Abstract
The diagnosis of Alzheimer’s disease (AD) in the oldest-old is complicated by the increasing prevalence of age-related neurofibrillary tangles, plaques and non-AD pathologies such as cerebrovascular disease (CVD), hippocampal sclerosis (HS), aging-related tau astrogliopathy (ARTAG), as well as TDP-43 and Lewy pathology. The contribution of these non-AD pathologies to dementia and cognitive resilience is unclear. We assessed the level of AD neuropathologic change (ADNPC) and non-AD pathology in 185 participants enrolled in The 90+ Study with available cognitive assessments and brain tissue. Logistic regression models—adjusting for age, sex and education—determined the association between each pathology and dementia or between subgroups. 53% had dementia, primarily AD or mixed AD; 23% had cognitive impairment without dementia (CIND); 23% were not impaired. Both AD and non-AD pathology was prevalent. 100% had tangles, 81% had plaques, and both tangles and plaques associated with dementia. ARTAG distributed across limbic (70%), brainstem (39%) and cortical regions (24%). 49% had possible CVD and 26% had definite CVD, while HS was noted in 15%. Cortical ARTAG, CVD and HS were each associated with dementia, but limbic and brainstem ARTAGs were not. TDP-43 and Lewy pathologies were found in 36 and 17% and both associated with dementia. No pathology distinguished CIND and the not impaired. By NIA-AA criteria and dementia status, the cohort was subdivided into four groups: those with minimal ADNPC included the not dementia (ND) and Not AD dementia groups; and those with significant ADNPC included the Resilient without dementia and AD dementia groups. Compared to the ND group, the Not AD dementia group had more HS, cortical ARTAG, TDP-43, and Lewy pathology. Compared to the AD dementia group, the Resilient group had less CVD, no HS and less cortical ARTAG, TDP-43 and Lewy pathology. Our findings imply that reductions in non-AD pathologies including CVD contribute to cognitive resilience in the oldest-old.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The neuropathological diagnosis of AD in the oldest-old—those older than 90 years—is complicated by the increasing prevalence of aging-related neurofibrillary tangles (NFTs) and plaques in individuals without clinical dementia [5]. In fact, those with AD pathology but no dementia may be pathologically indistinguishable from those with dementia who are vulnerable to a low burden of AD neuropathologic change (ADNPC). Similarly, others prove cognitively resilient to an intermediate level of ADNPC, while their peers develop dementia [34, 41].
Aging can also result in the accumulation of non-AD pathologies, including proteinopathies such as α-synuclein-positive Lewy bodies, TDP-43-positive inclusions and aging-related tau astrogliopathy (ARTAG), as well as cerebrovascular disease (CVD) and hippocampal sclerosis (HS) [15, 27, 33, 35]. Incidental Lewy bodies are generally absent in those under 60 years, but are found in 17–25% of aged brains without parkinsonism and clinical dementia. Lewy pathology occurs in distributions affecting amygdala-only, or brainstem, limbic and cortical areas. Similar to α-synuclein, TDP-43 protein aggregates is a frequent co-pathology in AD and is present in 13–36% of individuals without clinical amyotrophic lateral sclerosis or frontotemporal degeneration [15, 36, 38]. TDP-43 is staged as occurring first in the amygdala, then hippocampal and finally cortical areas [19]. CVD is the most frequent non-AD pathology in the brains of the oldest-old with 75–90% of individuals having some degree of CVD pathology [34]. While no consensus criteria for CVD exist, common approaches toward the pathological diagnosis of CVD involve reporting the extent of microvascular lesions (MVL) including arteriolosclerosis, Aβ-positive cerebral amyloid angiopathy, expanded Virchow–Robin spaces, pigmented macrophages, myelin loss and microinfarcts [13, 36]. We previously reported that just one of these MVL—microinfarcts—affected 51% of the oldest-old [10]. HS is another common age-related pathology that may directly be associated with TDP-43 co-pathology and with cognitive deficits [12, 18, 38]. ARTAG also rises in frequency with increasing age [23]. While rare under age 60 years, ARTAG has been found in almost half of brains over age 75 years [24, 26, 27]. ARTAG occurs as different types in multiple anatomical areas. Gray matter ARTAG might correlate with clinical symptoms [25, 31]; however, limbic and brainstem subpial, perivascular and white matter ARTAG has not been correlated with dementia [28, 46].
Cognitive impairment and dementia may be the consequence of any of these pathologies or others not listed here. Thus, the decreasing correlation between AD pathology and dementia in the oldest-old may be the result of the increasing prevalence of non-AD pathology with advancing age. Here, we describe the prevalence and severity of each pathology in The 90+ Study and use logistic regression to determine the association of each measure with the cognitively impaired, but no dementia (CIND) versus the not impaired (NI); the association of each measure with dementia; and the association of each measure with increasing burdens of plaques and tangles independent of dementia. Finally, we use NIA-AA criteria to examine the hypothesis that non-AD pathology associates with dementia independent of AD pathology.
Methods
Cohort
Participants were enrolled in The 90+ Study, a population-based study of people 90 years and older [9]. Study participants were followed in person every 6 months with neurological and physical examinations, neuropsychological testing, functional assessments and informant interviews. Of 212 individuals who came to autopsy before September 30, 2013, 185 had adequate limbic, brainstem and neocortical brain tissue available and cognitive assessments within 1 year of death.
The longitudinal neuropsychological battery included the MMSE, California Verbal Learning Test, Trail-making tests, clock drawing, verbal fluencies and other standardized measures of cognition. Additional information relevant to cognitive diagnoses included interviews with knowledgeable informants and review of all medical records including clinical neuroimaging and laboratory results. After a participant’s death, final clinical diagnoses were assigned during a multi-disciplinary consensus conference led by The 90+ Study principal investigators (CK and MC) while blinded to pathological evaluations. Using all available information, participants were cognitively classified as not impaired (NI), cognitively impaired but no dementia (CIND) or as having dementia. Dementia diagnosis was established using Diagnostic and Statistical Manual of Mental Disorders 4th Edition criteria [1]. Participants were classified as CIND if they showed cognitive or functional deficits that were not severe enough to meet criteria for dementia. In addition, for individuals with dementia or CIND, the presumed clinical etiologies—i.e., Alzheimer disease, Parkinson disease, vascular dementia, others—were assigned during this conference applying standard diagnostic criteria. MMSE scores reflected the differences between groups: NI (mean 27.5, IQR 27–29), CIND (mean 23.6, IQR 21–27) and dementia (mean 11.9, IQR 4–19). The mean number of months between death and last MMSE did not differ between groups: NI (mean 5.9, IQR 2.0–6.2), CIND (mean 5.5, IQR 3.1–6.3) and dementia (mean 6.3, IQR 2.7–7.0).
