Abstract
Background
Stretching a muscle not only increases the extensibility or range of motion (ROM) of the stretched muscle or joint but there is growing evidence of increased ROM of contralateral and other non-local muscles and joints.
Objective
The objective of this meta-analysis was to quantify crossover or non-local changes in passive ROM following an acute bout of unilateral stretching and to examine potential dose–response relations.
Methods
Eleven studies involving 14 independent measures met the inclusion criteria. The meta-analysis included moderating variables such as sex, trained state, stretching intensity and duration.
Results
The analysis revealed that unilateral passive static stretching induced moderate magnitude (standard mean difference within studies: SMD: 0.86) increases in passive ROM with non-local, non-stretched joints. Moderating variables such as sex, trained state, stretching intensity, and duration did not moderate the results. Although stretching duration did not present statistically significant differences, greater than 240-s of stretching (SMD: 1.24) exhibited large magnitude increases in non-local ROM compared to moderate magnitude improvements with shorter (< 120-s: SMD: 0.72) durations of stretching.
Conclusion
Passive static stretching of one muscle group can induce moderate magnitude, global increases in ROM. Stretching durations greater than 240 s may have larger effects compared with shorter stretching durations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
•Non-local (crossover or global) stretching effects from unilateral passive stretching can induce moderate magnitude ROM increases. | |
•This increased non-local ROM was not modified by the participant’s trained state, sex or stretching intensity. | |
•Although not statistically significant, there may be a trend for greater ROM improvements with longer durations of unilateral stretching. |
1 Introduction
There is an abundance of literature demonstrating stretch-induced acute increases in joint range of motion (ROM) [1,2,3,4]. Improvements in ROM permit more expansive movements with less resistance [2], enhance longer duration stretch–shortening cycle activity (prolonged amortization or transition phase) performance (i.e., longer distance running, rebound chest press and others) [5, 6] and have been reported to enhance muscle force output at longer muscle lengths [7,8,9,10], which is typically the environment where many musculotendinous injuries occur [1]. Moreover, from an injury prevention perspective, increased ROM has been shown to reduce the incidence of musculotendinous injuries specifically with explosive, high speed, and change of direction activities [1]. Mechanisms underlying the enhanced acute ROM have been attributed to an increased stretch tolerance, neural inhibition, thixotropic properties (i.e., decreased visco-elasticity) and muscle and tendon architectural modifications (i.e., changes in tissue compliance, stiffness, fascicle angles and rotation among others) [11,12,13,14,15,16,17,18,19,20,21,22]. Consequently, knowledge of the training methods that can be used to optimise ROM are of key importance.
Crossover, or non-local augmentation of ROM has also been reported when the stretching of one muscle produces an increased ROM in a homologous (crossover) or heterologous non-stretched muscle or joint [23,24,25,26,27,28,29,30,31,32,33,34,35]. Such a global or systemic response to stretching would have important implications for training and rehabilitation, particularly when a unilateral injury prevents training or activity. For example, prior to a rehabilitation exercise, an individual can perform unilateral stretching to access a greater contralateral ROM when performing the activity without fear of further injuring the affected muscles or tendons. The genesis of non-local stretching effects research originated with studies examining crossover or non-local muscle fatigue [36,37,38,39,40,41,42,43,44,45,46,47]. The term non-local muscle fatigue was incorporated to indicate a temporary deficit in performance of non-exercised homologous and heterologous muscle groups that could be located contralaterally, or ipsilaterally, as well as inferiorly or superiorly to the fatigued muscle groups [45]. The proposed mechanisms of non-local muscle fatigue have been ascribed to neural inhibition, biochemical (i.e., metabolite distribution of lactate, H+, K+ ions and others), biomechanical (trunk fatigue impairing stability), and psychological (i.e., mental energy deficit) factors [45]. Based on non-local muscle fatigue effects, some researchers were intrigued to examine whether similar global effects would occur following unilateral acute stretching. Clark [29] published the earliest evidence using six unilateral hamstrings static stretches of six seconds each and reported a 14.4% improvement in the straight leg raise ROM. The 10 other studies investigating non-local stretching effects have been published in the last five years (2016–2020). As the increased acute ROM of a non-stretched muscle cannot be attributed to peripheral or local mechanisms (i.e., thixotropic or architectural modifications), the non-local ROM changes would illustrate the extent of global mechanisms. Unlike non-local muscle fatigue [45], there would be little accumulation and distribution of metabolites, and a reduced chance of trunk muscle or core fatigue. Hence, non-local stretching effects would more likely be attributed to neural inhibition or psycho-physiological (i.e., increased stretch or pain tolerance) factors [1].
Furthermore, there are conflicts in the literature regarding variables that could moderate the effectiveness of stretching on ROM. For example, a variety of stretch intervention studies have shown similar improvements in ROM of the stretched limb or joint with 15 vs. 30 s [48], 120 vs. 240 s [49], and 3.5 vs. 7 vs. 10.5 min [50] of static stretching. On the other hand, greater increases in ROM were found by Roberts and Wilson [51] with 15 s versus 5 s duration stretches, Bandy and Irion [52] suggested that 30–60 s of static stretching was more effective than 15 s, with other researchers also suggesting greater than 30 s of static stretching achieves the greatest ROM [53, 54]. It is unknown if the duration of unilateral stretching impacts non-local ROM. The effect of unilateral stretching intensity on non-local ROM, which could impact sensory afferent neural influences or psychological (i.e., stretch or pain tolerance) factors, also needs to be evaluated in future studies. The influence of stretching intensity on the stretched limb’s ROM is also contradictory with evidence showing no substantial ROM differences when comparing stretching at submaximal intensity to maximal or near maximal ROM or discomfort [55,56,57] versus evidence of greater ROM when implementing low force prolonged static stretching [58, 59]. Again, there is no information on non-local responses to stretch intensity. In addition, there are a number of other possible moderating variables that could impact non-local ROM. For example, there are very few studies that recruited trained individuals and no studies that have used only female participants and thus the trained state and sex could play a role.
With no published systematic reviews or meta-analyses regarding the extent (magnitude) of stretching on non-local ROM, the objective of this meta-analytical review was to examine the literature to determine the relative effect of varying unilateral muscle stretching durations on non-stretched muscles or joint passive ROM in healthy untrained and trained adults under the age of 40 years, with the goal of strengthening the mechanistic underpinnings of acute stretching. We were additionally interested in the effect of moderating variables such as unilateral stretch duration, intensity, trained state, and participants’ sex. Based upon previous findings reporting the effects of acute stretching durations on ROM [51,52,53,54], we hypothesized that longer durations of acute stretching would induce greater increases in non-local joint ROM in healthy untrained and trained young adults. Furthermore, based on prior studies [55,56,57], it was also hypothesized that stretch intensity would not impact non-local ROM.
