Abstract
Rationale
Selective dorsal rhizotomy (SDR) is a surgical technique developed over the past decades to manage patients diagnosed with cerebral palsy suffering from spastic diplegia. It involves selectively lesioning sensory rootlets in an effort to maintain a balance between elimination of spasticity and preservation of function. Several recent long-term outcome studies have been published. In addition, shorter follow-up randomized controlled studies have compared the outcome of patients having undergone physiotherapy alone with those that received physiotherapy after selective dorsal rhizotomy.
Materials and methods
In this account, we will discuss the rationale and outcome after SDR. The outcome is addressed in terms of the gross motor function measurement scale (GMFM), degree of elimination of spasticity, strength enhancement, range of motion, fine motor skills, activity of daily living, spastic hip, necessity for postoperative orthopedic procedures, bladder and sphincteric function, and finally possible early or late complications associated with the procedure.
Conclusion
We conclude that SDR is a safe procedure, which offers durable and significant functional gains to properly selected children with spasticity related to cerebral palsy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Cerebral palsy
Cerebral palsy comprises a number of clinical syndromes associated with abnormalities of posture and movement resulting from injury to the immature brain. The neurological insult is not evolutive, and the injury to the central nervous system is static, but the clinical expression of cerebral palsy is subject to change as children and their developing nervous system mature. Characteristically, children suffer from a variable combination of spasticity, disorders of movement, hypotonia, ataxia, and/or rigidity. Paradoxically, despite progress in neonatal care, the incidence of cerebral palsy is increasing, affecting an estimated 500,000 children and adults in the USA with life-long disabilities [44, 62]. This increase is likely the result of increased survival of very-low-birth-weight infants as well as improved national registries [78].
The etiology of cerebral palsy has been traditionally associated to prenatal, perinatal, or postnatal events. The latter could go up to 3 years postnatally. Typical prenatal causes include supranumerary gestations, infection, antepartum vaginal bleeding, and maternal exposure to alcohol. Common perinatal factors include fetal anoxia, bradycardia, hemorrhage, and infection; all of which are usually more common in the premature and low-birth-weight infants. Early childhood factors include traumatic brain injury, near-drowning, as well as meningo-encephalitis. Prenatal factors are thought to be primarily responsible in 70–80% of cases [28].
In the immature brain, cerebral hypoperfusion tends to result in two well-known clinical outcomes: firstly, periventricular leukomalacia, which is the most important determinant of neurological compromise seen in the very-low-birth-weight infants [22] (<1,500 g), and secondly, the development of germinal matrix hemorrhages, which often will lead to asymmetric deficits. When the basal ganglia are affected, an extra pyramidal presentation with prominent dystonia and choreoathetosis is seen.
From weeks 26 to 34 of gestation, periventricular white matter areas near the lateral ventricles are more susceptible to hypoxic ischemic injury. It is not surprising that leg fibers are predominantly affected in these children who also demonstrate enlargement of the body of the lateral ventricles on imaging. Larger lesions will lead to quadraparesis with or without bulbar signs.
The extent and distribution of motor syndromes after the development of cerebral palsy is therefore variable. Five subtypes of presentations have been identified with a significant degree of overlap in any individual child being assessed [63]. Approximately 60% of children will exhibit predominance of spasticity either in the form of diplegia or asymmetric quadraparesis. This is the presentation that is most amenable to medical and surgical therapeutic measures. Twenty percent of children show a predominant athetosis and dystonia picture. There can also be chorea and hemiballistic movements in the presentation. In 10% of children, the predominant presentation is ataxia. Unless underlying hydrocephalus is contributing, very few therapeutic medical or surgical options are available to these children. In the remaining 10% of the cases, the presentation is more that of hemiplegia associated or not with a migration disorder, depending on the timing of the injury during gestation. Finally, it is important, as mentioned previously, to realize that most children have more than one form of cerebral palsy and therefore have a mixed presentation.
Treatment options
At present, there is no clinically meaningful intervention that will successfully repair existing damage to the brain areas that control muscle coordination and movement. Several interventions are available, however, to decrease the severity of impact of the deficits on activities of daily living and to improve overall functional performance of these children. These include physical and occupational therapy, oral medication, botulinum toxin, intrathecal Baclofen therapy, orthopedic surgery, and selective dorsal rhizotomies. Appropriate use of these therapeutic interventions is essential given that 95% of children with diplegia survive until the age of 30 years [20] and that the overall survival of all children with cerebral palsy until the age of 20 years is established at 90% [26, 42]. Surgical interventions remain in the realm of functional surgery. Therefore, a major emphasis is placed on quality-of-life issues. As such, the treatment is best optimized using a modern multidisciplinary approach.