AD neuropathology
Hippocampus and amygdala (limbic), midbrain and medulla (brainstem), and mid-frontal and superior temporal cortex (cortex) slides were immunostained for tau pathology with PHF-1 antibody, for Aβ plaques with Nab228 antibody, for α-synuclein pathology with Syn303 antibody and for TDP-43 pathology with 1D3 as described previously [38]. All antibodies are well characterized as specific detectors of the common aggregating proteins in neurodegenerative disease; each is made freely available to all Alzheimer’s disease centers (PHF-1, Nab228, Syn303) or are commercially available (1D3, Millipore, USA). All slides were available for review except for medulla in 36 subjects. Semi-quantitative scores (0–3+) for each slide with NFT and Aβ plaque pathology were used to assign to each case as I–VI Braak stage, 0–4 Thal phase (cerebellum was unavailable to stage the Thal phase 5) and 0–3 CERAD neuritic plaque scores as previously described [39]. Specifically, Braak stage was determined using the hippocampal section for Braak stages I–IV; Braak stage V was defined by the presence of NFT in either cortex slide; Braak stage VI (n = 4) was defined by the presence of severe NFT pathology in both cortical regions. Thal phase was determined using a simplified Thal phase schema (0–4) through examination of the hippocampus [44]; hippocampi without any plaques were assigned Thal phase 0 if both the frontal and temporal cortices were also free of plaques. Neuritic plaque burden was determined by tau immunohistochemistry to assign CERAD neuritic plaque scores [4, 42]. NIA-AA criteria [30] determined the level of ADNPC (low, intermediate, high) with the additional diagnoses of definite and possible primary age-related tauopathy (PART) also included [11]. Hence, definite PART is defined by Braak stages I–IV without plaques; possible PART by Braak stages I–IV with Thal phase 1–2 and CERAD 0–1; and the remaining levels of ADNPC as per NIA-AA criteria.
Non-AD neuropathology
TDP-43 stage was determined from TDP-43 pathology semi-quantitative scores for each slide. Cases were either TDP-43 negative (stage 0), TDP-43 positive in the amygdala-only (stage 1), TDP-43 positive in the amygdala and hippocampus (stage 2) or TDP-43 positive in the amygdala, hippocampus and frontal or temporal cortex (stage 3) [19].
A Lewy pathology distribution score was determined from α-synuclein semi-quantitative scores for each slide. α-synuclein-positive cases had Lewy pathology in the brainstem or amygdala only (stage 1), in the limbic areas in addition to stage 1 areas (stage 2) or in the cortical areas as well as stage 2 areas (stage 3) [29].
The tau-immunostained slides were used to determine the severity by semi-quantitative scores for each of the two signature ARTAG astrocytic tau inclusions: thorn-shaped astrocytes (TSA) and granular or fuzzy astrocytes (GFA). As per published consensus recommendations, the anatomical location of each pathology was also noted (i.e., subpial, subependymal, perivascular, white matter and gray matter) (Supplemental Table 1) [23]. For purposes of this paper, the maximum semi-quantitative ARTAG score—TSA or GFA, regardless of anatomical location—was used to determine separate ARTAG regional scores: limbic (hippocampus and amygdala), brainstem (midbrain and medulla) and cortical (mid-frontal and superior temporal cortex) ARTAG. This simplification—for example, a case with 2+ GFA in the amygdala and 1+ TSA in the hippocampus is scored 2+ limbic ARTAG—allowed us to compare the regional ARTAG burden across cases. Note that all brainstem ARTAG cases had limbic ARTAG, and cases with cortical ARTAG also had both limbic and brainstem ARTAG except rarely (n = 7/185).
CVD pathology was determined from the semi-quantitative grading of MVL (none, mild, moderate, severe) similar to Neltner et al. [36]: arteriolosclerosis, expanded Virchow–Robin spaces and pigment-laden macrophages in the perivascular spaces in hematoxylin and eosin-stained sections for all limbic and neocortical slides, myelin loss on hematoxylin and eosin-stained sections with Luxol fast blue for all neocortical slides, and cerebral amyloid angiopathy on Aβ immunohistochemistry for all neocortical slides. Finally, the presence of microinfarcts was also noted. These MVL included arteriolosclerosis (77%, n = 142), enlarged Virchow–Robin spaces (68%, n = 125), myelin loss (44%, n = 81), pigmented macrophages (37%, n = 69), Aβ amyloid angiopathy (16%, n = 30) and microinfarcts (8%, n = 15) (Supplemental Table 1; Supplemental Fig. 1). For each case, a CVD diagnosis was determined by summing all moderate to severe MVL and the presence of microinfarcts, similar to Deramecourt et al. [13]. From observed MVL counts of 0–12, definite CVD was defined by the presence of six or more MVL (26%, n = 48). For descriptive purposes, we also report possible CVD as the presence of 3–5 MVL (49%, n = 90) [30].
Finally, HS is determined from the presence of severe gliosis and neuronal loss in the CA1 sector and subiculum of the hippocampus. HS is frequently associated with TDP-43 pathology and “pure” HS occurs independently of NFT and Aβ plaque pathology. Here, the presence of HS was noted in the hematoxylin and eosin-stained hippocampus independently of TDP-43, tau or Aβ immunohistochemistry [6, 38].
Statistics
All statistical analyses used R version 3.3.2. All statistical tests were two-sided, reporting odds ratios (OR), 95% confidence intervals (CI) and p values. Multiple testing adjustments were not performed due to the exploratory nature of the study [2]. Statistical significance was set at < 0.05 level. Logistic regression models using the full range of each available pathology—adjusting for age at death, sex and education—determined the association between each pathological measure and the odds of dementia. A separate logistic regression determined the association between each pathological measure and the odds of being in the CIND versus NI sub-cohorts. For the subgroup analysis, participants were defined as belonging to one of four distinct subgroups according to their dementia status and the presence or absence of significant AD pathology. Statistically, the presence of significant AD pathology was defined by an intermediate or high level of ADNPC [11, 30]. The not dementia (ND) group had no dementia and no significant AD pathology; the Not AD dementia group had dementia and no significant AD pathology; the Resilient group had no dementia and significant AD pathology; the AD dementia group had dementia and AD pathology. By logistic regression, we estimated the odds of dementia for each non-AD pathology after adjusting for AD pathology as a binary covariate and the given non-AD pathology as predictor, as well as age at death and sex. To allow for the possibility of interactions between AD pathology and the given predictor (i.e., the effect of the predictor may be different in participants with AD pathology as opposed to participants without AD pathology), we also included in each model an interaction term between AD pathology and the given predictor [22]. If the interaction term between AD pathology and the given predictor was not significant, it was removed from the model. In such cases, the effect of the predictor on the odds of dementia is the same for the Resilient vs. AD dementia groups as well as the ND vs. dementia Not AD groups. For measures with a significant interaction, ORs were reported separately for AD and non-AD groups.
