Abstract
Introduction
Crohn’s disease can present with complex surgical pathologies, posing a significant risk of morbidity and mortality for patients. The implementation of a loop ileostomy for selected patients may help minimize associated risks.
Methods
In this retrospective cohort study, we investigated the utilization of temporary fecal diversion through the creation of a loop ileostomy in Crohn’s surgery. Closure of all ostomies involved a hand-sewn single-layer technique. We then conducted bivariate analysis on 30-day outcomes for closures, focusing on favorable recovery defined as the restoration of bowel continuity without the occurrence of two challenges in recovery: newly developed organ dysfunction or the necessity for reoperation.
Results
In total, 168 patients were included. The median age of the patients was 38 years (IQR 27–51). The most common indication for a loop ostomy was peritonitis (49%). After ileostomy closure, 163 patients (97%) achieved favorable recovery, while five encountered challenges; four (2.4%) underwent abdominal surgery, and one (0.6%) developed acute renal failure requiring dialysis. Two patients (1.2%) had a re-creation of ileostomy. Patients encountering challenges were older (56 [IQR 41–61] vs. 37 [IQR 27–50]; p 0.039) and more often required secondary intention wound healing (40% vs. 6.7%; p 0.049) and postoperative parenteral nutrition following their index surgery (83% vs. 26%; p 0.006).
Conclusion
Selectively staging the Crohn’s disease operations with a loop ileostomy is a reliable practice with low morbidity and high restoration rates of bowel continuity. Our hand-sewn single-layer technique proves effective in achieving successful surgical recovery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Complex Crohn’s Disease (CD) surgery has a high-risk potential for septic complications. Given the high postoperative mortality in emergency settings (3.6% for CD) [1] the decision by an inflammatory bowel disease (IBD) surgeon to cool off active inflammation with a diverting ostomy may be driven by more than anecdote. Despite the broadly accepted ability of diversion to provide a good window of opportunity to control a severe penetrating disease [2], correct malnutrition [3] and reduce immune suppression [3, 4], some patients and clinicians still remain adamant about rejecting a stoma or feel uncertain about its indication. A severe aversion or uncertainty of a diverting ostomy might be due to the fear of stigmatization or a disconnect among gastroenterologists, general surgeons and subspecialized IBD surgeons. However, the hazard inherent to the ostomy itself is less devastating than a full-blown surgical site contamination such as generalized peritonitis [5] and the stoma can be reversed in most cases with minimal additional complications [6].
Historically, multiple studies investigated the morbidity following closure of a diverting loop ileostomy (DLI), but it is still unclear what proportion of patients suffer from conditions as a result of their index surgery [7] Furthermore, from the most straight forward over-and-over stitches to the most advanced staplers, there are many predefined closure techniques for loop ileostomies [8]. The various different treatments of the opposite bowel walls of prospected anastomotic rim or the selection between continuous and interrupted placement of sutures contribute to this variability [9, 10]. Adding further complexity in the comparisons of outcomes, some surgeons prefer a double-layer anastomosis for closures of loop ileostomy [10,11,12].
Proven to be successful even in small-diameter bowel anastomosis of infancy [13] or Heinike-Mikulicz strictureplasty of IBD surgery [14], single-layer anastomosis fashioned in an interrupted style is an appropriate alternative technique to perform end-to-end anastomosis in the closures. Despite being widely used and having gained acceptance by some as a gold standard, diversity of suturing techniques still poses significant challenges in comparing and categorizing this method under a single, overarching label of “interrupted single layer anastomosis” [11, 15, 16].
Our focus on single-layered interrupted sutures is driven by Crohn’s disease’s unique mesenteric and antimesenteric polarization, aimed at mitigating uneven tension distribution across the suture line and reducing the risk of stricture formation. By clarifying the technical distinctions and providing a more nuanced understanding of the treatments of the opposite bowel walls at the anastomotic rim of the closures, we examined our selective use of DLIs in complex surgical situations related to CD and analyzed the outcomes of our hand-sewn single-layer technique for loop ileostomy closures.