Selective dorsal rhizotomies have revolutionized the management of spasticity related to cerebral palsy and the impact of the procedure on functional outcome of these children will be the subject of the remainder of this article.
Selective dorsal rhizotomies
Historical background
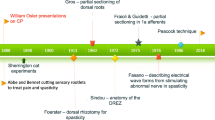
The impact of rhizotomies on tone has been known to the scientific community since Sherington’s experiments on the decerebrate cats. In humans, Abbe and Bennett in 1889 [1, 9] described the relief of pain by nerve root section. Foerster in 1913 [32] specifically described relief of spasticity after dorsal rhizotomy. Approximately one half of his patients had congenital spasticity. Foerster also was the first to propose intraoperative electrical stimulation to identify levels and to distinguish ventral from dorsal roots. Nevertheless, because of comorbidity associated with a rather radical procedure at the time, for approximately half a century, the procedure was largely abandoned. Gros et al. in the mid-1960s [35] contributed significantly to the evolution of the procedure by distinguishing beneficial spasticity from detrimental spasticity and performing partial rhizotomies. They also introduced electromyography (EMG) monitoring to permit identification of rootlets innervating more dysfunctional muscle groups. In the mid-1070s, Fasano et al. [29] developed a series of criteria based on abnormality of evoked motor responses to electrical stimulation, and finally, in the 1980s, Peacock and Arens proposed moving the operative site to the lumbo-sacral canal to allow more secure identification of the segmental level. Most centers continue to employ variations of the techniques originally described by Fasano et al. and Peacock et al. [29, 72]. Currently, the operation is carried either at the lumbo-sacral level or in certain centers at the level of the conus [70].
The field has evolved with respect to the degree of root lesioning required so as to maintain a balance between elimination of spasticity and preservation of strength. Currently, criteria relying on spread of the response and on incremental or multi-phasic patterns of the response to a 50-Hz train are used in most centers to optimize the balance between spasticity reduction and preservation of function. The ability of EMG-guided dorsal rhizotomies to reliably define which rootlets should be sectioned has become a matter of controversy. Contributing to this debate has been the lack of technical standardization [50, 51, 87], the variability of motor response after repeated nerve root stimulation [100, 101], the effect of anesthetic agents on intraoperative stimulation [17, 36], the natural variability known to exist in segmental innervation of the lower extremity muscles [76], and finally, the anecdotal reports of sustained contractions after stimulation in patients operated upon for reasons other than spasticity [19, 88, 92].
We value spread of the muscle response to the contralateral limb or upper extremity to a 50-Hz stimulation as a valid criterion to define rootlets contributing to disinhibited spinal circuitry in uncontrolled spasticity. Sustained contractions after the 50-Hz stimulation have been demonstrated in nonspastic patients [19, 88], but the pattern of spread contralaterally and suprasegmentally has not.
Using these criteria of spread, we demonstrated a definite statistical reliability in intraoperative stimulation to 50-Hz train of dorsal roots [60]. In our report, an absolute grade variation of 0 or 1 occurred in 93% of 752 roots, which were stimulated twice. Additionally, the correlation between the EMG determination of the pattern of spread and the physiotherapy determination correlated at greater than 90%. Therefore, we strongly advocate the routine use of intraoperative electrophysiological monitoring to identify those rootlets contributing significantly to the spastic process. Stimulation helps us attain a balance between significant reduction in spasticity and preservation of sensation, voluntary muscle control, and bladder function. In functional surgery, these guiding principles must prevail (Fig. 1).
Intraoperative photograph showing the cauda equina during a case of selective dorsal rhizotomy done via a narrow lumbosacral (L1–S2) laminotomy/laminoplasty. On the left hand side, a sacral root is being divided into an anterior motor component and a posterior sensory one. On the right hand side, the Sami knife and probe are holding the right fourth lumbar (L4) sensory root about to be separated into equal smaller rootlets as the other roots seen already separated in the background in preparation for stimulation followed by selective lesioning
Long-term outcome
Several groups have reported on the outcome of children undergoing selective dorsal rhizotomies with a variable follow-up. Over the years, several reliable quantitative outcome measures have been defined. These have been helpful in assessing lower extremity function, alignment, transition, upper extremity function, activities of daily living, bladder function, hip stability, complication rates, and the rate of postoperative orthopedic procedures.
