Abstract
Purpose
To show that heart rate variability (HRV) in the standing position better reflects the way in which athletes adapt to training in so-called intermittent sports than the indicator of resting parasympathetic tone usually employed in endurance sports.
Methods
Twenty professional soccer players (intermittent sport) took part in a 5-week training session divided into three successive periods: “Warm-up”, “Intensive training” and “Tapering”. At the beginning and end of each of the three periods, a stand test was carried out and the heart rate was recorded, beat by beat (Polar Team 2). We analysed HRV to determine the indicator mostly used to demonstrate training adaptation in endurance sports (lnRMSSD supine, natural logarithm of root mean square of the successive differences) as well as indicators obtained by means of spectral analysis in both supine and standing position.
Results
A decrease in heart rate was observed in the supine position at rest during training (−5.2 ± 1.3 bpm) while lnRMSSD and spectral analysis indicators remained unchanged. The “Warm-up” caused an increase in spectral analysis total power in standing position which was further highlighted by “Tapering” (3.39 ± 0.09, 3.61 ± 0.08 and 3.65 ± 0.09 log ms2, respectively). However, the autonomic changes are probably more complex than a change in autonomic activity or balance since spectral analysis autonomic indicators remained unchanged.
Conclusions
HRV in the standing position could monitor training adaptation in intermittent sports contrary to the indicator usually employed in endurance sports. However, the significance of the HRV change in the standing position during training remains unclear.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Before every competition, athletes must undergo a training period, which varies in length according to the discipline. It must allow them to reach a high level of performance for the forthcoming competition and, in some disciplines, for the entire competitive season. A complex juggling act is therefore, required to develop physical qualities and yet minimise fatigue due to the training load. To do this, trainers use several tools in an attempt to establish how athletes adapt to the training load. Amongst these, questionnaires, laboratory tests, neuromuscular tests and tracking (global positioning system, GPS) are mostly used by trainers (for a review of these tools, see Halson 2014). Some use heart rate variability (HRV) (Naranjo Orellana et al. 2015). This method indirectly reflects the autonomous nervous system (Task Force 1996). It can be used in two ways by analysing time on the one hand and frequency on the other (Task force 1996). Time analyses carried out when supine and resting are preferred in the training context as they appear to be less dependent on the respiratory cycle, less sensitive to signal noise and less complex to implement (Buchheit 2014). They have proved invaluable in endurance sports in which the main energy resources involve the aerobic pathways (especially the root mean square of the successive differences, RMSSD, and its logarithmic transformation, lnRMSSD to normalise distribution, Buchheit 2014). In fact, they reflect the parasympathetic branch of the autonomic nervous system. They can thus effectively direct the training load (Kiviniemi et al. 2007; Vesterinen et al. 2013), reflect a state of fatigue (Plews et al. 2013), a state of over-training (Le Meur et al. 2013) and are also correlated to performance (Atlaoui et al. 2007).
Many sporting disciplines, especially intermittent sports (e.g., soccer, basket-ball), are characterised by random alternations between resting/active cycles requiring both aerobic and anaerobic pathways. Furthermore, the physical and tactical constraints of these sports lend themselves to the fight or flight response which involves a reaction by the sympathetic branch of the autonomic nervous system (Jansen et al. 1995). In theory, it therefore, seems interesting to assess the sympathetic branch during an activation test (Olufsen et al. 2008). The stand test is a conventional activation test used to investigate the autonomic nervous system (Task Force 1996). Accurate quantification of sympathetic nervous activity is difficult and would not be practicable for in-field experiments. However, the low frequency domain (LF) of HRV spectral analysis is influenced by the sympathetic nervous system in addition to the influences of the parasympathetic nervous system and baroreflex (Task Force 1996; Rahman et al. 2011; Moak et al. 2009). Therefore, a change in sympathetic nervous system should lead to a change in LF. LF has not been studied very much in the training context since it is well known that it cannot reliably assesses the sympathetic nervous system when heart rate is high like during physical activity (Perini and Veicsteinas 2003, Pichon et al. 2004). However, LF (or its logarithmic transformation logLF) has been largely studied in the context of slight activation (associated with a slight increase in heart rate) as is seen during the stand test (Task force 1996). Moreover, Schmitt et al. (2013, 2015) showed that LF or logLF in the standing position provides additional information to HRV in the supine position in endurance sports and is involved in reflecting the state of fatigue in Nordic skiers (Schmitt et al. 2013, 2015) and in runners (Portier et al. 2001). Lastly, we recently showed that it correlates to the perception of form in professional soccer players in the intermittent sport context (personal communication).