Methods
In this retrospective cohort study, we conducted a manual review of operative and clinical notes of patients registered in our institutional review board–approved CD surgery database at New York University Langone Health (IRB no: i22-00112). Eligibility for the study was confined to CD patients who underwent a DLI creation and subsequently underwent a single-layer interrupted handsewn DLI closure between September 2016 and September 2021. Exclusions comprised individuals who were diagnosed with CD following pouch surgery (n = 25), did not undergo closure within the designated time frame (n = 17), had their closures performed at external healthcare facilities (n = 2), or were closed using a different technique (n = 1). Figure 1 illustrates the complete patient selection flow diagram, including the Montreal classifications [17] of the selected cohort.
We systematically analyzed all pertinent demographic and clinical factors, including patient demographics, disease characteristics, surgical details, estimated indications for diversion, immunosuppression, complications, and other major perioperative features. By incorporating covariates from both the stoma creation (the index surgery) and its subsequent closure, we conducted bivariate analysis on 30-day outcomes for loop ostomy closures. Our emphasis was on favorable recovery, defined as the restoration of bowel continuity without facing two challenges in recovery—newly developed organ dysfunction or the necessity for reoperation.
We defined ‘initial procedures’ as the primary staged interventions for the treatment of IBD, whereas ‘additional procedures’ comprised subsequent interventions performed after the primary procedure. During both types of procedures, DLIs were surgically created at the index surgery, with closure scheduled for a subsequent session as an integral component of the procedure’s culmination stage (e.g., the index surgery was the second stage in a three-stage ileal pouch anal anastomosis [IPAA] procedure).
An anastomotic leak was defined as a discontinuity of tissue apposition in the anastomosis confirmed by a combination of clinical, radiologic, endoscopic and or operative findings. The definition of malnutrition was based on the GLIM 2018 criteria [18]. Biochemical diagnostic tests, such as serum albumin, were not routinely used for malnutrition screening. Both peritonitis and preoperative malnutrition were considered a threat to anastomotic integrity and to preventing sepsis-associated organ failure, serving as rationales for fecal diversion. Severity of the postoperative complications were graded according to the Clavien–Dindo classification (CDC) [19]. Delayed postoperative ileus was defined as obstipation and intolerance of oral intake that persist for more than three days following surgery. Small bowel obstruction (SBO) was defined as the presence of symptoms, signs, and radiographic evidence of mechanical small intestinal obstruction. Short bowel syndrome was describing reduced absorptive surface area for nutrients resulting in dependency to recurrent intravenous hydration episodes or a home parenteral nutrition support. Readmission was defined as an unplanned patient admission to a hospital within 30 days after being discharged.
Surgical technique
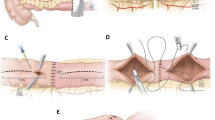
The ileostomy is closed by suturing the resultant antimesenteric defect in one layer using 3 − 0 absorbable braided polyglactin (2 − 0 if tissues are thick or inflamed) interrupted seromuscular sutures. In the case of poor tissue quality such as a large serosal defect, an enterotomy not amenable to repair or an ischemia, segmental small bowel resection is performed first and one layer end-to-end small bowel anastomosis is then performed using 3 − 0 or 2 − 0 absorbable braided polyglactin interrupted seromuscular sutures; Turnbull style sutures posteriorly and interrupted seromuscular sutures anteriorly (Fig. 2).
