Abstract
Purpose
Selective dorsal rhizotomy (SDR) reduces spasticity in children with cerebral palsy (CP) and is performed either through a lumbosacral multilevel laminectomy or a single-level laminectomy at the medullary conus. Spinal interventions generally involve the risk of subsequent instability depending on the extent of structural weakening. Destabilizing spasticity in CP might further increase this risk for both options. Laminoplasty is frequently applied to reduce instability through anatomical restoration, although the unavoidable interruption of interspinous ligaments might be a reason for inconsistent results. We report on a novel technique of laminoplasty, achieving complete restoration of the dorsal column.
Methods
One hundred sixteen ambulatory children with gross motor function classification scale (GMFCS) level I to III were submitted to SDR through a single-level approach. The lamina was reinserted with a previously unreported technique of laminoplasty. Osseous reintegration of the excised lamina was supposed, if its spinous process was located in place on late follow-up radiographs. Scoliosis was described via Cobb’s angle.
Results
At a mean follow-up of 33 months, radiographs were available from 72 children with a mean age at surgery of 7.2 years. Sixty-two out of the 72 reinserted laminae were supposed to be vital and reintegrated. Seven children developed a predominantly mild scoliosis. No association was found between development of scoliosis and GMFCS level or age.
Conclusions
This novel laminoplasty technique provides the least invasive approach for SDR. The incidence of scoliosis after this single-level approach is comparable to the natural history of ambulatory CP children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Selective dorsal rhizotomy (SDR) is a surgical procedure to reduce spasticity. Afferent lumbar and sacral sensory nerves are partially interrupted to decrease the hyperexcitable reflexes, which are predominantly responsible for the spasticity [1]. The goal of this procedure is to improve motor function in spastic patients. After a continuous evolution of the technique Foerster [2] first described in 1908, it became widely accepted as a treatment option for children with cerebral palsy in the 1980s [3–5]. The traditional multisegmental laminectomy, aiming at sectioning each dorsal nerve root at its very segmental level [5], has been modified in order to address the entirety of them through one monosegmental laminectomy at the level of the medullary conus [6].
Most centers stayed with the traditional multilevel laminectomy with or without laminoplasty because the segmental allocation of each nerve root seems to be easier at the site of its neuroforamen [5, 7–12].
Intraoperative neuromonitoring (IONM) is applied to select and classify the sensory nerve rootlets to be interrupted [13]. For this, the EMG signal after in situ stimulation is graded according to Phillips and Park [14]. To minimize undesirable side effects, about 50 to 70 % of the rootlets are selected and interrupted [10]. Results regarding the successful reduction of spasticity have been published copiously in the last 30 years [15–18].
Few authors point out a high risk of developing spinal deformities after SDR performed via multilevel approaches [9, 11, 12, 19–21]. An incidence of scoliosis of 16 to 55 % in their heterogeneous cohorts was reported, whereas Cobb angles of more than 40° were seen rarely. The risk for spinal deformities particularly increases when more than two laminae are removed [22, 23]. In order to reduce this risk and to decrease postoperative adhesions and pain, laminoplasty was added by some surgeons [9–11]. Unfortunately, multilevel laminoplasty does not reduce the risk of instability compared to laminectomy [9]. A previous comparison of monosegmental versus multisegmental approaches did not specifically study the incidence of spinal deformities [24] but, although instability after monosegmental laminectomy has been described, a current cohort of patients, that underwent SDR via monosegmental laminectomy, did not need treatment for spinal deformities so far [6].
The aim of this study is to report on the first experiences with a new method of laminoplasty which can be used for any extent of laminectomy. For this study, the application was restricted to monosegmental SDR, in order to monitor the reintegration of the lamina and the development or progression of scoliosis after this procedure.
