Abstract
Cranial vault thickness (CVT) and its variations provide valuable insights into an individual’s biological attributes such as age and sex. This paper aimed to assess the correlations of CVT with age and sex and develop a regression model for age estimation in Malaysian subadults using computed tomography (CT) images. A total of 521 CT images (male/female: 279/242; age range: 0–20 years; Malay/Chinese/Indian: 221/145/155) were included in the study. Correlations of CVT measurements with age and sex, including frontal bone thickness (FBT), occipital bone thickness (OBT), left parietal bone thickness (LPBT), and right parietal bone thickness (RPBT) were assessed and regression formulae were developed for age estimation in subadults. A significant correlation between CVT measurements and age was demonstrated (p < 0.001). Age estimation was most accurate in the younger age group (< 2 years) at frontal and occipital, and accuracy decreases in the older age groups. Additionally, sexual dimorphism was evident in the frontal and parietal bone thickness within the age range of 3–6 years and 16–20 years, respectively. In conclusion, the findings suggested CVT measurements could be used to corroborate other age estimation methods for subadults.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Skull is a valuable source of information for creating a person’s biological profile such as age and sex [1]. Cranial vault thickness (CVT) was shown to be correlated with age and sex, and remains as the subject of ongoing discussion and research [2]. However, various studies have produced inconsistent results, due to differences in measurement techniques and analysis methods employed [3,4,5]. One contributing factor to the inconsistencies is the variation in the specific landmarks chosen for skull measurements, even in similar bones. The choice of measurement landmarks may influence the obtained results, leading to significant variations in the correlation between CVT and the studied factors. Moreover, different measurement techniques, such as manual callipers, two-dimensional (2D) imaging, or three-dimensional (3D) imaging, could also contribute to discrepancies in findings. The variations in techniques and approaches across studies have made it difficult to compare and draw definitive conclusions about the relationships between CVT and different biological parameters [2].
The limited published data on subadults’ CVT underscores the need for additional research in this specific area. Most anthropometry methods were developed for adults and may not be suitable for subadult cases [3, 6,7,8]. The unique challenges in subadult CVT studies are due to the ongoing growth and complex development process during this period [9]. This includes sutural growth, bone remodelling, and changes in cranial proportions. Hence, it is essential to develop an accurate and reliable method for measuring CVT and establish comprehensive CVT data specifically for subadults. Addressing this gap will equip researchers and practitioners with the knowledge necessary for analysing and interpreting CVT in subadults, thereby enhancing outcomes across forensic anthropology, medical research, and clinical practice [10].
3D imaging technology, such as computed tomography (CT) images provides valuable insights in forensic anthropology, medical research, and clinical practice [7]. This technology allows non-invasive assessment of skull thickness and evaluation of larger samples [11]. Researchers are able to analyse more extensive and diverse populations due to availability and accessibility of clinical CT scans, facilitating the study of skull thickness variations in relation to age, sex, and various skull conditions [12]. This 3D approach may enhance the understanding of skull thickness variations across demographic groups and contributes to the development of potentially more accurate and reliable biological profiles in forensic anthropology [7]. Therefore, this paper aimed to assess the correlations of CVT with age and sex and develop a regression model for age estimation in Malaysian subadults using CT images.
Materials and methods
Approval for research was granted by the Medical Research Ethics Committee, UMMC (MREC ID NO: 202,147 − 10,039). All procedures conducted in this study involving human participants followed the guidelines and regulation standards of the national research committee and the 1964 Helsinki Declaration. The informed consent requirement was waived by the Medical Research Ethics Committee, UMMC because this is a retrospective study that used CT images obtained from the archives of the hospital. Research management fund (project no: RMF 0637–2021) from Universiti Malaya was used to fund this study.
A dataset containing 521 CT images of the cranium was retrospectively selected from University Malaya Medical Centre’s (UMMC) radiology department. The sample included of 279 male and 242 female Malaysian patients attending the UMMC from June 2020 to March 2021, aged between 0 and 20 years. Figure 1 shows the demographic profiles distribution.
The CT scan images were obtained using the Siemens SOMATOM (Forchheim, Germany) scanner. Samples with skull malformation, past trauma, surgical history, defects or pathological conditions, infectious, or neoplastic pathology were excluded. Tube voltage at 120 kV, 110–450 mAs, 0.4s exposure time, voxel size of 0.625 mm x 0.625 mm x 1 mm, and convolution kernel ranging from H40s to H60s were used to acquire images stored in DICOM format.