Results
The 185 participants were on average 98 years old at the time of death, 71% were women, and 49% had a college education (Table 1). Cognitively, 53% had dementia and the majority of those with dementia had a primary AD or mixed AD clinical diagnosis (84%, 84/99). Those without dementia included 23% with CIND and 23% with NI.
Frequency and severity of AD pathology
AD pathology was highly prevalent (Table 2). 100% (n = 185) had NFTs quantifiable by Braak stage, 81% (n = 150) had plaques that were measurable by Thal phase and 63% (n = 117) had neuritic plaques that could be assigned CERAD scores (21). A moderate distribution of NFTs was most frequent (Braak III-IV, 63%, n = 115), while the plaque burden was commonly low (Thal phase 0–2, 56%, n = 104; CERAD none or sparse, 62%, n = 115). By NIA-AA criteria, a level of ADNPC was assigned to each case. 48% (n = 88) had a low level of ADNPC, while 52% (n = 96) had an intermediate or high level of ADNPC. The majority of the cases with low AD pathology were best classified as PART cases (77/88), while the majority of the cases with more substantial AD pathology were assigned an intermediate level of ADNPC (83/96).
Frequency and severity of non-AD pathology
Non-AD pathology was also very prevalent in this cohort (Table 2). ARTAG affected 77% (n = 142) and was predominantly TSA pathology (75%, n = 138), but a minority also had GFA pathology (25%, n = 46) (Fig. 1). Subpial and white matter ARTAGs were commonly observed, particularly in limbic areas, but subependymal, perivascular and gray matter ARTAGs were also noted (Supplemental Table 1). Most frequently, ARTAG was observed in limbic regions (70%; n = 130), but was also present in brainstem regions (39%, n = 72) and cortical regions (24%, n = 44). The majority of cases with brainstem ARTAG had limbic ARTAG, and the majority of cortical ARTAG cases had both limbic and brainstem ARTAGs (data not shown). TDP-43 positive lesions were not uncommon and 36% (n = 66) had inclusions that could be staged [19]. In 10% (n = 18), TDP-43 was limited to the amygdala, while 22% (n = 41) had additional inclusions in the hippocampus, and 4% (n = 7) of the cases had neocortical TDP-43. Alpha-synuclein-positive Lewy pathology was less frequent and were observed in 17% (n = 31) of the cohort. A mild or greater burden of Lewy pathology was noted in the brainstem in 6% (n = 11) or as limbic pathology as well in 9% (n = 16). Rarely did Lewy pathology spread to the neocortex (1%, n = 2).
ARTAG pathology. Limbic (a–d), brainstem (e–g) and neocortical (h) ARTAG in The 90+ Study. a Severe GFA in gray matter of the amygdala. b Severe TSA in the temporal lobe. c ARTAG frequently co-exists with AD tau pathology. Substantial white matter TSA underlying a severe burden of NFTs in the entorhinal cortex. d Subependymal TSA in the lateral ventricle. e TSA in the medial lemniscus and inferior olive nucleus. f TSA and GFA in the spinal lemniscus in the mesencephalon. g Perivascular TSA around the cerebral aqueduct. h Gray matter GFA in the temporal cortex. Scale bar is 100 um in all images. ARTAG aging-related tau astrogliopathy, GFA granular fuzzy astrocytes, TSA thorn-shaped astrocytes
CVD affected the majority of the cohort (75%, n = 138). CVD was defined by the presence of three or more significant—moderate to severe—MVL in a given brain. Individually, MVL were common. Only accounting for significant modifications, arteriolosclerosis was detected in 45%, expanded Virchow–Robin spaces in 33%, myelin loss in 12%, pigmented macrophages in 8% and cerebral amyloid angiopathy in 8% (Supplemental Table 1). Additionally, microinfarcts were found in 8% of the cohort. Definite CVD, defined by the presence of six or more significant MVL, was assigned to 26% of cases (n = 48). The specific hippocampal cell loss and gliosis that defined HS were less common and noted in 15% (n = 28).
Associations with cognitive impairment and dementia
We first asked if any pathological measure was associated with either cognitive impairment or dementia. By logistic regression, neither the AD nor the non-AD measures distinguished CIND from NI (Table 3). Braak stage, Thal phase and CERAD score were statistically equivalent in the CIND and NI groups, as were TDP-43 stage, Lewy pathology, cortical ARTAG, limbic ARTAG, brainstem ARTAG, definite CVD and HS (all p values > 0.1). Since the CIND and NI groups were pathologically indistinguishable, we assigned both to a not dementia group for further analysis.
By logistic regression, most of the AD and non-AD pathologies were associated with the dementia group than the not dementia group (Table 3). Increasing Braak stage strongly associated with dementia (p value < 0.001) and increasing plaques burdens: Thal phase (p value = 0.002), and CERAD score (p value = 0.002). TDP-43 stage and Lewy pathology were higher in the dementia group than the not dementia group (p values < 0.001 and 0.018, respectively). HS (p value < 0.001), definite CVD (p value = 0.016) and cortical ARTAG (p value = 0.007) were associated with dementia, but limbic ARTAG (p value = 0.84) and brainstem ARTAG (p value = 0.62) were not.
Interactions between pathologies
Since both AD and non-AD pathologies were associated with dementia, we next asked if these variables were correlated. First, we divided the cohort by tau burden into low (I–III) and high (IV–VI) Braak stage groups (Fig. 2 and Supplemental Table 3). By logistic regression, the high Braak group had a higher Thal phase (OR 2.50, p value < 0.001) and CERAD score (OR 3.38, p value ≤ 0.001). In addition, several of the non-AD pathologies were also associated with the high Braak stage group. HS (OR 4.16, p value = 0.006) and definite CVD (OR 2.65, p value = 0.009) were more likely at higher Braak stages as was cortical ARTAG (OR 3.66, p value < 0.001) and TDP-43 stage (OR 1.92, p value < 0.001). Limbic ARTAG (OR 1.04, p value = 0.76), brainstem ARTAG (OR 1.16, p value = 0.41) and Lewy pathology did not significantly associate with a high Braak stage (p value = 0.11).
