Abstract
Introduction
Patients with non-aneurysmal subarachnoid hemorrhage (SAH) are considered to have an overall benign course of disease compared to patients suffering from aneurysmal SAH. Nevertheless, a small but significant number of such patients might only achieve unfavorable outcome. Therefore, the purpose of the present study was to determine if routine laboratory markers of acute phase response are associated with unfavorable outcome in patients with non-aneurysmal SAH.
Methods
From 2006 to 2017, 154 patients suffering from non-aneurysmal SAH were admitted to our institution. Patients were stratified according to the distribution of cisternal blood into patients with perimesencephalic SAH (pSAH) versus non-perimesencephalic SAH (npSAH). C-reactive protein (CRP) and white blood cells (WBC) assessments were performed within 24 h of admission as part of routine laboratory workup. Outcome was assessed according to the modified Rankin Scale (mRS) after 6 months and stratified into favorable (mRS 0–2) vs. unfavorable (mRS 3–6).
Results
The multivariate regression analysis revealed “CRP > 5 mg/l” (p = 0.004, OR 143.7), “WBC count > 12.1 G/l” (p = 0.006, OR 47.8), “presence of IVH” (p = 0.02, OR 13.5), “poor-grade SAH” (p = 0.01, OR 45.2) and “presence of CVS” (p = 0.003, OR 149.9) as independently associated with unfavorable outcome in patients with non-aneurysmal SAH.
Conclusion
Elevated C-reactive protein and WBC count at admission were associated with unfavorable outcome after non-aneurysmal SAH.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A ruptured intracranial aneurysm is usually the source of bleeding in patients suffering from spontaneous subarachnoid hemorrhage (SAH). Nevertheless, in approximately 15% of patients suffering from SAH, identification of the bleeding source is not possible [1,2,3]. This subgroup has been described as patients suffering from non-aneurysmal, non-traumatic, spontaneous subarachnoid hemorrhage previously [4]. Patients with non-aneurysmal SAH are considered to have an overall benign course of disease compared to patients suffering from aneurysmal SAH [2, 5, 6]. Nevertheless, some of these patients can develop delayed cerebral ischemia (DCI) during treatment course [3, 7]. Although the functional outcomes of most patients with non-aneurysmal SAH are favorable, a small but significant number of such patients might only achieve unfavorable outcome [1, 8, 9]. Thus, it is important to identify patients at risk of unfavorable functional outcomes during the early stage in the treatment course. Following, there is growing evidence of the prognostic value of c-reactive protein (CRP) and white blood cell (WBC) in patients with aneurysmal SAH [10, 11]. However, the independent value of CRP and WBC for risk prediction of unfavorable outcome after non-aneurysmal SAH has not been established. Therefore, the purpose of the present study was to determine if markers of acute phase response (CRP, WBC) are associated with unfavorable outcome in patients with non-aneurysmal SAH. Furthermore, we analyzed our institutional data in order to elucidate possible risk factors associated with unfavorable outcome in patients suffering from non-aneurysmal subarachnoid hemorrhage.
Materials and methods
Patients
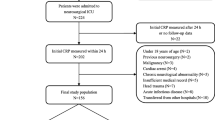
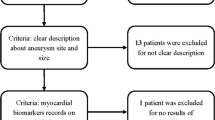
Between January 2006 and July 2017, 978 patients suffering from spontaneous SAH were admitted to our institution. SAH was diagnosed by computed tomography (CT) or lumbar puncture. All patients with spontaneous SAH underwent four-vessel digital subtraction angiography (DSA). All patients with confirmed bleeding source (intracranial aneurysm, vascular malformation) or traumatic origin of SAH were excluded from further analysis. In patients with angiogram-negative SAH, a spinal magnetic resonance imaging (MRI) scan was performed in order to rule out any spinal bleeding source of SAH. Furthermore, patients underwent a second four-vessel DSA 6 weeks after ictus if a potential bleeding source was not identified during the first diagnostic work-up. Patients with spontaneous, non-traumatic SAH without identification of a bleeding source after diagnostic screening were then included for further analysis in the present study.
