Abstract
Background
Static stretching (SS) can impair performance and increase range of motion of a non-exercised or non-stretched muscle, respectively. An underdeveloped research area is the effect of unilateral stretching on non-local force output.
Objective
The objective of this review was to describe the effects of unilateral SS on contralateral, non-stretched, muscle force and identify gaps in the literature.
Methods
A systematic literature search following preferred reporting items for systematic review and meta-analyses Protocols guidelines was performed according to prescribed inclusion and exclusion criteria. Weighted means and ranges highlighted the non-local force output response to unilateral stretching. The physiotherapy evidence database scale was used to assess study risk of bias and methodological quality.
Results
Unilateral stretching protocols from six studies involved 6.3 ± 2 repetitions of 36.3 ± 7.4 s with 19.3 ± 5.7 s recovery between stretches. The mean stretch-induced force deficits exhibited small magnitude effect sizes for both the stretched (−6.7 ± 7.1%, d = −0.35: 0.01 to −1.8) and contralateral, non-stretched, muscles (−4.0 ± 4.9%, d = , 0.22: 0.08 to 1.1). Control measures exhibited trivial deficits.
Conclusion
The limited literature examining non-local effects of prolonged SS revealed that both the stretched and contralateral, non-stretched, limbs of young adults demonstrate small magnitude force deficits. However, the frequency of studies with these effects were similar with three measures demonstrating deficits, and four measures showing trivial changes. These results highlight the possible global (non-local) effects of prolonged SS. Further research should investigate effects of lower intensity stretching, upper versus lower body stretching, different age groups, incorporate full warm-ups, and identify predominant mechanisms among others.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A preponderance of published literature has reported that prolonged static stretching (SS: > 60 s per muscle group) in isolation (without a full warm-up including prior and subsequent dynamic activities) can increase joint range of motion (ROM) and induce subsequent muscle strength (force), power, endurance, and sprint performance impairments in the stretched muscle or muscle groups (Behm and Chaouachi 2011; Behm et al. 2016a, 2020; Kay and Blazevich 2012). The mechanisms underlying SS-induced performance deficits have been attributed to both morphological and neural influences (Behm et al. 2020). Morphological mechanisms may include alterations in passive and active muscle force possibly indicating a reduction in the stiffness of the muscle’s parallel elastic components (Kay and Blazevich 2009; Kay et al. 2015; Konrad et al. 2017a, b), reduced myofibrillar Ca2+ sensitivity (i.e., due to metabolite accumulation) (Stephenson and Williams 1985; Sugi et al. 2013), and possibly changes in titin properties (Herzog 2014; Lee et al. 2007; Leonard and Herzog 2010). Neural mechanisms have been attributed to the exteroceptive reflex stimulation reducing excitatory sympathetic nervous activity (Guissard et al. 2001; Wu et al. 1999) and reduced strength of motoneurone dendrites persistent inward currents (PIC: (decreasing the amplification of central neural drive) (Trajano et al. 2017, 2020). Although stretch-induced neural inhibition is often hypothesised, a recent review by Behm and colleagues (Behm et al. 2020) found that SS effects on electromyographic (EMG) activity are conflicting, with both decrements and no changes reported, although when the EMG signal is normalised to the compound muscle action potential (M-wave) there are more consistent impairments. In addition, corticospinal excitability (Pulverenti et al. 2019) and cortical silent period (Pulverenti et al. 2020) are not substantially affected by stretching. Whereas morphological mechanisms can only apply to the stretched muscle (local effects), neural mechanisms can influence both local and non-local responses. Thus, in addition to the possible reductions in sympathetic nervous activity and PIC strength, an alternative method of examining the extent of global or non-local neural effects of SS would be to investigate crossover or non-local muscle performance responses.
There is evidence of non-local (contralateral homologous or heterologous muscles) improvements in ROM following a bout of unilateral stretching (Behm et al. 2016b, 2019; Caldwell et al. 2019a, b; Ce et al. 2020; Chaouachi et al. 2017; Clark et al. 1999; De-la-Cruz-Torres et al. 2020; Killen et al. 2019; Lima et al. 2014; Whalen et al. 2019; Wilke et al. 2016b, 2017). Global (whole body) mechanisms contributing to these non-local improvements in ROM have been postulated to be related to psycho-physiological influences (i.e., increased stretch or pain tolerance) (Magnusson et al. 1996, 1997) and the downregulation of sympathetic excitatory nerve stimulation (Behm 2018). Prolonged SS (i.e., 30 s or more) can also reduce motoneuron spindle reflexive (excitatory) activity (Avela et al. 1999; Guissard et al. 2001). Type I and II muscle spindle afferents innervate spinal motoneurons (Prochazka and Ellaway 2012) and project to the somatosensory and the primary motor cortex (Phillips et al. 1971; Rathelot and Strick 2009) and thus could have global consequences. The global impact of decreased sympathetic nervous system stimulation and disfacilitation of excitatory spindle afferents (neural inhibition) could potentially adversely affect the performance of non-stretched muscles (Behm 2018; Behm et al. 2020).
There is evidence of non-local SS-induced isometric maximum voluntary contraction (MVC) force (Behm et al. 2019), jump height (Marchetti et al. 2014) and muscle activation (Marchetti et al. 2017) deficits following unilateral SS. However, these results contrast with those that show no significant impairments of isokinetic torque following eight repetitions of 30 s each of unilateral hip flexion static stretching (Chaouachi et al. 2017). Jelmini et al. (2018) reported no change in handgrip force or EMG activity after three static stretching repetitions of 45 s each of the finger flexors, but did find a 10.8% decrease in rate of force generation. In contrast, Caldwell et al. (Caldwell et al. 2019a) found a near significant, small magnitude improvement (p = 0.06, d = 0.22, 12.1%) in contralateral drop jump height and no change in ground contact time following four repetitions of 30 s of unilateral SS. Hence, with this conflict in the literature it is not known whether there is sufficient evidence to substantiate that unilateral SS generally has global (contralateral or non-local) consequences (i.e., positive, negative or no significant effect) on performance.