(A) Handsewn closure without small bowel resection. (B) Handsewn closure after small bowel resection; Turnbull style sutures posteriorly (Step-by-step description: (1) inside-out full thickness, (2) outside-in full thickness, (3) inside-out mucosa, (4) outside-in mucosa) and interrupted seromuscular sutures anteriorly
Statistical analyses
The data was analyzed using the Statistical Package for Social Sciences 28.0 software for Windows (SPSS; IBM, Armonk, NY, USA). Sankey diagram was created using the networkD3 and tidyverse packages at R version 4.2.1. The Kolmogorov-Smirnov test was used to determine the normality of continuous variables. Median (interquartile range [IQR]) was used for the presentation of non-parametric distributions. For categorical variables, χ2 or Fisher exact test and for continuous variables, Student’s t test or Mann–Whitney U test were used depending on the test assumptions. The comparison of clinical characteristics between patients with favorable recovery and those encountering challenges after a DLI closure involved a bivariate analysis. Only traits with a significance level of p < 0.5 were considered for display in the tabular presentations, and results with p-values less than 0.05 were deemed statistically significant.
Results
A total of 168 CD patients (64 female and 104 males) underwent handsewn loop ostomy closure and were followed up for a median of 1.6 years (IQR 0.5–2.8). The median patient age was 35 years (IQR 26–51). The median patient body mass index (BMI) was 23 kg/m2 (IQR 20–26). There were 7 (4.2%) American Society of Anesthesiologists (ASA) class I, 136 (81%) class II and 25 (15%) class III patients. Median time between the index surgery and the closure was 3.0 months (IQR 3.0–4.0). The median duration of inflammatory bowel disease was 9.0 years (IQR 3.0–20). The diagnosis of Crohn’s disease was established following the conversion from ulcerative colitis (UC) in 10 patients.
The foremost goal of a DLI in 162 patients (96%) was to safeguard at least one simultaneously anastomosed bowel segment. Of the remaining six patients, ileostomy was dedicated to alleviating inflammation in three cases and addressing strictures in the other three. The most common indication for a DLI creation was peritonitis (49%). The prevalence of multiple indications was 42%. Table 1 delineates the estimated indications for the establishment of a DLI in Crohn’s disease. Furthermore, Fig. 3 illustrates the distribution of patients with different indications using a traceable multi-level Sankey chart, specifically highlighting groups comprising 3 or more individuals while excluding less populated groups.
A total of 119 (71%) CD patients had received immunosuppressive treatment at some point during the 12-week period preceding the creation of a DLI. Of these, 97 (58%) patients were prescribed biologics, 61 (36%) patients were receiving corticosteroid treatment, and 39 (23%) patients were receiving a combination of both biologics and corticosteroids. At the time of DLI closure, 84 (50%) CD patients were identified as having undergone a pattern of discontinuation of immunosuppressive agents. Among patients who had been on immunosuppression within 12 weeks prior to DLI creation, the rate of discontinuation, specifically the proportion of patients who had ceased immunosuppressive agents by the time of the closure surgery, was found to be 71% (84/119).
At the index surgery, preoperative malnutrition was present in 31 patients (18%), peritonitis was observed in 83 patients (49%), and at least one fistula takedown was carried out in 93 (55%) procedures; entero-colonic fistula was the most common fistula type (55/93). Of the patients with entero-colonic fistula, 64% (35/55) had aggressive Crohn’s disease involvement or complications at the descending colon/sigmoid and underwent anterior resection/left hemicolectomy. Among patients with perianal Crohn’s involvement at the index surgery, a perianal complex fistula served as the sole indication for DLI in 27% (3/11) of the cases; one of these three patients underwent a proctectomy. An ileocolic resection was necessary in 68% of patients with fistulizing disease (63/93). One patient underwent IPAA. Baseline clinical characteristics of patients at the DLI closure are shown in Table 2. Complications between the index surgery and the ileostomy closure are presented in Table 3.
At the time of the DLI closure surgery, 8 patients (4.8%) underwent an additional small bowel resection because their ostomies were not suitable for anastomosis after mobilization. The ileostomy was closed without a resection in 160 patients (95%). A regular circumstomal skin incision was enough in 159 patients (95%). One patient (0.6%) needed an extended circumstomal incision and 8 patients (4.8%) needed a circumstomal plus a midline incision.
