Abstract
Cerebellar mutism syndrome (CMS) is a well-known complication of posterior fossa (PF) tumour surgery. CMS has previously been reported in cases of non-tumour surgical aetiology in a limited number of publications. We report a case of a 10-year-old girl who suffered a cerebellar haemorrhage and subsequent CMS following surgical treatment of a ruptured arteriovenous malformation (AVM) in the cerebellar vermis. The AVM was removed acutely through a transvermian access, and hydrocephalus was treated with temporary external drainage. In the postoperative period, she suffered diffuse vasospasms of the anterior cerebral circulation and had a permanent shunt placed for hydrocephalus. Her mutism resolved after 45 days but severe ataxia persisted. To our knowledge, this is the first reported case of CMS related to a vermian haemorrhagic stroke with postoperative diffuse vasospasms. Based on this case, we present a literature review on CMS of non-tumour surgical origin in children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
CMS is a well-described complication of PF tumour surgery in children affecting up to 30% of cases. It typically arises within 1–10 days postoperatively [1] and consists of (1) transient mutism or speech reduced to a few words elicited only by vigorous stimulation, (2) emotional lability, (3) ataxia and (4) hypotonia [2]. Preoperative risk factors include tumour size, midline location, brain stem invasion and histopathology of medulloblastoma or atypical teratoid/rhabdoid tumour [1, 3].
Splitting the vermis (transvermian approach) has been suggested as a surgical risk factor of CMS, resulting in recommendations such as vermis-sparring surgical techniques (including the telovelar approach) to reduce the risk. Conflicting results have been published on this matter [4, 5].
It is broadly acknowledged that the dentate-thalamo-cortical pathway (DTCp) plays a central role in the development of CMS explained by proximal damage (either direct surgical trauma or subsequent postoperative oedema) to the dentate nucleus (DN) or superior cerebellar peduncle (SCP) leading to a disruption of the pathway resulting in cerebello-cerebral diaschisis and localized supratentorial hypoperfusion [6].
Traditionally, CMS has been related to PF tumour surgery; however, non-tumour surgical cases have been described including PF stroke, traumatic brain injury, cerebellar infection and inflammation as well as metabolic diseases [7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46]. A vascular aetiology like vasospasms underlying CMS has previously been hypothesized but not substantiated [47].
The aim of this study is to present a case of CMS following a haemorrhagic stroke from an AVM in the vermis in a paediatric patient. As the literature on CMS related to non-neoplastic pathologies is scarce, we additionally conducted a comprehensive literature review of paediatric cases of CMS of non-tumour surgical origin.
Case report
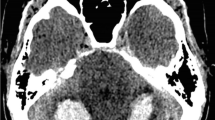
We present a 10-year-old girl, right-handed with unremarkable health and developmental history, who while watching TV suffered a sudden onset of frontal headache, quadriparesis and explosive vomiting followed by seizuring. She lost consciousness, was intubated and rushed to the hospital. On admission, she had isochoric pupils with normal light reflex. A computed tomography (CT) scan was performed showing a haemorrhagic lesion of the PF involving vermis with perforation to the ventricular system and hydrocephalus (Fig. 1).
The CT angiogram revealed an AVM in the vermis. The AVM was supplied by branches from both superior cerebellar arteries and classified as Spetzler-Martin grades 2–3 (Fig. 2).
Surgery was performed immediately after admission with the removal of the haematoma and AVM through a transvermian access. Hydrocephalus was initially treated with external ventricular drainage (EVD).
The girl was extubated on the first postoperative day uttering single words or short sentences. She had ataxia of the upper extremities and ocular motor control difficulties. She spoke few words on the 3rd postoperative day and underwent a postoperative digital subtraction angiography (DSA), which showed no sign of residual AV shunting.
On day 4, she had full onset mutism accompanied by agitation and emotional irritability. Her auditory and verbal comprehension was deemed intact as she was able to respond adequately to questions by squeezing either her right or left hand.