The possibility of non-local force impairments from unilateral stretching may impact training and rehabilitation. Sustained stretching of one muscle group not only improves ROM of other non-stretched muscles (Behm et al. 2016b) but may also impair force and power. The sequence of training may be a consideration since for example, lower body stretching could negatively affect subsequent upper body strength and vice versa. The objective of this scoping review was to investigate and summarize the literature on non-local stretching effects on force and power (i.e., jump height) output to determine whether the prolonged SS-induced impairments observed with stretched limbs are generally present in non-local, non-stretched, muscle groups. In addition, we aimed to identify gaps in the literature to facilitate future lines of enquiry with regard to this research area.
Methods
Search strategy
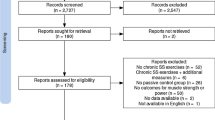
Literature searches following the Preferred Reporting Items for Systematic review and Meta-Analysis Protocols (PRISMA-P) review guidelines was performed by two co-authors (SA and DGB). These searches were conducted separately and independently using the PubMed, Scopus, SPORTDiscus, Web of Science, Cochrane, and CINHAL Plus databases. The topic was searched using a Boolean search strategy with the operator “OR” for the following title keywords: stretch, stretching, flexibility, range of motion, non-local, unilateral, contralateral, ipsilateral, crossover, or remote with the operator “AND” for terms such as performance, strength, force, power and jump (Fig. 1). Researchers’ personal computer databases were also examined for related articles. Related peer-reviewed publications written in English, spanned the period from 1999 to November 2020. The search syntax was saved in PubMed so that we received regular updates on the publication of further relevant studies. The selected articles were also cross referenced by the authors to identify relevant studies that might have been overlooked in the database search. In selecting studies for inclusion, a review of all relevant article titles was conducted before an examination of article abstracts and, then, full published articles (Fig. 1).
Data extraction
Data were extracted from gathered articles with a form created in Microsoft Excel. Where required data were not clearly or completely reported, article authors were contacted for clarification.
Inclusion and exclusion criteria (study selection)
Articles investigating non-local stretching and performance measures were included according to population, intervention, comparison and outcomes (PICOS) criteria (Table 1).
Systematic review analysis
Means and standard deviations for a strength (force or torque) performance measure were used to calculate an effect size (d = M1 −M2/SD pooled) (Cohen 1988). Effect sizes descriptors were trivial (< 0.2), small (0.2 to <0.5), moderate (0.5 to <0.8) and large (≥ 0.8) (Cohen 1988). With only six studies investigating the effects of unilateral stretching on non-local muscle performance (Table 2), the number of studies for a full, meaningful meta-analysis was considered low. Hence, weighted means ([effect size for study #1 × (number of participants/total participants) + effect size for study #2 × (number of participants/total participants) + …] = weighted mean of all selected studies) were calculated to illustrate the average response.
Assessment of risk of bias
The Physiotherapy Evidence Database (PEDro) scale was used to assess the risk of bias and methodological quality of the included studies (Table 3). This scale evaluates internal study validity on a scale from 0 (high risk of bias) to 10 (low risk of bias). Two reviewers (DGB and SA) independently rated each study. Any ratings that yielded different results were further adjudicated by a third reviewer. This rating was then used in the risk of bias scale. A median score of ≥ 6 represents the threshold for studies with a low risk of bias (Maher et al. 2003).
PEDro scale criteria are as follows:
-
Eligibility criteria were specified.
-
Subjects were randomly allocated to groups
-
Allocation was concealed.
-
The groups were similar at baseline regarding the most important prognostic indicators.
-
There was blinding of all subjects.
-
There was blinding of all therapists/researchers who administered the therapy/protocol.
-
There was blinding of all assessors who measured at least one key outcome.
-
Measures of at least one key outcome were obtained from more than 85% of the subjects initially allocated to groups.
-
All subjects for whom outcome measures were available received the treatment or control condition as allocated or, where this was not the case, data for at least one key outcome was analysed by “intention to treat”.
-
The results of between-group statistical comparisons are reported for at least one key outcome.
-
The study provides both point measures and measures of variability for at least one key outcome.
Results
Table 2 illustrates the individual study characteristics. The six studies were acute intervention studies with three studies involving males and three studies including both sexes. Five of the six studies were published in 2017 or thereafter. Participant characteristics and stretch intervention characteristics are provided in Tables 4 and 5, respectively. From the six studies, the average SS-induced force deficit of the stretched muscle (weighted means) was a small magnitude (−6.7 ± 7.1%, d = −0.35; 0.01 to −1.8), whereas the contralateral, non-stretched, muscle also showed a small magnitude effect size of (−4.0 ± 4.9%, d = −0.22; 0.08 to −1.1). The five control measures (no stretch intervention) from three studies (Behm et al. 2019; Caldwell et al. 2019a; Ce et al. 2020) exhibited a trivial strength deficit effect size of −0.03 (Fig. 2).
Discussion
The major finding from this scoping review was that both the stretched and contralateral, non-stretched limbs of the young adult participants in these studies experienced small magnitude, SS-induced force decrements. This finding highlights the global (non-local) effects of prolonged, unilateral, static stretching; in that the resultant force decrements experienced due to the stretching of a limb do not necessarily manifest only in that limb.
In accord with prior reviews, prolonged SS (> 60 s per muscle group) without a proper dynamic warm-up can induce small magnitude strength impairments (Behm 2018; Behm and Chaouachi 2011; Behm et al. 2016a, 2020; Chaabene et al. 2019; Kay and Blazevich 2012) in the stretched muscle. The typical stretching routine used in the studies in this review involved six repetitions of 36 s each for a total duration of approximately 216 s (3 min: 36 s), which exceeds the recommended maximum duration of 60 s of SS per muscle group. Furthermore, unlike the typical sport or training scenario, none of the studies included in this review included a full dynamic warm-up (prior aerobic activity and activity-specific dynamic activity following the stretching) as suggested in prior reviews (Behm et al. 2016a, 2020). Although these results should be expected for the stretched muscle based on the prolonged stretching interventions, this is the first review to report small magnitude, force impairments in a non-local, non-stretched muscle as a result of unilateral SS. Hence, the same SS routine characteristics that induce decrements in the stretched muscle can also incur deficits in a non-stretched muscle, but due to different mechanisms.