Gross motor function measurement scale
The gross motor function measurement scale (GMFM) is a criterion referenced observed measure that was developed and validated to assess children with cerebral palsy [79]. It contains 88 items that are scored on a four-point ordinal scale. Five dimensions are assessed. Dimension A contains 17 items that are performed in lying or rolling position. Dimension B contains 20 items observed in the sitting position. Dimension C assesses crawling and kneeling with 14 items. Dimension D assesses standing ability with 13 items, and finally, dimension E assesses walking, running, and jumping with 24 items. The total score is obtained by averaging the percent of scores across the five dimensions. As indicated by Russell et al. [79], clinical gains of 6% either in the total score or in one dimension are felt to be clinically significant in children having undergone a surgical intervention. Three randomized trials comparing physiotherapy alone and selective dorsal rhizotomies followed by physiotherapy were published in 1997 and 1998 [54, 90, 103]. The primary outcome measure was the GMFM score. Two of the three trials reported the improvement in GMFM scores after rhizotomies to be statistically more significant than in the control group receiving physiotherapy alone, whereas the third did not. A recent meta-analysis [53] of controlled trials revealed a statistically significant improvement in the patients undergoing rhizotomy plus physiotherapy compared to those having received physiotherapy alone [11, 53]. A recent study [25] comparing a nondisabled group of children to a group undergoing physiotherapy alone and a group undergoing rhizotomy followed by intensive physical therapy concluded that children with CP are weaker than individuals in the nondisabled group regardless of data collection type or intervention. However, the group having undergone selective dorsal rhizotomy obtained significantly greater improvement in gross motor function than the group undergoing physiotherapy alone.
Unfortunately, follow-up data from the randomized controlled trials and that of other reports does not extend beyond 2 years. Our group has been able to report a statistically significant clinical improvement in GMFM total scores at 1, 3, and 5 years postsurgery [57] (Fig. 2). Improvement continued to occur at 5 years, although the most important gains were noted 1 year postoperatively. Furthermore, the results were most dramatic in the two dimensions involving lower limb function that are dimensions D and E, which are the dimensions of importance in patients trying to attain ambulatory skills of better quality.
Line plots comparing GMFM total scores (left), dimension D (center), and dimension E (right) between the patients in the study according to the preoperative level of functioning. Patients were subdivided into five groups according to their preoperative locomotive abilities. Dimensions D and E primarily relate to lower-extremity motor function and are the primary outcome measures of the GMFM (adopted with permission, Mittal et al. [57])
In our series, children with mild to moderate degrees of ambulatory dysfunction that is with either ambulatory capacity, ambulatory capacity using assistive devices, or emerging locomotor function (ambulatory potential) represented nearly 95% of the study population. The better the level of preoperative motor function was, the greatest the gains were postoperatively (see Fig. 2). Paradoxically, these patients are often not referred or referred later because of the misconception that their potential for significant gains is low. As an example, in the group of independent ambulators, the average preoperative GMFM score was established at 84%, but gains in dimension D and E at 5 years after the procedure were 13 and 26%, respectively. These children reach near-normal levels of function to the greatest satisfaction of their families. The small group of more severely disabled nonambulatory patients was unlikely to have worthwhile improvement in motor function despite adequate elimination of lower extremity spasticity. This group of patients was too small to be analyzed statistically.
Elimination of spasticity
While elimination of spasticity is a means to an end given that it does not guarantee improvement in function, this remains a crude way of assessing outcome. Several groups [3, 14, 24, 34, 39, 46, 47, 54, 55, 57, 80, 85, 87, 89, 90, 103] have shown significant reduction in spasticity after selective dorsal rhizotomy. However, very few groups reported quantitative results at 5 years after rhizotomy [37, 57]. Our study at 1, 3, and 5 years demonstrated a long-term beneficial effect of elimination of spasticity after rhizotomy. The results were statistically significant in all lower extremity muscle groups tested and remained so over the 5-year study period.
Strength enhancement
In the previous section, we demonstrated the preservation of the reduction in tone over several years after rhizotomies. This allows for unmasking of potential strength in the affected muscles, which should, with proper therapy, replace the spasticity as the primary parameter of gait. Several studies have reported gains in lower limb strength [24, 37, 47, 57, 89, 90]. However, only two of these studies [37, 57] were able to establish gains in strength after the initial first year.
Strength is a difficult parameter to evaluate. Our group, using the Rusk Institute of Rehabilitation/New York University rhizotomy evaluation form, [3] looked at postural stability, transition from one position to another, and quality of alignment as parameters of motor function over 5 years postoperatively comparing the scores to the preoperative values. We were able to show clear gains in alignment scores, transition scores, and stability scores for these patients. The major gains occurred in the first postoperative year, but ongoing gains were clearly detectable up to 5 years post-treatment. Of note, because of the high functional level of most of the children involved in our studies, the most important improvements occurred in alignment scores given that the children were able to perform quite well in the sitting position and perform transition movements independently before rhizotomy.