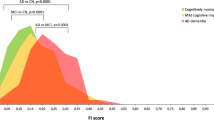
Pathological associations with Braak stages. Non-AD pathology and plaque pathology are graphed by increasing Braak stage. Non-AD pathology includes TDP-43 stage and Lewy pathology (lines, on the right axis) as well as HS, cortical ARTAG and CVD (histogram, frequency indicated by the right axis), while plaque pathology is represented as NIA-AA criteria A0-3 and C0-3 values (lines, on the left axis). At low Braak stages (I–III): definite CVD is prevalent in about 15% of individuals; HS and cortical ARTAGs are rare, as are TDP-43 and Lewy pathologies; plaques are restricted to cortical areas (A1) and are only rarely neuritic (C1). At high Braak stages (IV–VI): definite CVD increases to over 30% (p = 0.009); HS increases to over 20% (p = 0.006); cortical ARTAG increases to over 30% (p < 0.001); TDP-43 is commonly found in the amygdala (p < 0.001) but Lewy pathology remains rare; plaques are more widespread (Thal phase, p < 0.001) and neuritic (CERAD, p ≤ 0.001). See Supplemental Table 3 for the full statistics including OR and 95% CI for each measure’s association with Braak stage and Thal phase. ARTAG aging-related tau astrogliopathy, CVD cerebrovascular disease, HS hippocampal sclerosis
Next, we divided the cohort by plaque burden into low (0–2) and high (3–4) Thal phase groups (Supplemental Table 3). By plaque burden, the AD pathologies were strongly associated. The high Thal phase group had a higher Braak stage (OR 2.67, p value < 0.001) and higher CERAD scores (OR 2.69, p value = 0.001). The non-AD pathologies did not associate with the high Thal phase group compared to the low Thal phase group. The high Thal phase group was not more likely to have HS (OR 1.26, p value = 0.57), definite CVD (OR 1.21, p value = 0.58) or more TDP-43 (OR 1.24, p value = 0.18), Lewy pathology (OR 1.13, p = 0.57), cortical ARTAG (OR 1.18, p value = 0.52), limbic ARTAG (OR 0.90, p value = 0.44) or brainstem ARTAG (OR 0.75, p value = 0.12).
Resilient and Not AD dementia groups
The AD variables—Braak stage, amyloid phase and CERAD score—individually associated with dementia in The 90+ Study, but did not differentiate the NI and CIND groups. By NIA-AA criteria, the level of intermediate or high ADNPC accounted for 60% (n = 59) of the dementia group (Supplemental Table 2). Nonetheless, 32% (n = 32) were also diagnosed with definite or possible PART. In the cases without dementia, 61% (n = 26) of the NI cases had a PART diagnosis as did 54% (n = 19) of the CIND individuals. Conversely, 38% (n = 16) of the NI cases had an intermediate or high level of ADNPC along with 49% (n = 21) of CIND individuals. Overall, by NIA-AA criteria, an intermediate or high level of ADNPC had limited sensitivity (60%) and specificity (57%) for dementia in The 90+ Study. As a result, individuals could be subdivided into four groups based on the presence or absence of AD pathology and their dementia status (Table 4). An AD dementia group was composed of individuals with intermediate or high level of ADNPC and dementia, while a Resilient group also had an intermediate or high level of ADNPC but without dementia. Similarly, individuals without Aβ plaques or with a low level of ADNPC were separated into a not dementia (ND) group and a Not AD dementia group. By NIA-AA criteria, both the AD dementia and Resilient groups, and the ND and Not AD dementia groups were similarly defined by their plaque and tangle burdens. The AD dementia group was primarily composed of intermediate ADNPC cases (83%) as was the Resilient group without dementia (92%). Similarly, the ND group was mostly definite or possible PART cases (92%) as was the Not AD dementia group (80%).
We hypothesized that higher burdens of non-AD pathologies would associate with dementia independent of AD pathology. Logistic regression was performed with interaction terms to determine if the non-AD pathologies had differential effects in the ND and Not AD dementia groups, compared to the Resilient and AD dementia groups with AD pathology (Table 5). Definite CVD was the only measure to have a significant interaction. That is, while CVD pathology was comparable in the Not AD dementia and ND groups (p value = 0.93), definite CVD was increased in the AD dementia group compared to the Resilient group (p value = 0.003). For the other measures, the interaction term was non-significant and removed from the model. After adjusting for AD pathology, the presence of HS was strongly associated with dementia in the Not AD dementia group compared to the ND group, as well as in the AD dementia group compared to the Resilient group (p value < 0.001). TDP-43 stage (p value = 0.001) and Lewy pathology (p value = 0.024) also associated with dementia independent of AD pathology, as did cortical ARTAG (p value = 0.008). Limbic and brainstem ARTAGs did not associate with dementia in this model (p value > 0.7).
Discussion
While AD pathology is common in the oldest-old, our study develops two lines of evidence implicating non-AD pathologies in the development of dementia in this age group. First, dementia occurs in the absence of significant AD pathology when CVD, ARTAG, HS, TDP-43 and Lewy pathology act as multiple insults on the aged brain. Together, the combined pathologies are a sufficient correlation of dementia. Second, cognitive resilience occurs in the presence of a significant AD pathology when the non-AD pathological burden is reduced. Here, neuropathological resilience to the non-AD pathologies may result in cognitive resilience as AD pathology develops.
In The 90+ Study, both AD and non-AD pathologies are prevalent: NFTs were noted in 100%, plaques in 81%, ARTAG in 77% and the MVL that define CVD in 75%, while TDP-43 inclusions, Lewy pathology and HS were less abundant. Since age-related pathologies are frequent in the oldest-old, it is important to highlight that it is not just the presence but the severity of each pathology that associates with dementia. By analyzing only burdens of NFTs above Braak stage III (57%), plaques above Thal phase 2 (44%), ARTAG with a cortical distribution (24%) and MVL with a definite CVD diagnosis (26%) in many of our models, we attempted to disambiguate the pathologies most proximal to dementia from those pathologies that are age associated.