Although the average result indicated SS-induced non-local force impairments, an analysis of the individual studies demonstrates variable responses. Behm et al. (2019) imposed eight SS repetitions of 30 s each on the quadriceps and hamstrings and reported small magnitude (11.1%; d = −0.31) contralateral MVC force deficits. Similarly, 5 × 45 s repetitions of knee extensors SS generated large magnitude (−11.0%; d = −1.1) impairments of knee extensors MVC force (Ce et al. 2020). In contrast, there were trivial (1.2%; d = 0.08), non-significant contralateral knee flexion isokinetic torque changes following eight repetitions of 30 s each of unilateral hip flexion static stretching (Chaouachi et al. 2017), trivial (−4.9%; d = −0.17) knee flexion MVC force deficits following 10 × 30 s SS of the hamstrings (Killen et al. 2019) and trivial (−4.2%; d = −0.15) knee flexion MVC force deficits with four repetitions of 30 s SS of the hamstrings (Caldwell et al. 2019a). Jelmini et al. (2018) reported no change in handgrip force or EMG activity after three SS repetitions of 45 s each of the finger flexors, but did report a small magnitude, 10.8% (d = −0.29) decrease in rate of force generation.
Although jump performance (proxy measure of power) was not included in the present weighted mean results, there is also variability in these findings. Ten SS of 30 s each of the upper limbs (shoulder horizontal abduction) produced large magnitude deficits in countermovement jump ground reaction forces (−25%; d = 1.41) (Marchetti et al. 2014). In contrast, Caldwell et al. (2019a) found a near significant, small magnitude improvement (p = 0.06, d = 0.22, 12.1%) in contralateral drop jump height following four repetitions of 30 s of unilateral SS.
In summary, although, the weighted means illustrated a small magnitude, non-local force deficit, three measures indicated deficits in force output, while four measures showed only trivial changes. The substantially larger effect size of the Ce et al. (2020) study, in concert with the small magnitude impairments of the Behm et al. (2019) and Jelmini et al. (2018) studies counterbalanced the four trivial measures resulting in an overall mean small magnitude force deficit. Further research is necessary to strengthen the aggregate findings in the literature on the effect of different types of unilateral stretching on non-local force as well as power and other physiological and functional measures (i.e., sprint speed, sporting skills such as kicking and throwing). Morphological mechanisms would not be a consideration of non-local force or power impairments since the non-stretched muscle would not have experienced appreciable stress or strain. While Halperin et al. (2015) suggested four possible mechanisms contributing to non-local muscle fatigue including biochemical, biomechanical, neural or psycho-physiological factors, their relative contribution to non-local stretching force and power impairments may not be similar.
A fatigued muscle can accumulate a variety of metabolites such as potassium [alteration of electrochemical gradients inhibiting muscle excitability (Juel 1986; Nordsborg et al. 2003)], hydrogen ions (reduction in the force per cross bridge and myofibrillar Ca2 + sensitivity (Fitts 2008)), inorganic phosphates (Debold et al. 2016), reactive oxygen species (Reid 2016) among other metabolites. These metabolites dispersed from the exercised muscle can be transported to non-exercised muscles throughout the body (Bangsbo et al. 1996; Halperin et al. 2014; Johnson et al. 2014; Nordsborg et al. 2003) adversely affecting muscle contractility. However, a passive SS routine is unlikely to generate metabolic expenditures that would result in a substantial accumulation and global dispersal of force inhibiting metabolites (Colosio et al. 2020).
Biomechanical factors concern the fatigue of core or trunk muscles that stabilise the body when contracting a limb muscle. Decrements in core/trunk stability have been shown to substantially impair limb force output (Anderson and Behm 2005; Behm and Anderson 2006; Behm et al. 2010). This biomechanical mechanism is an unlikely factor as passive SS typically does not induce considerable disruptive, fatiguing torques to the trunk that need to be countered by active stabilization of the trunk muscles.
Neural inhibition has been postulated as a mechanism underlying stretch-induced performance impairments of the stretched limb (Behm 2018; Behm and Chaouachi 2011; Behm et al. 2016a, 2020) as well as a factor contributing to non-local muscle fatigue effects (Halperin et al. 2015). Using the interpolated twitch technique, voluntary activation of the contralateral quadriceps was reported to significantly decrease (−7%, d = −2.29) following 4 × 45 s of unilateral static stretching (Ce et al. 2020). However, EMG changes of the non-stretched muscles following unilateral stretching is conflicting. There are reports of very large (−11 to −22%, d = 2.25–3.9) (Ce et al. 2020) and small (−3.1%; d = −0.22) (Marchetti et al. 2017) magnitude reductions in EMG activity during a MVC, which contrast with reports of no significant changes in MVC EMG activity (Behm et al. 2019; Caldwell et al. 2019a; Jelmini et al. 2018; Killen et al. 2019). These difficulties in ascertaining EMG changes may be related to the curvilinearity of the force-EMG relationship with a plateau of EMG activity at higher force levels (Perry and Bekey 1981a, b). In the literature, there are conflicting effects of stretching on EMG activity of the stretched muscle (Behm et al. 2020), as well as a lack of substantial corticospinal excitability (Budini et al. 2017, 2019; Pulverenti et al. 2019) and intracortical inhibition (silent period) (Opplert et al. 2020; Pulverenti et al. 2019, 2020) effects. The short post-SS duration of inhibitory reflex activity and lack of corticospinal excitability changes, suggest the influence of neural inhibition on non-local strength is not conclusive.
