Abstract
Background
Although silica is a proven lung carcinogen, there is no convincing evidence linking crystalline silica to gastrointestinal malignancies.
Methods
We detailedly searched studies on the link between gastrointestinal malignancies and occupational silica exposure. Studies published between 1987 and 2023 were found by searching PubMed, Scopus, Cochrane Library, and Web of Science databases. Further studies were included from reference searching. We conducted a meta-analysis of the incidence and mortality of gastrointestinal malignancies and occupational silica exposure. We computed pooled-risk estimates using random effects models. Egger’s regression asymmetry test and a funnel plot were used to identify publication bias. Moreover, sensitivity analysis and subgroup analysis were out.
Results
We identified 40 research with individuals from 13 different countries. The results indicate that occupational silica exposure raises the risk of gastric and esophageal cancer incidence, with pooled standardized incidence ratio of 1.35 (95% CI 1.21–1.51, p < 0.001), 1.31 (95% CI 1.04–1.65, p = 0.023), respectively, but there was a lack of statistically significant relationship between standardized mortality ratio. In addition, we found that silica exposure did not increase the risk of colorectal and pancreatic cancers. Occupational silica exposure was found to increase the risk of liver cancer, with pooled SIR and SMR of 1.19 (95% CI 1.04–1.35, p = 0.009), 1.24 (95% CI 1.03–1.49, p = 0.026), respectively.
Conclusions
We discovered a link between occupational silica exposure and gastrointestinal malignancies, with cancers of the liver, stomach, and esophagus being the most prevalent. Colorectal and pancreatic cancer were not linked to occupational silica exposure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Silica is the main component of rocks, commonly used as a raw material in mining, construction, ceramic and pottery industries, glass manufacturing, metals, and many others (Si et al. 2016). The International Agency for Research on Cancer (IARC) first identified crystalline silica (CS) as a category 1 human carcinogen in 1987 and categorized it as such in 1997 (Wilbourn et al. 1997). However, to date, many people are still exposed to silica, especially workers in some factories. According to estimates, silica dust exposure in the workplace affects tens of millions of individuals globally (Chen et al. 2012). At the same time, gastrointestinal (GI) cancers are common worldwide. According to global cancer statistics for 2020, gastrointestinal cancers account for about one-third of all cancer incidence and mortality worldwide (Sung et al. 2020). Colorectal, stomach, esophageal, liver, and pancreatic cancers are the most prevalent gastrointestinal cancers.
There is evidence that exposure to silica can cause cancers in organs other than lung cancer, such as the liver, gastric, colorectal, esophageal, and other cancers (Goldberg et al. 2001; Døssing et al. 1997). Men who work in mining, quarries, construction, and metal processing are at an elevated risk, supporting the idea that stomach cancer may be caused by exposure to dusty settings (Aragonés et al. 2002). Workers exposed to quartz dust (IRR: 1.3, 95% CI 1.0–1.7) and cement dust (IRR: 1.5, 95% CI 1.1–2.1) had an increased risk of developing stomach cancer (Sjödahl et al. 2007). Lian et al. (2019) found that silica phytoliths can contaminate wheat flour, result in recurrent local lesions in the esophagus, and promote proliferation by acting as anchoring. A hospital-based multi-center case–control study in Spain found that miners, stonemasons, and carvers had a higher risk of esophageal cancer (OR 10.78, 95% CI 1.24–93.7) (Santibanez et al. 2008). A Swedish study found that among women, occupational groups such as glass, pottery, and tile workers had a statistically significant higher risk of pancreatic cancer (Alguacil et al. 2003). A population-based case–control study conducted in Canada to explore the association between hundreds of occupational settings and colon cancer in men found 21 occupational agent exposures that increase the risk of colon cancer in men, including fiberglass (Goldberg et al. 2001). A nested case–control study conducted in Denmark examined the hypothesis that occupational exposure to chemical agents causes primary liver cancer. Among them, workers in the stone, clay and glass industries had a higher risk of primary liver cancer (OR: 1.53, 95% CI 1.0–2.3) than those who did not work in these sites (OR 1.34, 95% CI 0.8–1.8) (Døssing et al. 1997).
Many epidemiological studies and meta-analyses have previously demonstrated the association between occupational silica exposure and lung cancer. However, there is limited information investigating the carcinogenicity of silica in non-respiratory organs. We are interested in those who work in an environment with chronic silica exposure and clinically confirmed gastrointestinal cancer diagnoses. We, therefore, conducted this study to discover if working with silica increases the risk of developing or dying from gastrointestinal cancers.