A follow-up magnetic resonance imaging (MRI) of the brain on the 11th postoperative day showed oedema and haematoma involving both superior cerebellar peduncles (SCP) and vermis, a small haematoma remnant in the surgical cavity and perifocal oedema surrounding the cavity (Fig. 3).
Her EVD was removed on day 17. Increased agitation was observed in the following days. On day 19, a CT cerebral angiography was performed due to sudden onset of right upper extremity paralysis showing narrowing of the anterior cerebral artery (ACA) and median cerebral artery (MCA) bilaterally as well as hydrocephalus. A diagnosis of vasospasms was suggested but not confirmed on a DSA. Furthermore, there was doubt whether the possible vasospasms were in fact the cause of her localized paresis. There was no angiographic evidence of AVM remnants. Hydrocephalus was treated with reimplantation of an EVD and replaced by a ventriculo-peritoneal (VP) shunt 3 days later. She was transferred to the neurointensive care unit for conservative treatment of vasospasms with calcium antagonists (nimodipine 1 g/kg × 6) for 7 days. A follow-up CT cerebral angiography on day 20 showed no signs of vasospasms. Due to dysphagia, she had a gastrostomy tube inserted on day 26. On day 33, she was transferred to a specialized neurorehabilitation unit. Her right upper extremity paresis had resolved but she suffered from global hypotonia.
Her mutism lasted 45 days and on day 49 she started producing single words. On day 77 she was discharged to outpatient rehabilitation, with ongoing symptoms of ataxic dysarthria and dysprosody. Prior to discharge, her neuropsychological evaluation with WISC-V (Wechsler Intelligence Scale for Children, 5th edition) and TOMAL-2 (Test of Memory and Learning, 2nd edition) revealed reduced verbal working memory and learning ability.
At 4-month follow-up, she was able to speak in full sentences, however still dysarthric. Furthermore, she suffered from severe upper limb ataxia and was dependent on a walker. The last follow-up was 12 months post ictus where cognition and language were re-established; however, she suffered from marginally slowed speech and slight dysarthria. She had ataxia and impaired balance primarily on the right side. She was able to write with her right hand, although compromised, and ate without dysphagia.
Literature review
Methods
A PubMed literature search until November 31, 2022, was conducted. The search generated 347 articles. The following MeSH search term was used: (((“Mutism”[Mesh] OR Mutism OR mute)) AND (“Adolescent”[Mesh] OR “Child”[Mesh] OR “Infant”[Mesh] OR Adolescent OR Child OR Infant OR Paediatric OR Pediatric)) AND ((“Cerebellar Diseases”[Mesh] OR Cerebellar OR Posterior Fossa)))).
Inclusion required the following: (1) mutism of cerebellar origin, (2) no cerebellar tumour surgery, (3) paediatric patients defined as < 18 years at diagnosis. All titles and, when relevant, abstracts and full articles were reviewed by the first author. Forty case reports were included in the literature review and previous relevant focused literature reviews were included as references in the discussion. Cross-checking references from included case reports yielded no additional relevant articles. Figure 4 shows a prisma diagram over the literature search and inclusion.
Results
Table 1 summarizes the clinical and paraclinical relevant findings of the case reports. Table 2 gives an overview of the demographics, predominant clinical and neuroradiographic features of the 3 largest case groups.
Included publications contained a total of 58 reported cases divided into groups depending on the probable or known cause of cerebellar mutism.
Mutism ranged from 2 days to > 180 days with a median of 14 days; however, some articles did not report the exact duration of mutism. Predominant accompanying symptoms involved limb or truncal ataxia, behavioural changes and hypotonia, as well as subsequent ataxic dysarthria. Predominant persisting neurological deficits at the final follow-up involved dysarthria, slow speech and gait or limb ataxia with follow-up ranging from 2 weeks to 6 years. One case died after 26 days from the primary incident, 2 articles did not include follow-up in the history and 5 articles did not report follow-up time.