We hypothesized that the low frequency domain of HRV spectral analysis during a stand test reflects the way in which professional soccer players adapt to the training load.
Methods
This study was carried out during preparations for the 2014–2015 competition season. Twenty professional soccer players in the French Second Division took part in this experiment. Informed consent was obtained from all individual participants included in the study. The procedures were approved by the ethic committee of Angers (France). The anthropometric and physical characteristics were as follows: age 24.9 ± 1.5 years (mean ± SEM), height 1.77 ± 0.03 m, weight 75.0 ± 1.3 kg.
Experimental design
The training programme was defined by the team’s technical staff and was not changed in any way by the investigators. Preparation for the season was carried out over 5 weeks. This was divided into three periods. The first so-called “Warm-up” period lasted 14 days. The second so-called “Intensive training” period also lasted 14 days. Finally, the third period, “Tapering”, lasted 8 days (Fig. 1).
Experimental design according to player agenda during training period. 5-week training time schedule comprising three successive periods: “Warm-up” (D0–D14), “Intensive training” (D15–D28) and “Tapering” (D29–D35). The aim of the “Warm-up” period is to develop aerobic capacity and football-specific training. The “Intensive training” period comprises four friendly matches and training in football techniques. The “Tapering” period comprises only one friendly match and training in football techniques. Stand tests are indicated (T0–T3)
Four stand tests were carried out during this preparatory phase. The first test was carried out prior to preparation (T0). This was followed by a test at the end of each of the three periods (T1, T2 and T3). Thus T1 was carried out at the end of the “Warm-up” period (D14), T2 at the end of the “Intensive training” period (D28) and T3 at the end of the “Tapering” period (D35). Each stand test was carried out in the morning (08:30) on an empty stomach. A stand test comprised one 10-min phase in the supine position followed by a 7-min standing phase. The soccer players did not have to move or speak throughout the stand test. Every player was fitted with a heart rate monitor (Polar Team 2) to register the RR-interval (the interval between two heart beats, i.e., the opposite to cardiac frequency) beat by beat with a 1 ms resolution.
Analysis of heart rate variability (HRV)
We carried out HRV time and frequency analyses to estimate the activity of the autonomic nervous system using Nevrokard software (aHRV version 11.0.4, Nevrokard Kiauta, Slovenia). We determined the spectral power density by means of spectral analysis of 512 RR-interval values after re-sampling at 2 Hz from the fifth minute of the 10-min phase in the supine position. Breathing rate was not controlled nor recorded and we did not record blood pressure during this in-field experiment. The filtering of artefacts and ectopic beats was performed manually in advance (aHRV software). The time domain analysis involved determining the RMSSD (parasympathetic indicator) and its related natural logarithm (lnRMSSD). As recommend by Task Force (1996), the frequency domain analysis involved obtaining the harmonic component spectrum of very low (VLF ≤0.04 Hz), Low (0.04 Hz ≤ LF ≤ 0.15 Hz) and High (0.15 Hz ≤ HF ≤ 0.40 Hz) Frequencies to establish the related spectral power (ms2/Hz) as well as total power (or overall HRV, T ≤ 0.4 Hz), normalised LF and HF power (LFnu and HFnu, respectively, no unit), and LF/HF ratio. Finally, the logarithm of related VLF, LF, HF, T, LFnu, HFnu and LF/HF was calculated (logVLF, logLF, logHF, logT, logLFnu, logHFnu, logLF/HF, respectively). According to the Task Force (1996), HF are under the parasympathetic influences while LF are under the influence of both parasympathetic and sympathetic nervous system. However, LF changes tracks sympathetic changes during a stand test. HF and LF could be normalised according to total power (T) to avoid the confounding effects of overall HRV and HR changes. Normalised HF and LF are indicators of parasympathetic and sympathetic influences, respectively (HFnu and LFnu). LF/HF ratio is an indicator of the autonomic balance between sympathetic and parasympathetic system. The meaning of VLF has remained unclear. These VLF could not be correctly assessed on short term recordings but it is recommended to mention them in any study that used spectral analysis even in short term (Task Force 1996). In the standing position, the same procedure was carried out on 512 consecutive RR-interval values re-sampled at 2 Hz as when lying down. The first 2 min of this phase used to adapt to the new position and stabilise the heart rate were routinely eliminated.