Methods
Literature search
We carried out a systematic review and meta-analysis of cohort studies of workers exposed to silica in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. The PRISMA checklist is included in the supplementary material (Table S1). Two authors (Shao and Zhang) systematically searched and analyzed the literature published from 1987 to February 2023. Another author (Fu) independently re-reviewed the search and analysis results and addressed and decided on potential disagreements during the literature search. Four academics databases (PubMed, Scopus, Cochrane Library, and Web of Science) were used for searching through the following keywords: (1) silica, quartz; (2) workplaces where silica is exposed over time, such as ceramics, glass, mining, metallurgy, etc.; (3) illnesses resulting from prolonged exposure to silica, such as pneumosilicosis, silicosis, etc.; (4) gastrointestinal cancers, such as liver cancer, stomach cancer, pancreatic cancer, etc. (details can be found in Table S2). EndNote X9 (Clarivate Analytics) collected, managed and identified duplicate citations. The sources listed in the chosen articles were also taken into account.
Before starting the review, the systematic review protocol was registered with PROSPERO (registration number: CRD42022337773).
Study selection
We found 46,896 potentially relevant studies through preliminary searching. We excluded duplicate articles through EndNote X9. Based on the title and abstract, two authors (Zhang and Shao) reviewed the publications and chose those that fulfilled the inclusion criteria. The screened articles were read in their full text. We included only those articles that met the screening criteria. Disagreements that arose during the title, abstract, and full-text screening process were resolved through discussion with another author (Fu).
The inclusion criteria included: (1) design of the study: cohort studies; (2) occupational exposure: workers with exclusive or predominant occupational exposure to silica; (3) outcome: gastrointestinal cancer incidence or mortality.
The exclusion criteria included: (1) occupational exposure: workers with possible low-level exposure to silica and exposure to some other known carcinogens, such as seamen, and mechanics (If the studies met the inclusion criteria, i.e., workers who were primarily or exclusively exposed to silica in their daily work, they would not be excluded because of the level of silica exposure); (2) outcome: studies lacking confidence intervals or outcome measures other than standardized mortality or incidence.
Data extraction
We extracted the following information: (1) first author; (2) year of publication; (3) type of industry or occupation of employment (e.g., foundry, ceramics, or mining); (4) geographical region, divided into Europe, North America, Asia, and Oceania; (5) source of diagnosis: cancer registration, death certificate; (6) composition of the cohort by gender: M for male, F for female, and MF for both male and female; (7) years of follow-up; (8) observed number of cases; (9) Standardized mortality rate (SMR) or Standardized incidence rate (SIR) and 95% confidence interval (CI) for specific GI cancer subtypes.
Mortality studies rely on data from death certificates, whereas incidence studies use information from cancer registries.
Study quality
Quality assessment (QA) was performed based on the NIH score for a quality assessment tool for Observational Cohort and Cross-Sectional Studies (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools.). We divided the studies into three categories based on the scores obtained: (1) high quality (8–10 points); (2) medium quality (5–7 points); (3) low quality (0–4 points) (total 10 criteria, one point for each criteria met).
Statistical analysis
We calculated overall pooled effect estimates and 95% confidence intervals (CI) using a random effects model. We performed a pooled assessment of the risk of morbidity or mortality for each gastrointestinal cancer subtype. In the analysis, we combined colon and rectal cancer as colorectal cancer because both the colon and rectum belong to the large intestine. We conducted subgroup analyses based on study features to explore potential sources of heterogeneity. Subgroup analyses were performed according to the following features: gender (male, female or mixed), geographic region (Asia, Europe, North America, or Oceania), workplace (mining, ceramics, glass, foundry, Silicon carbide, diatomaceous earth or mixed), publication years (1987–2000, 2001–2010, 2011–2023).
Sensitivity analyses were performed to assess the outcomes’ robustness, and meta-analyses were repeated after excluding each study. We also employed funnel plots and Egger’s regression asymmetry test to evaluate publication bias. The levels of significance for all statistical tests were assumed to be equal to or less than 0.05. STATA 15 software was used for all statistical analyses.
Results
Literature selection and study characteristics
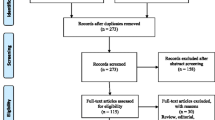
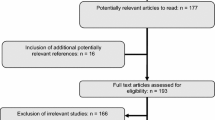
A systematic search of PubMed, Cochrane Library, Web of Science, and Scopus databases for articles published between 1987 and 2023 initially identified 46,896 articles. After removing 12,209 duplicates, the titles and abstracts of the remaining articles were reviewed, of which 34,390 were discarded after review. The remaining 297 articles were screened for full text based on inclusion and exclusion criteria; 257 did not meet the inclusion criteria [non-occupational exposure (n = 73), inconsistent or missing results (n = 110), wrong study design (n = 57), and conference paper (n = 17)]. The final 40 papers were selected in the meta-analysis since they satisfied the inclusion criteria. The flowchart in our meta-analysis is shown in Fig. 1.