Case reports of CMS related to infection
The most frequently reported causes were cerebellitis due to rotavirus (n = 17 in 5 case reports [16, 27, 34, 39, 41] followed by cerebellitis or encephalopathy from influenza virus A/B (n = 3 [19, 38, 42]), cerebellitis from varicella-zoster virus (n = 2 [10, 17]) and pneumococcal meningitis (n = 1 [15]). In 10 cases (8 case reports [9, 14, 29,30,31, 35,36,37]), cerebellitis and acute disseminated encephalomyelitis were described without an identified pathogen.
Duration of mutism lasted from 2 days to < 180 days. Two cases had hydrocephalus treated with EVD and 1 case was treated with a VP shunt. Case information from one article in Spanish was included only from the abstract.
Case reports of CMS related to haemorrhagic vascular pathology
AVM
Eight patients in 7 case reports were identified [7, 8, 12, 13, 25, 43, 44]. In all cases, the AVM was located in the vermis. All cases presented with a haemorrhage from the AVM. In 6 patients the AVM was surgically removed and in 2 patients the AVM was treated with embolization. One patient was treated with a VP shunt. None reported postoperative or posthaemorrhagic vasospasms.
Mutism lasted from 4 days to > 180 days (mute at last follow-up) and 1 article did not account for the duration of mutism.
Cavernous malformation
Two reports each with a single case describe CMS related to a cavernous malformation [21, 45], the first case located in the right pons and SCP and the second case located in the mesencephalon. Both cases presented with haemorrhage from the cavernoma. One case had immediate surgical removal of the cavernoma and 1 patient had surgical removal of the cavernoma 7 days post ictus. The duration of mutism was 12 days in both cases.
Haemorrhagic stroke
One case report described mutism in a single patient after suffering a spontaneous haemorrhagic stroke in the vermis [40] without apparent vascular abnormality. The haematoma was immediately evacuated. Mutism lasted 42 days.
Spontaneous intratumoural bleed
A single case reported mutism from a spontaneous intratumoural haemorrhage lasting a total of 10 days [20]. The tumour was surgically removed 8 weeks after the haemorrhage.
Case reports of CMS related to ischaemic vascular pathology
Five patients suffering mutism following a vascular ischaemic stroke were described in 3 case reports [23, 26, 32]. Three cases suffered ischaemic stroke subsequent to vertebral artery dissection, of which 1 was caused by trauma. One case suffered isolated spontaneous basilar artery occlusion and finally 1 patient suffered hyperleukocytosis-induced stroke during the onset of ALL. All were treated conservatively. The duration of mutism lasted from 10 to 42 days, with 1 article not accounting for the duration of mutism.
Case reports of CMS related to TBI
Eight patients in 7 case reports were identified [11, 18, 22, 24, 25, 28, 46]. One patient had immediate evacuation of an acute subdural haematoma in the PF; the remaining cases were either treated conservatively (5/8) or treatment was unaccounted for (2/8). Mutism lasted from a few days (without further specification) to 56 days. One included case was reported in Japanese with an English abstract with no documentation of mutism duration.
Other pathologies
We found one case of reported mutism related to a metabolic disorder (ornithine transcarbamylase deficiency). This X-linked urea cycle disorder results in neurotoxic hyperammonaemia with neurologic symptoms such as seizures, decreased consciousness and abnormal motor function [33]. In this reported case mutism lasted 4 days.
Discussion
This case of CMS in a 10-year-old girl following emergency surgery for a ruptured vermian AVM and the additional literature review support alternate aetiologies other than cerebellar tumour surgery to potentially result in CMS.
In our literature search, we encountered a few reviews on subgroups, but no previous comprehensive literature review of paediatric cases of CMS of non-tumour surgical origin. Baillieux et al. [8] reviewed the literature on CMS of vascular origin in 2007 and found 10 published cases. Makarenko et al. [29] reviewed literature on CMS of non-surgical origin in 2017 finding 20 patients. Lahirish et al. [28] reviewed the literature on CMS in paediatric head trauma in 2021 and included 6 patients. Thus, the current literature review contributes with an updated, comprehensive and collected overview for future reference. An important note is that the majority of case reports (n = 30/40) included in this literature overview were published prior to the Delphi Consensus definition of CMS [2] potentially leading to case heterogeneity as the definition of CMS was not consistent.