However, according to the recent Behm et al. (2020) review, there may be two possible neural mechanisms associated with ROM increases of a stretched muscle; exteroceptive reflex stimulation, which can attenuate excitatory sympathetic nervous activity (Guissard et al. 2001; Wu et al. 1999) and reductions in motoneurone dendrites persistent inward currents (PIC) (decreasing the amplification of central neural drive (Trajano et al. 2017, 2020). Exteroceptive reflexes arising from cutaneous receptors have polysynaptic innervations to motoneurones (Jenner and Stephens 1982; Kearney and Chan 1999), which can inhibit sympathetic nervous system excitation (Wu et al. 1999), contributing to reductions in muscle tone, heart rate and blood pressure (van den Berg and Cabri 1999; Wu et al. 1999). However, exteroceptive reflex activity only persists for several seconds after stretching desists (Delwaide et al. 1981; Guissard et al. 2001). A similar response duration has been observed with the attenuation of facilitatory reflex activity from proprioceptive Ia and Ib afferents and thus their activity also seems to be too transient to persistently impair subsequent strength and power output. Hence, while strength deficits of a stretched or non-stretched muscle detected within seconds of a SS might be partially attributed to exteroceptive reflex influence on the sympathetic nervous system, deficits in the stretched or non-stretched muscles shortly thereafter would likely not be related to exteroceptive or proprioceptive reflexes. However, monoamines from sympathetic nervous system activity such as noradrenaline can affect motoneurone PIC activity that might impact non-local responses.
PICs can amplify (motoneurones fire at higher frequencies) and prolong synaptic input from supraspinal and reflex pathways augmenting motor output fivefold (Lee and Heckman 2000). SS might affect PIC-dependent amplification of central drive to the stretched muscle, which would diminish force production (Trajano et al. 2017, 2020). It has been found that SS-induced reductions in PICs were fully recovered by 10 min post-SS (Trajano et al. 2014a). These durations of impairment and recovery closely match the SS-induced deficits outlined in studies undertaken by Pulverenti et al. (2019, 2020) and Trajano and colleagues (Trajano et al. 2014b). However, these findings apply to the stretched muscle’s performance and it is unknown whether the diminution of the PICs would also be experienced by contralateral motoneurones affecting non-local muscle strength. PIC activity is augmented by monoamines such as serotonin and noradrenaline (released predominantly from sympathetic nerve fibres) (Lee and Heckman 2000). With a static stretching-induced reduction in sympathetic activity (Inami et al. 2014; Thomas et al. 2021), there would be a decrease in noradrenaline release globally, affecting non-local motoneurone PIC activity. In summary, there are conflicting effects of stretching on EMG activity (Behm et al. 2020), a lack of substantial corticospinal excitability (Budini et al. 2017, 2019; Pulverenti et al. 2019) and intracortical inhibition (Opplert et al. 2020; Pulverenti et al. 2019; 2020) effects, which in combination with the short post-SS duration of inhibitory reflex activity, suggest the effect of these neural factors or measures may not be the primary mechanism but need further investigation. The effects of unilateral stretching on contralateral motoneuron PIC activity may be a possible mechanistic candidate but these PIC effects have not been examined in non-stretched muscles.
Sustaining focus or concentration during prolonged SS would involve a level of cognitive demand to maintain joint position and stretch intensity (Behm et al. 2020). Prolonged cognitive activity can induce mental fatigue (impaired cognitive functioning), which can negatively affect subsequent performance, principally with prolonged activities (Marcora et al. 2009; Pageaux et al. 2013; 2014). Mental fatigue can lead to the perception that an activity is more demanding, and therefore an individual may succumb earlier or exert less effort (Marcora et al. 2009; Pageaux et al. 2013, 2014). Mental fatigue would not be specific to a single muscle or muscle group. Halperin et al. (2015) postulated that mental fatigue could be a major contributor to non-local muscle fatigue as non-local muscle fatigue is more apparent with prolonged fatiguing tests that would require sustained concentration. Although speculative, the concentration necessary to maintain a prolonged SS to or near the point of discomfort could induce some mental fatigue that would impair the ability of the individual to subsequently fully activate and co-ordinate the contraction of multiple muscle groups (activate agonists, synergists and core stabilizers, and inhibit antagonists muscles) to achieve maximal force output. Nonetheless, as there is a paucity of literature on non-local stretching effects, more research is needed on both the non-local responses and the possible mechanisms (i.e., neural inhibition and cortical fatigue).
Another possible mechanism contributing to non-local force and power deficits may be myofascial chains. Myofascial connective tissue is reported to furnish a network of myofascial chains or meridians (Wilke et al. 2016a). Human (Norton-Old et al. 2013; van Wingerden et al. 1993; Vleeming et al. 1995) and animal (Schleip et al. 2012; Yahia et al. 1993) cadaveric studies reveal that fascia can modify its stiffness and transfer stress to nearby structures. The myofascial transfer of force is conducted through longitudinal (Benetazzo et al. 2011; Eng et al. 2014) and spiral (oblique) (Myers 2001) lines as well as with transversal orientations to synergists and antagonists (Huijing et al. 2007; Maas et al. 2001; 2005; Meijer et al. 2007). Improved acute cervical ROM after hamstrings (Wilke et al. 2017) and gastrocnemius (Wilke et al. 2016b) stretching was accredited to myofascial longitudinal and spiral lines. But the scope of these transfer effects has been contested. Krause et al. (2016) proposed that myofascial strain is transferred predominantly to neighbouring skeletal muscles. The myofascial literature typically report force transmission that is distal to proximal or vice versa (i.e., stretch the plantar flexors and increase cervical ROM). The unilateral, stretch-induced, non-local muscle force and power impairments reported in this review occur primarily in contralateral muscles. Whereas longitudinal or spiral chains may affect force output of neighbouring muscles or along a longitudinal kinetic chain, they are unlikely to significantly contribute to contralateral limb muscle impairments.
Stretch-induced non-local force deficits can have implications for both training and rehabilitation. Individuals should be aware that prolonged high intensity stretching of one muscle group can not only increase ROM of other non-stretched muscles (Behm et al. 2016b) but may also impair force and power. For example, with training or rehabilitation, extensive lower body stretching prior to training could affect subsequent upper body force output (i.e., bench press or shoulder press) or prolonged upper body stretching could negatively impact the ability to sprint, jump or kick a ball. Stretch-induced impairments of a stretched muscle are trivial when less than 60 s of SS and a full warm-up is incorporated (Behm 2018; Behm and Chaouachi 2011; Behm et al. 2016a; 2020; Chaabene et al. 2019; Kay and Blazevich 2012), but it is unknown if the same parameters would nullify non-local stretching effects. These issues provoke the need for further research.