We extracted 41 independent cohorts from 40 articles, of which 22 cohorts were from Europe (8 from Italy, 4 from the UK, 3 from Norway, 2 from the Czech Republic, and 1 each from Sweden, Poland, Germany, and the Netherlands), 8 from North America, 10 from Asia, and 1 from Oceania. They were all cohort studies, either retrospective or prospective. The follow-up period ranged from 10 to 20 years. One study evaluated the risk of gastrointestinal cancer incidence in terms of short-term versus long-term silica exposure, respectively (Bugge et al. 2010). A study also compared the risk of gastrointestinal cancers in patients with silicosis and those without silicosis (Tomaskova et al. 2012). The study found that non-silicosis workers had a higher risk of gastrointestinal cancers, while silicosis workers had a higher risk of lung cancer. Industries included in the studies covered mining, glass manufacturing, ceramics, smelting, silicon carbide, diatomaceous earth, and construction. Of these, 33 cohorts assessed the incidence of gastrointestinal cancers and 8 cohorts assessed mortality from gastrointestinal cancers. 28 cohorts reported the risk of gastric cancer; 23 cohorts reported the risk of liver cancer23 cohorts reported the risk of esophageal cancer; 22 cohorts reported the risk of pancreatic cancer, and 32 cohorts reported the risk of colorectal cancer. The particular characteristics of the studies included in the meta-analysis are summarized in Table 1.
Results of cancer incidence studies were obtained from cancer registries (n = 6). Most cancer mortality study results were obtained from death certificates (n = 32), and a few were obtained from population registries to determine the subject’s vital status (n = 2).
Meta-analysis
The results show that occupational silica exposure increases the risk of gastric and esophageal cancer incidence, with pooled SIR of 1.35 (95% CI 1.21–1.51, p < 0.001), 1.31 (95% CI 1.04–1.65, p = 0.023), respectively, but there was a lack of statistically significant relationship between mortality (pooled SMR for gastric cancer: 1.08, 95% CI 0.95–1.22, p = 0.228, pooled SMR for esophageal cancer: 0.88, 95% CI 0.64–1.21, p = 0.432). In addition, the study found a lack of statistically significant association between occupational silica exposure and mortality and incidence of colorectal and pancreatic cancer (pooled SIR for colorectal cancer: 0.98, 95% CI 0.88–1.08, p = 0.662, pooled SMR for colorectal cancer: 0.92, 95% CI 0.84–1.02, p = 0.101, pooled SIR for pancreatic cancer: 1.03, 95% CI 0.86–1.22, p = 0.767, pooled SMR for pancreatic cancer: 0.98, 95% CI 0.87–1.10, p = 0.747). Occupational silica exposure was found to increase the risk of liver cancer, with pooled SIR and SMR of 1.19 (95% CI 1.04–1.35, p = 0.009),1.24 (95% CI 1.03–1.49, p = 0.026), respectively. Figures 2 and 3 show the forest plots of occupational silica exposure and standardized incidence and mortality rate of gastrointestinal cancers, respectively.
Subgroup analysis
Gastric cancer
Figure 4 shows the results of a partial subgroup analysis of occupational silica exposure and gastrointestinal cancer risk. The results of the subgroup analysis of occupational silica exposure and risk of gastrointestinal cancer incidence and mortality are shown in Table S3 and Table S4, respectively. Subgroup analyses based on sex, industry, region, and year of literature publication showed that all subgroup analyses were consistent with the results of the meta-analysis, except that there was no significant association between female workers and foundry industry and gastric cancer incidence (SIR: 0.62, 95% CI 0.17–2.32; SIR: 0.82, 95% CI 0.47–1.43, respectively). The heterogeneity of all subgroup analyses was less than 10%. Mining and silicon carbide workers had a higher risk of gastric cancer, with pooled SIR results of 1.37 (95% CI 1.20–1.57) and 1.42 (95% CI 1.14–1.75), respectively. The risk of death from gastric cancer was significantly higher in the mining and foundry industry than in other sectors (SMR: 1.31, 95% CI 1.20–1.44; SMR: 1.25, 95% CI 1.08–1.45, respectively).
Esophageal cancer
The esophageal cancer subgroup analysis showed a significant association between the incidence of esophageal cancer and occupational silica exposure in men (SIR: 1.30, 95% CI 1.04–1.63). Similarly, there was a significant association between esophageal cancer incidence and occupational silica exposure in Asia (SIR: 1.76, 95% CI 1.25–2.47). The remaining subgroup analyses did not reveal a significant association with esophageal cancer incidence. Esophageal cancer mortality (SMR: 0.52, 95% CI 0.36–0.75) was significantly lower in the mixed male and female cohorts, but there was a large heterogeneity between studies. Esophageal cancer mortality rates (SMR: 0.56, 95% CI 0.38–0.83; SMR: 0.32, 95% CI 0.19–0.54, respectively) were also significantly lower in the pottery and metallurgical industries. The results of the remaining subgroup analyses were consistent with the meta-analysis.