2 Methods
2.1 Search Strategy
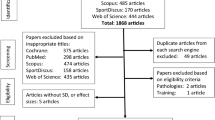
A literature search following Preferred Reporting Items for Systematic review and Meta-Analysis Protocols (PRISMA-P) review guidelines was performed by two co-authors (SA and SHA) separately and independently using PubMed, Scopus, SPORTDiscus, Web of Science, Cochrane, and CINAHL Plus databases. The topic was searched using a Boolean search strategy with the operators “OR” and “AND” and a combination of the following title keywords: stretch, stretching, flexibility, range of motion, non-local, unilateral, contralateral, ipsilateral, crossover, or remote (Fig. 1). Researchers’ personal computer databases were also examined for related articles. Related articles were found from 1999 to July 2020 (no date restrictions imposed). The selected articles were also cross referenced by the authors to identify relevant studies that might have been overlooked in the database search. In selecting studies for inclusion, a review of all relevant article titles was conducted before an examination of article abstracts and, then, full published articles. Only peer-reviewed articles were included in the meta-analysis.
2.2 Data Extraction
Data were extracted from gathered articles with a form created in Microsoft Excel. Where required data were not clearly or completely reported, article authors were contacted for clarification. When authors did not respond to our queries, the respective dataset was not considered for further analysis.
2.3 Inclusion and Exclusion Criteria (Study Selection)
Articles investigating non-local stretching were included if they fulfilled the following selection criteria: the study (1) was a randomized controlled trial or a controlled trial; (2) included a measure of acute (not cross-education training studies) pre- and post-stretching homologous or heterologous muscle or joint ROM, (3) was published in a peer-reviewed journal (abstracts and unpublished studies were excluded), (4) was published in English before July, 2020; and (5) the study participants were healthy, untrained, recreationally active, or trained individuals under the age of 40 years. Furthermore, non-exercised muscle group(s) could not be the antagonists to the stretched muscle groups (e.g., knee extensors and flexors), as movement of the stretched muscle could stretch the antagonist muscle, thus distorting the effect we desired to examine. Studies were excluded if means and standard deviations, or effect sizes were not available.
2.4 Systematic Review Analysis
Meta-analytical comparisons were carried out in RevMan version 5.3 (The Nordic Cochrane Centre. Cochrane Collaboration. 2014. p. 1–43). Means and standard deviations for a measure of joint range of motion were used to calculate an effect size. The outcome variable (ROM) was chosen based on a logically defensible rationale, an accepted method of study-inclusion justification in a meta-analysis [60]. To analyse the collected data, the inverse-variance random effects model for meta-analyses was used, because it allocates a proportionate weight to trials based on the size of their individual standard errors [61] and facilitates analysis while accounting for heterogeneity across studies [62], a common characteristic of intervention-based research. The analysis encompassed a primary analysis (all studies combined) as well as an evaluation of moderating variables such as stretch duration, intensity, participants’ trained state and sex. We presented effect sizes alongside 95% confidence intervals (CI) and these were interpreted using the conventions outlined for standardised mean difference by Hopkins et al. [63] (< 0.2 = trivial; 0.2–0.6 = small, 0.6–1.2 = moderate, 1.2–2.0 = large, 2.0–4.0 = very large, > 4.0 = extremely large).
To gauge the degree of heterogeneity amongst the included studies, the I2 statistic was calculated. This represents the percentage of variation across studies that is due to heterogeneity as opposed to chance [64]. Low, moderate, and high levels of heterogeneity correspond to I2 values of 25%, 50% and 75%, respectively; however, these thresholds are considered tentative [65]. The X2 (chi square) assesses if any observed differences in results are compatible with chance alone. A low p value, or a large chi-squared statistic relative to its degree of freedom, provides evidence of heterogeneity of intervention effects beyond those attributed to chance [63].
2.5 Assessment of Risk of Bias
The Physiotherapy Evidence Database (PEDro) scale was used to assess the risk of bias and methodological quality of the included studies (Table 1). This scale evaluates internal study validity on a scale from 0 (high risk of bias) to 10 (low risk of bias). Two reviewers independently rated each study. Any ratings that yielded different results were further adjudicated by a third reviewer. This rating was then used in the risk of bias scale. A median score of ≥ 6 represents the threshold for studies with a low risk of bias [66]. Furthermore, Figs. 2, 3, 4, 5 and 6 show symmetric funnel plots, which illustrate the absence of publication bias in the observed studies.
3 Results
3.1 Study Selection
In total, eleven studies met the inclusion criteria and were included in the meta-analysis (Table 2). They comprised 14 experimental groups. Mean study participant characteristics are provided in Table 3. The PRISMA flow diagram illustrating the number of studies excluded at each stage of the systematic review and meta-analysis is shown in Fig. 1. As only two studies examined the effect of unilateral stretching on heterologous muscle groups, it was not possible to statistically analyze heterologous versus homologous ROM responses.
3.2 Primary Analysis
For the primary analysis, there was a moderate, significant effect of passive static stretching on non-local ROM (Effect size: ES = 0.86 [95% CI 0.51, 1.20], Z = 4.83 [p < 0.01]). Between-study heterogeneity was moderate and significant (I2 = 66% [p < 0.01]). These results are displayed in Fig. 2.
3.3 Effect of Moderator Variables
The results of the moderator analysis are displayed in Table 4. Differences between subgroups were non-significant with low heterogeneity. For the moderator ‘trained state’, moderate magnitude effect sizes were found in trained (1.14 [95% CI 0.26, 2.01]) and untrained (0.79 [95% CI 0.36, 1.21]) participants (Fig. 3). For the moderator ‘stretch intensity’, ‘maximal’ stretching (0.81 [95% CI 0.09, 1.53]) had a marginally lower effect size than ‘submaximal’ (0.99 [95% CI 0.61, 1.37]) (Fig. 4). For stretching duration, > 240 s per stretch (1.24 [95% CI 0.46, 2.02]) resulted in a large magnitude effect size which exceeded the moderate magnitude effects found with 120–240 s (1.03 [95% CI 0.18, 1.88]) and < 120 s (0.72 [95% CI 0.35, 1.09]) (Fig. 5). For the moderator ‘sex’, it was not possible to compare a male group to a female group so, instead, a male group was compared to a combined male and female group. For this moderator variable, there was a marginally greater effect in males (1.10 [95% CI − 0.04, 2.24]) than in combined males and females (0.79 [95% CI 0.44, 1.14]), though both effect sizes were of moderate magnitude and there was an imbalance in the amount of studies allocated between the subgroups (Fig. 6).
4 Discussion
The major finding of this meta-analysis was that unilateral, acute, passive, static stretching induced moderate magnitude increases in non-local (non-stretched) joint ROM in healthy young adults (Fig. 2). Moderating variables such as trained state, stretching intensity, and sex did not modify the results (Figs. 3, 4, 6). Although stretching duration did not result in statistically significant differences, greater than 240-s of stretching exhibited large magnitude increases in non-local ROM compared to only moderate magnitude improvements with lower (< 240- and < 120-s) durations of stretching (Fig. 5).