Information, including patient characteristics on admission and during treatment course, radiological features, and functional neurological outcome were collected and entered into a computerized database. The World Federation of Neurological Surgeons (WFNS) scale was used in order to grade patients on admission [12, 13]. Patients suffering from spontaneous SAH were divided into good grade (WFNS grades I-III) versus (vs.) poor grade (WFNS grades IV-V) on admission. Patients with SAH were stratified into two groups according to the subarachnoid blood distribution into (1) patients suffering from perimesencephalic SAH (pSAH) and (2) patients suffering from non-perimesencephalic SAH (npSAH) [3, 4]. In addition, further radiological features were identified and collected, including presence of intraventricular hemorrhage (IVH), and new delayed cerebral ischemia (DCI) during treatment course. Furthermore, C-reactive protein (CRP) and white blood cells (WBC) assessments were performed within 24 h of admission as part of routine laboratory workup. WBC counts (normal range: 3.9–10.2 G/l) were dichotomized > 12.1 G/l to indicate moderate inflammation. CRP (normal range: 0–3 mg/l) was dichotomized > 5 mg/l to suggest moderate inflammation.
Patients with acute hydrocephalus (AH) were treated by ventriculostomy for external CSF diversion [14]. All patients with spontaneous SAH received nimodipine from the day of clinical admission. Screening for cerebral vasospasm (CVS) was performed daily using neurological examination and transcranial Doppler ultrasound (TCD) measurements. If CVS was suspected on the basis of TCD or delayed ischemic neurological deficit (DIND), CT-angiography/-perfusion (CT-A, CT-P) were performed in order to confirm CVS. Presence of clinical-relevant CVS was defined as CVS-associated DIND and/or CVS-related perfusion deficit in CT-P. In cases of onset of clinical-relevant CVS, hypertension was induced with catecholamines during treatment course [15]. DCI was defined as occurrence of new ischemic lesions on any radiological imaging during treatment course compared to the initial CT scan on admission.
Outcome was assessed according to the modified Rankin Scale (mRS) after 6 months and stratified into favorable (mRS 0–2) vs. unfavorable (mRS 3–6).
Statistics
Data analyses were performed using the computer software package SPSS (version 25, IBM Corp., Armonk, NY). Unpaired t test was used for parametric statistics. Categorical variables were analyzed in contingency tables using Fisher’s exact test. Results with p < 0.05 were considered statistically significant.
Furthermore, in order to find potential confounding factors between potentially independent predictors a multivariate analysis was performed to find independent predictors of unfavorable outcome in patients suffering from non-aneurysmal SAH using binary logistic regression. Variables with significant p values in the univariate analysis were considered as potentially independent variables in a multivariate analysis. A backward stepwise method was used to construct a multivariate logistic regression model in relation to unfavorable outcome as a dependent variable with an inclusion criterion of a p value < 0.05.
Results
Patient characteristics
Between January 2006 and July 2017, 978 patients suffering from spontaneous SAH were admitted to our institution, and in 154 patients of which, no bleeding source was found initially and throughout the follow-up (16%). Patient characteristics, including age, clinical admission status, radiological features, development of CVS and/or DCI, WBC count, CRP, presence of acute hydrocephalus, information concerning the use of anticoagulation agents prior SAH, and clinical outcome of the present series are shown in detail in Table 1.
Factors influencing functional outcome after non-aneurysmal SAH
Influence of clinical and radiological features
Overall, 138 patients (90%) suffering from non-aneurysmal SAH achieved favorable outcome. Patients with unfavorable outcome were significantly more often older than 65 years compared to patients who achieved favorable outcome (63% vs. 18%; p = 0.0003, OR 7.5, 95% CI 2.5–22.7). Additionally, patients with unfavorable outcome after non-aneurysmal SAH presented significantly more often with anticoagulation medication intake prior ictus compared to patients who achieved favorable outcome (50% vs. 21%; p = 0.03, OR 3.8, 95% CI 1.3–10.9).
Patients with non-aneurysmal SAH and IVH achieved significantly less often favorable outcome compared to patients with non-aneurysmal SAH without additional IVH (22% vs. 63%; p = 0.002, OR 5.8, 95% CI 1.9–17.1). Patients with unfavorable outcome presented significantly more often with Fisher 3 grade blood distribution compared to patients with favorable outcome (94% vs. 53%; p = 0.01, OR 10.1, 95% CI 1.3–78.7). Patients with favorable outcome presented significantly more often with perimesencephalic blood distribution compared to patients with unfavorable neurological outcome (50% vs. 0%; p < 0.0001, OR 33.0, 95% CI 1.9–561.3).
