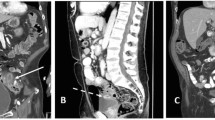
Abstract
Purpose
Exposure to immune checkpoint inhibitors (ICIs) can predispose to immune-related adverse events (irAEs) involving the gastrointestinal tract. The association between ICIs and bowel perforation has not been well studied. We aimed to describe the clinical course, complications, treatment, and outcomes of patients experiencing bowel perforation during or after ICI treatment.
Methods
This retrospective, single-center study included adult cancer patients with bowel perforation that occurred between the first dose of ICI treatment and up to 1 year thereafter between 1/1/2010 and 4/30/2021. Patients’ clinical course, imaging, treatment, and outcomes related to bowel perforation were collected and analyzed.
Results
Of the 13,991 patients who received ICIs during the study period, 90 (0.6%) met the inclusion criteria. A majority were male (54.4%), the most common cancer type was melanoma (23.3%), and most patients had received PD-1/L1 inhibitor treatment (58.8%). Onset of perforation occurred after a median of four ICI treatment cycles. The most common symptom was abdominal pain (95.5%). The colon was the most common location for the perforation (37.7%). Evidence of diverticulitis, enterocolitis, or appendicitis was seen in 32 (35.6%) patients, and 6 (6.6%) patients had luminal cancer involvement at the time of perforation. The overall hospitalization rate related to perforation was 95.5%, with mortality of 15.5% during the same admission. Antibiotics were given in 95% of our sample; 37.8% of patients also required surgical/interventional radiology intervention. Forty-six patients (51.1%) had perforation-related complications (e.g., sepsis, fistula, abscess), which were associated with a higher mortality rate (30%).
Conclusion
Our findings suggest a low incidence of bowel perforation after ICI treatment (0.6%), with 40% of patients having coexisting bowel inflammation as a potential contributing factor. Patients with bowel perforation had an aggressive disease course and high rates of hospitalization, complications, and mortality. Early recognition and prompt intervention is critical to improve patient outcomes. Future studies are warranted to further investigate the cause, predictive markers, and optimal treatment for this patient population.


Similar content being viewed by others
Data availability
The data sets used and analyzed in this study are available from the corresponding author on reasonable request.
References
Adhikari R, Ghose M, Tekin A, Singh S, Singh R (2021) Intestinal perforation: a rare complication of treatment with bevacizumab. Cureus 13(3):e14093. https://doi.org/10.7759/cureus.14093
Badgwell BD, Camp ER, Feig B, Wolff RA, Eng C, Ellis LM, Cormier JN (2008) Management of bevacizumab-associated bowel perforation: a case series and review of the literature. Ann Oncol 19(3):577–582. https://doi.org/10.1093/annonc/mdm508
Balogun OS, Osinowo A, Afolayan M, Olajide T, Lawal A, Adesanya A (2019) Acute perforated appendicitis in adults: management and complications in Lagos Nigeria. Ann Afr Med 18(1):36–41. https://doi.org/10.4103/aam.aam_11_18
Banaszkiewicz Z, Woda Ł, Tojek K, Jarmocik P, Jawień A (2014) Colorectal cancer with intestinal perforation—a retrospective analysis of treatment outcomes. Contemp Oncol (poznan, Poland) 18(6):414–418. https://doi.org/10.5114/wo.2014.46362
Bavaro MF (2002) Neutropenic enterocolitis. Curr Gastroenterol Rep 4:297–301
Beck T N (2020) Colonic Perforation after Dual Ipilimumab and Nivolumab Treatment. Case Reviews in Surgery. 2:9