Range of motion
Gait is a dynamic process. Range of motion is therefore considered an important criterion with respect to outcome measures. Several studies [3, 13, 14, 34, 37, 57, 64, 84, 85, 89] and in particular the two randomized controlled studies [90, 103] used goniometry as a study tool. These studies indicated a greater range of motion in the lower extremity joints in patients tested early, that is, between 9 months and 2 years after rhizotomy. Only few studies [37, 57] including ours showed significant improvement up to 5 years after rhizotomies. Using gait analysis, several studies also showed improved stride length and velocity in ambulatory patients [4, 13, 16, 74, 80, 92, 93, 96, 98, 102, 103].
In our study, the greatest improvements in range of motion occurred between the preoperative status and the first year postoperatively, although significant gains were still felt to occur between years 1 and 5 postoperatively particularly with respect to knee extension. On the other hand, the range of motion for ankle dorsiflexion and hip abduction improved to a lesser degree between years 1 and 5 postoperatively.
Fine motor skills
Whereas the expected primary impact of selective dorsal rhizotomies is on gait, several anecdotal reports of improvements in upper extremity function after rhizotomies have appeared both in the orthopedic and functional literatures. This question is particularly important because the advent of intrathecal Baclofen therapy as an alternative therapeutic option. Given Baclofen’s ability to diffuse throughout the intrathecal space, it would stand to reason to believe that it might have a more beneficial impact on upper extremity function than selective dorsal rhizotomies performed at the lumbo-sacral level. This theoretical consideration might have an impact on the choice of treatment modality selected for the patient if one of the established goals is to obtain gains in the upper extremities.
Several studies have reported reduction in spasticity of the upper extremities after selective dorsal rhizotomies [19, 47, 55, 73]. Whether this translated into better upper extremity function remained to be clarified. Seven studies [5, 6, 19, 30, 82, 85, 89] reported qualitative improvements in upper limb function. Furthermore, six studies reported quantitative improvements such as improved block stacking and manipulation patterns in the upper extremities [8, 14, 23, 43, 48, 59]. Two recent studies have looked at beneficial effects on upper extremity skills using quantitative validated tools [16, 49]. However, all of these studies had a very short follow-up of approximately 1 year.
We chose to focus on fine motor control and analyzed upper extremity skills from a developmental and functional perspective using a standardized validated test, the Peabody developmental motor scale (PDMS), to determine if selective posterior rhizotomy resulted in functional upper extremity gains. Folio and Fewell [33] designed the PDMS for use as both a discriminative and an evaluative measure of upper extremity function. The test provides a comprehensive assessment of a child’s gross and fine motor patterns and skills in relation to adaptive capacity. The large number of items tested require a child to use his/her motor capacity to adapt to specific situations. In this manner, the child’s strength and weaknesses can be analyzed accurately and thoroughly. Our report demonstrated clearly that the PDMS was capable of identifying longitudinal changes, up to 5 years postrhizotomies, in fine motor function in children with cerebral palsy. The two domains where most significant gains occur namely, eye/hand coordination and manual dexterity, are skills that have a major impact on the quality of life of affected children. Using percentile scores and Z scores to take into account maturation in analyzing progress, rhizotomies were shown to allow children to reach low normal levels of function in upper extremity, which even the higher functioning groups of children with cerebral palsy did not attain preoperatively. Furthermore, children with milder motor deficits, that is dependent or independent ambulators, were more likely to improve fine motor skills at 3 and 5 years after surgery than those who were unable to walk preoperatively. Therefore, children who have some baseline ambulatory function may potentially benefit the most from posterior rhizotomies, whereas the more severely disabled children who have no ambulatory potential are unlikely to have a worthwhile improvement in functional performance whether it be at the upper extremity or lower extremity level despite adequate elimination of spasticity.
Our results explicitly indicate that one should not favor Baclofen pump implantation over selective posterior rhizotomy because of a theoretical misconception that dorsal rhizotomy only addresses lower extremity impairments. Clearly, higher-functioning children can obtain excellent results in upper extremity fine motor control after rhizotomy, which remains the therapeutic gold standard for children who meet rigid preoperative criteria predictive of ambulatory gains. On the other hand, upper extremity spasticity and blunted fine motor skills seen in quadriplegic children may be addressed more readily with intrathecal Baclofen by placement of a catheter higher along the spinal cord [27, 65] when rhizotomy criteria are not met.
A comparative study between selective posterior rhizotomy and intrathecal Baclofen looking specifically at upper extremity gains would be interesting. However, such a study might be difficult to justify ethically given the growing body of literature demonstrating that the two operations are best suited for distinct subgroups of children with spastic cerebral palsy [99].