Influence of laboratory values
Furthermore, patients who achieved favorable outcome presented significantly more often with CRP ≤ 5 mg/l compared to patients with unfavorable outcome after non-aneurysmal SAH (76% vs. 13%; p < 0.0001, OR 22.3, 95% CI 4.8–103.4; Fig. 1A). Patients who achieved favorable outcome presented significantly more often with WBC counts ≤ 12.1 G/l compared to patients with unfavorable outcome after non-aneurysmal SAH (79% vs. 25%; p < 0.0001, OR 11.3, 95% CI 3.4–37.6; Fig. 1B).
Detailed results of univariate analysis concerning factors influencing functional outcome in patients suffering from non-aneurysmal SAH are given in Table 1.
Multivariate analysis
We performed a multivariate logistic regression analysis of those variables significantly associated with unfavorable outcome in patients suffering from non-aneurysmal SAH.
Multivariate analysis revealed the variables “CRP > 5 mg/l” (p = 0.004, OR 143.7, 95% CI 4.9–4144.2), “WBC count > 12.1 G/l” (p = 0.006, OR 47.8, 95% CI 3.1–747.8), “presence of IVH” (p = 0.02, OR 13.5, 95% CI 1.5–125.8), “poor-grade SAH” (p = 0.01, OR 45.2, 95% CI 2.5–818.9), and “presence of CVS” (p = 0.003, OR 149.9, 95% CI 5.5–4035.5) as significant and independent predictors for unfavorable outcome in patients with non-aneurysmal SAH (Nagelkerke’s R2 78%).
Discussion
In the present analysis of patients with non-aneurysmal SAH, it was found that initial elevated CRP values, increased WBC count, presence of intraventricular hemorrhage, poor clinical admission status, as well as presence of CVS are independent and significant predictors for unfavorable outcome in patients with non-aneurysmal SAH. Furthermore, the occurrence of CVS was the only independent and significant predictor for development of DCI during treatment course in patients with non-aneurysmal SAH. The present results are generally in line with the previous studies [3, 16,17,18]. However, potential influence of individual and routine laboratory tests (CRP, WBC) on functional outcome were not investigated in these reports.
Influence of CRP and WBC on outcome in patients with non-aneurysmal SAH
Despite well-known risk factors for unfavorable outcome after non-aneurysmal SAH, the present study revealed a strong and independent association between CRP, WBC count and functional outcome in these patients.
Several studies have reported that increased levels of acute inflammatory markers, such as CRP and WBC count are associated with increased risk of unfavorable outcome in patients with aneurysmal SAH [10, 11, 19,20,21,22]. However, data on inflammatory response in patients suffering from non-aneurysmal SAH is scarce. Muroi et al. reported lower interleukin-6-levels in patients with non-aneurysmal SAH compared to patients with aneurysmal SAH [23]. The present study demonstrate that elevated CRP values and WBC counts are independent of confounding factors predict an unfavorable outcome following non-aneurysmal SAH. The pathophysiological reason for an association between, CRP, WBC and outcome in patients with non-aneurysmal SAH is uncertain. Frontera et al. stated that inflammation markers are associated with early brain injury in patients with aneurysmal SAH [24]. However, CRP and WBC are relatively non-specific inflammatory markers [24]. Previously, Turner et al. reported an elevated baseline CRP as predictor for unfavorable outcome in patients with aneurysmal SAH and therefore as a potential selection tool for good grade patients with SAH who are at higher risk for unfavorable outcome [25]. Therefore, future studies should evaluate further inflammation markers in order to better comprehend potential correlation between inflammation and its impact on functional outcome in patients with non-aneurysmal SAH.
Nevertheless, CRP and WBC are routinely collected in patients presenting with SAH and are therefore, a potential simple way to determine the risk of unfavorable outcome for patients who are assumed to have a more benign illness compared to patients with aneurysmal SAH.