Beck TN, Kudinov AE, Dulaimi E et al (2019) Case report: reinitiating pembrolizumab treatment after small bowel perforation. BMC Cancer 19:379
Burdine L, Lai K (2014) Laryea J A (2014) Ipilimumab-induced colonic perforation. J Surg Case Reports 3:rju010. https://doi.org/10.1093/jscr/rju010
Celli R, Kluger HM, Zhang X (2018) Anti-PD-1 therapy-associated perforating colitis. Case Rep Gastrointest Med 2018:3406437. https://doi.org/10.1155/2018/3406437
Chang ST, Menias CO, Lubner MG, Mellnick VM, Hara AK, Desser TS (2017) Molecular and clinical approach to intra-abdominal adverse effects of targeted cancer therapies. Radiographics 37(5):1461–1482. https://doi.org/10.1148/rg.2017160162
Chen XL, Lei YH, Liu CF, Yang QF, Zuo PY, Liu CY, Chen CZ, Liu YW (2013) Angiogenesis inhibitor bevacizumab increases the risk of ischemic heart disease associated with chemotherapy: a meta-analysis. PLoS ONE 8(6):e66721. https://doi.org/10.1371/journal.pone.0066721
Cho S J (2019) Intraepithelial lymphocytes in the gastrointestinal tract: to celiac disease and beyond. Current Issues in Anatomic Pathology 1:19
Chow DC, Babaian M, Taubin HL (1997) Jejunoileal diverticula. Gastroenterologist 5(1):78–84
Delasos L, Desai A, Lopetegui Lia N, Kethireddy N, Ray C (2019) A case of immunotherapy-induced colitis complicated by perforation and treated with infliximab postoperatively. Case Rep Oncol Med 2019:9069354. https://doi.org/10.1155/2019/9069354
Dilling P, Walczak J, Pikiel P et al (2014) Multiple colon perforation as a fatal complication during treatment of metastatic melanoma with ipilimumab—case report. Pol Przegl Chir 86:94–96
Haanen J, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, Jordan K (2017) Management of toxicities from immunotherapy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 28(suppl_4):iv119–iv142. https://doi.org/10.1093/annonc/mdx225
Hapani S, Chu D, Wu S (2009) Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol 10(6):559–568. https://doi.org/10.1016/S1470-2045(09)70112-3
Kim HK, Baek SW, Jeong Y, Yang Y, Kwon J, Han HS, An JY, Woo CG, Lee OJ, Lee TG, Lee KH (2019) Pseudoprogression presenting as intestinal perforation in non-small cell lung cancer treated with anti-PD-1: a case report. Mol Clin Oncol 11(2):132–134. https://doi.org/10.3892/mco.2019.1871
Lee HT, Lee SH, Heo YS (2019) Molecular interactions of antibody drugs targeting PD-1, PD-L1, and CTLA-4 in immuno-oncology. Mol (basel, Switzerland) 24(6):1190. https://doi.org/10.3390/molecules24061190
Prieux-Klotz C, Dior M, Damotte D, Dreanic J, Brieau B, Brezault C, Abitbol V, Chaussade S, Coriat R (2017) Immune checkpoint inhibitor-induced colitis: diagnosis and management. Target Oncol 12(3):301–308. https://doi.org/10.1007/s11523-017-0495-4
Services UDoHaH (2017) common terminology criteria for adverse events (CTCAE) Version 5.0. Institute NIoHNC, Cary
Shah R, Witt D, Asif T, Mir FF (2017) Ipilimumab as a cause of severe pan-colitis and colonic perforation. Cureus 9(4):e1182. https://doi.org/10.7759/cureus.1182
Shaikh DH, Baiomi A, Mehershahi S, Abbas H, Gongati S, Nayudu SK (2020) Paclitaxel-induced bowel perforation: a rare cause of acute abdomen. Case Rep Gastroenterol 14(3):687–694. https://doi.org/10.1159/000510131
Shin R, Lee SM, Sohn B, Lee DW, Song I, Chai YJ, Lee HW, Ahn HS, Jung IM, Chung JK, Heo SC (2016) Predictors of morbidity and mortality after surgery for intestinal perforation. Ann Coloproctol 32(6):221–227. https://doi.org/10.3393/ac.2016.32.6.221