Colorectal cancer
In the subgroup analysis of colorectal cancer, we also performed separate meta-analyses for colon and rectal cancers. All subgroup analyses showed no significant association between occupational silica exposure and colorectal cancer incidence, and there was no significant heterogeneity across all subgroup analyses. The pooled results from the literature published between 2011 and 2023 suggest that occupational silica exposure leads to a colorectal cancer mortality rate (SMR: 0.84, 95% CI 0.74–0.95) significantly lower, with low heterogeneity between studies. A similar pattern was found in the male and female mixed cohorts and in the mixed industry. Moreover, mortality rates for colon cancer (SMR: 0.88, 95% CI 0.79–0.97) were significantly lower with low heterogeneity. The remaining SMR subgroup analysis results were consistent with the meta-analysis results and showed no significant heterogeneity.
Pancreatic cancer
All subgroup analyses showed no significant association between pancreatic cancer incidence and mortality and occupational silica exposure, and there was no significant heterogeneity among all subgroups. The results of the subgroup analyses were consistent with the results of the meta-analysis.
Liver cancer
The risk of liver cancer incidence (SIR: 1.16, 95% CI 1.02–1.33) and mortality (SMR: 1.63, 95% CI 1.17–2.28) was higher among mining workers than in other industries. Asian workers had a higher risk of liver cancer incidence (SIR: 1.18, 95% CI 1.01–1.37) and mortality (SMR: 1.24, 95% CI 1.02–1.50) than North America (SIR: 1.10, 95% CI 0.83–1.46; SMR: 0.90, 95% CI 0.50–1.62). There was no significant association between liver cancer incidence and mortality and silica exposure in European and North American workers. Pooled SIR results from the literature published from 2001 to 2010 showed no significant correlation between occupational silica exposure and liver cancer incidence (SIR: 1.51, 95% CI 0.70–3.25), but pooled SMR results showed a significant correlation between occupational silica exposure and liver cancer mortality (SMR: 2.09, 95% CI 1.86–2.35). In contrast, the pooled results of the literature published between 2011 and 2023 showed the opposite, with the SMR pooled and SIR pooled results of 1.08 (95% CI 0.97–1.21) and 1.18 (95% CI 1.03–1.34), respectively. We performed a subgroup analysis of liver cancer mortality studies according to the year of publication in the literature and found that the heterogeneity between groups was 0.
Publication bias and sensitivity analyses
Although there was no evidence of publication bias in the egger assay results of the Liver Cancer Mortality Study, the funnel plots showed some asymmetry. Funnel plots did not show asymmetry for all studies except the liver cancer mortality study (Figure S1). None of the sensitivity analyses affected the combined results by omitting any article, except for the liver cancer incidence, mortality, and esophageal cancer incidence studies (Figure S2).
Quality assessment
Study quality assessments were performed by two independent authors using the NIH score. We modified this tool to fit better the studies we reviewed (Table S5). We performed a quality assessment of all studies included in the meta-analysis. Only three studies were considered high quality (Bugge et al. 2010; Ciocan et al. 2022; Wang et al. 2021), while the remaining studies were of moderate quality (n = 37). The quality assessment of all studies involved in this meta-analysis is shown in Table S6.
Discussion
Many studies have linked the risk of gastrointestinal malignancies to occupational silica exposure, but this link hasn’t always been deemed statistically significant. Therefore, we conducted a systematic search and quantitatively assessed this association. According to our findings, occupational silica exposure may increase the risk of liver, gastric and esophageal cancers, but there is no statistical association with pancreatic and colorectal cancers. A recently published study that systematically analyzed the association between PM2.5 exposure and gastrointestinal cancer risk similarly showed that pollutant particle exposure increases the risk of liver cancer (Pritchett et al. 2022). Although the findings suggest that occupational silica exposure increases the risk of liver cancer mortality, the heterogeneity between studies was high. Using subgroup analysis, we found that the heterogeneity between studies was zero after subgroup analysis based on the year of publication of the literature. The pooled mortality results for liver cancer have decreased since 2011, explaining the high heterogeneity of the overall pooled mortality results.
The higher risk of both gastric cancer incidence and mortality among mining workers may be because miners are most likely to be highly exposed to respirable crystalline silica (Si et al. 2016). There was no statistical association between the incidence of gastric cancer and occupational silica exposure in female and foundry workers. This result may be because only one study was included when the subgroup analysis was performed (Hung et al. 2014; Westberg et al. 2013).