4.1 Moderating Variables: Trained State
There was no statistically significant difference between the non-local stretching response with both trained (SMD: 1.14) and untrained (SMD: 0.79) groups, demonstrating a mean, moderate magnitude increase in non-local ROM (Fig. 3). While the difference was not statistically significant, the chance of reporting a type II, false negative finding could be possible with only three studies conducted with trained individuals in the analysis. Although trained individuals might be expected to achieve greater ROM of a stretched muscle due to training-induced morphological adaptations to muscles and tendons [22, 49], the non-local response may be more dependent on psycho-physiological responses and thus may not exhibit trained state differences. More research is needed to ensure there are no type II errors in an analysis due to insufficient number of studies.
4.2 Moderating Variables: Stretching Intensity
There was no statistically significant effect of passive static stretching intensity with both maximal (SMD: 0.81) and submaximal (SMD: 0.99) stretching intensities showing moderate magnitude non-local effects (Fig. 4). This finding is in accord with studies examining varying stretch intensity effects on the stretched muscle or joint. For example, studies using 50 and 85% of point of discomfort (POD) [49], initial POD, maximum POD and 120% of maximum POD [67], initial POD and near maximal POD [68] or 4 weeks of low or high intensity stretch training [69], all reported similar increases in ROM. Although classified as submaximal for this analysis, most studies with this classification in Table 2 had participants stretch at 70–90% of maximal intensity or tolerance. Since possible differences in the degree of discomfort between 70 and 90% and maximal intensity stretching may elicit minimal differences in pain perception, the stretching intensities used in these studies could have elicited similar psycho-physiological responses.
The psycho-physiological ability to cope with stretch discomfort or pain may influence the global effects of static stretching and illustrate a possible non-local flexibility mechanism. The concept of increased stretch tolerance suggests the musculotendinous unit can tolerate greater stress without a change in tension for a given length [14, 16]. Muscle sympathetic nervous activity has been shown to be elevated with stretching [70], facilitating stretch reflexes [71] influencing both the exercised and non-exercised muscles [72]. Guissard and Duchateau [73], indicated that joint ROM is highly influenced by the muscle resistance caused by tonic reflexes. Roatta et al. [74] provided evidence for sympathetic-induced increases in motor unit discharge frequency and reduced twitch ½ relaxation time demonstrating adrenergic stimulation of muscle fibre contractility. Hence, sympathetic-induced (fight or flight) reflexive and motor unit-induced effects on muscle tonicity could increase resistance to global muscle extensibility.
While passive static stretching may not seem excessively dangerous, the objective of pain perception is to alert the individual to potential threats or damage [75] and thus excite the sympathetic nervous system [76]. We speculate, that in the case of sustained stretching near or at the POD, the individual would soon sense that the discomfort of stretching was not a serious threat or harm and the fight or flight response (increased sympathetic excitation) would be downregulated over time to allow the individual to relax and perhaps extend the muscle even farther (greater stretch tolerance). This cortical perceived lack of threat or injury resulting in decreased sympathetic activation (neural and endocrine responses) and increased stretch tolerance should translate to a global response (increased non-local muscle or joint ROM). The sympathetic nervous system responses affect the entire body and are not specific to a single muscle or region [70, 77] and thus would impact non-local muscles [72].
In addition, increased global stretch tolerance might be related to global pain modulatory systems such as diffuse noxious inhibitory control (DNIC) [78,79,80]. Stretching to the POD can induce some prolonged (i.e., 30–60 s) mild or moderate pain. DNIC is activated by nociceptive (pain) stimuli (i.e., mechanical stress) [78,79,80]. The nociceptive receptor activity is transmitted to multi-modal, dynamic range convergent cortical neurons in the subnucleus reticularis dorsalis where it can suppress pain transmission monoaminergically [78,79,80], reducing pain perception not only at the painful location but also globally [80, 81]. DNIC suppresses pain sensitivity with the widespread distribution of monoamines such as endorphins, enkephalins and other compounds to contribute to global analgesia [80, 81], thus contributing to greater non-local stretch or pain tolerance. Stove et al. [82] demonstrated this global analgesia/increased non-local stretch (pain) tolerance effect by applying a painful cold pressor test to the hand and wrist and found an increase in passive knee extension ROM.
4.3 Moderating Variables: Stretching Duration
Similar to the trained state variable, there was no statistically significant difference between low (< 120-s), moderate (< 240-s) and long (> 240-s) duration stretching interventions (Fig. 5). However, there was a SMD numerical trend for increasing effect sizes with increasing stretch durations, which agrees with the findings of Reid et al. [83], who reported greater ROM with 120 versus 60 s, and both durations had greater ROM than 30 s of static stretching. The statistically significant ROM differences associated with stretch duration reported by Reid et al. [83] might be attributed to the augmented benefits of performing the stretching within a full warm-up including prior aerobic activity and post-stretch dynamic activities. While low [26, 29, 30, 33, 35] and medium [24, 26,27,28] duration, unilateral stretch interventions demonstrated moderate (0.72 and 1.03, respectively) magnitude increases in non-local ROM, long (1.24) duration stretches [23, 31, 32] provided increasingly larger magnitude non-local ROM increases. With a low number of studies (short: 6 studies, medium: 4 studies, long duration: 3 studies), the interpretation of the statistics is fraught with difficulties. Both the medium and long duration stretch durations had one study each with an outlier effect size that was substantially higher than the other 2–3 studies in the medium and long duration groups, respectively. If the numerical differences do represent a trend, it could be speculated that the longer duration of discomfort might contribute to an augmented stretch (pain) tolerance. However, once again, more stretch duration studies are needed to strengthen statistical interpretations.
The aforementioned role of sympathetic stimulation on non-local ROM can originate not only from cortical efferents but also from peripheral afferents. Thus, another mechanism that might impact changes in non-local ROM are myofascial and skin sensory receptors [84, 85] that are sensitive to skin stimulation, tangential forces and lateral stretch [86]. These exteroceptive (E-) reflexes possess multi-synaptic innervations to motoneurons [87, 88] that inhibit sympathetic excitation (decrease muscle tone) [89]. Furthermore, stretching can activate skin and joint receptors, which also project to the somatosensory cortex and thalamus, which could also affect motor output from the primary motor cortex [90]. However, exteroceptive reflex effects cease within seconds after stretching and human [73] and animal (cat) studies [91,92,93], have suggested that exteroceptive receptors’ inhibition contribute only a minor component to ROM increases with larger amplitude stretches.