The foremost non-AD, age-associated pathology is CVD. Since CVD is the third leading cause of death, it is not surprising that the MVL that develop into CVD are observed in 60–90% of individuals with and without dementia [37, 40]. In this study, we used a simple additive model of moderate to severe MVL including arteriolosclerosis, expanded Virchow–Robin spaces, white matter myelin loss, pigmented macrophages, cerebral amyloid angiopathy and microinfarcts. To date, there are no consensus neuropathologic criteria to diagnose CVD, although previous studies have assessed MVL with similar scoring methodologies and have reported on their association with vascular impairment, old age and dementia [13, 16, 36]. In an earlier study, we reported on just one MVL and found that 51% of participants had microinfarcts, but dementia was only associated with the 13% that had frontal, temporal or parietal cortical microinfarcts [10]. Our study confirms the high prevalence of MVL affecting 75% of the cohort and concludes that the 26% of individuals with definite CVD have increased odds for dementia. We also report that AD-associated dementia may have an increased prevalence of CVD or, alternatively, that cognitive resilience to AD-associated dementia is more common in individuals without CVD.
ARTAG was also common in this cohort and, to our knowledge, this is the first study to show an association of ARTAG with clinical dementia in a large cohort of elderly individuals [27, 28, 43, 46]. While the importance of CVD and HS is increasingly recognized [20], ARTAG’s clinical relevance has only been suggested in a small cohort of individuals [25]. ARTAG may originate in the medial temporal lobe in the white matter or subpial areas adjacent to the amygdala [23, 26, 43]. Our data are consistent with an outward spread of ARTAG from limbic to the brainstem areas and then to the neocortical areas, since only occasionally did we see cortical ARTAG independent of both limbic and brainstem ARTAGs also being present (4%, 7/185). Both Braak stage and cortical ARTAG were associated with dementia in this cohort and the frequency of cortical ARTAG increased with increasing Braak stage (Fig. 2). The templated spread of pathological tau as NFTs is now well understood and, since there is experimental evidence for the spread of neuronal and glial tau pathology following intracerebral injections of proteopathic tau seeds into mice brains [3, 8, 32], it is plausible that ARTAG may undergo templated spread as well. Nonetheless, how ARTAG spreads is still unknown, but our data are consistent with the intriguing implication that neuronal tau pathology and astrocytic tau pathology are related in the oldest-old.
The prevalence of HS increases with age and may be more directly associated with cognitive deficits than the other more frequent pathologies reported here [6, 21]. In The 90+ Study, HS prevalence was 15%, but represented 26% of dementia cases. Importantly, HS was strongly associated with dementia independent of AD pathology and the absence of HS was associated with the Resilient group. There are some indications that HS may be related to TDP-43 pathology and even to arteriolosclerosis [35, 38]. In our cohort, 71% (20/28) of the HS cases also had hippocampal TDP-43 inclusions, although we did not see a relationship with arteriolosclerosis (data not shown). For such a clinically relevant pathology, a closer look at the mechanisms related to the pathology of HS bears further scrutiny.
In this study, TDP-43 pathology prevalence was 36% and associated with dementia in individuals with and without AD pathology. Importantly, the amount of TDP-43 pathology described here was not diagnostic of frontotemporal degeneration and amyotrophic lateral sclerosis and none of the cohort was clinically characterized with those symptoms. Instead, TDP-43 pathology may be accumulating in the aged brain downstream of increasing AD pathologies [19] or, as already mentioned, downstream of HS [35].
The occurrence of incidental α-synuclein-positive Lewy bodies and Lewy neurites is increasingly being reported [7, 15, 27, 33, 35] and the 17% prevalence in The 90+ Study is in line with other cohorts without overt parkinsonism. Intriguingly, Lewy pathology was associated with dementia even though there was a weak correlation between the clinical diagnosis of Lewy body disease and Lewy pathology: 12 cases were diagnosed with LBD or AD/LBD, of which only 4 had Lewy pathology. Rather than directly correlating with clinical dementia, Lewy pathology may accumulate as a co-pathology in AD dementia [30], or as one of many insults that contribute to cognitive decline (Table 5).
NFTs were present in all individuals in The 90+ Study and may drive the additional co-pathologies observed in this cohort. Braak stage was associated with dementia along with Thal phase and CERAD score (Table 3). However, at Braak stage IV or higher, there is an increased prevalence of non-AD pathologies (Fig. 2), an association not seen at high Thal phases. These results are correlative and not causal; nonetheless, the recent demonstration that pathological tau isolated from AD brain increases the plaque burden and induces widespread tau pathology in animal models implies that pathological tau interacts with the Aβ plaque environment to drive the increase in tau and Aβ pathology [17]. In particular, the increase from Braak stage III to IV may represent an important transition from age-associated pathology to dementia-associated pathology. According to NIA-AA criteria, Braak stages III and IV are pathological equivalent for diagnostic purposes [30], but this simplification may overlook clinically relevant pathological changes in the oldest-old [14].
This study has several limitations. First, cognitive impairment and dementia may be the consequence of any of the pathologies we examined here or others not examined such as increased gliosis, blood brain barrier changes, etc. Second, while we reported ARTAG scores that were significantly associated with dementia, our severity and distribution scores were simple, additive measures based on a limited sample of brain regions. Our understanding of ARTAG as a pathological correlate for cognitive decline needs further observations of the specific constellations of ARTAG in vulnerable regions with greater discernment of the pathogenic spread of ARTAG into additional brain regions. Third, we reported an association of CVD with dementia, but there are no consensus criteria for CVD. Going forward, the staging of MVL will have to be examined in a rigorous, reproducible way.
Previous studies converge around two ideas related to the prevalence of AD pathology with advancing age: that the oldest-old with dementia have fewer NFTs and plaques than do their younger peers with dementia, and that those without dementia have more age-related NFTs and plaques than the younger-old [5, 14, 38, 41]. This raises the possibility that additional, non-AD pathologies underlie vulnerability to dementia and that a significant portion have cognitive resilience to AD pathology. We conclude that non-AD pathologies such as HS, TDP-43 inclusions, cortical ARTAG and Lewy pathology may increase the aged brain’s vulnerability to dementia independent of AD pathology, and that individuals with minimal CVD and the other non-AD pathologies may be more cognitively resilient to AD neuropathology. Although it is unknown what factors influence the accumulation of many of these pathologies, healthy aging choices may lower the burden of CVD [45] and could reduce a person’s dementia risk in old age.