Conclusions
The limited scope of literature examining non-local effects of prolonged SS reveal that both the stretched and contralateral, non-stretched limbs of young adult participants demonstrate on average, small magnitude, strength deficits (Fig. 3). However, this assertion must be viewed with caution as the frequency of these effects were nearly equally divided with three measures demonstrating deficits, and four measures showing trivial changes. Possible mechanisms underlying SS-induced non-local force deficits may involve a combination of factors such as a decrease in sympathetic activation affecting overall central nervous system excitation and motoneurone PIC activity as well as a mental energy deficit (cortical fatigue) adversely affecting concentration to maximally activate and coordinate muscle contractions. Myofascial chains may affect the force output of neighbouring muscles or more distant muscles located along a longitudinal or spiral meridian. Unfortunately, there is insufficient evidence to identify a predominant mechanism(s). This review will hopefully act as a clarion to motivate researchers to add and contribute to the area of non-local stretching effects as it has implications for training and rehabilitation as well as insights into psychological and physiological mechanisms. Areas for future investigation include the volume, intensity and type of stretching that might elicit non-local force, power, speed and other performance impairments, whether similar to a stretched muscle; would a full warm-up nullify possible decrements, upper versus lower body stretching effects, possible differences associated with age, sex, trained state, muscle type, joint, and the underlying mechanisms.
a Mean characteristics of stretching protocols used in the reviewed studies. b Effects of unilateral static stretching (SS) on the stretched and contralateral non-stretched muscles. c Mechanisms potentially underpinning the SS-induced performance impairment. ES: mean effect size. d Gaps in the literature to be investigated by future research. CMJ: countermovement jump, DJ: drop jump, EMG: electromyography, GRF: ground reaction force
Data availability statement
All pertinent data are provided in the listed tables (PEDro scale analysis, individual and mean study characteristics) and figures (PRISMA flow chart).
Abbreviations
- EMG:
-
Electromyography
- MVC:
-
Maximal voluntary contraction
- PEDro:
-
Physiotherapy evidence database
- PIC:
-
Persistent inward currents
- PICOS:
-
Population, intervention, comparison and outcomes
- PRISMA-P:
-
Preferred reporting items for systematic review and meta-analysis protocols
- ROM:
-
Range of motion
- SS:
-
Static stretching
References
Anderson K, and Behm DG (2005) The impact of instability resistance training on balance and stability. Sports Med 35(1):43–53. http://www.ncbi.nlm.nih.gov/pubmed/15651912
Avela J, KyrîlÑinen H, Komi PV (1999) Altered reflex sensitivity after repeated and prolonged passive muscle stretching. J Appl Physiol 86(4):1283–1291
Bangsbo J, Madsen K, Kiens B, Richter EA (1996) Effect of muscle acidity on muscle metabolism and fatigue during intense exercise in man. J Physiol 495(Pt 2):587–596. https://doi.org/10.1113/jphysiol.1996.sp021618
Behm, DG (2018) The science and physiology of flexibility and stretching: implications and applications in sport performance and health, chaps 3–6. Routledge Publishers, London, UK
Behm DG, Anderson KG (2006) The role of instability with resistance training. J Strength Cond Res 20(3):716–722. https://doi.org/10.1519/R-18475.1
Behm DG, Chaouachi A (2011) A review of the acute effects of static and dynamic stretching on performance. Eur J Appl Physiol 111(11):2633–2651. https://doi.org/10.1007/s00421-011-1879-2
Behm DG, Drinkwater EJ, Willardson JM, Cowley PM (2010) The use of instability to train the core musculature. Appl Physiol Nutr Metab 35(1):91–108. https://doi.org/10.1139/H09-127
Behm DG, Blazevich AJ, Kay AD, McHugh M (2016a) Acute effects of muscle stretching on physical performance, range of motion, and injury incidence in healthy active individuals: a systematic review. Appl Physiol Nutr Metab 41(1):1–11. https://doi.org/10.1139/apnm-2015-0235
Behm DG, Cavanaugh T, Quigley P, Reid JC, Nardi PS, Marchetti PH (2016b) Acute bouts of upper and lower body static and dynamic stretching increase non-local joint range of motion. Eur J Appl Physiol 116(1):241–249. https://doi.org/10.1007/s00421-015-3270-1
Behm DG, Lau RJ, O’Leary JJ, Rayner MCP, Burton EA, Lavers L (2019) Acute effects of unilateral self-administered static stretching on contralateral limb performance. J Perform Health Res 3(1):1–7. https://doi.org/10.2503/jphr.2019.3.1.behm
Behm DG, Kay AD, Trajano GS, Blazevich AJ (2020) Mechanisms underlying performance impairments following prolonged static stretching without a comprehensive warm-up. Eur J Appl Physiol. https://doi.org/10.1007/s00421-020-04538-8
Benetazzo L, Bizzego A, De Caro R, Frigo G, Guidolin D, Stecco C (2011) 3D reconstruction of the crural and thoracolumbar fasciae. Surg Radiol Anat 33(10):855–862. https://doi.org/10.1007/s00276-010-0757-7
Budini F, Gallasch E, Christova M, Rafolt D, Rauscher AB, Tilp M (2017) One minute static stretch of plantar flexors transiently increases H reflex excitability and exerts no effect on corticospinal pathways. Exper Physiol 102(8):901–910