After the closure, 21 patients (12%) had postoperative complications within thirty days of the operation. Of these patients, 11 patients (6.5%) experienced only minor deviations (e.g. a mild episode of self-limited nausea/vomiting) from the normal postoperative course (CDC-I). Two patients (1.2%) needed medication, blood transfusion or total parenteral nutrition without any interventional procedures or sepsis (CDC-II). Seven patients (4.2%) needed a surgical, endoscopic or radiologic intervention but didn’t have sepsis (CDC-III). One patient (0.6%) had sepsis with single organ dysfunction (CDC-IV).
Thirty days after the DLI closure, 163 patients (97.0%) achieved favorable recovery. Restoration of bowel continuity was possible in 166 patients (98.8%). Five patients encountered challenges; four (2.4%) underwent abdominal surgery, and one (0.6%) developed acute renal failure requiring dialysis. Two of the four patients who underwent abdominal surgery had a re-creation of ileostomy due to anastomotic leak. Postoperative 30-day outcomes after the DLI closures are displayed in Table 4. Patients encountering challenges were older (57 [IQR 43–62] vs. 35 [IQR 26–49]; p 0.040), more often required secondary intention wound healing at the index surgery (40% vs. 6.7%; p 0.049) and more frequently needed postoperative parenteral nutrition within the 30-day period following their index surgery (100% vs. 28%; p 0.002) (Table 5).
Discussion
Specifically tailored for patients confronting the intricacies of CD, our study represents a pivotal exploration into the safety and efficacy of the interrupted single-layer loop ileostomy closure technique. Remarkably, 97% of cases achieved a favorable recovery. This signifies the restoration of bowel continuity without the emergence of newly developed organ dysfunction or the necessity for subsequent reoperation.
Traditionally in CD surgery, a deliberate temporary DLI serves as a bridge to definitive bowel resection, effectively mitigating inflammation and optimizing patient well-being and nutrition [20]. The plan for reversal within 3-to-6 months can prove unattainable in a significant proportion of initially designated transient loop enterostomies [21]. However, recent advancements in CD management and the utilization of sophisticated surgical techniques, incorporating upstream temporary stomas, have resulted in a decrease in the incidence of permanent stomas [22]. The incorporation holds the potential to maintain the integrity of bowel anastomosis distal to the diversion site. This potential aligned significantly with the primary goal of implementing a DLI in our study. Among patients exclusively deemed eligible for closure and who underwent reversal surgery, only 1.2% had their ileostomy recreated.
With the integration of biologics into CD treatment, many aspects of diversion, including stoma reversal rates [22, 23], the psychosocial impacts [24], disease recurrence [20, 25], and stoma-related complications [21], are thoroughly reassessed. However, even in the era of biologics, there exists a significant gap in our understanding of the outcomes precisely related to the intended plan of closing the temporary stoma when patients proceed to surgery for closure. This gap is further enlarged by contemporary medical ethics constraints, preventing the safe conduct of a well-matched controlled study to compare the protective effect size of temporary diversion in the immediate postoperative phase of bowel resections in CD cohorts. Consequently, our study significantly enriches the literature by showcasing the absence of mortality in 168 staged complex CD procedures, coupled with a tangible outcome of 98.8% bowel continuity. This stands as a testament to the safety and preeminence of staging complex CD operations with a DLI.
In our study, anastomotic dehiscence occurred in 2 patients (1.2%), and postoperative bowel obstruction developed in 9 patients (5.4%), with 8 out of 9 responding to conservative treatment. These complication rates were slightly lower than those reported in a recent systematic review of morbidity, which included 2146 ileostomy closures (56.6% handsewn closure, 3.5% CD) [26]. The review, without specifying the type of anastomosis, noted rates of 2.0% for anastomotic dehiscence and 7.6% for postoperative bowel obstruction after closures.