The anatomical substrate of CMS is broadly acknowledged to be a cerebello-cerebral efferent pathway, namely the DTCp, where proximal damage to the tract results in diminished cerebellar input to the cerebral cortex. Additionally, recent research has suggested that the fastigial nucleus and the periaqueductal grey matter may play an underappreciated role in this syndrome [48]. Numerous theories of the pathophysiology of DTCp disruption have been suggested. The main theory is that the surgical approach may cause direct injury to the DN or SCP. Other theories involve postoperative oedema affecting regions of interest, axonal injury and degeneration due to a cytotoxic environment, thermal injury related to the use of an ultrasonic surgical aspirator and cerebellar perfusion deficits due to vasospasms [47]. Potentially, tissue damage involving the same regions due to either primary cerebellar insults or cerebellar surgery for other pathologies than neoplasia could also trigger pathophysiologcal mechanisms hypothesized for CMS. The potential role of hydrocephalus in CMS risk and severity has yet to be substantially elucidated.
The reason for CMS in our case is likely to be multifactorial supporting the theory of CMS originating from various pathophysiological mechanisms. Our patient suffered tissue trauma—both from the stroke and surgery—to presumed critical structures of the syndrome; her imaging showed blood and oedema involving SCP and the deep cerebellar structures bilaterally. In addition, she had possible diffuse vasospasms of the anterior cerebral circulation and suffered from hydrocephalus, treated initially with an EVD and then with a permanent shunt after a failed attempt to relieve her from the EVD.
Postsurgical CMS
Our comprehensive literature review supports the theory of CMS being due to damage of anatomical substrates, regardless of aetiology, rather than being a direct consequence of PF tumour surgery. Concomitantly, several cases reported CMS after non-tumoural PF surgery, both endovascular [7, 12] and open resection [13, 21, 22, 40, 45]. It has previously been suggested that a vermis-sparing, telovelar approach to the 4th ventricle reduced the risk of CMS. However, a prospective study on 500 children with PF tumours found no difference in the risk of postoperative speech impairment between the telovelar and transvermian approach [1]. CMS in the presented case occurred after applying a transvermian access for evacuating the haematoma and excising the AVM, with a delayed onset similar in time to CMS following PF tumour surgery. In our case, the postoperative MRI on day 11th revealed oedema and blood in the bilateral SCP. Interestingly, studies report conflicting results on lateralized damage to the SCP and risk of CMS; however, bilateral damage to the SCP as in our case seem to increase the risk [49]. As in previously published cases, it is difficult to determine whether the injury from the initial haemorrhage, the subsequent surgical evacuation or a combination of the two caused CMS in our case.
During the admission, our case experienced a sudden onset of right upper extremity paralysis with a CT cerebral angiography suggestive of vasospasms, however not confirmed by DSA. A CT cerebral angiography the following day showed normalization of the artery caliber. Diffuse vasospasms in PF tumour surgery have previously been reported in cases [47] but to our knowledge not in context with paediatric PF AVM resection. A recent case from Deghedy et al. showed basilar artery spasm and subsequent mutism in a patient who underwent PF tumour resection 3 days prior to the insult. Thus, CMS due to vasospasms of the microcirculation seems plausible due to the syndrome’s delayed onset from surgery, yet no substantial study supporting this hypothesis has been published. Treatment with a calcium antagonist was applied to alleviate the diffuse vasospasms in our case, although mutism persisted long after the therapy was stopped. In our opinion, it is speculative yet interesting whether calcium antagonists could alleviate symptoms of CMS if vasospasms prove to play a central part in the aetiology of CMS.
No substantial pharmacological treatment of CMS has been published. Fluoxetine, zolpidem, bromocriptine and donepezil have been suggested but only reported in a small number of cases [50]. Although, a recent study using metformin to promote neurogenesis showed promising results on neurocognitive outcome [51], current suggested treatment is limited to rehabilitation of speech, neurocognition and motor skills.