As for the results of the partial subgroup analysis that found a reduced risk of mortality from esophageal cancer may be due to the progressive improvement in the prognosis of esophageal cancer in many countries in recent decades. Endoscopic procedures have been increasingly used to treat precancerous and early-stage esophageal cancer (Lagergren et al. 2017). There is a lack of statistical association between esophageal cancer incidence and silica exposure in women, as there is only one relevant study on women (Hung et al. 2014). The same lack of statistical association between incidence and silica exposure was observed among workers in Europe and North America. This may be due to some differences in occupational dust exposure limits between countries, with developed countries having lower occupational exposure limits for silica than developing countries. Compared to China, where the occupational exposure limit for respirable crystalline silica is 0.7 mg/m3, Canada has a 0.05 mg/m3 restriction (Vanka et al. 2022).
Pooled results from studies published between 2011 and 2023 found no statistically significant association between colorectal cancer incidence rates but a significant decrease in mortality. The increased use of sigmoidoscopy and colonoscopy in conjunction with polypectomy, which has a 5-year relative survival rate of over 65% in high-income nations like the United States, Europe, and Canada, may be to blame for this (Brenner et al. 2014).
In Europe and North America, there was no significant association between liver cancer incidence and mortality and occupational silica exposure, but a significant association was found for liver cancer incidence and mortality in Asia. This is probably attributable to the fact that the occupational exposure limits for silica in developed countries are lower than those in developing countries (Vanka et al. 2022). A significant association between morbidity and mortality and occupational silica exposure was only found in mine workers, possibly because miners are most likely to be highly exposed to respirable crystalline silica (Si et al. 2016). Primary liver cancer is not easily detected in its early stages and has a poor prognosis. As medical conditions have improved, liver cancer detection techniques as well as prognoses have evolved considerably. Thus, occupational silica exposure was observed unrelated to liver cancer incidence but associated with mortality from 2001 to 2010, while the opposite was observed from 2011 to 2023.
One motivation for our review was recognizing a biologically plausible association between occupational silica exposure and the development of gastrointestinal cancers. There are a number of possible mechanisms explaining this relationship. Silicon dioxide can enter the human body through respiratory inhalation, digestive intake, and skin contact. Among them, inhalation through breathing is the main route of human exposure. Silica accumulates most significantly in the lungs. Silica with a pneumatic diameter of less than 5 μm is more likely to reach the distal airways and alveoli, and some silica can enter the blood circulation through the air-blood barrier and reach the organs of the gastrointestinal tract. The pharynx is a common structure of the respiratory and digestive systems, and some of the gases may enter the digestive system by mistake when they enter the respiratory system (Gross et al. 2008). In addition, it has been found that occupational dust in the interstitial spaces of fine bronchi and alveoli can reach the organs of the gastrointestinal tract through mucus cilia clearance (Munkholm and Mortensen 2014). Zande et al. found that high concentrations of silica particles are more likely to accumulate in the stomach as aggregates and are difficult to be digested and absorb by the body (Zande et al. 2014). Numerous studies have demonstrated that certain nanoparticles may travel from the primary exposure locations (such as the lung or gut) to various secondary organs, including the liver. More than 90% of the translocated nanoparticles remain in the liver, making it a secondary exposure site that preferentially accumulates nanoparticles compared to other organs (Kermanizadeh et al. 2014). Liver damage, aberrant liver metabolism, and liver fibrosis can all be brought on by amorphous silica nanoparticles. Duan et al. (2018) found that silica nanoparticles (Si-NPs) caused moderate steatosis and inflammatory cell infiltration in the liver. Si-NPs prompted hyperlipemia and hepatic steatosis via the TLR5-signaling pathway. Liver steatosis and cellular inflammation are the main causes of cirrhosis and hepatocellular carcinoma in nonalcoholic fatty liver disease.
This comprehensive review and meta-analysis have various advantages, such as the first systematic review of occupational silica exposure and gastrointestinal cancer risk, and the quality assessment results of all involved literature were above moderate quality. The primary findings of our meta-analysis are further supported by the low degree of heterogeneity in research results, the consistency of results across subgroups, and the absence of publication bias. The present study has some limitations. Few of the studies we included specifically addressed occupational silica exposure in women (n = 3), which also led to some possible bias in our gender-based subgroup analysis results. Most studies on occupational silica exposure had focused on respiratory diseases and neglected the effect of occupational silica exposure on gastrointestinal diseases, which also led to the low number of literatures included in some groups when we performed subgroup analysis.
Conclusion
We discovered a link between occupational silica exposure and gastrointestinal malignancies, with cancers of the liver, stomach, and esophagus being the most prevalent. Colorectal and pancreatic cancer was not linked to occupational silica exposure. The carcinogenic effect of silica on the gastrointestinal system and its carcinogenic mechanism needs more studies to confirm.