Limitations
The present study has several limitations. Statistical analysis and data collection were retrospective and the present data represent only a single-center experience. Furthermore, there is only a small number of patients with unfavorable outcome in the present patient collective, which might also influence statistical results. Although our data show a strong association between CRP, WBC count and outcome, a causal relationship remains to be established. Nevertheless, the results of the present study should be considered for further validation in future studies.
Conclusions
Elevated C-reactive protein and WBC count at admission were associated with unfavorable outcome after non-aneurysmal SAH. The results of the present study provide valuable information on functional outcome in patients suffering from non-aneurysmal SAH and should be taken in consideration during management of these patients.
References
Rinkel GJ, Wijdicks EF, Vermeulen M, Hasan D, Brouwers PJ, van Gijn J (1991) The clinical course of perimesencephalic nonaneurysmal subarachnoid hemorrhage. Ann Neurol 29(5):463–468
van Gijn J, van Dongen KJ, Vermeulen M, Hijdra A (1985) Perimesencephalic hemorrhage: A nonaneurysmal and benign form of subarachnoid hemorrhage. Neurology 35(4):493–497
Schuss P, Hadjiathanasiou A, Brandecker S, Wispel C, Borger V, Güresir Á, Vatter H, Güresir E (2018) Risk factors for shunt dependency in patients suffering from spontaneous, non-aneurysmal subarachnoid hemorrhage. Neurosurg Rev. https://doi.org/10.1007/s10143-018-0970-0.
Rinkel GJ, Wijdicks EF, Hasan D, Kienstra GE, Franke CL, Hageman LM, Vermeulen M, van Gijn J (1991) Outcome in patients with subarachnoid haemorrhage and negative angiography according to pattern of haemorrhage on computed tomography. Lancet 338(8773):964–968
Elhadi AM, Zabramski JM, Almefty KK, Mendes GA, Nakaji P, McDougall CG, Albuquerque FC, Preul MC, Spetzler RF (2015) Spontaneous subarachnoid hemorrhage of unknown origin: Hospital course and long-term clinical and angiographic follow-up. J Neurosurg 122(3):663–670
Konczalla J, Schmitz J, Kashefiolasl S, Senft C, Platz J, Seifert V (2016) Non-aneurysmal non-perimesencephalic subarachnoid hemorrhage: effect of rehabilitation at short-term and in a prospective study of long-term follow-up. Top Stroke Rehabil 23(4):261–268
Konczalla J, Kashefiolasl S, Brawanski N, Lescher S, Senft C, Platz J, Seifert V (2016) Cerebral vasospasm and delayed cerebral infarctions in 225 patients with non-aneurysmal subarachnoid hemorrhage: the underestimated risk of fisher 3 blood distribution. J Neurointerventional Surg 8(12):1247–1252
Gupta SK, Gupta R, Khosla VK, Mohindra S, Chhabra R, Khandelwal N, Gupta V, Mukherjee KK, Tewari MK, Pathak A, Mathuriya SN (2009) Nonaneurysmal nonperimesencephalic subarachnoid hemorrhage: is it a benign entity? Surg Neurol 71(5):566–571; (discussion 571,571–562,572)
Al-Mufti F, Merkler AE, Boehme AK, Dancour E, May T, Schmidt JM, Park S, Connolly ES, Lavine SD, Meyers PM, Claassen J, Agarwal S (2018) Functional outcomes and delayed cerebral ischemia following nonperimesencephalic angiogram-negative subarachnoid hemorrhage similar to aneurysmal subarachnoid hemorrhage. Neurosurgery 82(3):359–364
Juvela S, Kuhmonen J, Siironen J (2012) C-reactive protein as predictor for poor outcome after aneurysmal subarachnoid haemorrhage. Acta Neurochir 154(3):397–404
Al-Mufti F, Misiolek KA, Roh D, Alawi A, Bauerschmidt A, Park S, Agarwal S, Meyers PM, Connolly ES, Claassen J, Schmidt JM (2018) White blood cell count improves prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurgery. https://doi.org/10.1093/neuros/nyy045
Rosen DS, Macdonald RL (2004) Grading of subarachnoid hemorrhage: modification of the world world federation of neurosurgical societies scale on the basis of data for a large series of patients. Neurosurgery 54(3):566–575 (discussion 575–566)