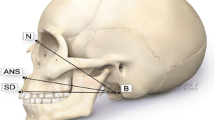
The skull model was reconstructed into multiplanar (axial, coronal, and sagittal) views. Bregma, lambda, left and right euryon landmarks were identified, and the coordinate values (x, y, and z) were generated automatically by the system for each landmark within the 3D coordinate framework. Four CVT landmarks: frontal bone thickness (FBT) = 1 cm in front of the bregma; occipital bone thickness (OBT) = 1 cm behind the lambda; left parietal bone thickness (LPBT) = left euryon, and right parietal bone thickness (RPBT) = right euryon were measured on sagittal and axial views (Fig. 2) [6].
The mean and standard deviation of all age groups were obtained and correlation between CVT measurements and age groups was evaluated with Pearson’s correlation coefficient (R) (p < 0.05) [13]. R values approaching, or close to -0.2/0.2, -0.5/0.5, -0.8/0.8, and − 1/1 indicated weak, moderate, strong, and perfect correlations, respectively. Models for age estimation were developed individually using stepwise linear regression for all age groups (below 2 years, 3–6 years, 7–9 years, 10–12 years, 13–15 years, and 16–20 years). Normality for each variable was tested using skewness and kurtosis. Differences in CVT measurements between sexes were then compared using independent sample t-test.
Intra-observer reliability was tested by repeated measurements of 30 cranial datasets of different age groups and sexes chosen at random, with an interval of 3 weeks. Another set of 30 cranial images, analysed by an experienced oral and maxillofacial radiologist (> 7 years) was compared for inter-observer reliability. Intra- and inter-observer reliability of the x, y, and z coordinates for all landmarks were assessed using intraclass correlation coefficients (ICC). Reliability was categorised as poor for ICC lower than 0.50, moderate (0.50–0.75), good (0.75–0.90), and excellent for ICC was higher than 0.90 [14]. In addition, the technical error of measurement (TEM), relative technical error of measurement (rTEM), and the coefficient of reliability (R) were obtained to determine the measurement errors. The acceptable values for rTEM and R were < 1.0% and > 0.95, respectively [15]. IBM SPSS Statistics version 26 was used to perform all statistical analyses.
Results
Descriptive statistics
Descriptive statistics (mean thickness and standard deviation) of CVT for different age groups are presented in Table 1. The overall mean thickness and standard deviation of the frontal and occipital bones were 5.37 ± 1.84 mm and 6.82 ± 2.23 mm, respectively. The right parietal bone mean thickness was 4.74 ± 19.93 mm, and 3.88 ± 1.17 mm for the left parietal bone. CVT increased with age at all four landmarks. CVT values were higher at the occipital than the frontal and parietal regions areas.
Age
The correlations between CVT and age are presented in Table 2. The highest number of correlations was observed in the 0–2 years group in FBT (R = 0.605, p < 0.001), RPBT (R = 0.513, p < 0.001), LPBT (R = 0.557, p < 0.001), and OBT (R = 0.596, p < 0.001). In the age group of 3–6 years, a correlation was observed only in OBT (R = 0.379, p < 0.001). Meanwhile, in the age group of 7–9 years, correlations were demonstrated in RPBT (R = 0.528, p < 0.001) and LPBT (R = 0.490, p < 0.001).
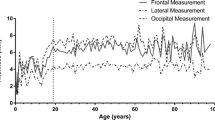
Figure 3 illustrates positive correlations between CVT and age (R = 0.098–0.785, p < 0.05). Regression models developed for all age groups are summarised in Table 3. Based on R² and the standard error of estimate (SEE), age estimation was most accurate in the 0–2 years group (R²=0.629, SEE = 0.303) at the frontal and occipital regions. Lower R² values of 0.133, 0.344, 0.016, 0.051, and 0.035, along with higher SEE values of 1.038, 0.667, 0.819, 0.780, and 1.402, were observed in the age groups of 3–6 years, 7–9 years, 10–12 years, 13–15 years, and 16–20 years, respectively. In addition, variance inflation factor (VIF) values for the regression models of all age groups were below 5, indicating reliability.
Sexual dimorphism
Independent t-test results of CVT for males and females are presented in Table 4. Sexual dimorphism in both frontal (t = 2.572, p = 0.012) and parietal (t = 2.579, p = 0.011) bone thickness was evident in the age groups 3–6 years and 16–20 years, respectively (p < 0.05).
Intra- and inter-observer errors
The ICC values for intra- and inter-observer reliability are presented in Table 5. The values for the x, y, and z coordinates of all landmarks were higher than 0.90, suggesting nearly perfect agreement between observers. Table 6 shows the TEM, rTEM, and R values for intra– and inter–observer measurement errors. All rTEM values for intra–observer error (0.142–0.246%) and inter–observer error (0.151–0.259%) were lower than 1%. Additionally, all R values exceeded 0.95 (0.966–0.996), indicating reliability.