Activities of daily living
The ultimate goal of reduction in spasticity, improvement in joint range of motion, and increased motor strength after selective dorsal rhizotomy is the improvement in activities of daily living such as oral/facial hygiene, grooming, and feeding with emphasis on upper and lower extremity dressing and toileting. Assessments of trunk control and mobility issues such as independent sitting and moving in bed, transfers onto chairs, toilets, bathtubs, or cars, as well as indoor and outdoor walking skills are also part of activities of daily living. The sum of every small improvement in different spheres of activities of daily living is what determines the level of improved patient independence.
Several investigators, using nonvalidated outcome assessment tools, have reported improvements in activities of daily living [6, 8, 10, 37, 39, 46, 52, 54, 61, 64, 85, 89, 90, 104]. The quantitative validated tool most widely used to assess activities of daily living in children is the pediatric evaluation of disability inventory (PEDI). Four studies have used the PEDI in a prospective fashion and reported statistically significant improvements in self-care and mobility domains after dorsal rhizotomies [12, 23, 58, 65]. Only one of these studies had follow-up data beyond 2 years after rhizotomies. The result of our own study using the PEDI score reveals important gains in functional performance as early as 6 months after dorsal rhizotomies with continued gains occurring up to 5 years after rhizotomies. As expected, higher-functioning children with independent or dependent ambulatory potential were more likely to improve functional performances at 3 and 5 years after rhizotomies than those who were unable to walk preoperatively. Therefore, nonambulatory patients are likely to have more modest improvements in activities of daily living despite adequate elimination of lower extremity spasticity after rhizotomies. The modest gains are likely to be equally achievable in these more involved children with a less invasive and/or reversible treatment such as intrathecal Baclofen.
The spastic hip
Analyzing 88 hips both preoperatively and 1 year postoperatively (unpublished data), we have noted stabilization of progressive hip deformity after selective posterior rhizotomies as long as the Reimer Index of femoral uncovering does not exceed 50%. In fact, 95% of the hips studied appeared to stabilize or improve as long as the index was less than 50% before the rhizotomies, and other groups have reported similar results [25, 38, 71]. Changes in hip spasticity and strength postrhizotomies have been implicated in this stabilization [25].
Therefore, our practice is to allow the child to undergo rhizotomy if all selection criteria are met and the Reimer Index does not exceed 50%. If the Reimer Index does exceed 50%, then an orthopedic hip surgery may be needed to precede the rhizotomy by approximately 6–12 months.
Adjunctive orthopedic procedures
In several centers, a competition exists between the orthopedic management of spasticity and selective dorsal rhizotomies. Selective dorsal rhizotomies have undergone extensive investigations as a treatment option with most studies concluding of the long-term beneficial effects as described above. In fact, selective dorsal rhizotomies have emerged as the only treatment of spastic cerebral palsy meeting the rigid criterion of class I evidence in terms of reduction in spasticity when combined with intensive physiotherapy [25, 66].
Most centers who have developed a multidisciplinary approach to the treatment of spasticity whereby collaboration exists between orthopedics and neurosurgery have adopted the rational strategy of treating the cause (that is, the spasticity) rather than treating the consequences of soft tissue and joint conditions as an initial step. One study [15] in 2004 compared the outcome after selective dorsal rhizotomies vs orthopedic procedures. Both approaches lead to multidimensional benefits for ambulatory children with spastic diplegia, but clearly, qualitative changes in movement, self-care, and social function gains were greater in the selective dorsal rhizotomy group than the orthopedic surgery group. Finally, an immuno-histochemical study looking at the “nerve cell adhesion molecule” has concluded that this molecule is upregulated in response to denervation and is present in larger quantities in muscle biopsy specimens obtained from patients with cerebral palsy. This could therefore suggest that early intervention might have beneficial effects that would prevent the need for later treatment of soft tissue or joint deformities [51]. Through four different publications, the St. Louis group [18, 66–68] has shown that the frequency of orthopedic surgeries after selective dorsal rhizotomies is not as high as was initially reported by Subramanian et al. [93]. When operated at a young age, (between 2 and 5 years of age), children with spastic diplegia and in particular independent walkers appeared to have a significantly lower rate of subsequent orthopedic surgery requirements than patients who are either operated at a later age, need assistance, or have a quadraparetic pattern of spasticity. The greatest reductions occur in soft tissue releases, whereas surgeries for joint deformities tend to be required more consistently in both groups.