Prolonged passive static stretching (i.e., 30-s or more) can impede reflexive (excitatory) activity of motoneuron spindles [1, 2, 94, 95], which project to spinal motoneurons as well as to the somatosensory and the primary motor cortex [96,97,98]. Extensive transcollosal and inter-cortical projections have been proposed as a major mechanism underlying cross-education training effects [99,100,101]. A lack of stretch-induced changes in corticospinal excitability have been reported when testing muscles at rest [102,103,104], with MVCs [102] and 20% MVC [105]. In contrast, Opplert et al. [106] reported motor evoked potential amplitude increases with a 30% MVC after 5 sets of 20 s of passive stretching. Hence, with five of six stretching-related studies demonstrating no motor evoked potential changes, the inhibitory effects of passive stretching on corticospinal excitability with non-local stretching ROM changes are likely to be minimal. Similarly, the cortical silent period, which provides insight into GABAb inhibitory interneuron effects on intra-cortical inhibition, reveals no changes during MVC [102], 30% [106] and 20% [105] MVCs after passive stretching. Thus, with a lack of stretching-induced changes in corticospinal excitability or intra-cortical inhibition, the role of stretch reflex-induced neural inhibition on non-local or global stretching effects and performance is quite likely trivial compared to stretch tolerance (reduced sympathetic excitation and global pain modulatory systems) mechanisms.
A further contributing mechanism to non-local increases in ROM might be the influence of myofascial chains. As myofascia envelopes all muscles, it has been postulated that they provide an extensive network of myofascial chains or meridians [107]. Animal [108, 109] and human cadaveric studies [110,111,112] demonstrate that fascia can alter its stiffness and transfer stress to adjacent structures. The force transferability of myofascial chains may occur through longitudinal [113, 114] and spiral (oblique) [115] lines as well as with transversal orientations to synergists and antagonists [116,117,118,119]. Wilke [34] suggested that myofascial and muscle strain can be transferred from remote body regions, promoting global connectivity. However, the extent of these transfer effects has been debated. Krause et al. [120] suggested that fascial strain is transferred primarily to neighbouring skeletal muscles. The increased cervical ROM after hamstrings [34] and gastrocnemius [35] stretching was ascribed to fascial longitudinal and spiral lines and suggests the possible substantial range of these myofascial chains. However, as Wilke [34] did not directly measure any fascial structural changes, the non-local effects could possibly be due to stretch tolerance or reduced sympathetic excitation, without fascial chain contributions. Even with the acceptance of myofascial chains or meridians, the force transfer is primarily distal to proximal or vice versa (i.e., stretch the hamstrings and increase cervical ROM) and thus longitudinal or spiral chains would not likely contribute substantially to an increased extensibility of a contralateral homologous limb muscle (i.e., hamstrings to hamstrings). Hence myofascial chains may contribute to the longitudinal transfers of myofascial extensibility to improve remote ROM, but probably are not major contributing factors outside the longitudinal and spiral lines. More research is needed to verify these assumptions.
4.4 Moderating Variable: Sex Differences
This meta-analysis did not show statistically significant differences between studies involving only men (SMD: 1.1) versus studies using both men and women (SMD: 0.79), with both groups exhibiting moderate or near moderate magnitude improvements of non-local ROM (Fig. 6). However, there is evidence in the research for sex differences in stretch tolerance. Marshall and Siegler [121] reported no sex differences in musculotendinous passive stiffness, but females showed greater hamstrings extensibility and lower pain scale scores. Similarly, Hoge et al. [122] did not find any sex differences in passive stiffness, although females demonstrated greater flexibility. Therefore, the generally greater flexibility of females over males may be attributed to higher female stretch pain tolerance [123]. Nonetheless, this lack of sex difference finding should be viewed with caution as this analysis compared studies that utilized men compared to studies that recruited both men and women. There were no comparative studies that incorporated only women participants.
4.5 Study Limitations
We acknowledge that this meta-analysis has some limitations that warrant discussion. First, the overall number of identified studies to be included in the quantitative analysis is rather small (11 studies with 14 experimental groups), which could impact the moderator analyses. However, with reference to the Cochrane Handbook for Systematic Reviews (The Nordic Cochrane Centre. Cochrane Collaboration. 2014. p. 1–43), this number is large enough to aggregate findings in the form of a meta-analysis. In addition, this is the first meta-analysis of non-local acute stretching effects on range of motion in healthy adults, which is why we are confident that this manuscript will advance our knowledge in this relatively new area of research. Second, a large variety of outcome measures assessed ROM in the included studies. This provided heterogeneity in the form of I2 values ranging between 0 and 68%. Yet, this range is still below the critical cut-off of 75%. It would also have been interesting to compare heterologous versus homologous ROM responses to unilateral stretching; however there were only two studies that examined heterologous responses [23, 35]. Further studies are needed on this topic to ascertain whether there are statistically significant differences between the heterologous ROM increases of 8.1% (SMD: 0.72) [23] and 5.0% (SMD: 0.34) [35], respectively, versus an average 9.6% (SMD 0.91) increase in the homologous ROM of the other nine studies [24, 26,27,28,29,30,31,32,33] in this review.
5 Conclusions
Non-local (crossover or global) stretching effects derived from unilateral passive static stretching can induce moderate magnitude ROM increases. This augmented non-local ROM was not modified by the participant’s trained state, sex or stretching intensity. Although not statistically significant, there may be a trend for greater contralateral ROM improvements with longer durations of unilateral stretching. If an individual is involved in rehabilitation and cannot stretch an uninjured limb or does not have the time or inclination to stretch all major muscle groups, even stretching a limited number of muscles will promote acute global increases in ROM.
References
Behm DG, Blazevich AJ, Kay AD, McHugh M. Acute effects of muscle stretching on physical performance, range of motion, and injury incidence in healthy active individuals: a systematic review. Appl Physiol Nutr Metab. 2016;41(1):1–11. https://doi.org/10.1139/apnm-2015-0235.
Behm DG, Chaouachi A. A review of the acute effects of static and dynamic stretching on performance. Eur J Appl Physiol. 2011;111(11):2633–51. https://doi.org/10.1007/s00421-011-1879-2.
Kay AD, Blazevich AJ. Effect of acute static stretch on maximal muscle performance: a systematic review. Med Sci Sports Exerc. 2012;44(1):154–64. https://doi.org/10.1249/MSS.0b013e318225cb27.
Chaabene H, Behm DG, Negra Y, Granacher U. Acute effects of static stretching on muscle strength and power: an attempt to clarify previous caveats. Front Physiol. 2019;10:1468. https://doi.org/10.3389/fphys.2019.01468.
Godges JJ, MacRae H, Longdon C, Tinberg C. The effects of two stretching procedures on the economy of walking and jogging. J Orthop Sport Physical Ther. 1989;7(3):350–7.
Wilson G, Elliot B, Wood G. Stretching shorten cycle performance enhancement through flexibility training. Med Sci Sports Exerc. 1992;24:116–23.
Herda TJ, Cramer JT, Ryan ED, McHugh MP, Stout JR. Acute effects of static versus dynamic stretching on isometric peak torque, electromyography, and mechanomyography of the biceps femoris muscle. J Strength Cond Res. 2008;22(3):809–17.
McHugh MP, Nesse M. Effect of stretching on strength loss and pain after eccentric exercise. Med Sci Sports Exerc. 2008;40(3):566–73. https://doi.org/10.1249/MSS.0b013e31815d2f8c.
Nelson AG, Allen JD, Cornwell A, Kokkonen J. Inhibition of maximal voluntary isometric torque production by acute stretching is joint-angle specific. Res Q Exerc Sport. 2001;72(1):68–70. https://doi.org/10.1080/02701367.2001.10608934.