Patients and methods
Patients and study design
One hundred thirty-two children with a mainly spastic type of lower-limb involvement of cerebral palsy and a mean age of 7.2 ± 2.9 years (min-max 2.7–17.1 years) at the time of surgery underwent selective dorsal rhizotomy applying the technique described below between 01/2007 and 02/2015 in a single level-one center for pediatric neurosurgery. Inclusion for surgery followed the criteria established by Park and Johnston [6], requesting independent ambulation corresponding to gross motor functioning classification scale (GMFCS) level I to III [25]. A preoperative Cobb angle above 20° was an exclusion criterion. The study design included x-ray examinations of the spine preoperatively, as well as 1, 2, and 5 years postoperatively. Within our prospective cohort study, 72 patients had at least one preoperative and one follow-up anterior-posterior x-ray of the spine and were included for spinal deformity evaluations as well as for the evaluation of successful laminoplasty.
Operative technique
Prior to surgery, the level of the medullary cone is determined under MRI guidance using enhancing skin markers. Needles are placed and fixed for EMG recording of reference muscles from L2 to S2. Surgery then is performed under general anesthesia with the patient in a prone position. After soft-tissue preparation, the laminectomy is performed in a conventional technique providing an interlaminar access and then using a standard craniotome to interrupt the lamina on both sides. The following oblique transsection of the spinal processes above and below the interrupted segment is performed with a Ruskin bone splitting forceps with long blades (Fig. 1). Then, the disconnected complex containing the lamina, its spinal process, and the posterior ligament complex holding the adjoining parts of the split processes is harvested and stored in damp cloth.
Through a midline dural incision, the sensory rootlets L1 to S2 are identified and separated from the motor rootlets, which remain covered by a synthetic pad throughout the procedure. In a L1 laminectomy, L1 can be identified directly, following the respective rootlets down to their dural exit. Fifty percent of the sensory rootlets of L1 are selected randomly as there is no reference muscle. From L2 to S1, the roots are separated into four to eight rootlets. Fifty to 60 % of the rootlets are cut according to EMG evaluation. Finally, S2 is divided into two parts and the fascicle showing less pudendus activity while stimulating the bulbo-cavernous reflex arc is severed. The dura is closed tightly and a caudally directed peridural catheter is put in place.
The lamina is refixed with a single multi-hole miniplate (MatrixNEURO™, DePuy Synthes, Zuchwil, Switzerland; Fig. 2) on both sides. Then, the interrupted spinal processes are readapted with a compression suture using ethibond 0 with a cutting needle which either penetrates through the readapted parts of the spinal process or—in older children—is conducted twice around the process fragments through the interspinous ligament and then closed tightly.
Finally, the muscle fascia is closed, readapting it with appropriate stitches to the interspinous and the supraspinous ligament and the wound is closed in layers.
Postoperative management
Combined intravenous and peridural analgesia is provided for 3 days postoperatively. During 3 days of bed rest, passive movement exercises are performed. Afterwards, rapid mobilization is pursued. An intensive 6 weeks in-patient rehabilitation program aiming at improving gait and strength is started approximately 1 week postoperatively. To avoid substantial mechanical loads on the laminoplasty, bending exercises and whole body vibration are not recommended for 3 months postoperatively; horseback riding and impact sports should be paused for 6 months postoperatively. No restrictions are made concerning regular daily activities. This postoperative protocol was based on the intention to encourage walking and activities of daily living while restricting activities which might interfere with spinal healing. The protocol was applied universally and was not modified for the level of laminoplasty, age at surgery, or gender.
Methods of evaluation
The primary aim of our x-ray analysis was the measurement of Cobb angles preoperatively and postoperatively to confirm the required inclusion criteria as well as to monitor the incidence of short and mid-term coronal plane deformities. Furthermore, the successful reintegration of the resected lamina could be determined reviewing the visibility of the respective spinous process (Fig. 3). The development of scoliosis was measured according to Cobb. The degree of scoliosis was classified into “no,” “mild,” or “moderate” scoliosis referring to a Cobb angle of ≤10°, 11–20°, or 21–40°. All measurements were performed independently by each author and blinded to the patients’ data. In cases of discrepancy, the highest pathologic value was used. Perioperative complications, especially urogenital dysfunction and wound infections, were recorded. At each follow-up, children and parents were surveyed for pain incidents during the interval with regard to location, duration and treatment.