Budini F, Kemper D, Christova M, Gallasch E, Rafolt D, Tilp M (2019) Five minutes static stretching influences neural responses at spinal level in the background of unchanged corticospinal excitability. J Musculoskel Neuronal Interact 19(1):30–36
Caldwell SL, Bilodeau RLS, Cox MJ, Peddle D, Cavanaugh T, Young JD et al (2019a) Unilateral hamstrings static stretching can impair the affected and contralateral knee extension force but improve unilateral drop jump height. Eur J Appl Physiol 119(9):1943–1949. https://doi.org/10.1007/s00421-019-04182-x
Caldwell SL, Bilodeau RLS, Cox MJ, Behm DG (2019b) Twice daily, self-administered band stretch training improves quadriceps isometric force and drop jump characteristics. J Sport Sci Med 18:544–551
Ce E, Coratella G, Bisconti AV, Venturelli M, Limonta E, Doria C et al (2020) Neuromuscular versus mechanical stretch-induced changes in contra- versus ipsilateral muscle. Med Sci Sports Exerc. https://doi.org/10.1249/MSS.0000000000002255
Chaabene H, Behm DG, Negra Y, Granacher U (2019) Acute effects of static stretching on muscle strength and power: an attempt to clarify previous caveats. Front Physiol 10:1468–1472. https://doi.org/10.3389/fphys.2019.01468
Chaouachi A, Padulo J, Ben KS, Othmen A, Chatra M, Behm DG (2017) Unilateral static and dynamic hamstrings stretching increases contralateral hip flexion range of motion. Clin Physiol Funct Imaging 37(1):23–29. https://doi.org/10.1111/cpf.12263
Clark S, Christiansen A, Hellman DF, Hugunin JW, Hurst KM (1999) Effects of ipsilateral anterior thigh soft tissue stretching on passive unilateral straight-leg raise. J Orthop Sports Phys Ther 29(1):4–12. https://doi.org/10.2519/jospt.1999.29.1.4
Cohen J (1988) Statistical power analysis for the behavioural sciences. L. Erbraum Associates, Hillside, pp 48–96
Colosio AL, Teso M, Pogliaghi S (2020) Prolonged static stretching causes acute, nonmetabolic fatigue and impairs exercise tolerance during severe-intensity cycling. Appl Physiol Nutr Metab 45(8):902–910. https://doi.org/10.1139/apnm-2019-0981
Debold EP, Fitts RH, Sundberg CW, Nosek TM (2016) Muscle fatigue from the perspective of a single crossbridge. Med Sci Sports Exerc 48(11):2270–2280
De-la-Cruz-Torres B, Carrasco-Iglesias C, Minaya-Munoz F, Romero-Morales C (2020) Crossover effects of ultrasound-guided percutaneous neuromodulation on contralateral hamstring flexibility. Acupunct Med: https://doi.org/10.1177/0964528420920283
Delwaide PJ, Toulouse P, Crenna P (1981) Hypothetical role of long-loop reflex pathways. Appl Neurophysiol 44(1–3):71–176. http://www.ncbi.nlm.nih.gov/pubmed/7294778
Eng CM, Pancheri FQ, Lieberman DE, Biewener AA, Dorfmann L (2014) Directional differences in the biaxial material properties of fascia lata and the implications for fascia function. Ann Biomed Eng 42(6):1224–1237. https://doi.org/10.1007/s10439-014-0999-3
Fitts RH (2008) The cross-bridge cycle and skeletal muscle fatigue. J Appl Physiol 104(2):551–558. https://doi.org/10.1152/japplphysiol.01200.2007
Guissard N, Duchateau J, Hainaut K (2001) Mechanisms of decreased motoneurone excitation during passive muscle stretching. Exp Brain Res 137:163–169
Halperin I, Aboodarda SJ, Behm DG (2014) Knee extension fatigue attenuates repeated force production of the elbow flexors. Eur J Sport Sci 14(8):823–829. https://doi.org/10.1080/17461391.2014.911355
Halperin I, Chapman DW, Behm DG (2015) Non-local muscle fatigue: effects and possible mechanisms. Eur J Appl Physiol 115(10):2031–2048. https://doi.org/10.1007/s00421-015-3249-y
Herzog W (2014) The role of titin in eccentric muscle contraction. J Exp Biol 217(Pt 16):2825–2833. https://doi.org/10.1242/jeb.099127
Huijing PA, van de Langenberg RW, Meesters JJ, Baan GC (2007) Extramuscular myofascial force transmission also occurs between synergistic muscles and antagonistic muscles. J Electromyogr Kinesiol 17(6):680–689. https://doi.org/10.1016/j.jelekin.2007.02.005
Inami T, Shimizu T, Baba R, Nakagaki A (2014) Acute changes in autonomic nerve activity during passive static stretching. Amer J Sports Sci Med 2(4):166–170
Jelmini JD, Cornwell A, Khodiguian N, Thayer J, Araujo AJ (2018) Acute effects of unilateral static stretching on handgrip strength of the stretched and non-stretched limb. Eur J Appl Physiol 118(5):927–936. https://doi.org/10.1007/s00421-018-3810-6
Jenner JR, Stephens JA (1982) Cutaneous reflex responses and thier central nervous pathways studied in man. J Physiol 333:405–419
Johnson MA, Mills DE, Brown PI, Sharpe GR (2014) Prior upper body exercise reduces cycling work capacity but not critical power. Med Sci Sports Exerc 46(4):802–808. https://doi.org/10.1249/MSS.0000000000000159
Juel C (1986) Potassium and sodium shifts during in vitro isometric muscle contraction, and the time course of the ion-gradient recovery. Pflugers Arch 406(5):458–463. https://doi.org/10.1007/bf00583367
Kay AD, Blazevich AJ (2009) Isometric contractions reduce plantar flexor moment, Achilles tendon stiffness, and neuromuscular activity but remove the subsequent effects of stretch. J Appl Physiol 107(4):1181–1189. https://doi.org/10.1152/japplphysiol.00281.2009
Kay AD, Blazevich AJ (2012) Effect of acute static stretch on maximal muscle performance: a systematic review. Med Sci Sports Exerc 44(1):154–164. https://doi.org/10.1249/MSS.0b013e318225cb27
Kay AD, Husbands-Beasley J, Blazevich AJ (2015) Effects of contract-relax, static stretching, and isometric contractions on muscle-tendon mechanics. Med Sci Sports Exerc 47(10):2181–2190. https://doi.org/10.1249/MSS.0000000000000632