Discussion
The human skull stands as one of the most thoroughly investigated structures of the skeletal system. The ability of morphological characteristics to describe age and sex, demonstrated the human skull as a highly valuable source of information for constructing a comprehensive biological profile of an unidentified individual [1, 16].
During the initial stages of ontogeny, the skull undergoes substantial growth and changes in bone thickness, with additional periods of accelerated growth during childhood and adolescence [17,18,19]. The current study showed that CVT gradually increased during growth, with significant variability in the first 6 years and plateaued towards the end of adolescence up to 20 years. The most substantial change in thickness observed in the cranial vault was around the occipital bone with the largest increase occurring between ages 0 and 2. Fewer changes were observed thereafter. CVT values in the occipital area specifically were much larger than the frontal and parietal areas, consistent with the findings of Li, Park [20]. In addition, several studies reported a significant increase in skull thickness during early infancy and childhood, leading to the attainment of adult skull dimensions by the end of skeletal maturation [18,19,20,21,22,23]. Furthermore, right and left parietal bone thickness were highly correlated, which is in agreement with previous studies [6, 24].
The relationship between CVT and age has been a subject of considerable investigations [4, 25]. The current study established that CVT has a positive correlation with developmental age in the subadult’s cranium with a significant increase during the first 2 years. This may be associated with the overall increased growth in skeletal bone mass, influenced by systemic factors such as hormones and nutrition [26,27,28]. De Boer, Van der Merwe [4] and Calderbank, Morgan [10] found positive correlations between age and frontal, temporal, and occipital measurements in subadults younger than 19. However, a study on 43 men and 21 women aged older than 16 years measuring skull thickness from lambda, bregma, and left and right euryon reported no significant correlation between CVT and age [3]. This could be due to the dynamics of vault bones that may undergo alterations due to additional functional matrices, including muscles and the shape of the brain as ontogeny progresses into adulthood [19]. This long-term association between CVT and age establishes the potential groundwork to corroborate other age estimation methods for subadults [25].
Age estimation methods for subadults, including dental development, pelvis size, and lengths of long bones such as femur and tibia, have been extensively studied [29,30,31,32,33,34,35]. Various researchers investigated the relationship between CVT and chronological age [3, 4, 10], however, most did not consider development of regression models to estimate age for subadults. The age estimation regression models developed in the present study were most accurate in the younger age group (< 2 years) at lambda and bregma. At these landmarks, the models consistently demonstrated the highest accuracy and precision. In contrast, Kamnikar [25] reported that age estimation is feasible for juveniles under the age of 6 at vertex and lambda points, but not at bregma. Therefore, this study indicated that CVT measurements could be utilised as a supplement to other methods to estimate age for subadults.
Sexual dimorphism in CVT has been a subject of interest for understanding cranial development and potential differences [4]. Significant sexual dimorphism was observed in frontal and parietal bone thickness for age groups 3–6 years and 16–20 years, respectively. In contrast, De Boer, Van der Merwe [4] and Anzelmo, Ventrice [19] reported no significant correlations between CVT and sex in subadults [4, 19]. While Zaafrane, Ben Khelil [2], Moreira-Gonzalez, Papay [36] and Hatipoglu, Ozcan [37] found significant sexual dimorphism in adults and this was theorised to be due to hyperostosis frontalis interna (HFI). Ross et al. [38] posited that cranial thickness is sexually dimorphic by the time HFI happens. This study also showed significantly thicker frontal and occipital bones in males, except for the euryon which is consistent with findings by Zaafrane, Ben Khelil [2] and Lynnerup, Astrup [3].
The ability to measure the human skull using CT images has a significant practicality in various fields, including anatomy, clinical medicine, biomechanical studies, and forensic anthropology. Traditionally, measurements of CVT were conducted during autopsies [3]. However, CT scans have emerged as a preferred modality for bone imaging due to their high-fidelity representations of cranial bones [7]. CT scans provide detailed and accurate 3D images of the skull, allowing for precise measurements of various cranial parameters, including skull thickness [2]. Measurement of CVT on 3D images has been shown to be reliable, convenient, and often superior to conventional measurement methods [39,40,41]. These studies have shown that measurements obtained from CT scans correlate with similar measurements obtained from human specimens [3, 5, 42]. This indicates that the measurements derived from CT scans are comparable and provide reliable information about skull thickness [36, 43].