Data availability
The author confirms that all data generated or analysed during this study are included in this published article.
Abbreviations
- GI:
-
Gastrointestinal
- CI:
-
Confidence intervals
- SIR:
-
Standardized incidence ratio
- SMR:
-
Standardized mortality ratio
- IARC:
-
The International Agency for Research on Cancer
- CS:
-
Crystalline silica
- PRISMA:
-
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- QA:
-
Quality assessment
- Si-NPs:
-
Silica nanoparticles
References
Adzersen KH, Becker N, Steindorf K, Frentzel-Beyme R (2003) Cancer mortality in a cohort of male German iron foundry workers. Am J Ind Med 43(3):295–305
Alguacil J, Pollán M, Gustavsson P (2003) Occupations with increased risk of pancreatic cancer in the Swedish population. Occup Environ Med 60(8):570–576
Allen EM, Alexander BH, MacLehose RF, Nelson HH, Ramachandran G, Mandel JH (2015) Cancer incidence among Minnesota taconite mining industry workers. Ann Epidemiol 25(11):811–815
Aragonés N, Pollán M, Gustavsson P (2002) Stomach cancer and occupation in Sweden: 1971–89. Occup Environ Med 59(5):329–337
Bartoli D, Battista G, De Santis M, Iaia TE, Orsi D, Tarchi M, Pirastu R, Valiani M (1998) Cohort study of art glass workers in Tuscany, Italy: mortality from non-malignant diseases. Occup Med 48(7):441–445
Berry G, Rogers A, Yeung P (2004) Silicosis and lung cancer: a mortality study of compensated men with silicosis in New South Wales, Australia. Occup Med 54(6):387–394
Brenner H, Kloor M, Pox CP (2014) Colorectal cancer. Lancet 383(9927):1490–1502
Brown TP, Rushton L (2005) Mortality in the UK industrial silica sand industry: 2. A retrospective cohort study. Occup Environ Med 62(7):446–452
Bugge MD, Kjuus H, Martinsen JI, Kjærheim K (2010) Cancer incidence among short- and long-term workers in the Norwegian silicon carbide industry. Scand J Work Environ Health 36(1):71–79
Carta P, Cocco PL, Casula D (1991) Mortality from lung cancer among Sardinian patients with silicosis. Br J Ind Med 48(2):122–129
Carta P, Aru G, Manca P (2001) Mortality from lung cancer among silicotic patients in Sardinia: an update study with 10 more years of follow up. Occup Environ Med 58(12):786–793
Checkoway H, Heyer NJ, Demers PA, Breslow NE (1993) Mortality among workers in the diatomaceous earth industry. Br J Ind Med 50(7):586–597
Checkoway H, Heyer NJ, Seixas NS, Welp EAE, Demers PA, Hughes JM, Weill H (1997) Dose–response associations of silica with nonmalignant respiratory disease and lung cancer mortality in the diatomaceous earth industry. Am J Epidemiol 145(8):680–688
Chen J, McLaughlin JK, Zhang JY, Stone BJ, Luo J, Chen RA, Dosemeci M, Rexing SH, Wu Z, Hearl FJ et al (1992) Mortality among dust-exposed Chinese mine and pottery workers. J Occup Med 34(3):311–316
Chen W, Yang J, Chen J, Bruch J (2006) Exposures to silica mixed dust and cohort mortality study in tin mines: exposure-response analysis and risk assessment of lung cancer. Am J Ind Med 49(2):67–76
Chen W, Liu Y, Wang H, Hnizdo E, Sun Y, Su L, Zhang X, Weng S, Bochmann F, Hearl FJ, Chen J, Wu T (2012) Long-term exposure to silica dust and risk of total and cause-specific mortality in Chinese workers: a cohort study. PLoS Med 9(4):e1001206
Cherry NM, Burgess GL, Turner S, McDonald JC (1998) Crystalline silica and risk of lung cancer in the potteries. Occup Environ Med 55(11):779–785
Ciocan C, Pira E, Coggiola M, Franco N, Godono A, La Vecchia C, Negri E, Boffetta P (2022) Mortality in the cohort of talc miners and millers from Val Chisone, northern Italy: 74 years of follow-up. Environ Res 203:111865
Døssing M, Petersen KT, Vyberg M, Olsen JH (1997) Liver cancer among employees in Denmark. Am J Ind Med 32(3):248–254
Duan J, Liang S, Feng L, Yu Y, Sun Z (2018) Silica nanoparticles trigger hepatic lipid-metabolism disorder in vivo and in vitro. Int J Nanomed 13:7303–7318
Gallagher LG, Park RM, Checkoway H (2015) Extended follow-up of lung cancer and non-malignant respiratorydisease mortality among California diatomaceous earth workers. Occup Environ Med 72(5):360–365