Balle SS, Magnusson SP, McHugh MP. Effects of contract-relax vs static stretching on stretch-induced strength loss and length-tension relationship. Scand J Med Sci Sports. 2015;25(6):764–9. https://doi.org/10.1111/sms.12399.
Kay AD, Blazevich AJ. Moderate-duration static stretch reduces active and passive plantar flexor moment but not Achilles tendon stiffness or active muscle length. J Appl Physiol. 2009;106(4):1249–56. https://doi.org/10.1152/japplphysiol.91476.2008.
Kay AD, Husbands-Beasley J, Blazevich AJ. Effects of contract-relax, static stretching, and isometric contractions on muscle-tendon mechanics. Med Sci Sports Exerc. 2015;47(10):2181–90. https://doi.org/10.1249/MSS.0000000000000632.
Magnusson SP. Passive properties of human skeletal muscle during stretch maneuvers. A review. Scand J Med Sci Sports. 1998;8(2):65–77.
Magnusson SP, Simonsen EB, Aagaard P, Boesen J, Johannsen F, Kjaer M. Determinants of musculoskeletal flexibility: viscoelastic properties, cross-sectional area, EMG and stretch tolerance. Scand J Med Sci Sports. 1997;7(4):195–202.
Magnusson SP, Simonsen EB, Aagaard P, Gleim GW, McHugh MP, Kjaer M. Viscoelastic response to repeated static stretching in the human hamstring muscle. Scand J Med Sci Sports. 1995;5(6):342–7.
Magnusson SP, Simonsen EB, Aagaard P, Sorensen H, Kjaer M. A mechanism for altered flexibility in human skeletal muscle. J Physiol. 1996;497(Pt 1):291–8.
Magnusson SP, Simonsen EB, Dyhre-Poulsen P, Aagaard P, Mohr T, Kjaer M. Viscoelastic stress relaxation during static stretch in human skeletal muscle in the absence of EMG activity. Scand J Med Sci Sports. 1996;6(6):323–8.
Trajano GS, Nosaka K, Seitz LB, Blazevich AJ. Intermittent stretch reduces force and central drive more than continuous stretch. Med Sci Sports Exerc. 2014;46(5):902–10. https://doi.org/10.1249/MSS.0000000000000185.
Trajano GS, Seitz L, Nosaka K, Blazevich AJ. Contribution of central vs. peripheral factors to the force loss induced by passive stretch of the human plantar flexors. J Appl Physiol (1985). 2013;115(2):212–8. https://doi.org/10.1152/japplphysiol.00333.2013.
Trajano GS, Seitz LB, Nosaka K, Blazevich AJ. Can passive stretch inhibit motoneuron facilitation in the human plantar flexors? J Appl Physiol (1985). 2014;117(12):1486–92. https://doi.org/10.1152/japplphysiol.00809.2014.
Blazevich AJ, Cannavan D, Waugh CM, Fath F, Miller SC, Kay AD. Neuromuscular factors influencing the maximum stretch limit of the human plantar flexors. J Appl Physiol. 2012;113(9):1446–55. https://doi.org/10.1152/japplphysiol.00882.2012.
Blazevich AJ, Cannavan D, Waugh CM, Miller SC, Thorlund JB, Aagaard P, et al. Range of motion, neuromechanical, and architectural adaptations to plantar flexor stretch training in humans. J Appl Physiol (1985). 2014;117(5):452–62. https://doi.org/10.1152/japplphysiol.00204.2014.
Behm DG, Cavanaugh T, Quigley P, Reid JC, Nardi PS, Marchetti PH. Acute bouts of upper and lower body static and dynamic stretching increase non-local joint range of motion. Eur J Appl Physiol. 2016;116(1):241–9. https://doi.org/10.1007/s00421-015-3270-1.
Behm DG, Lau RJ, O’Leary JJ, Rayner MCP, Burton EA, Lavers L. Acute effects of unilateral self-administered static stretching on contralateral limb performance. J Perform Health Res. 2019;3(1):1–7. https://doi.org/10.25036/jphr.2019.3.1.behm.
Caldwell SL, Bilodeau RLS, Cox MJ, Peddle D, Cavanaugh T, Young JD, et al. Unilateral hamstrings static stretching can impair the affected and contralateral knee extension force but improve unilateral drop jump height. Eur J Appl Physiol. 2019;119(9):1943–9. https://doi.org/10.1007/s00421-019-04182-x.
Caldwell SL, Bilodeau RLS, Cox MJ, Behm DG. Cross education training effects are evident with twice daily, self-administered band stretch training. J Sport Sci Med. 2019;18:544–51.
Ce E, Coratella G, Bisconti AV, Venturelli M, Limonta E, Doria C, et al. Neuromuscular versus mechanical stretch-induced changes in contra- versus ipsilateral muscle. Med Sci Sports Exerc. 2020. https://doi.org/10.1249/MSS.0000000000002255.
Chaouachi A, Padulo J, Kasmi S, Othmen AB, Chatra M, Behm DG. Unilateral static and dynamic hamstrings stretching increases contralateral hip flexion range of motion. Clin Physiol Funct Imaging. 2017;37(1):23–9. https://doi.org/10.1111/cpf.12263.
Clark S, Christiansen A, Hellman DF, Hugunin JW, Hurst KM. Effects of ipsilateral anterior thigh soft tissue stretching on passive unilateral straight-leg raise. J Orthop Sports Phys Ther. 1999;29(1):4–9. https://doi.org/10.2519/jospt.1999.29.1.4 (discussion 10-2).
De-la-Cruz-Torres B, Carrasco-Iglesias C, Minaya-Munoz F, Romero-Morales C. Crossover effects of ultrasound-guided percutaneous neuromodulation on contralateral hamstring flexibility. Acupunct Med. 2020. https://doi.org/10.1177/0964528420920283.
Killen BS, Zelizney KL, Ye X. Crossover effects of unilateral static stretching and foam rolling on contralateral hamstring flexibility and strength. J Sport Rehabil. 2018. https://doi.org/10.1123/jsr.2017-0356.
Lima BN, Lucareli PR, Gomes WA, Silva JJ, Bley AS, Hartigan EH, et al. The acute effects of unilateral ankle plantar flexors static- stretching on postural sway and gastrocnemius muscle activity during single-leg balance tasks. J Sports Sci Med. 2014;13(3):564–70.
Whalen A, Farrell K, Roberts S, Smith H, Behm DG. Topical analgesic improved or maintained ballistic hip flexion range of motion with treated and untreated legs. J Sports Sci Med. 2019;18(3):552–8.
Wilke J, Vogt L, Niederer D, Banzer W. Is remote stretching based on myofascial chains as effective as local exercise? A randomised-controlled trial. J Sports Sci. 2017;35(20):2021–7. https://doi.org/10.1080/02640414.2016.1251606.
Wilke J, Niederer D, Vogt L, Banzer W. Remote effects of lower limb stretching: preliminary evidence for myofascial connectivity? J Sports Sci. 2016;34(22):2145–8. https://doi.org/10.1080/02640414.2016.1179776.