A descriptive data analysis was performed. To find associations between continuous and ordinal variables, the Kruskal-Wallis test was applied, for statistical analysis of two nominal or ordinal variables Kendall’s tau was used. Significance level was set at p < 0.05.
Results
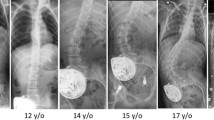
The parents of 116 out of the 132 children who underwent SDR between 1/2007 and 2/2015 consented the inclusion of their children into this prospective study. The main reason for declining consent was high travel expenses. In the studied cohort, no cases of superficial or deep wound infection or persistent liquor leakage were noted. Another 20 children were not yet due for their first follow-up examination at the time of data extraction (04/2015). In 72 (75 %) of the remaining 96 children, at least one follow-up x-ray was available. A summary of their demographic, functional, and radiographic data is given in Table 1. The mean age of this group was 7.2 ± 2.6 years at the time of surgery (min 2.7, max 15.4 years). The GMFCS level was I in 15, II in 30, and III in 27 patients. At follow-up, only two patients had improved by one level on the GMFC scale (one from III to II and one from II to I). The results of all 72 patients showed that preoperatively, no child had a scoliosis of more than 10°. The most common level of laminectomy was L1 (n = 51) followed by Th 12 (n = 16), L2 (n = 4), and L3 (n = 1). Figure 3 shows the distribution of age with regard to the level of laminectomy corresponding to the level of the conus medullaris. No persistent sensory alterations or urogenital dysfunctions were reported by the children or their caretakers at the 12-month follow-up. Applying the Kruskal-Wallis test, no significant association was found regarding the distribution of age and the level of laminectomy (p = 0.489).
The mean follow-up was 33 months (range 12–81 months). Postoperatively, 6 out of the 72 children developed a mild scoliosis, while one boy developed a thoracolumbar scoliosis of 37° between the 2nd and the 5th year of follow-up after successful laminoplasty. He showed a GMFCS II functional level and was 12.7 years old at the time of the diagnosis of his scoliosis. In total, the prevalence of scoliosis increased from 0 to 10 % while the age of the children increased from 7.2 to 10.0 years. No association was found between the development of scoliosis, the level of function, and age (p = 0.548, p = 0.217; Kruskal-Wallis test). Table 2 shows the distribution of age and the degree of scoliosis according to the functional levels. Furthermore, no association was found between the GMFCS categories and the Cobb angle categories (p = 0.101, Kendall’s tau). After an average of 33 months after SDR, the evaluation of the laminoplasty demonstrated that in 10 out of the 72 children (16 %), the spinous process of the reinserted lamina was no longer visible. In 62 children, the spinous processes remained vital and the mini-plates and screws in place during follow-up x-rays (Fig. 4). Out of the 7 children who developed a scoliosis, six had their laminoplasty at L1 and one at Th12. The reinserted lamina was described to be in place six out of the seven children with postoperative scoliosis. This child who is a functional level GMFCS III had its latest follow-up at 24 months and an L1 laminoplasty. One out of the 10 children in whose x-rays the lamina could not be identified at follow-up complained of back pain at the surgery site associated with athletic activity (rowing) at the 5-year follow-up. The pain resolved after a short period of rest without further therapy. Another one of these 10 children showed a mild scoliosis with a Cobb angle of 14°.
Discussion
SDR continued to become more popular during the last decades and evidence-based analysis supports the use of the procedure [26]. A recent study confirmed its value in a multimodal treatment concept. The reduction of spasticity through SDR achieves a superior improvement of gait when compared to osteotomies or soft-tissue surgery [27]. Having confirmed the success of the operation, the focus of many surgeons turned to minimizing the risk of the procedure, while the aim of the interdisciplinary team remains to improve the treatment modalities for each individual [28]. The importance of electrophysiological monitoring in both, improving the result and diminishing the risks, is supported by many authors but remains under discussion [10, 13, 29, 30].