Goldberg MS, Parent ME, Siemiatycki J, Desy M, Nadon L, Richardson L, Lakhani R, Latreille B, Valois MF (2001) A case–control study of the relationship between the risk of colon cancer in men and exposures to occupational agents. Am J Ind Med 39(6):531–546
Goldsmith DF, Beaumont JJ, Morrin LA, Schenker MB (1995) Respiratory cancer and other chronic disease mortality among silicotics in California. Am J Ind Med 28(4):459–467
Gross RD, Atwood CW Jr, Ross SB, Eichhorn KA, Olszewski JW, Doyle PJ (2008) The coordination of breathing and swallowing in Parkinson’s disease. Dysphagia 23(2):136–145
Hobbesland A, Kjuus H, Thelle DS (1999) Study of cancer incidence among 8530 male workers in eight Norwegian plants producing ferrosilicon and silicon metal. Occup Environ Med 56(9):625–631
Hung YP, Teng CJ, Liu CJ, Hu YW, Hung MH, Tzeng CH, Liu CY, Yeh CM, Chen TJ, Chiou TJ (2014) Cancer risk among patients with coal workers’ pneumoconiosis in Taiwan: a nationwide population-based study. Int J Cancer 134(12):2910–2916
Kermanizadeh A, Gaiser BK, Johnston H, Brown DM, Stone V (2014) Toxicological effect of engineered nanomaterials on the liver. Br J Pharmacol 171(17):3980–3987
Lagergren J, Smyth E, Cunningham D, Lagergren P (2017) Oesophageal cancer. Lancet 390(10110):2383–2396
Lian C, Zuo X, Tian L (2019) A possible role of biogenic silica in esophageal cancer in North China? Environ Sci Pollut Res Int 26(8):8340–8343
Marinaccio A, Scarselli A, Gorini G, Chellini E, Mastrantonio M, Uccelli R, Altavista P, Pirastu R, Merlo DF, Nesti M (2006) Retrospective mortality cohort study of Italian workers compensated for silicosis. Occup Environ Med 63(11):762–765
Merlo F, Costantini M, Reggiardo G, Ceppi M, Puntoni R (1991) Lung cancer risk among refractory brick workers exposed to crystalline silica: a retrospective cohort study. Epidemiology 2(4):299–305
Miller BG, MacCalman L (2010) Cause-specific mortality in British coal workers and exposure to respirable dust and quartz. Occup Environ Med 67(4):270–276
Munkholm M, Mortensen J (2014) Mucociliary clearance: pathophysiological aspects. Clin Physiol Funct Imaging 34(3):171–177
Ng TP, Chan SL, Lee J (1990) Mortality of a cohort of men in a silicosis register: further evidence of an association with lung cancer. Am J Ind Med 17(2):163–171
Pritchett N, Spangler EC, Gray GM, Livinski AA, Sampson JN, Dawsey SM, Jones RR (2022) Exposure to outdoor particulate matter air pollution and risk of gastrointestinal cancers in adults: a systematic review and meta-analysis of epidemiologic evidence. Environ Health Perspect 130(3):36001
Romundstad P, Andersen A, Haldorse T (2001) Cancer incidence among workers in the Norwegian silicon carbide industry. Am J Epidemiol 153(10):978–986
Santibanez M, Vioque J, Alguacil J, Barber X, Garcia de la Hera M, Kauppinen T, PANESOES Study Group (2008) Occupational exposures and risk of oesophageal cancer by histological type: a case–control study in eastern Spain. Occup Environ Med 65(11):774–781
Scarselli A, Binazzi A, Forastiere F, Cavariani F, Marinaccio A (2011) Industry and job-specific mortality after occupational exposure to silica dust. Occup Med 61(6):422–429
Si S, Carey RN, Reid A, Driscoll T, Glass DC, Peters S, Benke G, Darcey E, Fritschi L (2016) The Australian work exposures study: prevalence of occupational exposure to respirable crystalline silica. Ann Occup Hyg 60(5):631–637
Sjödahl K, Jansson C, Bergdahl IA, Adami J, Boffetta P, Lagergren J (2007) Airborne exposures and risk of gastric cancer: a prospective cohort study. Int J Cancer 120(9):2013–2018
Sorahan T, Faux AM, Cooke MA (1994) Mortality among a cohort of United Kingdom steel foundry workers with special reference to cancers of the stomach and lung, 1946–90. Occup Environ Med 51(5):316–322
Starzyński Z, Marek K, Kujawska A, Szymczak W (1996) Mortality among different occupational groups of workers with pneumoconiosis: results from a register-based cohort study. Am J Ind Med 30(6):718–725
Steenland K, Brown D (1995) Mortality study of gold miners exposed to silica and nonasbestiform amphibole minerals: an update with 14 more years of follow-up. Am J Ind Med 27(2):217–229