References
Bell CC (1994) DSM-IV: diagnostic and statistical manual of mental disorders. JAMA 272:828–829. https://doi.org/10.1001/jama.1994.03520100096046
Bender R, Lange S (2001) Adjusting for multiple testing–when and how? J Clin Epidemiol 54:343–349
Boluda S, Iba M, Zhang B, Raible KM, Lee VM-Y, Trojanowski JQ (2015) Differential induction and spread of tau pathology in young PS19 tau transgenic mice following intracerebral injections of pathological tau from Alzheimer’s disease or corticobasal degeneration brains. Acta Neuropathol (Berl) 129:221–237. https://doi.org/10.1007/s00401-014-1373-0
Boluda S, Toledo JB, Irwin DJ, Raible KM, Byrne MD, Lee EB, Lee VM-Y, Trojanowski JQ (2014) A comparison of Aβ amyloid pathology staging systems and correlation with clinical diagnosis. Acta Neuropathol (Berl) 128:543–550. https://doi.org/10.1007/s00401-014-1308-9
Braak H, Thal DR, Ghebremedhin E, Del Tredici K (2011) Stages of the pathologic process in Alzheimer disease: age categories from 1 to 100 years. J Neuropathol Exp Neurol 70:960–969. https://doi.org/10.1097/NEN.0b013e318232a379
Brenowitz WD, Monsell SE, Schmitt FA, Kukull WA, Nelson PT (2014) Hippocampal sclerosis of aging is a key Alzheimer’s disease mimic: clinical-pathologic correlations and comparisons with both alzheimer’s disease and non-tauopathic frontotemporal lobar degeneration. J Alzheimers Dis JAD 39:691–702. https://doi.org/10.3233/JAD-131880
Buchman AS, Shulman JM, Nag S, Leurgans SE, Arnold SE, Morris MC, Schneider JA, Bennett DA (2012) Nigral pathology and parkinsonian signs in elders without Parkinson’s disease. Ann Neurol 71:258–266. https://doi.org/10.1002/ana.22588
Clavaguera F, Akatsu H, Fraser G, Crowther RA, Frank S, Hench J, Probst A, Winkler DT, Reichwald J, Staufenbiel M, Ghetti B, Goedert M, Tolnay M (2013) Brain homogenates from human tauopathies induce tau inclusions in mouse brain. Proc Natl Acad Sci USA 110:9535–9540. https://doi.org/10.1073/pnas.1301175110
Corrada MM, Berlau DJ, Kawas CH (2012) A population-based clinicopathological study in the oldest-old: the 90+ study. Curr Alzheimer Res 9:709–717
Corrada MM, Sonnen JA, Kim RC, Kawas CH (2016) Microinfarcts are common and strongly related to dementia in the oldest-old: the 90 + study. Alzheimers Dement J Alzheimers Assoc 12:900–908. https://doi.org/10.1016/j.jalz.2016.04.006
Crary JF, Trojanowski JQ, Schneider JA, Abisambra JF, Abner EL, Alafuzoff I, Arnold SE, Attems J, Beach TG, Bigio EH, Cairns NJ, Dickson DW, Gearing M, Grinberg LT, Hof PR, Hyman BT, Jellinger K, Jicha GA, Kovacs GG, Knopman DS, Kofler J, Kukull WA, Mackenzie IR, Masliah E, McKee A, Montine TJ, Murray ME, Neltner JH, Santa-Maria I, Seeley WW, Serrano-Pozo A, Shelanski ML, Stein T, Takao M, Thal DR, Toledo JB, Troncoso JC, Vonsattel JP, White CL, Wisniewski T, Woltjer RL, Yamada M, Nelson PT (2014) Primary age-related tauopathy (PART): a common pathology associated with human aging. Acta Neuropathol (Berl) 128:755–766. https://doi.org/10.1007/s00401-014-1349-0
Cykowski MD, Powell SZ, Schulz PE, Takei H, Rivera AL, Jackson RE, Roman G, Jicha GA, Nelson PT (2017) Hippocampal sclerosis in older patients: practical examples and guidance with a focus on cerebral age-related TDP-43 with sclerosis. Arch Pathol Lab Med 141:1113–1126. https://doi.org/10.5858/arpa.2016-0469-SA
Deramecourt V, Slade JY, Oakley AE, Perry RH, Ince PG, Maurage C-A, Kalaria RN (2012) Staging and natural history of cerebrovascular pathology in dementia. Neurology 78:1043–1050. https://doi.org/10.1212/WNL.0b013e31824e8e7f
Dolan D, Troncoso J, Resnick SM, Crain BJ, Zonderman AB, O’Brien RJ (2010) Age, Alzheimer’s disease and dementia in the Baltimore Longitudinal study of ageing. Brain J Neurol 133:2225–2231. https://doi.org/10.1093/brain/awq141
Elobeid A, Libard S, Leino M, Popova SN, Alafuzoff I (2016) Altered proteins in the aging brain. J Neuropathol Exp Neurol 75:316–325. https://doi.org/10.1093/jnen/nlw002
Flanagan M, Larson EB, Latimer CS, Cholerton B, Crane PK, Montine KS, White LR, Keene CD, Montine TJ (2016) Clinical-pathologic correlations in vascular cognitive impairment and dementia. Biochim Biophys Acta 1862:945–951. https://doi.org/10.1016/j.bbadis.2015.08.019
He Z, Guo JL, McBride JD, Narasimhan S, Kim H, Changolkar L, Zhang B, Gathagan RJ, Yue C, Dengler C, Stieber A, Nitla M, Coulter DA, Abel T, Brunden KR, Trojanowski JQ, Lee VM-Y (2017) Amyloid-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation. Nat Med. https://doi.org/10.1038/nm.4443
Hokkanen SRK, Hunter S, Polvikoski TM, Keage HAD, Minett T, Matthews FE, Brayne C, MRC CFAS and CC75C Study Group (2017) Hippocampal sclerosis, hippocampal neuron loss patterns and Tdp-43 in the aged population. Brain Pathol Zurich Switz. https://doi.org/10.1111/bpa.12556
Josephs KA, Murray ME, Whitwell JL, Parisi JE, Petrucelli L, Jack CR, Petersen RC, Dickson DW (2014) Staging TDP-43 pathology in Alzheimer’s disease. Acta Neuropathol (Berl) 127:441–450. https://doi.org/10.1007/s00401-013-1211-9
Kapasi A, DeCarli C, Schneider JA (2017) Impact of multiple pathologies on the threshold for clinically overt dementia. Acta Neuropathol (Berl). https://doi.org/10.1007/s00401-017-1717-7