At our Center, we strongly believe that neurosurgical and orthopedic treatment options for spastic cerebral palsy are complementary rather than mutually exclusive. Our results indicate that long-term functional gains are statistically and clinically significant and durable in all functional measures and that this occurs by the 1-year postrhizotomy assessment. This is before any subsequent orthopedic intervention [57]. Furthermore, at 3 years postrhizotomy, only 8.5% of our patients had undergone orthopedic procedures, and only 12.7% had received lower extremity botulinum toxin injections. Even when these patients having received orthopedic interventions were excluded from the analyses, gains in dimensions D and E of the GMFM scores remained statistically very significant up to 5 years postoperatively. There is no doubt that orthopedic interventions can procure further gains to the children, but the bulk of the gains likely have occurred independent of adjunctive treatments. Like other groups, we find orthopedic procedures to correct joint deformities prevailing over soft tissue lengthening particularly with the adjuvant use of Botox. Botox injected in the postoperative phase seems to have a more profound and durable effect compared to injections done preoperatively. It seems clear therefore that the best outcomes occur in young spastic diplegic patients having undergone rhizotomies. In that group of patients, orthopedic procedures may be required in the long term at the frequency of 15–20% of patients [66]. The liberal use of Botox, which seems to have a synergistic effect with the rhizotomies when used postoperatively, serves to further improve the esthetics of the gait pattern. Older and more involved children, whether treated with rhizotomies or with Baclofen pump implantation, are likely to demonstrate a higher need for adjunctive orthopedic procedures.
Bladder and sphincteric function
The bladder of children with spastic cerebral palsy is likely to demonstrate a spastic profile given the loss of inhibitory cortical innervation seen under this condition. In a study looking at bladder capacity and compliance through urodynamic evaluation [40], we demonstrated that preoperatively, a large number of patients, even if they have achieved continence, have markedly elevated intravesical pressures. The range of pressures in this cohort of patients was determined to be at a level that would cause chronic upper urinary tract and renal disease in the long run.
Those performing rhizotomies at the upper sacral level for the relief of ankle spasticity have been torn between the necessity for relief of spasticity to improve motor function and the risk of deterioration in the bladder profile as a result of excessive lesioning. To that effect, several groups have tried to better identify pudendal afferents within several roots [21, 38, 40, 41, 45, 71, 83, 95]. Our additional report of elevated bladder pressures despite achievement of urinary continence in these children pre-operatively further complicated decision-making at that particular level during a selective posterior rhizotomy procedure. Not only would the surgeon want to preserve bladder function, but he/she would optimally like to reduce the spasticity of the bladder to avoid chronic urinary problems.
Clear identification of S2, S3, and S4 roots by counting from the “cul de sac” up and by using electrophysiological parameters comparing S2 and S1 motor responses coupled to limited lesioning of up to two thirds of S2 on either sides, but more frequently to a total of 50% of S2 rootlets bilaterally as a strategy has met with success in our group. All patients who were toilet trained before the procedure remained so, whereas those who were not achieved continence over time postoperatively. Furthermore, detailed urodynamic studies done pre- and postoperatively demonstrated improved bladder capacity and decreased bladder compliance with a secondary (bonus) protective effect on upper renal tracts in our patients to the level of “minimally acceptable” values for age. It therefore seems clear that preservation of one third to one half of dorsal rootlets at S2 and 100% preservation of clearly identified S3 and S4 roots optimizes the balance between improved ankle function and improved bladder parameters without any incidence on worsening of bladder function.
With the introduction of epidural morphine analgesia postoperatively, after having performed 40 selective dorsal rhizotomies without any urinary retention using intravenous postoperative analgesia, we noticed an increased rate of transient urinary retention. Removing the epidural catheter at postoperative day 3 and the urinary catheter 24 h later to let the effect of epidural morphine wear off has corrected this problem.
Overall, with approximately 250 interventions, we have had no permanent urinary deterioration using these two strategies.
Complications
This operation for a major intervention is qualified as quite safe and associated with relatively few complications. However, if complications occur, they will occur in the intraoperative, early postoperative, and late postoperative periods.
Intraoperative complications
In the late 1980s and early 1990s, significant concern was raised with respect to intraoperative bronchospasm [2, 7, 94] and the development of immediate postoperative aspiration pneumonias as a result of pre-existing respiratory distress syndrome and gastro-esophageal reflux. The “light” anesthetic conditions necessary for monitoring were thought to contribute significantly to this problem. The premorbid state, particularly in more involved children with lower functional status was also thought to play an important role.
Anesthetic protective measures such as perioperative broncho-dilators and H2 blockers were suggested by some to reduce the risk of aspiration pneumonia and bronchospasm [2, 7, 91, 97]. In the last 10–15 years with the advent of propofol anesthesia and the selection of dorsal rhizotomies for higher-functioning children, these intraoperative problems have become very rare.