In addition, and to have an objective view of the physical activity of the players over the 5-week training period, five players (a striker, a wide midfielder, a defensive midfielder, a full-back and a central defender) were fitted with a GPS system (SPI Proc X, 5 Hz, GPSport, Canberra, Australia) to quantify the distances covered at different speeds, for the duration of each training session.
Statistical analysis
Data are presented as mean ± SEM. We performed a between-period comparison by means of analysis of variance (ANOVA) for repeated measurements after a Bartlett’s test for equality of variances. When appropriate, a post hoc t test for paired data with Bonferroni correction was applied (Prism 5.01, GraphPad Software, San Diego, CA, USA). We set statistical significance at p ≤ 0.01 because of the large number of comparisons.
Results
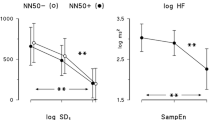
All of the data in the supine and standing positions are shown in Table 1. Heart rate (HR) at rest in the supine and standing positions fell highly significant in the tests carried out during and after the preparatory phase (T1, T2 and T3), compared to the control test before preparation started (T0) with no further changes (T1 vs. T2 vs. T3), Table 1. We did not observe any significant difference in the indicator, mostly used to demonstrate training adaptation in endurance sports (lnRMSSD in the supine position) as well as in spectral analysis in the supine position (Fig. 2 , differences in LnRMSSD and in logT were almost significant). We observed significant differences only in spectral analysis in the standing position. logLF increased at T3 but this change came with increase in both logT and logHF. We observed an increase of logT, logVLF, and logHF at T1 while only logHF is altered at T2 (Table 1). However, all these changes reflected the change in logT (Fig. 2) since we did not observe any difference in normalised units (logLFnu and logHFnu) nor in LF/HF (Table 1).
Heart rate variability during 5 weeks of training by professional soccer players. a Logarithmic transformation of the root mean square of the successive differences (lnRMSSD) supine, b Logarithmic transformation of the total power of heart rate Variability spectral analysis in supine position, and c the same in standing position. T0, T1, T2 and T3 correspond to the initial evaluation and these at the end of the three training phases (“Warm-up”, “Intensive training” and “Tapering”, respectively) over 5 weeks of training. lnRMSSD natural logarithm of root mean square of the successive differences; **with p < 0.01 vs. T0, ***with p < 0.001 vs. T0, no further differences (T1 vs. T2 vs. T3) have been observed

The distances covered at various speeds by the five subjects are presented in Table 2. The distances covered increased gradually between weeks 1 and 2, for all speeds. During weeks 3 and 4 (“Intensive training” period), the greatest distances were covered for speeds exceeding 16 km/h. Finally, during week 5 (“Tapering” period), there was a real decrease in the volume of work for speeds exceeding 16 km/h compared to weeks 3 and 4.
Discussion
According to our hypothesis, we showed a change in Low Frequency domain of spectral analysis in the standing position during a soccer training period. However, contrary to our hypothesis, this change was not clear due to a sympathetic alteration since the other spectral analysis domains were also altered (T, VLF and HF). This change reflected an overall increase in heart rate variability (total power of spectral analysis, T) and its meaning remains unclear. Nevertheless, our study shows the advantage of evaluating HRV by spectral analysis in standing position when monitoring training of intermittent sport disciplines. It also shows that the parasympathetic indicator, lnRMSSD in the supine position, the merits of which have been broadly demonstrated in endurance sports, is not useful in intermittent sports.
Training programme and HRV response
GPS data (Table 2) validate the objectives defined in advance by the trainers. In fact, for all speeds, the distances covered increased between weeks one and two (“Warm-up” period). The greatest distances were also covered at speeds below 16 km/h, equating to aerobic work, and the lowest distances for speeds exceeding 16 km/h corresponding to high intensity work. Conversely, the greatest distances were covered between weeks 3 and 4 (“Intensive training” period), for all speeds greater than 16 km/h. Finally, during the fifth week (“Tapering” period), the distances covered decreased for all speeds compared to previous weeks. These data therefore, confirm the coaches’ aim to have three successive training periods.