CMS following non-surgical cerebellar insults
Our review reveals that several cases of CMS of non-surgical aetiology have been published, ranging from infection and subsequent cerebellitis over spontaneous intratumoural bleeding and stroke-induced to direct cerebellar trauma. Predominant radiological findings included lesions in the vermis and diffusion restriction in DN. A study by Di Rocco et al. [52] on 34 children with PF tumours found preoperative language impairment in 11 patients, suggestive of a subclinical state of CMS even before surgical intervention. Furthermore, the subgroup with preoperative language impairment had a higher incidence of tumour invasion of the DN. The involvement of the DN is in accordance with the current understanding of the anatomical substrates of CMS. It seems conspicuous that multiple incidents can cause CMS, surgical trauma being just one of many. In our opinion, it is crucial to understand the phenomenon that we move beyond defining CMS as a postoperative complication to PF tumour surgery.
CMS severity related to AVM
Even though no analytical statistics were applied in this article, there may be a tendency toward mutism duration lasting longer in AVM cases. The median duration of mutism was 29 days in the AVM group, whereas the median duration of mutism in the TBI and cerebellitis group was 14 days corresponding well with the median duration of 16 days found in the study of PF tumours by Grønbæk et al. [1]. Perhaps this difference could be explained by both the spontaneous bleeding and the surgical trauma causing additive injury to the cerebellum and related deep structures, thus amplifying the impact. Furthermore, it is likely that only the most severe cases generate published case reports.
Neuropsychological consequences of CMS
Mutism is the most apparent syndrome of CMS, but long-term neuropsychological consequences in children suffering from PF tumours with subsequent CMS have gained increased focus in the recent years. In our case, the patient had a remarkable performance in school prior to the ictus according to her parents. During her in-hospital rehabilitation around day 75 postoperatively, TOMAL-2 and WISC-V showed reduced verbal working memory and reduced learning ability suggesting long-term neuropsychological problems as consequences of her disease. Children with CMS seem to experience a significant decline in their intellect [53], although long-term neuropsychological deficits of PF surgery and CMS are currently scarcely elucidated and possibly underappreciated. A recent study on the rehabilitation of children treated for PF tumours [54] concluded that advancements in computer technology and digital tools have led to targeted rehabilitation of neurocognitive deficits. However, future studies evaluating treatment and rehabilitation protocol efficacy are needed.
Conclusion
This case presentation along with the accompanying literature review provides evidence that CMS can be of non-tumour surgical origin, symptoms being similar to CMS related to PF tumour surgery. Furthermore, our presentation suggests that a broader range of pathologic factors than previously acknowledged may have the potential to cause CMS including a potential role of vasospasms. Clarifying such factors could point at treatment options to alleviate symptoms. In this way, PF pathology of non-tumoural aetiology may be helpful in explaining this severe and devastating syndrome.
Data availability
Not applicable.
References
Gronbaek JK et al (2021) Postoperative speech impairment and surgical approach to posterior fossa tumours in children: a prospective European multicentre cohort study. Lancet Child Adolesc Health 5(11):814–824. https://doi.org/10.1016/S2352-4642(21)00274-1