Concerning the surgical access, the correlation of invasiveness and risk cannot be substantiated by the existing literature as the only direct comparison of outcomes after single-level and multilevel laminectomy did not focus on risk management [24]. Ou and colleagues reported a shorter in-patient stay, less need for opioid analgesics and a tendency toward earlier mobilization out of bed for the single-level group [24]. In addition to minimizing operative trauma, biomechanical reconstruction aims at the reduction of long-term adverse events. With regard to SDR, this effect has been studied for multisegmental procedures by Johnson and co-workers, who did not find significant differences between laminectomy and laminoplasty when focusing on spinal deformities [9]. On the other hand, back pain of musculoskeletal origin was reported to be increased after laminectomy only, although no mechanical instability or spinal deformity was observed [31]. Whether the complaint of back pain after rowing in one child of our cohort was associated with SDR remains unanswered. While no need for treatment of spinal deformities after single-level laminectomy was reported by Park and Johnston without specification of parameters describing deformities [6], groups performing multilevel procedures found incidences of scoliosis after SDR between 16 and 55 % [9, 11, 12, 19–21].
This study describes a modified technique of laminoplasty first used in selective dorsal rhizotomy which was developed to completely restore the biomechanical properties of the surgically manipulated motion segment, avoiding the irreversible interruption of the dorsal tension band. The limitations of this first evaluation lie in the restricted information provided by a single anterior-posterior total spine x-ray. In order to keep exposure to radiation as low as clinically necessary, no computer tomography was applied to further describe the osseointegration or resorption in this study setting. For the same reason, lateral spine x-rays were not included in the study protocol. We hence accepted the bias toward a negative outcome through counting each lamina as resorbed if any doubt arose during evaluation. A possible bias toward positive outcome might have originated from the event that the reinserted lamina did neither integrate nor resorb during the time of follow-up. Furthermore, the sparing use of radiography in our study protocol restricted us to a systematic evaluation of scoliosis without the possibility to analyze sagittal profile pathologies.
The development of scoliosis in 10 % of our patients after a monosegmental procedure with a complete restoration of the biomechanical properties of the segment appears to be rather high. Though, it corresponds to the natural history of scoliosis in spastic cerebral palsy [32], which occurs with a prevalence of 1.3 to 38 % in ambulatory children depending on the method of evaluation, starting to develop around the age of 8 years and rarely exceeding a Cobb angle of 40° [33–36]. It does not follow the typical time frame for the development and progression of idiopathic scoliosis, as it starts before the pubertal growth spurt and does not necessarily show the highest progression rates during adolescence. Although it is known that the risk for scoliosis in cerebral palsy (CP) increases with increasing functional impairment, the evidence concerning etiology and pathophysiology of the development of the deformity is sparse [34, 37]. Asymmetrical body positioning, weakness, trunk instability, and muscular dysbalance are held responsible for promoting scoliosis and pelvic obliquity in children and adolescents with CP [38]. The influence of SDR on these parameters has to be studied to differentiate between natural history and surgically induced deformity. An association between the development of scoliosis and the osseous reintegration of the reinserted lamina could not be derived from the data, reporting the coincidence of scoliosis and probably resorbed lamina in one child only.
In conclusion, the introduced monosegmental laminoplasty technique is the least destructive dorsal spinal midline approach that has been described to date. No substantial adverse effects have been observed. Our hypothesis, that any surgically induced risk of instability in ambulatory children with bilateral spastic CP can be avoided by this approach, is supported but not sufficiently proven by our preliminary results and needs further long-term investigation.