HRV in the supine position
We did not observe any difference in lnRMSSD in the supine position at any time during the training period while this marker has proved invaluable in endurance sports. We expected a significant increase of this marker between T0 and T1, i.e., between the end of a period of inactivity and the end of a gradual resumption of soccer (“Warm-up” period, Carter et al. 2003; Mourot et al. 2004a; Buchheit et al. 2010; Vesterinen et al. 2015). Professional soccer players are highly trained and the period of inactivity was perhaps not long enough to detrain them significantly. We also expected a decrease while training load increased (“Intensive training”, T1 to T2) and an increase at the end of the training period (end of “Tapering”, T3, Oliveira et al. 2013; Boullosa et al. 2013). The lack of significant change in lnRMSSD in the supine position during this training period could be a consequence of heterogeneity induced in our study group by the training itself. All the players followed the same training programme while it is known that their positions (striker or side defender) alter training load on the first hand (Ingebrigtsen et al. 2015) and that training adaptation depend upon individuals on the second hand (Vesterinen et al. 2015). Moreover, several patterns of fatigue have been described including by means of HRV (Schmitt et al. 2015). Therefore, the training might have led to a heterogeneous group that included players well adapted and players with several fatigue patterns. As expected, HRV spectral analysis in the supine position did not provide additional information and remained unchanged despite a good reproducibility of this method over several weeks (Kowalewski and Urban 2004).
HRV in the standing position
In the standing position, training altered LF with highly significant differences but only at the end of the whole training period (end of “Tapering”, T3). The intensity of the second period (“Intensive training”) was appropriate in terms of players’ conditioning according to soccer standards but it was probably not intensive enough to lead to spectral analysis changes. However, this LF alteration is not likely linked to a change in the sympathetic nervous system since usual witnesses of sympathetic activation during a stand test remained unchanged (LFnu, LF/HF). Moreover, differences in total power (overall heart rate variability, T) were also highly significant, as well as in cases of VLF and HF. Hence, training increased the overall heart rate variability but with no clear changes of the spectral indicators linked to sympathetic or to parasympathetic influences. The first possible explanation that comes to mind is that changes in T simply reflect the HR adaptation to training and that spectral analysis is not sensitive enough to detect the associated autonomic changes. However, the total power increase was not likely as a simple reflection of training HR decrease, since HRV did not parallel HR during 5 weeks of training (Table 1). HR decreased at T1, T2, and T3 in comparison with T0 while logT increased significantly only at T1 and T3 but not at T2 in comparison with T0 (Table 1). A second possible explanation is that breathing frequency adaptation to training influenced spectral analysis results and masked autonomics changes. We did not record breathing during our experiment. However, resting breathing is not supposed to change during a several week training (Manzi et al. 2009). Moreover, moderate alteration of resting breathing patterns does not influence spectral analysis results (Saboul et al. 2013). A third explanation is that the view on the cardiovascular regulation through autonomic activity and balance is too simple and does not reflect the complexity of the system. It is today clear that the traditional homeostatic view on the cardiovascular system should be revisited (Fortrat et al. 2016). More than 80 % of heart rate variability is not linked to the VLF, LF, and HF harmonics and is made of fractal noise, and other not yet well defined components (Yamamoto and Hughson 1994; Fortrat et al. 2013). Our results suggest that training influences the complex non-harmonic components of HRV and opens the way to further studies to characterize this adaptation and to identify an efficient tool to monitor training adaptation.
HRV and training load
The overall HRV (T) in the standing position increased when the training load was low (“Warm-up” T0 to T1 and “Tapering” T2 to T3, Table 1). By contrast, overall HRV (total power, T) remained unchanged when training load increased (“Intensive training” T1 to T2). We expected a decrease of LF in the standing position in response to the training load increase (Uusitalo et al. 2000; Hedelin et al. 2000; Mourot et al. 2004b). Portier et al. (2001) showed a 25 % LF decrease at the end of an intensive training period while Schmitt et al. (2013, 2015) demonstrated that fatigue is associated with a more than 40 % LF decrease. But the goal of the training period was to allow the players to reach a high level of performance at the beginning of the forthcoming competition. Therefore, when the training programme is analysed more accurately, we can see that each high intensity training day was followed by 2 days with a low training load, i.e., 72 h’ recovery between each high intensity session. This allowed players to avoid a build-up of fatigue. Furthermore, in an attempt to aid their recovery, tools such as cold baths, massages and specific drinks, were made available to the players. These tools stimulate the autonomic nervous system (Al Haddad et al. 2010; Hansen et al. 2014; Wälchli et al. 2014). Moreover, this period of intensive training could also lead to heterogeneity in our group as in the case of HRV in the supine position.