Doix AC, Lefevre F, Colson SS. Time course of the cross-over effect of fatigue on the contralateral muscle after unilateral exercise. PLoS ONE. 2013;8(5):e64910. https://doi.org/10.1371/journal.pone.0064910.
Doix AM, Wachholz F, Marterer N, Immler L, Insam K, Federolf PA. Is the cross-over effect of a unilateral high-intensity leg extension influenced by the sex of the participants? Biol Sex Differ. 2018;9(1):29. https://doi.org/10.1186/s13293-018-0188-4.
Martin PG, Rattey J. Central fatigue explains sex differences in muscle fatigue and contralateral cross-over effects of maximal contractions. Pflugers Archiv Eur J Physiol. 2007;454(6):957–69. https://doi.org/10.1007/s00424-007-0243-1.
Rattey J, Martin PG, Kay D, Cannon J, Marino FE. Contralateral muscle fatigue in human quadriceps muscle: evidence for a centrally mediated fatigue response and cross-over effect. Pflugers Archiv Eur J Physiol. 2006;452(2):199–207. https://doi.org/10.1007/s00424-005-0027-4.
Aboodarda SJ, Copithorne DB, Power KE, Drinkwater E, Behm DG. Elbow flexor fatigue modulates central excitability of the knee extensors. Appl Physiol Nutr Metab. 2015;40(9):924–30. https://doi.org/10.1139/apnm-2015-0088.
Aboodarda SJ, Sambaher N, Millet GY, Behm DG. Knee extensors neuromuscular fatigue changes the corticospinal pathway excitability in biceps brachii muscle. Neuroscience. 2017;340:477–86. https://doi.org/10.1016/j.neuroscience.2016.10.065.
Ben Othman A, Chaouachi A, Hammami R, Chaouachi MM, Kasmi S, Behm DG. Evidence of nonlocal muscle fatigue in male youth. Appl Physiol Nutr Metab. 2017;42(3):229–37. https://doi.org/10.1139/apnm-2016-0400.
Halperin I, Aboodarda SJ, Behm DG. Knee extension fatigue attenuates repeated force production of the elbow flexors. Eur J Sport Sci. 2014;14(8):823–9. https://doi.org/10.1080/17461391.2014.911355.
Halperin I, Copithorne D, Behm DG. Unilateral isometric muscle fatigue decreases force production and activation of contralateral knee extensors but not elbow flexors. Appl Physiol Nutr Metab. 2014;39(12):1338–44. https://doi.org/10.1139/apnm-2014-0109.
Halperin I, Chapman DW, Behm DG. Non-local muscle fatigue: effects and possible mechanisms. Eur J Appl Physiol. 2015;115(10):2031–48. https://doi.org/10.1007/s00421-015-3249-y.
Grant MC, Robergs R, Baird MF, Baker JS. The effect of prior upper body exercise on subsequent wingate performance. Biomed Res Int. 2014;2014:329328. https://doi.org/10.1155/2014/329328.
Elmer SJ, Amann M, McDaniel J, Martin DT, Martin JC. Fatigue is specific to working muscles: no cross-over with single-leg cycling in trained cyclists. Eur J Appl Physiol. 2013;113(2):479–88. https://doi.org/10.1007/s00421-012-2455-0.
Donti O, Tsolakis C, Bogdanis GC. Effects of baseline levels of flexibility and vertical jump ability on performance following different volumes of static stretching and potentiating exercises in elite gymnasts. J Sports Sci Med. 2014;13(1):105–13.
Marchetti PH, Miyatake MMS, Magalhaes RA, Gomes WA, Da Silva JJ, Brigatto FA, et al. Different volumes and intensities of static stretching affect the range of motion and muscle force output in well-trained subjects. Sports Biomech. 2019. https://doi.org/10.1080/14763141.2019.1648540.
Kurtdere MK, Curt C, Ozsu Nebioglu I. Acute static stretching with different volumes improves hamstring flexibility but not reactive strength index and leg stiffness in well trained judo athletes. J Hum Sport Exerc. 2020. https://doi.org/10.14198/jhse.2021.164.03.
Roberts JM, Wilson K. Effect of stretching duration on active and passive range of motion in the lower extremity. Br J Sports Med. 1999;33(4):259–63.
Bandy WD, Irion JM. The effect of time on the static stretch of the hamstrings muscles. Phys Ther. 1994;74(9):845–50.
Feland JB, Myrer JW, Schulthies SS, Fellingham GW, Measom GW. The effect of duration of stretching of the hamstring muscle group for increasing range of motion in people aged 65 years or older. Phys Ther. 2001;81(5):1110–7.
Chan SP, Hong Y, Robinson PD. Flexibility and passive resistance of the hamstrings of young adults using two different static stretching protocols. Scand J Med Sci. 2001;11:81–6.
Knudson D, Bennett K, Corn R, Leick D, Smith C. Acute effects of stretching are not evident in the kinematics of the vertical jump. J Strength Cond Res. 2001;15(1):98–101.
Manoel ME, Harris-Love MO, Danoff JV, Miller TA. Acute effects of static, dynamic, and proprioceptive neuromuscular facilitation stretching on muscle power in women. J Strength Cond Res. 2008;22(5):1528–34 (Available from PM:18714235).
Young W, Elias G, Power J. Effects of static stretching volume and intensity on plantar flexor explosive force production and range of motion. 73. J Sports Med Phys Fit. 2006;46(3):403–11.
Apostolopoulos N. Performance flexibility. In: Foran B, editor. High-performance sports conditioning. Champaign: Human Kinetics; 2001. p. 49–61.
Warren CG, Lehmann JF, Koblanski JN. Elongation of rat tail tendon: effect of load and temperature. Arch Phys Med Rehabil. 1971;52(10):465–474 passim. Available from https://www.ncbi.nlm.nih.gov/pubmed/5116032.
Turner HM, Bernard RM. Calculating and synthesizing effect sizes. Contemp Issues Commun Sci Disord. 2006;33(1):42–55.
Deeks JJ, Higgins JP, Altman DG. Analysing data and undertaking meta-analyses. Cochrane handbook for systematic reviews of interventions. Cochrane Book Series; 2008. p. 243–96.
Kontopantelis E, Springate DA, Reeves D. A re-analysis of the Cochrane Library data: the dangers of unobserved heterogeneity in meta-analyses. PLoS ONE. 2013;8(7):e69930. https://doi.org/10.1371/journal.pone.0069930.
Hopkins WG, Marshall SW, Batterham AM, Hanin J. Progressive statistics for studies in sports medicine and exercise science. Med Sci Sports Exerc. 2009;41(1):3–12. https://doi.org/10.1249/Mss.0b013e31818cb278.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-34. https://doi.org/10.1016/j.jclinepi.2009.06.006.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60. https://doi.org/10.1136/bmj.327.7414.557.
Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther. 2003;83(8):713–21.