Kawas CH, Kim RC, Sonnen JA, Bullain SS, Trieu T, Corrada MM (2015) Multiple pathologies are common and related to dementia in the oldest-old: the 90+ study. Neurology 85:535–542. https://doi.org/10.1212/WNL.0000000000001831
Kent DM, Rothwell PM, Ioannidis JPA, Altman DG, Hayward RA (2010) Assessing and reporting heterogeneity in treatment effects in clinical trials: a proposal. Trials 11:85. https://doi.org/10.1186/1745-6215-11-85
Kovacs GG, Ferrer I, Grinberg LT, Alafuzoff I, Attems J, Budka H, Cairns NJ, Crary JF, Duyckaerts C, Ghetti B, Halliday GM, Ironside JW, Love S, Mackenzie IR, Munoz DG, Murray ME, Nelson PT, Takahashi H, Trojanowski JQ, Ansorge O, Arzberger T, Baborie A, Beach TG, Bieniek KF, Bigio EH, Bodi I, Dugger BN, Feany M, Gelpi E, Gentleman SM, Giaccone G, Hatanpaa KJ, Heale R, Hof PR, Hofer M, Hortobágyi T, Jellinger K, Jicha GA, Ince P, Kofler J, Kövari E, Kril JJ, Mann DM, Matej R, McKee AC, McLean C, Milenkovic I, Montine TJ, Murayama S, Lee EB, Rahimi J, Rodriguez RD, Rozemüller A, Schneider JA, Schultz C, Seeley W, Seilhean D, Smith C, Tagliavini F, Takao M, Thal DR, Toledo JB, Tolnay M, Troncoso JC, Vinters HV, Weis S, Wharton SB, White CL, Wisniewski T, Woulfe JM, Yamada M, Dickson DW (2016) Aging-related tau astrogliopathy (ARTAG): harmonized evaluation strategy. Acta Neuropathol (Berl) 131:87–102. https://doi.org/10.1007/s00401-015-1509-x
Kovacs GG, Lee VM, Trojanowski JQ (2017) Protein astrogliopathies in human neurodegenerative diseases and aging. Brain Pathol Zurich Switz 27:675–690. https://doi.org/10.1111/bpa.12536
Kovacs GG, Molnár K, László L, Ströbel T, Botond G, Hönigschnabl S, Reiner-Concin A, Palkovits M, Fischer P, Budka H (2011) A peculiar constellation of tau pathology defines a subset of dementia in the elderly. Acta Neuropathol (Berl) 122:205–222. https://doi.org/10.1007/s00401-011-0819-x
Kovacs GG, Robinson JL, Xie SX, Lee EB, Grossman M, Wolk DA, Irwin DJ, Weintraub D, Kim CF, Schuck T, Yousef A, Wagner ST, Suh E, Van Deerlin VM, Lee VM-Y, Trojanowski JQ (2017) Evaluating the patterns of aging-related tau astrogliopathy unravels novel insights into brain aging and neurodegenerative diseases. J Neuropathol Exp Neurol 76:270–288. https://doi.org/10.1093/jnen/nlx007
Kovacs GG, Xie SX, Lee EB, Robinson JL, Caswell C, Irwin DJ, Toledo JB, Johnson VE, Smith DH, Alafuzoff I, Attems J, Bencze J, Bieniek KF, Bigio EH, Bodi I, Budka H, Dickson DW, Dugger BN, Duyckaerts C, Ferrer I, Forrest SL, Gelpi E, Gentleman SM, Giaccone G, Grinberg LT, Halliday GM, Hatanpaa KJ, Hof PR, Hofer M, Hortobágyi T, Ironside JW, King A, Kofler J, Kövari E, Kril JJ, Love S, Mackenzie IR, Mao Q, Matej R, McLean C, Munoz DG, Murray ME, Neltner J, Nelson PT, Ritchie D, Rodriguez RD, Rohan Z, Rozemuller A, Sakai K, Schultz C, Seilhean D, Smith V, Tacik P, Takahashi H, Takao M, Rudolf Thal D, Weis S, Wharton SB, White CL, Woulfe JM, Yamada M, Trojanowski JQ (2017) Multisite assessment of aging-related tau astrogliopathy (ARTAG). J Neuropathol Exp Neurol 76:605–619. https://doi.org/10.1093/jnen/nlx041
Lace G, Ince PG, Brayne C, Savva GM, Matthews FE, de Silva R, Simpson JE, Wharton SB (2012) Mesial temporal astrocyte tau pathology in the MRC-CFAS ageing brain cohort. Dement Geriatr Cogn Disord 34:15–24. https://doi.org/10.1159/000341581
McKeith IG, Boeve BF, Dickson DW, Halliday G, Taylor J-P, Weintraub D, Aarsland D, Galvin J, Attems J, Ballard CG, Bayston A, Beach TG, Blanc F, Bohnen N, Bonanni L, Bras J, Brundin P, Burn D, Chen-Plotkin A, Duda JE, El-Agnaf O, Feldman H, Ferman TJ, Ffytche D, Fujishiro H, Galasko D, Goldman JG, Gomperts SN, Graff-Radford NR, Honig LS, Iranzo A, Kantarci K, Kaufer D, Kukull W, Lee VMY, Leverenz JB, Lewis S, Lippa C, Lunde A, Masellis M, Masliah E, McLean P, Mollenhauer B, Montine TJ, Moreno E, Mori E, Murray M, O’Brien JT, Orimo S, Postuma RB, Ramaswamy S, Ross OA, Salmon DP, Singleton A, Taylor A, Thomas A, Tiraboschi P, Toledo JB, Trojanowski JQ, Tsuang D, Walker Z, Yamada M, Kosaka K (2017) Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB consortium. Neurology. https://doi.org/10.1212/WNL.0000000000004058
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW, Duyckaerts C, Frosch MP, Masliah E, Mirra SS, Nelson PT, Schneider JA, Thal DR, Trojanowski JQ, Vinters HV, Hyman BT, National Institute on Aging, Alzheimer’s Association (2012) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol (Berl) 123:1–11. https://doi.org/10.1007/s00401-011-0910-3
Munoz DG, Woulfe J, Kertesz A (2007) Argyrophilic thorny astrocyte clusters in association with Alzheimer’s disease pathology in possible primary progressive aphasia. Acta Neuropathol (Berl) 114:347–357. https://doi.org/10.1007/s00401-007-0266-x
Narasimhan S, Guo JL, Changolkar L, Stieber A, McBride JD, Silva LV, He Z, Zhang B, Gathagan RJ, Trojanowski JQ, Lee VMY (2017) Pathological tau strains from human brains recapitulate the diversity of tauopathies in nontransgenic mouse brain. J Neurosci Off J Soc Neurosci 37:11406–11423. https://doi.org/10.1523/JNEUROSCI.1230-17.2017