Study limitations
We did not check the players’ lifestyle on ethical grounds. Lifestyle impacts on HRV in both the supine and standing positions (Gonçalves et al. 2015). Thus training was not the only parameter to affect HRV. Furthermore, GPS concerned only five players and only speeds were recorded. We know that quantification of speeds alone underestimates the training load because many highly intense actions (jumps, tackles, duels, etc.) are not taken into consideration (Carling et al. 2015). The design of our study could not include a control group since not any elite soccer player would agree to not train and to be deliberately not fit for the upcoming competition season. The group size is small. However, it matched with most of the other studies that use spectral analysis of HRV. Moreover, the group size was large enough to show highly significant differences of some spectral analysis indicators (Table 1). We studied professional soccer players who are not representative of recreational soccer players. However, focusing on elites athletes we reached clear conclusion about the interest of HRV in the standing position and the non usefulness of lnRMSSD in training adaptation monitoring in cases of intermittent sports. Our study included repeated measurements in the supine and in the standing position. Such a design may benefit from a two way analysis of variance to also study the change of position effect (from supine to standing). However, our study focused on the training effect and on the comparison of lnRMSSD while supine and HRV while standing. Our aim was not to study the position change effect. This one is well known including in the training context (Task Force 1996; Kowalewski and Urban 2004). Readers interested in standing adaptation could refer to the supplemental material (Table S1).
Conclusion
This study highlights the merits of evaluating HRV spectral analysis in standing in so-called intermittent sporting disciplines. It confirms that the autonomic nervous system influences can be evaluated overall to monitor training adaptation in an accurate manner. It also shows that the lnRMSSD parasympathetic indicator in the supine position is not useful during longitudinal follow-up of training in intermittent sporting disciplines.
Abbreviations
- ANOVA:
-
Analysis of variance
- Bpm:
-
Beats per minute
- D0 to D35:
-
Day 0 to day 35 of training period
- GPS:
-
Global positioning system
- HF:
-
High frequencies of heart rate variability
- HFnu:
-
High frequencies in normalised units
- HR:
-
Heart rate
- HRV:
-
Heart rate variability
- LF:
-
Low frequencies of heart rate variability
- LFnu:
-
Low frequencies in normalised units
- LF/HF:
-
Low frequencies/high frequencies ratio
- lnRMSSD:
-
Natural logarithm of root mean square of the successive differences
- Log:
-
Indicates the logarithm transformation of the associated variable
- n.u:
-
Normalied units
- RMSSD:
-
Root mean square of the successive differences
- SEM:
-
Standard deviation to the mean
- T :
-
Total power
- T0–T3:
-
Stand test before the first training period and at the end of each
- VLF:
-
Very low frequencies in normalised units
- W1–W5:
-
Week 1 to week 5 of training
References
Al Haddad H, Laursen PB, Chollet D, Lemaitre F, Ahmaidi S, Buchheit M (2010) Effect of cold or thermoneutral water immersion on post-exercise heart rate recovery and heart rate variability indices. Auton Neurosci 156:111–116. doi:10.1016/j.autneu.2010.03.017
Atlaoui D, Pichot V, Lacoste L, Barale F, Lacour JR, Chatard JC (2007) Heart rate variability, training variation and performance in elite swimmers. Int J Sports Med 28:394–400
Boullosa DA, Abreu L, Nakamura FY, Muñoz VE, Domínguez E, Leicht AS (2013) Cardiac autonomic adaptations in elite Spanish soccer players during preseason. Int J Sports Physiol Perform 8:400–409
Buchheit M (2014) Monitoring training status with HR measures: do all roads lead to Rome? Front Physiol 5:73. doi:10.3389/fphys.2014.00073
Buchheit M, Chivot A, Parouty J, Mercier D, Al Haddad H, Laursen PB, Ahmaidi S (2010) Monitoring endurance running performance using cardiac parasympathetic function. Eur J Appl Physiol 108:1153–1167. doi:10.1007/s00421-009-1317-x