Several limitations were identified in the present study. First, this study lacked equal distribution of sample across age, sex, and ethnicity. This limitation is inherent in any study using MSCT scans, especially in studies involving subadults. Given the radiation risk to patients, it was challenging to obtain MSCT scans with an appropriate level of resolution and the correct landmarks. Next, the sample for this study came from a very specific part of the Malaysian population, which could misrepresent the population and limit the applicability of the findings to other regions within Malaysia. Thus, it is vital to acknowledge the regional specificity of the sample and to interpret the results with caution when applying to the broader population.
Conclusions
This study found a significant correlation between CVT measurements and age. CVT gradually increases during the early years of life, particularly within the first 6 years. As individuals progress through adolescence, CVT reaches a plateau towards the end of adolescence and up to 20 years. This finding indicates that changes in CVT occur mostly during the earlier stages of growth and development. Additionally, age estimation is most accurate with bregma and lambda in children under 2 years old; however, accuracy decreases in the older age groups. This study contributes to the body of literature to support and further strengthen the age estimated by other methods for subadults.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Iscan MY, Steyn M (2013) The human skeleton in forensic medicine. Charles C Thomas
Zaafrane M et al (2018) Sex determination of a Tunisian population by CT scan analysis of the skull. Int J Legal Med 132(3):853–862
Lynnerup N, Astrup JG, Sejrsen B (2005) Thickness of the human cranial diploe in relation to age, sex and general body build. Head Face Med 1:1–7
De Boer H, Van der Merwe A, Soerdjbalie-Maikoe V (2016) Human cranial vault thickness in a contemporary sample of 1097 autopsy cases: relation to body weight, stature, age, sex and ancestry. Int J Legal Med 130:1371–1377
Ross M, Lee, KAP, W, Castle (1976) Skull thickness of black and white races. South Afr Med J 50(16):635–638
Lynnerup N (2001) Cranial thickness in relation to age, sex and general body build in a Danish forensic sample. Forensic Sci Int 117(1–2):45–51
Lillie EM et al (2016) Evaluation of skull cortical thickness changes with age and sex from computed tomography scans. J Bone Miner Res 31(2):299–307
Lillie EM et al (2015) Estimation of skull table thickness with clinical CT and validation with microCT. J Anat 226(1):73–80
McDowell JL, L’Abbé EN, Kenyhercz MW (2012) Nasal aperture shape evaluation between black and white South africans. Forensic Sci Int 222(1–3):397e1-397. e6
Calderbank T et al (2016) An investigation of juvenile cranial thickness-analysis of skull morphometrics across the complete developmental age range. J Forensic Radiol Imaging 4:70–75
Prevrhal S et al (2003) Accuracy of CT-based thickness measurement of thin structures: modeling of limited spatial resolution in all three dimensions. Med Phys 30(1):1–8
Poole KE et al (2012) Cortical thickness mapping to identify focal osteoporosis in patients with hip fracture. PLoS ONE 7(6):e38466
Zou KH, Tuncali K, Silverman SG (2003) Correlation and simple linear regression. Radiology 227(3):617–628
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15(2):155–163
Weinberg SM et al (2005) Intraobserver error associated with measurements of the hand. Am J Hum Biology: Official J Hum Biology Association 17(3):368–371
Maat GJR, Panhuysen RlGAM, Mastwijk R (2002) Manual for the physical anthropological report. Barge’s Anthropologica
Domenech-Fernandez P et al (2021) Analysis of skull bone thickness during growth: an anatomical guide for safe pin placement in halo fixation. Eur Spine J 30:410–415
Letts M, Kaylor D, Gouw G (1988) A biomechanical analysis of halo fixation in children. J Bone Joint Surg Br Vol 70(2):277–279
Anzelmo M et al (2015) Ontogenetic changes in cranial vault thickness in a modern sample of H omo Sapiens. Am J Hum Biology 27(4):475–485
Li Z et al (2015) A statistical skull geometry model for children 0–3 years old. PLoS ONE 10(5):e0127322
Delye H et al (2015) Creating a normative database of age-specific 3D geometrical data, bone density, and bone thickness of the developing skull: a pilot study. J Neurosurgery: Pediatr 16(6):687–702
Maves MD, Matt BH (1986) Calvarial bone grafting of facial defects. Otolaryngology—Head Neck Surg 95(4):464–470
Gajawelli N et al (2020) Neurocranium thickness mapping in early childhood. Sci Rep 10(1):16651
Roche A (1953) Increase in cranial thickness during growth. Hum Biol 25(2):81