Gudrunardottir T et al (2016) Consensus paper on post-operative pediatric cerebellar mutism syndrome: the Iceland Delphi results. Childs Nerv Syst 32(7):1195–1203. https://doi.org/10.1007/s00381-016-3093-3
Gudrunardottir T, Sehested A, Juhler M, Schmiegelow K (2011) Cerebellar mutism: review of the literature. Childs Nerv Syst 27(3):355–363. https://doi.org/10.1007/s00381-010-1328-2
Cobourn K et al (2020) Cerebellar mutism syndrome: current approaches to minimize risk for CMS. Childs Nerv Syst 36(6):1171–1179. https://doi.org/10.1007/s00381-019-04240-x
Renne B et al (2020) Cerebellar mutism after posterior fossa tumor resection in children: a multicenter international retrospective study to determine possible modifiable factors. Childs Nerv Syst 36(6):1159–1169. https://doi.org/10.1007/s00381-019-04058-7
Catsman-Berrevoets CE, Aarsen FK (2010) The spectrum of neurobehavioural deficits in the posterior fossa syndrome in children after cerebellar tumour surgery. Cortex 46(7):933–946. https://doi.org/10.1016/j.cortex.2009.10.007
Al-Anazi A, Hassounah M, Sheikh B, Barayan S (2001) Cerebellar mutism caused by arteriovenous malformation of the vermis. Br J Neurosurg 15(1):47–50. https://doi.org/10.1080/026886901300004094
Baillieux H, Weyns F, Paquier P, De Deyn PP, Marien P (2007) Posterior fossa syndrome after a vermian stroke: a new case and review of the literature. Pediatr Neurosurg 43(5):386–395. https://doi.org/10.1159/000106388
Barragan-Martinez D et al (2017) Language disorders in acute cerebellitis: beyond dysarthria. Rev Neurol 64(1):27–30
Brito T, Oliveira I, Silva L, Parente S (2019) Transient mutism, pathological laughter and cerebellar ataxia after primary varicella-zoster virus infection. BMJ Case Rep 12(11). https://doi.org/10.1136/bcr-2019-230683
Chivet A, Delestret I, Brodar C, Vinchon M (2022) Cerebellar mutism syndrome in pediatric head trauma with cerebellar injury. Childs Nerv Syst 38(4):759–766. https://doi.org/10.1007/s00381-021-05422-2
Demartini Z Jr et al (2020) Cerebellar mutism after embolization of vermian arteriovenous malformation. Childs Nerv Syst 36(6):1301–1305. https://doi.org/10.1007/s00381-019-04483-8
Dietze DD Jr., Mickle JP (1990) Cerebellar mutism after posterior fossa surgery. Pediatr Neurosurg 16(1):25–31; discussion 31. https://doi.org/10.1159/000120499
Dimova PS, Bojinova VS, Milanov IG (2009) Transient mutism and pathologic laughter in the course of cerebellitis. Pediatr Neurol 41(1):49–52. https://doi.org/10.1016/j.pediatrneurol.2009.01.013
Drost G, Verrips A, Thijssen HO, Gabreels (2000) Cerebellar involvement as a rare complication of pneumococcal meningitis. Neuropediatrics 31(2):97–9. https://doi.org/10.1055/s-2000-7474
Engan M, Bjorlykke JA, Moen G, Lund KB, Njolstad G (2016) A 4-year-old girl with diarrhoea, paresis and mutism. Tidsskr Nor Laegeforen 136(17):1458–1460. https://doi.org/10.4045/tidsskr.16.0070
Erol I, Ozkale Y, Saygi S, Alehan F (2014) Cerebellar mutism caused by primary varicella infection in an immunocompetent child. J Child Neurol 29(6):830–832. https://doi.org/10.1177/0883073813477202
Ersahin Y, Mutluer S, Saydam S, Barcin E (1997) Cerebellar mutism: report of two unusual cases and review of the literature. Clin Neurol Neurosurg 99(2):130–134
Fluss J et al (2010) Mild influenza-associated encephalopathy/encephalitis with a reversible splenial lesion in a Caucasian child with additional cerebellar features. Eur J Paediatr Neurol 14(1):97–100. https://doi.org/10.1016/j.ejpn.2009.09.007
Frassanito P, Massimi L, Caldarelli M, Di Rocco C (2009) Cerebellar mutism after spontaneous intratumoral bleeding involving the upper cerebellar vermis: a contribution to the physiopathogenic interpretation. Childs Nerv Syst 25(1):7–11. https://doi.org/10.1007/s00381-008-0711-8