While various techniques exist for ileostomy closure following IBD surgery [12], the end-to-end handsewn ileostomy closure stands out as one of the most commonly employed methods in loop ileostomy reversal [27]. Its appeal lies in its capacity to preserve bowel anatomy with minimal loss, offering the IBD surgeon the opportunity for reoperative procedures, including potential redo pouch surgeries [28]. This aspect significantly influenced our surgical strategy. The consideration of employing techniques other than end-to-end handsewn ileostomy closure underscores potential challenges, especially in the context of recurrent CD pathology. Notably, a stapled side-to-side anastomosis may entail additional bowel length loss or hinder patients from undergoing advanced procedures, particularly in the setting of reoperations [29]. This consideration gains significance for patients undergoing IPAA, given the observed 20% failure rate for initial pouches at the 10-year mark post-proctocolectomy in individuals with CD [28].
A comprehensive meta-analysis of randomized controlled trials stands out as the main reference point for proponents of stapled closures [8]. By comparing stapled anastomoses to handsewn anastomoses it has demonstrated that stapled closures were reducing the 30-day rate of bowel obstruction; 6.4% with stapler and 12.9% with handsewn. In our study, we observed an overall bowel obstruction rate of 5.4%. This leads us to posit that, with the expertise gained in high-volume centers, the occurrence of bowel obstruction events can be minimized, potentially approaching the rates seen with stapled anastomoses.
It’s important to note that none of the randomized controlled trials [30,31,32,33] included in this meta-analysis [8] delved into the specifics of complications at the index surgery, a limitation also found in large retrospective national studies comparing stoma closure techniques [34]. If future comparative studies are designed specifically for CD, a meticulous exploration is warranted, scrutinizing the intricacies of index surgery in the context of CD involvement. Furthermore, it is imperative for upcoming studies to allocate interventions judiciously, avoiding unnecessary risks of additional bowel loss while safeguarding the potential for future ileal pouch creation.
According to one of the largest morbidity and mortality series of loop ileostomy closures from the United Kingdom, a complication occurring after the index surgery was associated with anastomotic leakage at closures [35]. Although the principle was being developed in the context of this UK series, complications were lacking a deep level of granularity but presented as a single risk group. In another RCT, on the other hand, the index complications were granular and scaled similar to our study but it was not designed to address the impact of previous complications on the closure outcomes but to compare early versus late closures [36]. Besides, only patients without clinical or radiological signs of adverse events after the index operation were included and randomized in the trial. Although the complete picture remains to be fully understood, the influence of the role of the index surgery was evident in our findings. Thus, we believe close surveillance of complicated patients from the index CD surgery is the key for an enhanced planning of the following DLI closure surgery. Poor adherence to surveillance may lead to adverse consequences particularly for elderly patients, those requiring secondary intention wound healing at the index surgery and patients needing postoperative parenteral nutrition within the 30-day period following their index surgery.
In navigating the spectrum of diversion options— from a split stoma to an end stoma with a mucous fistula or a blind stump — IBD surgeons must possess a nuanced understanding, preserving these tools for specialized scenarios, particularly when a patient’s tolerance for anastomotic complications is minimal [37]. Yet, it’s crucial to recognize that alternatives to a DLI can heighten the risk of stoma complications, like retraction. Employing a DLI with a stoma rod, a fixture routinely embraced by our surgical team, serves as a barrier against the pull of remnant phlegmonous tissues and anticipated postoperative edema, thereby promoting a mesenteric tension around the stoma. Even amidst scenarios favoring end ileostomies for Crohn’s, opting for a loop-end ileostomy may present as a clinically prudent course of action, especially when crafting an end ileostomy proves technically demanding. This holds particularly true in scenarios where accessing the mesentery poses challenges, such as restricted reach [38].
