The nose and Jacobson’s organ. In: Gans C, Parsons TS (eds) Biology of the reptilia, vol 2. Academic Press, New York, pp 99–191
Piovano S, Balletto E, Di Marco S, Dominici A, Giacoma C, Zanetti A (2004) Loggerhead turtle (Caretta caretta) bycatches on long-lines: the importance of olfactory stimuli. Ital J Zool 71:213–216. https://doi.org/10.1080/11250000409356638
Rivière S, Challet L, Fluegge D, Spehr M, Rodriguez I (2009) Formyl peptide receptor-like proteins are a novel family of vomeronasal chemosensors. Nature 459:574–577. https://doi.org/10.1038/nature08029
Rodolfo-Masera T (1943) Su lésistenza di un particolare organo olfactivo nel setto nasale della cavia e di altri roditori. Arch Ital Anat Embryol 48:157–212
Saito K, Shoji T, Uchida I, Ueda H (2000) Structure of the olfactory and vomeronasal epithelia in the loggerhead turtle Caretta caretta. Fish Sci 66:409–411. https://doi.org/10.1046/j.1444-2906.2000.00065.x
Sato Y, Miyasaka N, Yoshihara Y (2005) Mutually exclusive glomerular innervation by two distinct types of olfactory sensory neurons revealed in transgenic zebrafish. J Neurosci 25:4889–4897. https://doi.org/10.1523/JNEUROSCI.0679-05.2005
Schwenk K (2008) Comparative anatomy and physiology of chemical senses in nonavian aquatic reptiles. In: Thewissen JGM and Nummela S (eds) Sensory evolution on the threshold, Univ. of California Press, Berkley, pp 65–81. https://doi.org/10.1525/california/9780520252783.003.0005
Taniguchi K, Taniguchi K (2014) Phylogenic studies on the olfactory system in vertebrates. J Vet Med Sci 76:781–788. https://doi.org/10.1292/jvms.10-0316
Wakabayashi Y, Ichikawa M (2008) Localization of G protein alpha subunits and morphology of receptor neurons in olfactory and vomeronasal epithelia in Reeve’s turtle, Geoclemys reevesii. Zoolog Sci 25:178–187. https://doi.org/10.2108/zsj.25.178
Wakabayashi Y, Ohkura S, Okamura H, Mori Y, Ichikawa M (2007) Expression of a vomeronasal receptor gene (V1r) and G protein α subunits in goat, Capra hircus, olfactory receptor neurons. J Comp Neurol 503:371–380. https://doi.org/10.1002/cne.21394
Wang Z, Pascual-Anaya J, Zadissa A, Li W, Niimura Y, Huang Z, Li C, White S, Xiong Z, Fang D, Wang B, Ming Y, Chen Y, Zheng Y, Kuraku S, Pignatelli M, Herrero J, Beal K, Nozawa M, Li Q, Wang J, Zhang H, Yu L, Shigenobu S, Wang J, Liu J, Flicek P, Searle S, Wang J, Kuratani S, Yin Y, Aken B, Zhang G, Irie N (2013) The draft genomes of soft-shell turtle and green sea turtle yield insights into the development and evolution of the turtle-specific body plan. Nat Genet 45:701–706. https://doi.org/10.1038/ng.2615
Zhang X, Firestein S (2002) The olfactory receptor gene superfamily of the mouse. Nat Neurosci 5:124–133. https://doi.org/10.1038/nn800
Acknowledgments
We thank Haruka Sasaki for technical assistance with primer designing, tissue sectioning, and tissue processing. We thank Asahi Nakamuta for expert assistance with phylogenetic analysis.
Author information
Authors and Affiliations
Contributions
Conceptualization, SN, NN; Sample preparation, MM, MI, MK.; Investigation, SN; Data analysis, NN; Wrote paper, NN; Supervision, MM, YY. All authors have read and agreed to the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Animal Ethics Committee of Iwate University (approval no. A202143).
Informed consent
Not Applicable
Conflict of interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakamuta, S., Mori, M., Ito, M. et al. In situ hybridization analysis of olfactory receptor expression in the sea turtle olfactory organ. Cell Tissue Res 393, 253–264 (2023). https://doi.org/10.1007/s00441-023-03782-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-023-03782-6