Carling C, Gregson W, McCall A, Moreira A, del Wong P, Bradley PS (2015) Match running performance during fixture congestion in elite soccer: research issues and future directions. Sports Med 45:605–613. doi:10.1007/s40279-015-0313-z
Carter JB, Banister EW, Blaber AP (2003) The effect of age and gender on heart rate variability after endurance training. Med Sci Sports Exerc 35:1333–1340
Fortrat JO, Baum C, Jeanguillaume C, Custaud MA (2013) Noisy fluctuation of heart rate indicates cardiovascular system instability. Eur J Appl Physiol 113:2253–2261. doi:10.1007/s00421-013-2655-2
Fortrat JO, Levrard T, Courcinous S, Victor J (2016) Self-organization of blood pressure regulation: experimental evidence. Front Physiol 7:112. doi:10.3389/fphys.2016.00112
Gonçalves TR, Farinatti Pde T, Gurgel JL, da Silva Soares PP (2015) Correlation between cardiac autonomic modulation in response to orthostatic stress and indicators of quality of life, physical capacity, and physical activity in healthy individuals. J Strength Cond Res 29:1415–1421. doi:10.1519/JSC.0000000000000769
Halson SL (2014) Monitoring training load to understand fatigue in athletes. Sports Med 2:S139–S147. doi:10.1007/s40279-014-0253-z
Hansen AL, Dahl L, Olson G, Thornton D, Graff IE, Frøyland L, Thayer JF, Pallesen S (2014) Fish consumption, sleep, daily functioning, and heart rate variability. J Clin Sleep Med 10:567–575. doi:10.5664/jcsm.3714
Hedelin R, Wiklund U, Bjerle P, Henriksson-Larsén K (2000) Pre- and post-season heart rate variability in adolescent cross-country skiers. Scand J Med Sci Sports 10:298–303
Ingebrigtsen J, Dalen T, Hjelde GH, Drust B, Wisløff U (2015) Acceleration and sprint profiles of a professional elite football team in match play. Eur J Sport Sci 15:101–110. doi:10.1080/17461391.2014.933879
Jansen AS, Nguyen XV, Karpitskiy V, Mettenleiter TC, Loewy AD (1995) Central command neurons of the sympathetic nervous system: basis of the fight-or-flight response. Science 270:644–659
Kiviniemi AM, Hautala AJ, Kinnunen H, Tulppo MP (2007) Endurance training guided individually by daily heart rate variability measurements. Eur J Appl Physiol 101:743–751
Kowalewski MA, Urban M (2004) Short- and long-term reproducibility of autonomic measures in supine and standing positions. Clin Sci (Lond) 106:61–66
Le Meur Y, Pichon A, Schaal K, Schmitt L, Louis J, Gueneron J, Vidal PP, Hausswirth C (2013) Evidence of parasympathetic hyperactivity in functionally overreached athletes. Med Sci Sports Exerc 45:2061–2071. doi:10.1249/MSS.0b013e3182980125
Manzi V, Castagna C, Padua E, Lombardo M, D’Ottavio S, Massaro M, Volterrani M, Iellamo F (2009) Dose–response relationship of autonomic nervous system responses to individualized training impulse in marathon runners. Am J Physiol 296:H1733–H1740. doi:10.1152/ajpheart.00054.2009
Moak JP, Goldstein DS, Eldadah BA, Saleem A, Holmes C, Pechnik S, Sharabi Y (2009) Supine low-frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation. Cleve Clin J Med 76(Suppl 2):S51–S59. doi:10.3949/ccjm.76.s2.11
Mourot L, Bouhaddi M, Perrey S, Rouillon JD, Regnard J (2004a) Quantitative Poincaré plot analysis of heart rate variability: effect of endurance training. Eur J Appl Physiol 91:79–87
Mourot L, Bouhaddi M, Tordi N, Rouillon JD, Regnard J (2004b) Short- and long-term effects of a single bout of exercise on heart rate variability: comparison between constant and interval training exercises. Eur J Appl Physiol 92:508–517
Naranjo Orellana J, de la Cruz Torres B, Sarabia Cachadiña E, de Hoyo M, Domínguez Cobo S (2015) Two new indexes for the assessment of autonomic balance in elite soccer players. Int J Sports Physiol Perform 10:452–457. doi:10.1123/ijspp.2014-0235
Oliveira RS, Leicht AS, Bishop D, Barbero-Álvarez JC, Nakamura FY (2013) Seasonal changes in physical performance and heart rate variability in high level futsal players. Int J Sports Med 34:424–430. doi:10.1055/s-0032-1323720