Kearney RE, Chan CWY (1999) Reflex response of human arm muscles to cutaneous stimulation of the foot. Brain Res 170:214–217
Killen BS, Zelizney KL, Ye X (2019) Crossover effects of unilateral static stretching and foam rolling on contralateral hamstring flexibility and strength. J Sport Rehabil 28(6):533–539. https://doi.org/10.1123/jsr.2017-0356
Konrad A, Stafilidis S, Tilp M (2017a) Effects of acute static, ballistic, and PNF stretching exercise on the muscle and tendon tissue properties. Scand J Med Sci Sports 27(10):1070–1080. https://doi.org/10.1111/sms.12725
Konrad A, Budini F, Tilp M (2017b) Acute effects of constant torque and constant angle stretching on the muscle and tendon tissue properties. Eur J Appl Physiol 117(8):1649–1656. https://doi.org/10.1007/s00421-017-3654-5
Krause F, Wilke J, Vogt L, Banzer W (2016) Intermuscular force transmission along myofascial chains: a systematic review. J Anat 228(6):910–918. https://doi.org/10.1111/joa.12464
Lee RH, Heckman C (2000) Adjustable amplification of synaptic input in the dendrites of spinal motoneurons in vivo. J Neurosci 20(17):6734–6740
Lee EJ, Joumaa V, Herzog W (2007) New insights into the passive force enhancement in skeletal muscles. J Biomech 40(4):719–727. https://doi.org/10.1016/j.jbiomech.2006.10.009
Leonard TR, Herzog W (2010) Regulation of muscle force in the absence of actin-myosin-based cross-bridge interaction. Am J Physiol Cell Physiol 299(1):C14-20. https://doi.org/10.1152/ajpcell.00049.2010
Lima BN, Lucareli PR, Gomes WA, Silva JJ, Bley AS, Hartigan EH, et al (2014) The acute effects of unilateral ankle plantar flexors static- stretching on postural sway and gastrocnemius muscle activity during single-leg balance tasks. J Sports Sci Med 13(3):564–570. http://www.ncbi.nlm.nih.gov/pubmed/25177183
Maas H, Baan GC, Huijing PA (2001) Intermuscular interaction via myofascial force transmission: effects of tibialis anterior and extensor hallucis longus length on force transmission from rat extensor digitorum longus muscle. J Biomech 34(7):927–940. https://doi.org/10.1016/s0021-9290(01)00055-0
Maas H, Meijer HJ, Huijing PA (2005) Intermuscular interaction between synergists in rat originates from both intermuscular and extramuscular myofascial force transmission. Cells Tissues Organs 181(1):38–50. https://doi.org/10.1159/000089967
Magnusson SP, Simonsen EB, Aagaard P, Sorensen H, Kjaer M (1996) A mechanism for altered flexibility in human skeletal muscle. J Physiol 497(Pt 1):291–298 (PM:8951730)
Magnusson SP, Simonsen EB, Aagaard P, Boesen J, Johannsen F, Kjaer M (1997) Determinants of musculoskeletal flexibility: viscoelastic properties, cross-sectional area, EMG and stretch tolerance. Scand J Med Sci Sports 7(4):195–202. https://www.ncbi.nlm.nih.gov/pubmed/9241023
Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M (2003) Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther 83(8):713–721. https://www.ncbi.nlm.nih.gov/pubmed/12882612
Marchetti PH, Silva FH, Soares EG, Serpa EP, Nardi PS, Vilela Gode B, et al (2014) Upper limb static-stretching protocol decreases maximal concentric jump performance. J Sports Sci Med 13(4):945–950. http://www.ncbi.nlm.nih.gov/pubmed/25435789
Marchetti PH, Reis RG, Gomes WA, da Silva WA, Soares EG, de Freitas FS, Behm DG (2017) Static-stretching of the pectoralis major decreases tríceps brachii activation during a maximal isometric bench press. Gazz Med Ital 176:212–218. https://doi.org/10.23736/S0393-3660.17.03452-0
Marcora SM, Staiano W, Manning V (2009) Mental fatigue impairs physical performance in humans. J Appl Physiol 106(3):857–864. https://doi.org/10.1152/japplphysiol.91324.2008
Meijer HJ, Rijkelijkhuizen JM, Huijing PA (2007) Myofascial force transmission between antagonistic rat lower limb muscles: effects of single muscle or muscle group lengthening. J Electromyogr Kinesiol 17(6):698–707. https://doi.org/10.1016/j.jelekin.2007.02.006
Myers TW (2001) Anatomy trains: Myofascial meridians for manual and movement therapists. Chirchill Livingstone Publishers, Edinburgh, pp 73–157
Nordsborg N, Mohr M, Pedersen LD, Nielsen JJ, Langberg H, Bangsbo J (2003) Muscle interstitial potassium kinetics during intense exhaustive exercise: effect of previous arm exercise. Am J Physiol Regul Integr Comp Physiol 285(1):R143-148. https://doi.org/10.1152/ajpregu.00029.2003
Norton-Old KJ, Schache AG, Barker PJ, Clark RA, Harrison SM, Briggs CA (2013) Anatomical and mechanical relationship between the proximal attachment of adductor longus and the distal rectus sheath. Clin Anat 26(4):522–530. https://doi.org/10.1002/ca.22116
Opplert J, Paizis C, Papitsa A, Blazevich AJ, Cometti C, Babault N (2020) Static stretch and dynamic muscle activity induce acute similar increase in corticospinal excitability. PLoS ONE 15(3):e0230388. https://doi.org/10.1371/journal.pone.0230388
Pageaux B, Marcora SM, Lepers R (2013) Prolonged mental exertion does not alter neuromuscular function of the knee extensors. Med Sci Sports Exerc 45(12):2254–2264. https://doi.org/10.1249/MSS.0b013e31829b504a
Pageaux B, Lepers R, Dietz KC, Marcora SM (2014) Response inhibition impairs subsequent self-paced endurance performance. Eur J Appl Physiol 114(5):1095–1105. https://doi.org/10.1007/s00421-014-2838-5
Perry J, Bekey GA (1981a) EMG-force relationships in skeletal muscle. CRC Crit Rev Biomed Eng 7:1–21
Perry J, Bekey GA (1981b) EMG-force relationships in skeletal muscle. CRC Critical Rev Biomedical Engin 7(1):1–21