Teasdale GM, Drake CG, Hunt W, Kassell N, Sano K, Pertuiset B, De Villiers JC (1988) A universal subarachnoid hemorrhage scale: report of a committee of the world federation of neurosurgical societies. J Neurol Neurosurg Psychiatry 51(11):1457
Schuss P, Wispel C, Borger V, Güresir Á, Vatter H, Güresir E (2018) Accuracy and safety of ventriculostomy using two different procedures of external ventricular drainage: a single-center series. J Neurol Surg Part A Central Eur Neurosurg 79(3):206–210
Raabe A, Beck J, Keller M, Vatter H, Zimmermann M, Seifert V (2005) Relative importance of hypertension compared with hypervolemia for increasing cerebral oxygenation in patients with cerebral vasospasm after subarachnoid hemorrhage. J Neurosurg 103(6):974–981
Boswell S, Thorell W, Gogela S, Lyden E, Surdell D (2013) Angiogram-negative subarachnoid hemorrhage: outcomes data and review of the literature. J Stroke Cerebrovasc Dis 22(6):750–757
Konczalla J, Schuss P, Platz J, Vatter H, Seifert V, Güresir E (2015) Clinical outcome and prognostic factors of patients with angiogram-negative and non-perimesencephalic subarachnoid hemorrhage: benign prognosis like perimesencephalic sah or same risk as aneurysmal sah? Neurosurg Rev 38(1):121–127 (discussion 127)
Jabbarli R, Reinhard M, Roelz R, Shah M, Niesen WD, Kaier K, Taschner C, Weyerbrock A, Van Velthoven V (2015) Outcome prediction after non-aneurysmal non-traumatic subarachnoid hemorrhage. Curr Neurovasc Res 12(3):269–276
McGirt MJ, Mavropoulos JC, McGirt LY, Alexander MJ, Friedman AH, Laskowitz DT, Lynch JR (2003) Leukocytosis as an independent risk factor for cerebral vasospasm following aneurysmal subarachnoid hemorrhage. J Neurosurg 98(6):1222–1226
Rothoerl RD, Axmann C, Pina AL, Woertgen C, Brawanski A (2006) Possible role of the c-reactive protein and white blood cell count in the pathogenesis of cerebral vasospasm following aneurysmal subarachnoid hemorrhage. J Neurosurg Anesthesiol 18(1):68–72
Dhar R, Diringer MN (2008) The burden of the systemic inflammatory response predicts vasospasm and outcome after subarachnoid hemorrhage. Neurocritical Care 8(3):404–412
Tam AK, Ilodigwe D, Mocco J, Mayer S, Kassell N, Ruefenacht D, Schmiedek P, Weidauer S, Pasqualin A, Macdonald RL (2010) Impact of systemic inflammatory response syndrome on vasospasm, cerebral infarction, and outcome after subarachnoid hemorrhage: Exploratory analysis of conscious-1 database. Neurocritical Care 13(2):182–189
Muroi C, Bellut D, Coluccia D, Mink S, Fujioka M, Keller E (2011) Systemic interleukin-6 concentrations in patients with perimesencephalic non-aneurysmal subarachnoid hemorrhage. J Clin Neurosci 18(12):1626–1629
Frontera JA, Provencio JJ, Sehba FA, McIntyre TM, Nowacki AS, Gordon E, Weimer JM, Aledort L (2017) The role of platelet activation and inflammation in early brain injury following subarachnoid hemorrhage. Neurocritical Care 26(1):48–57
Turner CL, Budohoski K, Smith C, Hutchinson PJ, Kirkpatrick PJ, Murray GD, collaborators S (2015) Elevated baseline c-reactive protein as a predictor of outcome after aneurysmal subarachnoid hemorrhage: data from the simvastatin in aneurysmal subarachnoid hemorrhage (STASH) trial. Neurosurgery 77(5):786–792; (discussion 792–783)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The present study was approved by the local ethics committee.
Informed consent
Informed consent was not sought as a retrospective study design was used.
Rights and permissions
About this article
Cite this article
Schuss, P., Hadjiathanasiou, A., Brandecker, S. et al. Elevated C-reactive protein and white blood cell count at admission predict functional outcome after non-aneurysmal subarachnoid hemorrhage. J Neurol 265, 2944–2948 (2018). https://doi.org/10.1007/s00415-018-9091-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-9091-5