References
Young RR (1989) Treatment of spastic paresis. N Engl J Med 320:1553–1555
Foerster O (1908) Über eine neue operative Methode der Behandlung spastischer Lähmungen mittels Resektion hinterer Rückenmarkswurzeln. Z Orthop Chir 22:203–223
Gros C, Ouaknine G, Vlahovitch B, Frerebeau P (1967) La radicotomie selective posterieure dans le traitment neurochirurgical de I’ hypertonie pyramidale. Neurochirurgie 13:505–518
Fasano VA, Broggi G, Barolat-Romana G, Sguazzi A (1978) A surgical treatment of spasticity in cerebral palsy. Child Brain 4:289–305
Peacock WJ, Arens LJ (1982) Selective posterior rhizotomy for the relief of spasticity in cerebral palsy. S Afr Med J 62:119–124
Park TS, Johnston JM (2006) Surgical techniques of selective dorsal rhizotomy for spastic cerebral palsy. Technical note. Neurosurg Focus 21:e7
Cole GF, Farmer SE, Roberts A, Stewart C, Patrick JH (2007) Selective dorsal rhizotomy for children with cerebral palsy: the Oswestry experience. Arch Dis Child 92:781–785
Farmer JP, Sabbagh AJ (2007) Selective dorsal rhizotomies in the treatment of spasticity related to cerebral palsy. Childs Nerv Syst 23:991–1002
Johnson MB, Goldstein L, Thomas SS, Piatt J, Aiona M, Sussman M (2004) Spinal deformity after selective dorsal rhizotomy in ambulatory patients with cerebral palsy. J Pediatr Orthop 24:529–536
Mittal S, Farmer JP, Poulin C, Silver K (2001) Reliability of intraoperative electrophysiological monitoring in selective posterior rhizotomy. J Neurosurg 95:67–75
Spiegel DA, Loder RT, Alley KA, Rowley S, Gutknecht S, Smith-Wright DL, Dunn ME (2004) Spinal deformity following selective dorsal rhizotomy. J Pediatr Orthop 24:30–36
Steinbok P, Hicdonmez T, Sawatzky B, Beauchamp R, Wickenheiser D (2005) Spinal deformities after selective dorsal rhizotomy for spastic cerebral palsy. J Neurosurg 102:363–373
Staudt LA, Nuwer MR, Peacock WJ (1995) Intraoperative monitoring during selective posterior rhizotomy: technique and patient outcome. Electroencephalogr Clin Neurophysiol 97:296–309
Phillips LH, Park TS (1989) Electrophysiologic studies of selective posterior rhizotomy patients. In: Park TS, Phillips LH, Peacock WJ (eds) Neurosurgery: state of the Art reviews: management of spasticity in cerebral palsy and spinal cord injury. Hanley & Belfus, Philadelphia, pp 459–469
Dudley RW, Parolin M, Gagnon B, Saluja R, Yap R, Montpetit K, Ruck J, Poulin C, Cantin MA, Benaroch TE, Farmer JP (2013) Long-term functional benefits of selective dorsal rhizotomy for spastic cerebral palsy. J Neurosurg Pediatr 12:142–150
Engsberg JR, Ross SA, Collins DR, Park TS (2006) Effect of selective dorsal rhizotomy in the treatment of children with cerebral palsy. J Neurosurg 105:8–15
McLaughlin J, Bjornson K, Temkin N, Steinbok P, Wright V, Reiner A, Roberts T, Drake J, O’Donnell M, Rosenbaum P, Barber J, Ferrel A (2002) Selective dorsal rhizotomy: meta-analysis of three randomized controlled trials. Dev Med Child Neurol 44:17–25
van Schie PE, Schothorst M, Dallmeijer AJ, Vermeulen RJ, van Ouwerkerk WJ, Strijers RL, Becher JG (2011) Short- and long-term effects of selective dorsal rhizotomy on gross motor function in ambulatory children with spastic diplegia. J Neurosurg Pediatr 7:557–562
Golan JD, Hall JA, O’Gorman G, Poulin C, Benaroch TE, Cantin MA, Farmer JP (2007) Spinal deformities following selective dorsal rhizotomy. J Neurosurg 106:441–449
Peter JC, Hoffman EB, Arens LJ, Peacock WJ (1990) Incidence of spinal deformity in children after multiple level laminectomy for selective posterior rhizotomy. Childs Nerv Syst 6:30–32
Turi M, Kalen V (2000) The risk of spinal deformity after selective dorsal rhizotomy. J Pediatr Orthop 20:104–107