Shivaji UN, Jeffery L, Gui X, Smith S, Ahmad OF, Akbar A, Ghosh S, Iacucci M (2019) Immune checkpoint inhibitor-associated gastrointestinal and hepatic adverse events and their management. Ther Adv Gastroenterol 12:1756284819884196. https://doi.org/10.1177/1756284819884196
Smith FO, Goff SL, Klapper JA, Levy C, Allen T, Mavroukakis SA, Rosenberg SA (2007) Risk of bowel perforation in patients receiving interleukin-2 after therapy with anti-CTLA 4 monoclonal antibody. J Immunother (hagerstown Md: 1997) 30(1):130. https://doi.org/10.1097/01.cji.0000211334.06762.89
Tang T, Abu-Sbeih H, Ma W, Lu Y, Luo W, Foo WC, Richards DM, Halperin DM, Ge PS, Wang Y (2020) Gastrointestinal injury related to antiangiogenesis cancer therapy. Clin Colorectal Cancer 19(3):e117–e123. https://doi.org/10.1016/j.clcc.2020.03.002
Tian Y, Abu-Sbeih H, Wang Y (2018) Immune checkpoint inhibitors-induced colitis. Adv Exp Med Biol 995:151–157. https://doi.org/10.1007/978-3-030-02505-2_7
Valerio L, Giani C, Agate L, Molinaro E, Viola D, Bottici V, Matrone A, Puleo L, Lorusso L, Cappagli V, Ribechini A, Elisei R (2021) Prevalence and risk factors of developing fistula or organ perforation in patients treated with lenvatinib for radioiodine-refractory thyroid cancer. Eur Thyroid J 10(5):399–407. https://doi.org/10.1159/000514182
Wu J, Shu P, He H, Li H, Tang Z, Sun Y, Liu F (2021) Predictors of mortality in patients with acute small-bowel perforation transferred to ICU after emergency surgery: a single-centre retrospective cohort study. Gastroenterol Report 10:goab054. https://doi.org/10.1093/gastro/goab054
Yasuda K, Tanaka T, Ishihara S, Otani K, Nishikawa T, Kiyomatsu T, Kawai K, Hata K, Nozawa H, Masui Y, Shintani Y, Watanabe T (2017) Intestinal perforation after nivolumab immunotherapy for a malignant melanoma: a case report. Surgical Case Reports 3(1):94. https://doi.org/10.1186/s40792-017-0370-7
Yeun SC (2022) EC: oncologic emergencies. BC Decker Inc, Hamilton
Zhou WF, Joseph SC, Berry R (2021) S1856 a life-threatening cause of diarrhea: a case of immune checkpoint inhibitor colitis. Am J Gastroenterol 116:S1856
Zou F, Abu-Sbeih H, Ma W, Peng Y, Qiao W, Wang J, Shah AY, Glitza Oliva IC, Piha-Paul SA, Thompson JA, Zhang HC, Thomas AS, Wang Y (2020) Association of chronic immune-mediated diarrhea and colitis with favorable cancer response. J Natl Compr Cancer Netw 19(6):700–708. https://doi.org/10.6004/jnccn.2020.7647
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
YW was the senior author of the study; she developed the concept, designed the study, interpreted the results, ensured the preservation of data accuracy and integrity at all stages, agreed to be accountable for all aspects of the study, oversaw the overall direction and planning of the study, and contributed to the writing of the manuscript, with input from all authors. APM collected the data for the study, conducted and interpreted the analysis, and wrote the manuscript. MS and CL helped with data collection and analysis. All authors critically revised the final version of the manuscript as well as had access to the study data and reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not received any financial consideration from any person or organization to support the preparation, analysis, results or discussion of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pizuorno Machado, A., Shatila, M., Liu, C. et al. Characteristics, treatment, and outcome of patients with bowel perforation after immune checkpoint inhibitor exposure. J Cancer Res Clin Oncol 149, 5989–5998 (2023). https://doi.org/10.1007/s00432-022-04569-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04569-y