Steenland K, Sanderson W (2001) Lung cancer among industrial sand workers exposed to crystalline silica. Am J Epidemiol 153(7):695–703
Sun J, Shibata E, Hisanaga N, Kamijima M, Ichihara G, Huang J, Toida M, Takeuchi Y (1997) A cohort mortality study of construction workers. Am J Ind Med 32(1):35–41
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Swaen GMH, Meijers JMM, Slangen JJM (1995) Risk of gastric cancer in pneumoconiotic coal miners and the effect of respiratory impairment. Occup Environ Med 52(9):606–610
Tomaskova H, Jirak Z, Splichalova A, Urban P (2012) Cancer incidence in Czech black coal miners in association with coalworkers’ pneumoconiosis. Int J Occup Med Environ Health 25(2):137–144
Ulm K, Gerein P, Eigenthaler J, Schmidt S, Ehnes H (2004) Silica, silicosis and lung-cancer: results from a cohort study in the stone and quarry industry. Int Arch Occup Environ Health 77(5):313–318
Vacek PM, Verma DK, Graham WG, Callas PW, Gibbs GW (2011) Mortality in Vermont granite workers and its association with silica exposure. Occup Environ Med 68(5):312–318
van der Zande M, Vandebriel RJ, Groot MJ, Kramer E, Herrera Rivera ZE, Rasmussen K, Ossenkoppele JS, Tromp P, Gremmer ER, Peters RJ, Hendriksen PJ, Marvin HJ, Hoogenboom RL, Peijnenburg AA, Bouwmeester H (2014) Sub-chronic toxicity study in rats orally exposed to nanostructured silica. Part Fibre Toxicol 11:8
Vanka KS, Shukla S, Gomez HM, James C, Palanisami T, Williams K, Chambers DC, Britton WJ, Ilic D, Hansbro PM, Horvat JC (2022) Understanding the pathogenesis of occupational coal and silica dust-associated lung disease. Eur Respir Rev 31(165):210250
Wang Z, Dong D, Liang X, Qu G, Wu J, Xu X (1996) Cancer mortality among silicotics in China’s metallurgical industry. Int J Epidemiol 25(5):913–917
Wang D, Yang M, Ma J, Zhou M, Wang B, Shi T, Chen W (2021) Association of silica dust exposure with mortality among never smokers: a 44-year cohort study. Int J Hyg Environ Health 236:113793
Westberg H, Andersson L, Bryngelsson IL, Ngo Y, Ohlson CG (2013) Cancer morbidity and quartz exposure in Swedish iron foundries. Int Arch Occup Environ Health 86(5):499–507
Wilbourn JD, McGregor DB, Partensky C, Rice JM (1997) IARC reevaluates silica and related substances. Environ Health Perspect 105(7):756–758
Yoon JH, Ahn YS (2014) Cause-specific mortality due to malignant and non-malignant disease in Korean foundry workers. PLoS ONE 9(2):e88264
Yu ITS, Tse LA, Wong TW, Leung CC, Tam CM, Chan ACK (2005) Further evidence for a link between silica dust and esophageal cancer. Int J Cancer 114(3):479–483
Zambon P, Simonato L, Mastrangelo G, Winkelmann R, Saia B, Crepet M (1987) Mortality of workers compensated for silicosis during the period 1959–1963 in the Veneto region of Italy. Scand J Work Environ Health 13(2):118–123
Zhang X, Wang H, Zhu X, Liu Y, Wang L, Dai Q, Cai N, Wu T, Chen W (2008) Cohort mortality study in three ceramic factories in Jingdezhen in China. J Huazhong Univ Sci Technol Med Sci = Hua Zhong Ke Ji Da Xue Xue Bao. Yi Xue Ying De Wen Ban = Huazhong Keji Daxue Xuebao Yixue Yingdewen Ban 28(4):386–390
Acknowledgements
The authors acknowledge all the participants and administrators of this study.
Funding
This work was supported by National Natural Science Foundation of China (42177419).
Author information
Authors and Affiliations
Contributions
JYS conducted the literature search, reviewed the articles, conducted the statistical analyses, and drafted the manuscript. PFF and XZ made substantial contributions to interpretation of data, and were involved in drafting the manuscript or revising it critically for important intellectual content. SCW and HC performed the data collation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
No ethics approval.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shao, J., Fu, P., Wang, S. et al. Occupational exposure to silica and risk of gastrointestinal cancers: a systematic review and meta-analysis of cohort studies. Int Arch Occup Environ Health 97, 231–251 (2024). https://doi.org/10.1007/s00420-024-02045-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00420-024-02045-3