Takeuchi K, Nakamura M. Influence of high intensity 20-second static stretching on the flexibility and strength of hamstrings. J Sports Sci Med. 2020;19(2):429–35.
Santos CXB, Barros Beltro N, Torres Piraua AL, Quagliotto Durigan JL, Behm DG, Cappato de Araujo R. Static stretching intensity does not influence acute range of motion, passive torque, and muscle architecture. J Sport Rehabil. 2020;29:1–6.
de Araujo VA, Oleivera Soares B, Remigio Calcacante B, Barros Beltrao N, Santos Nascimeto VY, Rodarti Pitangui AC, Cappato de Araujo R. Does the stretching intensity matter when targeting a range of motion gains? A Randomized trial. Motriz Rio Claro. 2020;26(2):26–31. https://doi.org/10.1590/s1980-6574202000018019.
Cui J, Blaha C, Moradkhan R, Gray KS, Sinoway LI. Muscle sympathetic nerve activity responses to dynamic passive muscle stretch in humans. J Physiol. 2006;576(Pt 2):625–34.
Kamibayashi K, Nakazawa K, Ogata H, Obata H, Akai M, Shinohara M. Invariable H-reflex and sustained facilitation of stretch reflex with heightened sympathetic outflow. J Electromyogr Kinesiol. 2009;19(6):1053–60.
Ray CA, Mark AL. Sympathetic nerve activity to nonactive muscle of the exercising and nonexercising limb. Med Sci Sports Exerc. 1995;27(2):183–7.
Guissard N, Duchateau J. Neural aspects of muscle stretching. Exerc Sport Sci Rev. 2006;34(4):154–8.
Roatta S, Arendt-Nielsen L, Farina D. Sympathetic-induced changes in discharge rate and spike-triggered average twitch torque of low-threshold motor units in humans. J Physiol. 2008;586(Pt 22):5561–74. https://doi.org/10.1113/jphysiol.2008.160770.
Moayedi M, Davis KD. Theories of pain: from specificity to gate control. J Neurophysiol. 2013;109(1):5–12. https://doi.org/10.1152/jn.00457.2012.
Berne RM, Levy MN. Physiology. Toronto: C.V. Mosby Publishers; 1983. p. 112–65.
Donadio V, Karlsson T, Elam M, Wallin BG. Interindividual differences in sympathetic and effector responses to arousal in humans. J Physiol. 2002;544(Pt 1):293–302.
Mense S. Neurobiological concepts of fibromyalgia—the possible role of descending spinal tracts. Scand J Rheumatol Suppl. 2000;113:24–9.
Sigurdsson A, Maixner W. Effects of experimental and clinical noxious counterirritants on pain perception. Pain. 1994;57(3):265–75.
Pud D, Granovsky Y, Yarnitsky D. The methodology of experimentally induced diffuse noxious inhibitory control (DNIC)-like effect in humans. Pain. 2009;144(1–2):16–9. https://doi.org/10.1016/j.pain.2009.02.015.
Le Bars D, Villanueva L, Bouhassira D, Willer JC. Diffuse noxious inhibitory controls (DNIC) in animals and in man. Patol Fiziol Eksp Ter. 1992;4:55–65.
Stove MP, Hirata RP, Palsson TS. Muscle stretching—the potential role of endogenous pain inhibitory modulation on stretch tolerance. Scand J Pain. 2019;19(2):415–22. https://doi.org/10.1515/sjpain-2018-0334.
Reid JC, Greene R, Young JD, Hodgson DD, Blazevich AJ, Behm DG. The effects of different durations of static stretching within a comprehensive warm-up on voluntary and evoked contractile properties. Eur J Appl Physiol. 2018;118(7):1427–45. https://doi.org/10.1007/s00421-018-3874-3.
Schleip R. Fascial plasticity—a new neurobiological explanation: Part I. J Bodyw Mov Ther. 2003;7(1):11–9.
Schleip R. Fascial plasticity—a new neurobiological explanation: Part 2. J Bodyw Mov Ther. 2003;7(2):104–16.
Kruger L. Cutaneous sensory system. Encyclopedia of neuroscience. Boston: Birkhauser; 1987. p. 64–127.
Jenner JR, Stephens JA. Cutaneous reflex responses and thier central nervous pathways studied in man. J Physiol. 1982;333:405–19.
Kearney RE, Chan CWY. Reflex response of human arm muscles to cutaneous stimulation of the foot. Brain Res. 1999;170:214–7.
Wu G, Ekedahl R, Stark B, Carlstedt T, Nilsson B, Hallin RG. Clustering of Pacinian corpuscle afferent fibres in the human median nerve. Exper Brain Res. 1999;126:399–409.
Canedo A. Primary motor cortex influences on the descending and ascending systems. Prog Neurobiol. 1997;51(3):287–335.
Cleland CL, Hayward L, Rymer WZ. Neural mechanisms underlying the clasp-knife reflex in the cat. II. Stretch-sensitive muscular-free nerve endings. J Neurophysiol. 1990;64(4):1319–30. https://doi.org/10.1152/jn.1990.64.4.1319.
Cleland CL, Rymer WZ. Neural mechanisms underlying the clasp-knife reflex in the cat. I. Characteristics of the reflex. J Neurophysiol. 1990;64(4):1303–18. https://doi.org/10.1152/jn.1990.64.4.1303.
Cleland CL, Rymer WZ. Functional properties of spinal interneurons activated by muscular free nerve endings and their potential contributions to the clasp-knife reflex. J Neurophysiol. 1993;69(4):1181–91. https://doi.org/10.1152/jn.1993.69.4.1181.
Behm DG, Bambury A, Cahill F, Power K. Effect of acute static stretching on force, balance, reaction time, and movement time. Med Sci Sports Exerc. 2004;36(8):1397–402.
Behm DG, Button DC, Butt JC. Factors affecting force loss with prolonged stretching. Can J Appl Physiol. 2001;26(3):261–72.
Carroll TJ, Herbert RD, Munn J, Lee M, Gandevia SC. Contralateral effects of unilateral strength training: evidence and possible mechanisms. J Appl Physiol (1985). 2006;101(5):1514–22. https://doi.org/10.1152/japplphysiol.00531.2006.
Hortobagyi T. Cross education and the human central nervous system: Mechanisms of unilateral interventions producing contralateral adaptations. IEEE Eng Med Biol. 2005;24:22–8.
Hortobagyi T, Richardson SP, Lomarev M, Shamim E, Meunier S, Russman H, et al. Interhemispheric plasticity in humans. Med Sci Sports Exerc. 2011;43(7):1188–99. https://doi.org/10.1249/MSS.0b013e31820a94b8.
Prochazka A, Ellaway P. Sensory systems in the control of movement. Compr Physiol. 2012;2(4):2615–27. https://doi.org/10.1002/cphy.c100086.
Phillips C, Powell T, Wiesendanger M. Projection from low-threshold muscle afferents of hand and forearm to area 3a of baboon’s cortex. J Physiol. 1971;217(2):419–46.