Nelson PT, Alafuzoff I, Bigio EH, Bouras C, Braak H, Cairns NJ, Castellani RJ, Crain BJ, Davies P, Del Tredici K, Duyckaerts C, Frosch MP, Haroutunian V, Hof PR, Hulette CM, Hyman BT, Iwatsubo T, Jellinger KA, Jicha GA, Kövari E, Kukull WA, Leverenz JB, Love S, Mackenzie IR, Mann DM, Masliah E, McKee AC, Montine TJ, Morris JC, Schneider JA, Sonnen JA, Thal DR, Trojanowski JQ, Troncoso JC, Wisniewski T, Woltjer RL, Beach TG (2012) Correlation of Alzheimer disease neuropathologic changes with cognitive status: a review of the literature. J Neuropathol Exp Neurol 71:362–381. https://doi.org/10.1097/NEN.0b013e31825018f7
Nelson PT, Jicha GA, Schmitt FA, Liu H, Davis DG, Mendiondo MS, Abner EL, Markesbery WR (2007) Clinicopathologic correlations in a large Alzheimer disease center autopsy cohort: neuritic plaques and neurofibrillary tangles “do count” when staging disease severity. J Neuropathol Exp Neurol 66:1136–1146. https://doi.org/10.1097/nen.0b013e31815c5efb
Nelson PT, Trojanowski JQ, Abner EL, Al-Janabi OM, Jicha GA, Schmitt FA, Smith CD, Fardo DW, Wang W-X, Kryscio RJ, Neltner JH, Kukull WA, Cykowski MD, Van Eldik LJ, Ighodaro ET (2016) “New Old Pathologies”: Ad, PART, and Cerebral Age-Related TDP-43 With Sclerosis (CARTS). J Neuropathol Exp Neurol 75:482–498. https://doi.org/10.1093/jnen/nlw033
Neltner JH, Abner EL, Jicha GA, Schmitt FA, Patel E, Poon LW, Marla G, Green RC, Davey A, Johnson MA, Jazwinski SM, Kim S, Davis D, Woodard JL, Kryscio RJ, Van Eldik LJ, Nelson PT (2016) Brain pathologies in extreme old age. Neurobiol Aging 37:1–11. https://doi.org/10.1016/j.neurobiolaging.2015.10.009
Neuropathology Group. Medical Research Council Cognitive Function and Aging Study (2001) Pathological correlates of late-onset dementia in a multicentre, community-based population in England and Wales. Neuropathology Group of the Medical Research Council Cognitive Function and Ageing Study (MRC CFAS). Lancet Lond Engl 357:169–175
Robinson JL, Geser F, Corrada MM, Berlau DJ, Arnold SE, Lee VM-Y, Kawas CH, Trojanowski JQ (2011) Neocortical and hippocampal amyloid-β and tau measures associate with dementia in the oldest-old. Brain J Neurol 134:3708–3715. https://doi.org/10.1093/brain/awr308
Robinson JL, Molina-Porcel L, Corrada MM, Raible K, Lee EB, Lee VM-Y, Kawas CH, Trojanowski JQ (2014) Perforant path synaptic loss correlates with cognitive impairment and Alzheimer’s disease in the oldest-old. Brain J Neurol 137:2578–2587. https://doi.org/10.1093/brain/awu190
Santos CY, Snyder PJ, Wu W-C, Zhang M, Echeverria A, Alber J (2017) Pathophysiologic relationship between Alzheimer’s disease, cerebrovascular disease, and cardiovascular risk: a review and synthesis. Alzheimers Dement Diagn Assess Dis Monit 7:69–87. https://doi.org/10.1016/j.dadm.2017.01.005
Savva GM, Wharton SB, Ince PG, Forster G, Matthews FE, Brayne C, Medical Research Council Cognitive Function and Ageing Study (2009) Age, neuropathology, and dementia. N Engl J Med 360:2302–2309. https://doi.org/10.1056/nejmoa0806142
Schmidt ML, DiDario AG, Lee VM, Trojanowski JQ (1994) An extensive network of PHF tau-rich dystrophic neurites permeates neocortex and nearly all neuritic and diffuse amyloid plaques in Alzheimer disease. FEBS Lett 344:69–73
Schultz C, Ghebremedhin E, Del Tredici K, Rüb U, Braak H (2004) High prevalence of thorn-shaped astrocytes in the aged human medial temporal lobe. Neurobiol Aging 25:397–405. https://doi.org/10.1016/S0197-4580(03)00113-1
Thal DR, Capetillo-Zarate E, Del Tredici K, Braak H (2006) The development of amyloid beta protein deposits in the aged brain. Sci Aging Knowl Environ SAGE KE 2006:re1. https://doi.org/10.1126/sageke.2006.6.re1
Vos SJB, van Boxtel MPJ, Schiepers OJG, Deckers K, de Vugt M, Carrière I, Dartigues J-F, Peres K, Artero S, Ritchie K, Galluzzo L, Scafato E, Frisoni GB, Huisman M, Comijs HC, Sacuiu SF, Skoog I, Irving K, O’Donnell CA, Verhey FRJ, Visser PJ, Köhler S (2017) Modifiable risk factors for prevention of dementia in midlife, late life and the oldest-old: validation of the LIBRA index. J Alzheimers Dis JAD 58:537–547. https://doi.org/10.3233/JAD-161208
Wharton SB, Minett T, Drew D, Forster G, Matthews F, Brayne C, Ince PG, MRC Cognitive Function and Ageing Neuropathology Study Group (2016) Epidemiological pathology of Tau in the ageing brain: application of staging for neuropil threads (BrainNet Europe protocol) to the MRC cognitive function and ageing brain study. Acta Neuropathol Commun 4:11. https://doi.org/10.1186/s40478-016-0275-x
Acknowledgements
We wish to thank the participants and their relatives, testers and examiners of The 90+ Study, and the staff of the UCI brain repository for making this study possible. We also thank Terry Schuck and Katie Casalnova for their assistance with this study. Funding from the National Institutes of Health (AG10124, AG16573, AG17586, R01AG021055, R01AG042444, P50AG16573 and MH64045) supported this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Robinson, J.L., Corrada, M.M., Kovacs, G.G. et al. Non-Alzheimer’s contributions to dementia and cognitive resilience in The 90+ Study. Acta Neuropathol 136, 377–388 (2018). https://doi.org/10.1007/s00401-018-1872-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-018-1872-5