We note that this study has several limitations due to its single center setting and retrospective design despite the use of prospectively collected data. The higher proportion of patients with abdominal Crohn’s disease undergoing surgical procedures including the creation of a loop ileostomy at our center may be attributed to our role as a quaternary center specializing in IBD. As a result, we often receive referrals for more complex cases requiring surgical intervention. In addition, it’s crucial to note that our study exclusively comprises patients deemed eligible for DLI closure and who proceeded to reversal surgery, meaning that the reported bowel restoration rates do not account for those patients who had a fecal diversion but failed to undergo restoration surgery. This limitation underscores the need for further exploration to comprehensively evaluate the outcomes of loop ileostomy in the broader context of stoma management in CD patients. Regarding the shift in IBD diagnosis post-pouch surgery, the majority of IPAA patients experienced a transition to Crohn’s disease, emphasizing the inherent complexities of Crohn’s disease pathophysiology. However, with advancements in our understanding of the underlying biological pathways, we anticipate that such limitations may gradually attenuate. Additionally, it’s important to acknowledge that our study evaluates corticosteroid use as a single group, without distinguishing based on dosages. This limitation restricts our ability to infer the impact of dosage variations on postoperative outcomes.
The detailed DLI creation and closure strategy set forth in the present paper contributes to the evidence on optimal management of CD patients. The end-to-end, interrupted and single-layer handsewn closures for DLIs created using a liberal and selective approach demonstrated safety and yielded positive outcomes. These included a 98.8% restoration of bowel continuity and a 97.0% recovery without the occurrence of adverse events such as newly developing organ dysfunction or the need for reoperation. In the face of the series of challenges brought on by intense inflammatory conditions encountered at a quaternary IBD referral center, we remain reluctant to consider the liberal use of diversions in CD surgery to be an “overused” practice.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Change history
05 September 2024
This article has been updated to correct the author’s family name from “Moreira” to “da Luz Moreira”.
References
Singh S, Al-Darmaki A, Frolkis AD et al (2015) Postoperative mortality among patients with inflammatory Bowel diseases: a systematic review and Meta-analysis of Population-Based studies. Gastroenterology 149:928–937. https://doi.org/10.1053/j.gastro.2015.06.001
Strong S, Steele SR, Boutrous M et al (2015) Clinical practice guideline for the surgical management of Crohn’s disease. Dis Colon Rectum 58:1021–1036. https://doi.org/10.1097/DCR.0000000000000450
Adamina M, Bonovas S, Raine T et al (2020) ECCO Guidelines on therapeutics in Crohn’s Disease: Surgical Treatment. J Crohns Colitis 14:155–168. https://doi.org/10.1093/ecco-jcc/jjz187
Coccolini F, Improta M, Sartelli M et al (2021) Acute abdomen in the immunocompromised patient: WSES, SIS-E, WSIS, AAST, and GAIS guidelines. World J Emerg Surg 16:1–21. https://doi.org/10.1186/s13017-021-00380-1
Lightner AL, Pemberton JH (2017) The role of Temporary Fecal Diversion. Clin Colon Rectal Surg 30:178–183. https://doi.org/10.1055/s-0037-1598158
Brown SR, Fearnhead NS, Faiz OD et al (2018) The Association of Coloproctology of Great Britain and Ireland consensus guidelines in surgery for inflammatory bowel disease. Colorectal Dis 20:3–117. https://doi.org/10.1111/codi.14448