Early postoperative complications
Although several potential complications exist with this operation in the early postoperative phase, they tend to be quite rare. Cerebrospinal fluid leak and infection have been reported. Sensory deficits [31, 75, 91] have also been reported in the 1980s likely as a result of the extensive lesioning rates (of the order of 70–80%), which took place at that time. Currently, at our center, the lesioning rate is of the order of 45–55% and never exceeds 60%. This is generally the lesioning rate reported by most groups currently [69, 85]. At that lesioning rate and by taking particular attention never to section an entire dorsal root, sensory deficits are rare, and most cooperative patients are able to perform a normal or near-normal sensory examination when tested formally several years later.
The incidence of bladder dysfunction has been reported previously [56, 77] and can be significantly reduced or eliminated by the use of careful lesioning guidelines in the upper sacral level [40].
As mentioned previously, transient urinary retention likely related to epidural morphine administration can also be reduced to a minimum by leaving the Foley catheter 24 h beyond the cessation of epidural morphine administration.
Most children experience early transient weakness [74, 75] related to a change in tone particularly at the trunk and the lower extremity levels. Similarly, a number of children (approximately 30% in our experience) have symptomatology reminiscent of restless leg syndrome, which responds well to bedtime Diazepam and never exceeds a period of 3 weeks postoperatively.
Late postoperative complications
There is a high rate of spinal deformities inherent to the natural history of cerebral palsy. Saito et al. [81] studied institutionalized patients with severe spastic cerebral palsy and determined that most scoliosis occurred before the age of 10, advanced most rapidly after onset, and progressed continuously up to and beyond age 20. Madigan and Wallace studying institutionalized patients found a scoliosis rate of greater than 10° in 44% of independent ambulators, 54% of dependent ambulators, and 76% of bedridden patients. Other important deformities are those of hyperlordosis and those of thoracolumbar kyphosis where the natural history would suggest progressive deformity as well. Finally, pelvic obliquity as a result of hip flexion contractures has been implicated in the increased incidence of spondylolisthesis in cerebral palsy patients.
Given the longevity of the follow-ups now available and given the fact that most early patients are now adults, several groups have paid attention to the incidence of spinal deformities in operated patients to try to determine whether the intervention influenced positively or negatively the natural history of the disease. In 2005, Steinbok et al. [86] reported nearly 55% of 104 patients having scoliosis defined as greater than 10° of coronal curvature. The incidence of kyphosis was established at 39% and the incidence of hyperlordosis at 21%. Two patients required spinal stabilization procedures.
We studied a group of 101 patients with a mean follow-up of nearly 6 years looking at coronal alignment as well as sagittal alignment at the thoracic, upper thoracolumbar, lower thoracolumbar, and lumbar levels. We also assessed the rate of spondylolisthesis and spondylolysis. Scoliosis was defined as a coronal angle of greater than 10°. All films were obtained in the weight-bearing position, which was not the case in other similar studies. We postulated that weight bearing might increase the magnitude of the curve changes reported.
At the level of the coronal alignment, our rate of curves greater than 10° defined as scoliosis was 45% but significantly higher in dependent ambulators than in independent ambulators. Nevertheless, only two patients had curves above 20°, and no patients required any bracing or fusion. The main features of our analysis at the level of the sagittal alignment was that patients who had surgery at an age greater than 5 years had more significant lumbar lordosis than those operated at a younger age. The difference of 56 vs 42° (which is in the normal range) was statistically significant. Fourteen patients demonstrated grade 1 spondylolisthesis, and four demonstrated grade 2 spondylolisthesis, but none were symptomatic, and at least four patients of this group had preoperative findings of spondylolisthesis on X-ray.
We concluded that selective dorsal rhizotomies did not influence negatively the natural history of spinal deformities and that possibly the procedure had a protective effect if done before the age of 5 with respect to the lordosis. Park [69] has proposed an operation at the conus medullaris level as an option to reduce incidence of the late postoperative spinal deformity after this procedure. We have been hesitant to adopt this approach, first because the rate of complication and of spinal deformity using a narrow laminoplasty approach appears to be quite acceptable and comparable (likely better than) to the natural history of the disease. Second, we strongly believe in the benefits of intraoperative stimulation in our decision making and prefer to decide how to lesion a given root when all rootlets have been stimulated and patterns of spread analyzed keeping in mind the primary surgical objective of balancing elimination of spasticity and preservation of strength. Finally, our other concern with the operation at the conus is the need for a wide exposure and a laminectomy at the junction of the rigid thoracic and mobile lumbar segments. The incidence of long-term orthopedic consequences and in particular thoracolumbar kyphosis, if any, have yet to be analyzed in a controlled fashion to clarify this important point.