Kamnikar KR (2016) Investigating the utility of age-dependent cranial vault thickness as an aging method for juvenile skeletal remains on dry bone, radiographic and computed tomography scans. Mississippi State University
Ballabriga A (2000) Morphological and physiological changes during growth: an update. Eur J Clin Nutr 54(1):S1–S6
Hall BK (2005) Bones and cartilage: developmental and evolutionary skeletal biology. Elsevier
Lieberman DE (1996) How and why humans grow thin skulls: experimental evidence for systemic cortical robusticity. Am J Phys Anthropology: Official Publication Am Association Phys Anthropologists 101(2):217–236
Bunyarit SS et al (2020) Dental age estimation of Malaysian Chinese children and adolescents: Chaillet and Demirjian’s method revisited using artificial multilayer perceptron neural network. Australian J Forensic Sci 52(6):681–698
Fife RL (2014) Juvenile Age Estimation Using Cranial Ratios
Smith DE, Humphrey LT, Cardoso HF (2021) Age estimation of immature human skeletal remains from mandibular and cranial bone dimensions in the postnatal period. Forensic Sci Int 327:110943
Yang F, Jacobs R, Willems G (2006) Dental age estimation through volume matching of teeth imaged by cone-beam CT. Forensic Sci Int 159:S78–S83
Pham CV et al (2021) Age estimation based on 3D post-mortem computed tomography images of mandible and femur using convolutional neural networks. PLoS ONE 16(5):e0251388
El-Din EAA et al (2019) Magnetic resonance imaging of the proximal tibial epiphysis: could it be helpful in forensic age estimation? Forensic Sci Med Pathol 15:352–361
Li Y et al (2019) Forensic age estimation for pelvic X-ray images using deep learning. Eur Radiol 29:2322–2329
Moreira-Gonzalez A, Papay FE, Zins JE (2006) Calvarial thickness and its relation to cranial bone harvest. Plast Reconstr Surg 117(6):1964–1971
Hatipoglu HG et al (2008) Age, sex and body mass index in relation to calvarial diploe thickness and craniometric data on MRI. Forensic Sci Int 182(1–3):46–51
Ross AH, Jantz RL, McCormick WF (1998) Cranial thickness in American females and males. J Forensic Sci 43(2):267–272
Connor S et al (2007) Precision and accuracy of low-dose CT protocols in the evaluation of skull landmarks. Dentomaxillofacial Radiol 36(5):270–276
van Lindert EJ et al Validation of cephalic index measurements in scaphocephaly Child’s nervous system, 2013. 29: pp. 1007–1014
Citardi MJ et al (2001) Comparison of scientific calipers and computer-enabled CT review for the measurement of skull base and craniomaxillofacial dimensions. Skull Base 11(01):005–012
Adeloye A, Kattan KR, Silverman FN (1975) Thickness of the normal skull in the American blacks and whites. Am J Phys Anthropol 43(1):23–30
Pillai P, Sammet S, Ammirati M (2008) Application accuracy of computed tomography-based, image-guided navigation of temporal bone. Neurosurgery, 63(4 Suppl 2): p. 326 – 32; discussion 332.
Acknowledgements
The first author would like to thank the Radiology Department, University Malaya Medical Centre (UMMC) for helping in obtaining MSCT scans.
Funding
Research management fund (project no: RMF 0637–2021) from Universiti Malaya was used to fund this study.
Author information
Authors and Affiliations
Contributions
Sharifah Nabilah Syed Mohd Hamdan, Norliza Ibrahim, and Rabiah Al-Adawiyah Rahmat contributed to the conception and design of the work; acquisition, analysis, and interpretation of data; drafting of the manuscript; and final approval for the version to be published. Amir Hazwan Abdul Rahim contributed to the editing and drafting of the manuscript and provided final approval for the version to be published. Zamri Radzi contributed to the critical revision of the draft and provided final approval for the version to be published.
Corresponding author
Ethics declarations
Ethical approval
This research was performed in accordance with the Declaration of Helsinki and human research ethics approval was granted by the Medical Research Ethics Committee, University Malaya Medical Centre (MREC ID NO: 202147 − 10039). Informed consent requirement was given a waiver by the Medical Research Ethics Committee, UMMC because this is a retrospective study that used CT images obtained from the archives of the hospital.
Consent to participate
Informed consent requirement was given a waiver by the Medical Research Ethics Committee, UMMC because this is a retrospective study that used CT images obtained from the archives of the hospital.
Human ethics
Not applicable.
Conflict of interest
The authors have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Syed Mohd Hamdan, S.N., Radzi, Z., Abdul Rahim, A.H. et al. Computed tomography study of cranial vault thickness in Malaysian subadult population. Int J Legal Med (2024). https://doi.org/10.1007/s00414-024-03276-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00414-024-03276-2