Papagelopoulos PJ, Peterson HA, Ebersold MJ, Emmanuel PR, Choudhury SN, Quast LM (1997) Spinal column deformity and instability after lumbar or thoracolumbar laminectomy for intraspinal tumors in children and young adults. Spine (Phila Pa 1976) 22:442–451
Yasuoka S, Peterson HA, MacCarty CS (1982) Incidence of spinal column deformity after multilevel laminectomy in children and adults. J Neurosurg 57:441–445
Ou C, Kent S, Miller S, Steinbok P (2010) Selective dorsal rhizotomy in children: comparison of outcomes after single-level versus multi-level laminectomy technique. Can J Neurosci Nurs 32:17–24
Palisano R, Rosenbaum P, Walter S, Russell D, Wood E, Galuppi B (1997) Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev Med Child Neurol 39:214–223
Novak I, McIntyre S, Morgan C, Campbell L, Dark L, Morton N, Stumbles E, Wilson SA, Goldsmith S (2013) A systematic review of interventions for children with cerebral palsy: state of the evidence. Dev Med Child Neurol 55:885–910
Feger MA, Lunsford CD, Sauer LD, Novicoff W, Abel MF (2015) Comparative effects of multilevel muscle tendon surgery, osteotomies, and dorsal rhizotomy on functional and gait outcome measures for children with cerebral palsy. PM R 7:485–493
Grunt S, Fieggen AG, Vermeulen RJ, Becher JG, Langerak NG (2014) Selection criteria for selective dorsal rhizotomy in children with spastic cerebral palsy: a systematic review of the literature. Dev Med Child Neurol 56:302–312
Huang JC, Deletis V, Vodusek DB, Abbott R (1997) Preservation of pudendal afferents in sacral rhizotomies. Neurosurgery 41:411–415
Lang FF, Deletis V, Cohen HW, Velasquez L, Abbott R (1994) Inclusion of the S2 dorsal rootlets in functional posterior rhizotomy for spasticity in children with cerebral palsy. Neurosurgery 34:847–853
Cobb MA, Boop FA (1994) Replacement laminoplasty in selective dorsal rhizotomy: possible protection against the development of musculoskeletal pain. Pediatr Neurosurg 21:237–242
Grunt S, Becher JG, Vermeulen RJ (2011) Long-term outcome and adverse effects of selective dorsal rhizotomy in children with cerebral palsy: a systematic review. Dev Med Child Neurol 53:490–498
Balmer GA, MacEwen GD (1970) The incidence and treatment of scoliosis in cerebral palsy. J Bone Joint Surg (Br) 52B:134–137
Persson-Bunke M, Hägglund G, Lauge-Pedersen H, Wagner P, Westbom L (2012) Scoliosis in a total population of children with cerebral palsy. Spine (Phila Pa 1976) 37:E708–E713
Robson P (1968) Prevalence of scoliosis in adolescents and young adults with cerebral palsy. Dev Med Child Neurol 10:447–452
Rosenthal RK, Levine DB, McCarver CL (1974) Occurrence of scoliosis in cerebral palsy. Dev Med Child Neurol 16:664–667
Galasko CS (1997) Progression of scoliosis. J Pediatr Orthop 17:407
Loeters MJ, Maathuis CG, Hadders-Algra M (2010) Risk factors for emergence and progression of scoliosis in children with severe cerebral palsy: a systematic review. Dev Med Child Neurol 52:605–611
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received with regard to this study.
Conflict of interest
The authors declare that they have no conflict of interest with regard to this study.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from the parents or the legal guardians of the participants included in this study and if possible with regard to age and mental ability from the participants themselves.
Rights and permissions
About this article
Cite this article
Funk, J.F., Haberl, H. Monosegmental laminoplasty for selective dorsal rhizotomy—operative technique and influence on the development of scoliosis in ambulatory children with cerebral palsy. Childs Nerv Syst 32, 819–825 (2016). https://doi.org/10.1007/s00381-016-3016-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3016-3