Frim DM, Ogilvy CS (1995) Mutism and cerebellar dysarthria after brain stem surgery: case report. Neurosurgery 36(4):854–857. https://doi.org/10.1227/00006123-199504000-00031
Fujisawa H et al (2005) Mutism after evacuation of acute subdural hematoma of the posterior fossa. Childs Nerv Syst 21(3):234–236. https://doi.org/10.1007/s00381-004-0999-y
Hashem H, Muhsen BA (2021) Hyperleukocytosis-induced stroke and tonsillar herniation: case report. Ann Med Surg (Lond). 70:102776. https://doi.org/10.1016/j.amsu.2021.102776
Kariyattil R, Rahim MI, Muthukuttiparambil U (2015) Cerebellar mutism following closed head injury in a child. Sultan Qaboos Univ Med J 15(1):e133–e135
Koh S, Turkel SB, Baram TZ (1997) Cerebellar mutism in children: report of six cases and potential mechanisms. Pediatr Neurol 16(3):218–219. https://doi.org/10.1016/s0887-8994(97)00018-0
Kossorotoff M et al (2010) A cognitive and affective pattern in posterior fossa strokes in children: a case series. Dev Med Child Neurol 52(7):626–631. https://doi.org/10.1111/j.1469-8749.2010.03616.x
Kubota T et al (2011) Chronological diffusion-weighted imaging changes and mutism in the course of rotavirus-associated acute cerebellitis/cerebellopathy concurrent with encephalitis/encephalopathy. Brain Dev 33(1):21–27. https://doi.org/10.1016/j.braindev.2010.04.007
Lahirish IAM et al (2021) Cerebellar mutism following head trauma: a case report and literature review. Surg Neurol Int 12:446. https://doi.org/10.25259/SNI_307_2021
Makarenko S, Singh N, McDonald PJ (2018) Non-surgical transient cerebellar mutism-case report and systematic review. Childs Nerv Syst 34(3):535–540. https://doi.org/10.1007/s00381-017-3643-3
McAndrew S, Listernick R, Kuntz N (2014) Cerebellar mutism in acute disseminating encephalomyelitis. Pediatr Neurol 50(5):511–514. https://doi.org/10.1016/j.pediatrneurol.2014.01.020
Mewasingh LD, Kadhim H, Christophe C, Christiaens FJ, Dan B (2003) Nonsurgical cerebellar mutism (anarthria) in two children. Pediatr Neurol 28(1):59–63. https://doi.org/10.1016/s0887-8994(02)00503-9
Miyakita Y, Taguchi Y, Sakakibara Y, Matsuzawa M, Kitagawa H (1999) Transient mutism resolving into cerebellar speech after brain stem infarction following a traumatic injury of the vertebral artery in a child. Acta Neurochir (Wien) 141(2):209–213. https://doi.org/10.1007/s007010050288
Nedermeijer SC, van den Hout J, Geleijns C, de Klerk H, Catsman-Berrevoets CE (2015) Posterior fossa syndrome in a patient with an ornithine transcarbamylase deficiency. Eur J Paediatr Neurol 19(3):364–366. https://doi.org/10.1016/j.ejpn.2014.12.001
Paketci C et al (2020) Rotavirus encephalopathy with concomitant acute cerebellitis: report of a case and review of the literature. Turk J Pediatr 62(1):119–124. https://doi.org/10.24953/turkjped.2020.01.017
Papavasiliou AS, Kotsalis C, Trakadas S (2004) Transient cerebellar mutism in the course of acute cerebellitis. Pediatr Neurol 30(1):71–74. https://doi.org/10.1016/s0887-8994(03)00420-x
Parrish JB, Weinstock-Guttman B, Yeh EA (2010) Cerebellar mutism in pediatric acute disseminated encephalomyelitis. Pediatr Neurol 42(4):259–266. https://doi.org/10.1016/j.pediatrneurol.2009.11.003
Riva D (1998) The cerebellar contribution to language and sequential functions: evidence from a child with cerebellitis. Cortex 34(2):279–287. https://doi.org/10.1016/s0010-9452(08)70755-x
Sanders PJ, van Waardenburg DA, Vermeulen RJ (2020) Influenza A-associated acute necrotising encephalopathy in a 10-year-old child. BMJ Case Rep 13(8). https://doi.org/10.1136/bcr-2019-233541
Shiihara T et al (2007) Rotavirus associated acute encephalitis/encephalopathy and concurrent cerebellitis: report of two cases. Brain Dev 29(10):670–673. https://doi.org/10.1016/j.braindev.2007.04.005