Chow A, Tilney HS, Paraskeva P et al (2009) The morbidity surrounding reversal of defunctioning ileostomies: a systematic review of 48 studies including 6,107 cases. Int J Colorectal Dis 24:711–723. https://doi.org/10.1007/s00384-009-0660-z
Löffler T, Rossion I, Gooßen K et al (2015) Hand suture versus stapler for closure of loop ileostomy—a systematic review and meta-analysis of randomized controlled trials. Langenbecks Arch Surg 400:193–205. https://doi.org/10.1007/s00423-014-1265-8
Chen C (2012) The art of bowel anastomosis. Scand J Surg 101:238–240. https://doi.org/10.1177/145749691210100403
Sajid MS, Siddiqui MRS, Baig MK (2012) Single layer versus double layer suture anastomosis of the gastrointestinal tract. Cochrane Database Syst Reviews. https://doi.org/10.1002/14651858.cd005477.pub4
Shikata S, Yamagishi H, Taji Y et al (2006) Single- versus two- layer intestinal anastomosis: a meta-analysis of randomized controlled trials. BMC Surg 6. https://doi.org/10.1186/1471-2482-6-2
Luglio G, Pendlimari R, Holubar SD et al (2011) Loop Ileostomy reversal after Colon and rectal surgery: a single institutional 5-year experience in 944 patients. Arch Surg 146:1191–1196. https://doi.org/10.1001/archsurg.2011.234
Ross AR, Hall NJ, Ahmed SA, Kiely EM (2016) The extramucosal interrupted end-to-end intestinal anastomosis in infants and children; a single surgeon 21 year experience. J Pediatr Surg 51:1131–1134. https://doi.org/10.1016/j.jpedsurg.2015.11.018
Pocivavsek L, Efrati E, Lee KYC, Hurst RD (2013) Three-dimensional geometry of the heineke-mikulicz strictureplasty. Inflamm Bowel Dis 19:704–711. https://doi.org/10.1097/MIB.0b013e3182802be3
Leslie A, Steele RJC (2003) The interrupted serosubmucosal anastomosis - still the gold standard. Colorectal Dis 5:362–366. https://doi.org/10.1046/j.1463-1318.2003.00460.x
Wong KS, Remzi FH, Gorgun E et al (2005) Loop ileostomy closure after restorative proctocolectomy: Outcome in 1,504 patients. Dis Colon Rectum 48:243–250
Satsangi J, Silverberg MS, Vermeire S, Colombel JF (2006) The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut 55:749–753
Cederholm T, Jensen GL, Correia MITD et al (2019) GLIM criteria for the diagnosis of malnutrition – A consensus report from the global clinical nutrition community. J Cachexia Sarcopenia Muscle 10:207–217. https://doi.org/10.1002/jcsm.12383
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Mennigen R, Heptner B, Senninger N, Rijcken E (2015) Temporary fecal diversion in the management of colorectal and perianal Crohn’s disease. Gastroenterol Res Pract 2015. https://doi.org/10.1155/2015/286315
Hirsch A, Yarur AJ, Dezheng H et al (2015) Penetrating Disease, narcotic use, and Loop Ostomy are Associated with Ostomy and IBD-related complications after ostomy surgery in Crohn’s Disease patients. J Gastrointest Surg 19:1852–1861. https://doi.org/10.1007/s11605-015-2908-y
Coscia M, Gentilini L, Laureti S et al (2013) Risk of permanent stoma in extensive Crohn’s colitis: the impact of biological drugs. Colorectal Dis 15:1115–1122. https://doi.org/10.1111/codi.12249
Müller C, Bergmann M, Stift A et al (2020) Restoration of intestinal continuity after stoma formation for Crohn’s disease in the era of biological therapy: a retrospective cohort study. Wien Klin Wochenschr 132:12–18. https://doi.org/10.1007/s00508-019-01586-9
Knowles SR, Wilson J, Wilkinson A et al (2013) Psychological well-being and quality of life in Crohn’s disease patients with an ostomy a preliminary investigation. J Wound Ostomy Cont Nurs 40:623–629. https://doi.org/10.1097/01.WON.0000436670.56153.7b