Rathelot J-A, Strick PL. Subdivisions of primary motor cortex based on cortico-motoneuronal cells. Proc Natl Acad Sci. 2009;106(3):918–23.
Pulverenti TS, Trajano GS, Kirk BJ, Blazevich AJ. The loss of muscle force production after muscle stretching is not accompanied by altered corticospinal excitability. Eur J Appl Physiol. 2019;119(10):2287–99.
Budini F, Gallasch E, Christova M, Rafolt D, Rauscher AB, Tilp M. One minute static stretch of plantar flexors transiently increases H reflex excitability and exerts no effect on corticospinal pathways. Exper Physiol. 2017;102(8):901–10.
Budini F, Kemper D, Christova M, Gallasch E, Rafolt D, Tilp M. Five minutes static stretching influences neural responses at spinal level in the background of unchanged corticospinal excitability. J Musculoskelet Neuronal Interact. 2019;19(1):30.
Pulverenti TS, Trajano GS, Walsh A, Kirk BJC, Blazevich AJ. Lack of cortical or Ia-afferent spinal pathway involvement in muscle force loss after passive static stretching. J Neurophysiol. 2020;123(5):1896–906. https://doi.org/10.1152/jn.00578.2019.
Opplert J, Paizis C, Papitsa A, Blazevich AJ, Cometti C, Babault N. Static stretch and dynamic muscle activity induce acute similar increase in corticospinal excitability. PLoS ONE. 2020;15(3):e0230388.
Wilke J, Krause F, Vogt L, Banzer W. What is evidence-based about myofascial chains: a systematic review. Arch Phys Med Rehabil. 2016;97(3):454–61. https://doi.org/10.1016/j.apmr.2015.07.023.
Schleip R, Duerselen L, Vleeming A, Naylor IL, Lehmann-Horn F, Zorn A, et al. Strain hardening of fascia: static stretching of dense fibrous connective tissues can induce a temporary stiffness increase accompanied by enhanced matrix hydration. J Bodyw Mov Ther. 2012;16(1):94–100. https://doi.org/10.1016/j.jbmt.2011.09.003.
Yahia LH, Pigeon P, DesRosiers EA. Viscoelastic properties of the human lumbodorsal fascia. J Biomed Eng. 1993;15(5):425–9. https://doi.org/10.1016/0141-5425(93)90081-9.
Norton-Old KJ, Schache AG, Barker PJ, Clark RA, Harrison SM, Briggs CA. Anatomical and mechanical relationship between the proximal attachment of adductor longus and the distal rectus sheath. Clin Anat. 2013;26(4):522–30. https://doi.org/10.1002/ca.22116.
van Wingerden JP, Vleeming A, Snijders CJ, Stoeckart R. A functional-anatomical approach to the spine-pelvis mechanism: interaction between the biceps femoris muscle and the sacrotuberous ligament. Eur Spine J. 1993;2(3):140–4. https://doi.org/10.1007/BF00301411.
Vleeming A, Pool-Goudzwaard AL, Stoeckart R, van Wingerden JP, Snijders CJ. The posterior layer of the thoracolumbar fascia. Its function in load transfer from spine to legs. Spine (Phila Pa 1976). 1995;20(7):753–8.
Benetazzo L, Bizzego A, De Caro R, Frigo G, Guidolin D, Stecco C. 3D reconstruction of the crural and thoracolumbar fasciae. Surg Radiol Anat. 2011;33(10):855–62. https://doi.org/10.1007/s00276-010-0757-7.
Eng CM, Pancheri FQ, Lieberman DE, Biewener AA, Dorfmann L. Directional differences in the biaxial material properties of fascia lata and the implications for fascia function. Ann Biomed Eng. 2014;42(6):1224–37. https://doi.org/10.1007/s10439-014-0999-3.
Myers TW. Anatomy trains: myofascial meridians for manual and movement therapists. Edinburgh: Chirchill Livingstone Publishers; 2001.
Huijing PA, van de Langenberg RW, Meesters JJ, Baan GC. Extramuscular myofascial force transmission also occurs between synergistic muscles and antagonistic muscles. J Electromyogr Kinesiol. 2007;17(6):680–9. https://doi.org/10.1016/j.jelekin.2007.02.005.
Maas H, Baan GC, Huijing PA. Intermuscular interaction via myofascial force transmission: effects of tibialis anterior and extensor hallucis longus length on force transmission from rat extensor digitorum longus muscle. J Biomech. 2001;34(7):927–40. https://doi.org/10.1016/s0021-9290(01)00055-0.
Maas H, Meijer HJ, Huijing PA. Intermuscular interaction between synergists in rat originates from both intermuscular and extramuscular myofascial force transmission. Cells Tissues Organs. 2005;181(1):38–50. https://doi.org/10.1159/000089967.
Meijer HJ, Rijkelijkhuizen JM, Huijing PA. Myofascial force transmission between antagonistic rat lower limb muscles: effects of single muscle or muscle group lengthening. J Electromyogr Kinesiol. 2007;17(6):698–707. https://doi.org/10.1016/j.jelekin.2007.02.006.
Krause F, Wilke J, Vogt L, Banzer W. Intermuscular force transmission along myofascial chains: a systematic review. J Anat. 2016;228(6):910–8. https://doi.org/10.1111/joa.12464.
Marshall PW, Siegler JC. Lower hamstring extensibility in men compared to women is explained by differences in stretch tolerance. BMC Musculoskelet Disord. 2014;15:223. https://doi.org/10.1186/1471-2474-15-223.
Hoge KM, Ryan ED, Costa PB, Herda TJ, Walter AA, Stout JR, et al. Gender differences in musculotendinous stiffness and range of motion after an acute bout of stretching. J Strength Cond Res. 2010;24(10):2618–26. https://doi.org/10.1519/JSC.0b013e3181e73974.
Fillingim RB, Edwards RR, Powell T. The relationship of sex and clinical pain to experimental pain responses. Pain. 1999;83(3):419–25. https://doi.org/10.1016/s0304-3959(99)00128-1.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article.
Conflict of interest
David G. Behm, Shahab Alizadeh, Saman Hadjizadeh Anvar, Ben Drury, Urs Granacher and Jason Moran declare that they have no conflicts of interest relevant to the content of this review.
Authorship contributions
DGB, SA, SHA conducted the literature search and collected and collated the data. DGB wrote the first draft of the manuscript. JM analyzed and interpreted the data. BD and UG provided input into the analysis and revised the original manuscript. All authors read and approved the final manuscript.
Data availability statement
All pertinent data are provided in the listed tables (PEDro scale analysis, individual and mean study characteristics and moderator analyses) and figures (PRISMA flow chart and funnel plots).
Rights and permissions
About this article
Cite this article
Behm, D.G., Alizadeh, S., Anvar, S.H. et al. Non-local Acute Passive Stretching Effects on Range of Motion in Healthy Adults: A Systematic Review with Meta-analysis. Sports Med 51, 945–959 (2021). https://doi.org/10.1007/s40279-020-01422-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-020-01422-5