Olufsen MS, Alston AV, Tran HT, Ottesen JT, Novak V (2008) Modeling heart rate regulation-part I: sit-to-stand versus head-up tilt. Cardiovasc Eng 8:73–87
Perini R, Veicsteinas A (2003) Heart rate variability and autonomic activity at rest and during exercise in various physiological conditions. Eur J Appl Physiol 90:317–325
Pichon AP, de Bisschop C, Roulaud M, Denjean A, Papelier Y (2004) Spectral analysis of heart rate variability during exercise in trained subjects. Med Sci Sports Exerc 36:1702–1708
Plews DJ, Laursen PB, Stanley J, Kilding AE, Buchheit M (2013) Training adaptation and heart rate variability in elite endurance athletes: opening the door to effective monitoring. Sports Med 43:773–781. doi:10.1007/s40279-013-0071-8
Portier H, Louisy F, Laude D, Berthelot M, Guézennec CY (2001) Intense endurance training on heart rate and blood pressure variability in runners. Med Sci Sports Exerc 33:1120–1125
Rahman F, Pechnik S, Gross D, Sewell L, Goldstein DS (2011) Low frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation. Clin Auton Res 21:133–141. doi:10.1007/s10286-010-0098-y
Saboul D, Pialoux V, Hautier C (2013) The impact of breathing on HRV measurements: implications for the longitudinal follow-up of athletes. Eur J Sport Sci 13:534–542. doi:10.1080/17461391.2013.767947
Schmitt L, Regnard J, Desmarets M, Mauny F, Mourot L, Fouillot JP, Coulmy N, Millet G (2013) Fatigue shifts and scatters heart rate variability in elite endurance athletes. PLoS One 8(8):e71588. doi:10.1371/journal.pone.0071588 (eCollection 2013)
Schmitt L, Regnard J, Parmentier AL, Mauny F, Mourot L, Coulmy N, Millet GP (2015) Typology of “fatigue” by heart rate variability analysis in elite Nordic-skiers. Int J Sports Med 36:999–1007. doi:10.1055/s-0035-1548885
Task Force of The European Society of Cardiology and The North American Society of Pacing and Electrophysiology (1996) Heart rate variability standards of measurement, physiological interpretation, and clinical use. Eur Heart J 17:354–381
Uusitalo AL, Uusitalo AJ, Rusko HK (2000) Heart rate and blood pressure variability during heavy training and overtraining in the female athlete. Int J Sports Med 21:45–53
Vesterinen V, Häkkinen K, Hynynen E, Mikkola J, Hokka L, Nummela A (2013) Heart rate variability in prediction of individual adaptation to endurance training in recreational endurance runners. Scand J Med Sci Sports 23:171–180. doi:10.1111/j.1600-0838.2011.01365.x
Vesterinen V, Häkkinen K, Laine T, Hynynen E, Mikkola J, Nummela A (2015) Predictors of individual adaptation to high-volume or high-intensity endurance training in recreational endurance runners. Scand J Med Sci Sports. doi:10.1111/sms.12530 (In press)
Wälchli C, Saltzwedel G, Krüerke D, Kaufmann C, Schnorr B, Rist L, Eberhard J, Decker M, Simões-Wüst AP (2014) Physiologic effects of rhythmical massage: a prospective exploratory cohort study. J Altern Complement Med 20:507–515. doi:10.1089/acm.2012.0833
Yamamoto Y, Hughson RL (1994) On the fractal nature of heart rate variability in humans: effects of data length and beta-adrenergic blockade. Am J Physiol 266:R40–R49
Acknowledgments
We wish to thank the players for taking part in this study. We also wish to thank the Stade Lavallois Mayenne FC which allowed and facilitated this study. We extend our thanks to the Centre Hospitalier Universitaire d’Angers (Angers University Hospital) and the Centre de Recherche Clinique of the Centre Hospitalier-Universitaire d’Angers (Angers University Hospital Clinical Research Centre) for their help with the administrative procedures for biomedical research projects. JOF benefits from the support of the Centre National d’Études Spatiales (National Centre for Spatial Studies) (CNES, Grant # 2014/4800000763).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Communicated by Massimo Pagani.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ravé, G., Fortrat, JO. Heart rate variability in the standing position reflects training adaptation in professional soccer players. Eur J Appl Physiol 116, 1575–1582 (2016). https://doi.org/10.1007/s00421-016-3416-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-016-3416-9