Koriche D, Gower-Rousseau C, Chater C et al (2017) Post-operative recurrence of Crohn’s disease after definitive stoma: an underestimated risk. Int J Colorectal Dis 32:453–458. https://doi.org/10.1007/s00384-016-2707-2
Mennigen R, Sewald W, Senninger N, Rijcken E (2014) Morbidity of Loop Ileostomy Closure after Restorative Proctocolectomy for Ulcerative Colitis and familial adenomatous polyposis: a systematic review. J Gastrointest Surg 18:2192–2200. https://doi.org/10.1007/s11605-014-2660-8
Markides GA, Wijetunga IU, Brown SR, Anwar S (2015) Meta-analysis of handsewn versus stapled reversal of loop ileostomy. ANZ J Surg 85:217–224. https://doi.org/10.1111/ans.12684
Fazio VW, Kiran RP, Remzi FH et al (2013) Ileal pouch anal anastomosis: analysis of outcome and quality of life in 3707 patients. Ann Surg 257:679–685. https://doi.org/10.1097/SLA.0b013e31827d99a2
Schwartzberg DM, Esen E, Remzi FH (2020) Thoughtful ileostomy creation in patients undergoing Redo IPAA. Dis Colon Rectum 63:117–120. https://doi.org/10.1097/DCR.0000000000001535
Hull TL, Kobe I, Fazio VW (1996) Comparison of handsewn with stapled loop ileostomy closures. Dis Colon Rectum 39:1086–1089. https://doi.org/10.1007/BF02081405
Hasegawa H, Radley S, Morton DG, Keighley MR (2000) Stapled versus sutured closure of loop ileostomy: a randomized controlled trial. Ann Surg 231:202–204. https://doi.org/10.1097/00000658-200002000-00008
Löffler T, Rossion I, Bruckner T et al (2012) HAnd suture Versus STApling for Closure of Loop Ileostomy (HASTA Trial): results of a multicenter randomized trial (DRKS00000040). Ann Surg 256:826–828. https://doi.org/10.1097/SLA.0b013e318272df97
Shelygin YA, Chernyshov SV, Rybakov EG (2010) Stapled ileostomy closure results in reduction of postoperative morbidity. Tech Coloproctol 14:19–23. https://doi.org/10.1007/s10151-009-0550-y
de Paula TR, Nemeth S, Kiran RP, Keller DS (2020) Predictors of complications from stoma closure in elective colorectal surgery: an assessment from the American College of Surgeons National Surgical Quality Improvement Program (ACSNSQIP). Tech Coloproctol 24:1169–1177. https://doi.org/10.1007/s10151-020-02307-5
Saha AK, Tapping CR, Foley GT et al (2009) Morbidity and mortality after closure of loop ileostomy. Colorectal Dis 11:866–871. https://doi.org/10.1111/j.1463-1318.2008.01708.x
Danielsen AK, Park J, Jansen JE et al (2017) Early closure of a temporary ileostomy in patients with rectal cancer: a multicenter randomized controlled trial. Ann Surg 265:284–290. https://doi.org/10.1097/SLA.0000000000001829
Myrelid P, Söderholm JD, Olaison G et al (2012) Split Stoma in resectional surgery of high-risk patients with ileocolonic Crohn’s disease. Colorectal Dis 14:188–193. https://doi.org/10.1111/j.1463-1318.2011.02578.x
Shen B (2018) Chap. 12 - Endoscopic Evaluation of Surgically Altered Bowel in Patients With Inflammatory Bowel Diseases. In: Shen B (ed) Interventional Inflammatory Bowel Disease: Endoscopic Management and Treatment of Complications. Academic Press, pp 141–159
Funding
No funding was used for this study.
Author information
Authors and Affiliations
Contributions
V.D., U.A. E.E. and F.H.R. drafted the manuscript, with V.D. also preparing figures and both V.D. and U.A. preparing tables, followed by all authors collectively reviewing, revising, and giving the manuscript its final shape.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doğru, V., Akova, U., Esen, E. et al. Temporary diverting loop ileostomy in Crohn’s disease surgery; indications and outcome. Langenbecks Arch Surg 409, 247 (2024). https://doi.org/10.1007/s00423-024-03404-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-024-03404-x