Phillips C, Powell T, Wiesendanger M (1971) Projection from low-threshold muscle afferents of hand and forearm to area 3a of baboon’s cortex. J Physiol 217(2):419–446
Prochazka A, Ellaway P (2012) Sensory systems in the control of movement. Comprehensive Physiol 2:2615–2627
Pulverenti TS, Trajano GS, Kirk BJC, Blazevich AJ (2019) The loss of muscle force production after muscle stretching is not accompanied by altered corticospinal excitability. Eur J Appl Physiol 119(10):2287–2299. https://doi.org/10.1007/s00421-019-04212-8
Pulverenti TS, Trajano GS, Walsh A, Kirk BJC, Blazevich AJ (2020) Lack of cortical or Ia-afferent spinal pathway involvement in muscle force loss after passive static stretching. J Neurophysiol 123(5):1896–1906. https://doi.org/10.1152/jn.00578.2019
Rathelot JA, Strick PL (2009) Subdivisions of primary motor cortex based on cortico-motoneuronal cells. Proc Natl Acad Sci 106(3):918–923
Reid MB (2016) Reactive oxygen species as agents of fatigue. Med Sci Sports Exerc 48(11):2239–2246. https://doi.org/10.1249/MSS.0000000000001006
Schleip R, Duerselen L, Vleeming A, Naylor IL, Lehmann-Horn F, Zorn A et al (2012) Strain hardening of fascia: static stretching of dense fibrous connective tissues can induce a temporary stiffness increase accompanied by enhanced matrix hydration. J Bodyw Mov Ther 16(1):94–100. https://doi.org/10.1016/j.jbmt.2011.09.003
Stephenson DG, Williams DA (1985) Temperature-dependent calcium sensitivity changes in skinned muscle fibres of rat and toad. J Physiol 360:1–12. https://doi.org/10.1113/jphysiol.1985.sp015600
Sugi H, Abe T, Kobayashi T, Chaen S, Ohnuki Y, Saeki Y et al (2013) Enhancement of force generated by individual myosin heads in skinned rabbit psoas muscle fibers at low ionic strength. PLoS ONE 8(5):e63658. https://doi.org/10.1371/journal.pone.0063658
Thomas E, Bellafiore M, Gentile A, Paoli A, Palma A, Bianco A (2021) Cardiovascular responses to muscle stretching: a systematic review and meta-analysis. Int J Sports Med. https://doi.org/10.1055/a-1312-7131
Trajano GS, Seitz LB, Nosaka K, Blazevich AJ (2014a) Can passive stretch inhibit motoneuron facilitation in the human plantar flexors? J Apply Physiol 117(12):1486–1492
Trajano GS, Nosaka K, Seitz LB, Blazevich AJ (2014b) Intermittent stretch reduces force and central drive more than continuous stretch. Med Sci Sports Exerc 46(5):902–910. https://doi.org/10.1249/MSS.0000000000000185
Trajano GS, Nosaka K, Blazevich AJ (2017) Neurophysiological mechanisms underpinning stretch-induced force loss. Sports Med 47(8):1531–1541. https://doi.org/10.1007/s40279-017-0682-6
Trajano GS, Taylor JL, Orssatto LBR, McNulty CR, Blazevich AJ (2020) Passive muscle stretching reduces estimates of persistent inward current strength in soleus motor units. J Exp Biol. https://doi.org/10.1242/jeb.229922
van den Berg F, Cabri J (1999) Angewandte Physiologie–Das Bindegewebe des Bewegungsapparates verstehen und beeinflussen. Georg Thieme Verlag, Stuttgart, pp 17–48
van Wingerden JP, Vleeming A, Snijders CJ, Stoeckart R (1993) A functional-anatomical approach to the spine-pelvis mechanism: interaction between the biceps femoris muscle and the sacrotuberous ligament. Eur Spine J 2(3):140–144. https://doi.org/10.1007/BF00301411
Vleeming A, Pool-Goudzwaard AL, Stoeckart R, van Wingerden JP, Snijders CJ (1995) The posterior layer of the thoracolumbar fascia. Its function in load transfer from spine to legs. Spine 20(7): 53–758. https://www.ncbi.nlm.nih.gov/pubmed/7701385
Whalen A, Farrell K, Roberts S, Smith H, Behm DG (2019) Topical Analgesic Improved or Maintained Ballistic Hip Flexion Range of Motion with Treated and Untreated Legs. J Sports Sci Med 18(3):552–558. https://www.ncbi.nlm.nih.gov/pubmed/31427878
Wilke J, Krause F, Vogt L, Banzer W (2016a) What Is evidence-based about myofascial chains: a systematic review. Arch Phys Med Rehabil 97(3):454–461. https://doi.org/10.1016/j.apmr.2015.07.023
Wilke J, Niederer D, Vogt L, Banzer W (2016b) Remote effects of lower limb stretching: preliminary evidence for myofascial connectivity? J Sports Sci 34(22):2145–2148. https://doi.org/10.1080/02640414.2016.1179776
Wilke J, Vogt L, Niederer D, Banzer W (2017) Is remote stretching based on myofascial chains as effective as local exercise? A randomised-controlled trial. J Sports Sci 35(20):2021–2027. https://doi.org/10.1080/02640414.2016.1251606
Wu G, Ekedahl R, Stark B, Carlstedt T, Nilsson B, Hallin RG (1999) Clustering of Pacinian corpuscle afferent fibres in the human median nerve. Exp Brain Res 126:399–409
Yahia LH, Pigeon P, DesRosiers EA (1993) Viscoelastic properties of the human lumbodorsal fascia. J Biomed Eng 15(5):425–429. https://doi.org/10.1016/0141-5425(93)90081-9
Funding
Partial funding was provided by the Natural Science and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception, writing and revisions to the manuscript. DGB, SA, conducted the literature search and collected and collated the data. DGB wrote the first draft of the manuscript. JM, BD and UG provided input into the analysis and revised the original manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest with the contents of this manuscript.
Additional information
Communicated by Michael Lindinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Behm, D.G., Alizadeh, S., Drury, B. et al. Non-local acute stretching effects on strength performance in healthy young adults. Eur J Appl Physiol 121, 1517–1529 (2021). https://doi.org/10.1007/s00421-021-04657-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-021-04657-w