Sinha AK, Rajender Y, Dinakar I (1998) Transient cerebellar mutism after evacuation of a spontaneous vermian haematoma. Childs Nerv Syst 14(9):460–462. https://doi.org/10.1007/s003810050261
Takanashi J et al (2010) Clinical and radiological features of rotavirus cerebellitis. AJNR Am J Neuroradiol 31(9):1591–1595. https://doi.org/10.3174/ajnr.A2131
Thabet FI, Khalil S, Naz F, Dyme IZ (2013) Cerebellar mutism and reversible cytotoxic edema in influenza B-associated encephalopathy. Pediatr Neurol 49(6):489–492. https://doi.org/10.1016/j.pediatrneurol.2013.06.014
Turkel SB et al (2004) Case series: acute mood symptoms associated with posterior fossa lesions in children. J Neuropsychiatry Clin Neurosci 16(4):443–445. https://doi.org/10.1176/jnp.16.4.443
Vandeinse D, Hornyak JE (1997) Linguistic and cognitive deficits associated with cerebellar mutism. Pediatr Rehabil 1(1):41–44. https://doi.org/10.3109/17518429709060941
Wang MC, Winston KR, Breeze RE (2022) Cerebellar mutism associated with a midbrain cavernous malformation. Case report and review of the literature. J Neurosurg 96(3):607–10. https://doi.org/10.3171/jns.2002.96.3.0607
Yokota H, Nakazawa S, Kobayashi S, Taniguchi Y, Yukihide T (1990) Clinical study of two cases of traumatic cerebellar injury. No Shinkei Geka 18(1):67–70
Avula S, Mallucci C, Kumar R, Pizer B (2015) Posterior fossa syndrome following brain tumour resection: review of pathophysiology and a new hypothesis on its pathogenesis. Childs Nerv Syst 31(10):1859–1867. https://doi.org/10.1007/s00381-015-2797-0
McAfee SS et al (2022) Fastigial nuclei surgical damage and focal midbrain disruption implicate PAG survival circuits in cerebellar mutism syndrome. Neuro Oncol. https://doi.org/10.1093/neuonc/noac168
Gronbaek JK et al (2022) Left-handedness should not be overrated as a risk factor for postoperative speech impairment in children after posterior fossa tumour surgery: a prospective European multicentre study. Childs Nerv Syst 38(8):1479–1485. https://doi.org/10.1007/s00381-022-05567-8
Ashida R, Nazar N, Edwards R, Teo M (2021) Cerebellar mutism syndrome: an overview of the pathophysiology in relation to the cerebrocerebellar anatomy, risk factors, potential treatments, and outcomes. World Neurosurg 153:63–74. https://doi.org/10.1016/j.wneu.2021.06.065
Ayoub R et al (2020) Assessment of cognitive and neural recovery in survivors of pediatric brain tumors in a pilot clinical trial using metformin. Nat Med 26(8):1285–1294. https://doi.org/10.1038/s41591-020-0985-2
Di Rocco C et al (2011) Heralding cerebellar mutism: evidence for pre-surgical language impairment as primary risk factor in posterior fossa surgery. Cerebellum 10(3):551–62. https://doi.org/10.1007/s12311-011-0273-2
Moxon-Emre I et al (2014) Impact of craniospinal dose, boost volume, and neurologic complications on intellectual outcome in patients with medulloblastoma. J Clin Oncol 32(17):1760–1768. https://doi.org/10.1200/JCO.2013.52.3290
Chieffo DPR et al (2022) Posterior fossa tumor rehabilitation: an up-to-date overview. Children (Basel) 9(6). https://doi.org/10.3390/children9060904
Funding
Open access funding provided by Royal Danish Library.
Author information
Authors and Affiliations
Contributions
All authors took part in the first draft of the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable. We thank the patient and parents for giving consent to publish this case.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laustsen, A.F., Børresen, M.L., Hauerberg, J. et al. Cerebellar mutism syndrome of non-tumour surgical aetiology—a case report and literature review. Childs Nerv Syst 39, 2201–2213 (2023). https://doi.org/10.1007/s00381-023-05947-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-023-05947-8