Finally, whether the incidence of long-term degenerative changes such as disc herniation, facet hypertrophy, and spinal stenosis will occur at a younger age in these children remains to be determined. In our group, one patient known to have a congenitally narrowed spinal canal at the time of the initial intervention developed spinal stenosis limited to L4 and L5 12 years postoperatively and required decompressive laminectomy.
Conclusion
This paper has concentrated on outcomes after selective posterior rhizotomies for the treatment of spastic cerebral palsy. Several key points have been addressed; there is class 1 evidence supporting the efficiency of selective dorsal rhizotomies in the treatment of spastic cerebral palsy. Quantitative outcome measures demonstrate that higher functioning children with ambulatory capacity or potential for ambulation operated at a young age do best with respect to outcome measures such as GMFM, PDMS, PEDI, and Weefim scores. Their postoperative quality of life is also better with respect to requiring adjunctive orthopedic procedures less frequently. They also show a lower incidence of spinal deformities, in particular, with respect to severity of scoliosis and of hyperlordosis. Finally, the effects of rhizotomies are durable, and quantitative improvements still continue to occur at least up to 5 years postoperatively.
In contrast, older and more involved children show more modest gains and a higher rate of postoperative adjunctive therapy and late spinal deformities. Multidisciplinary teams should direct these patients toward alternative modes of therapy namely, intrathecal Baclofen therapy and/or orthopedic intervention. Children with spastic cerebral palsy are best treated in a multidisciplinary environment where all therapeutic options are available in a complementary rather than in a competitive framework. Continued multidisciplinary interaction will be needed to further advance the field. As for any other functional neurosurgery intervention, careful analysis of outcome by objective measures over long periods is the method by which multidisciplinary teams will obtain crucial answers to the important questions raised.
References
Abbe R (1889) A contribution to surgery of the spine. Med Rec (NY) 35:149–152
Abbott R (1992) Complications with selective posterior rhizotomy. Pediatr Neurosurg 18:43–47
Abbott R, Johann-Murphy M, Shiminski-Maher T, Quartermain D, Forem SL, Gold JT, Epstein FJ (1993) Selective dorsal rhizotomy: outcome and complications in treating spastic cerebral palsy. Neurosurgery 33:851–857
Adams J, Cahan LD, Perry J, Beeler LM (1995) Foot contact pattern following selective dorsal rhizotomy. Pediatr Neurosurg 23:76–81
Albright AL, Barry MJ, Fasick MP, Janosky J (1995) Effects of continuous intrathecal baclofen infusion and selective posterior rhizotomy on upper extremity spasticity. Pediatr Neurosurg 23:82–85
Arens LJ, Peacock WJ, Peter J (1989) Selective posterior rhizotomy: a long-term follow-up study. Child’s Nerv Syst 5:148–152
Bader D, Ramos AD, Lew CD, Platzker AC, Stabile MW, Keens TG (1987) Childhood sequelae of infant lung disease: exercise and pulmonary function abnormalities after bronchopulmonary dysplasia. J Pediatr 110:693–699
Beck AJ, Gaskill SJ, Marlin AE (1993) Improvement in upper extremity function and trunk control after selective posterior rhizotomy. Am J Occup Ther 47:704–707
Bennet WH (1889) A case in which acute spasmodic pain in the left lower extremity was completely relieved by subsural division of the posterior rootlets of certain spinal nerves. Med Chir Trans 72:329–348
Berman B, Vaughan CL, Peacock WJ (1990) The effect of rhizotomy on movement in patients with cerebral palsy. Am J Occup Ther 44:511–516
Bjornson KF, McLaughlin JF (2001) The measurement of health-related quality of life (HRQL) in children with cerebral palsy. Eur J Neurol 8(Suppl 5):183–193
Bloom KK, Nazar GB (1994) Functional assessment following selective posterior rhizotomy in spastic cerebral palsy. Child’s Nerv Syst 10:84–86
Boscarino LF, Ounpuu S, Davis RB III, Gage JR, DeLuca PA (1993) Effects of selective dorsal rhizotomy on gait in children with cerebral palsy. J Pediatr Orthop 13:174–179
Buckon CE, Sienko TS, Aiona MD, Piatt JH (1996) Assessment of upper-extremity function in children with spastic diplegia before and after selective dorsal rhizotomy. Dev Med Child Neurol 38:967–975
Buckon CE, Thomas SS, Piatt JH Jr, Aiona MD, Sussman MD (2004) Selective dorsal rhizotomy versus orthopedic surgery: a multidimensional assessment of outcome efficacy. Arch Phys Med Rehabil 85:457–465
Cahan LD, Adams JM, Perry J, Beeler LM (1990) Instrumented gait analysis after selective dorsal rhizotomy. Dev Med Child Neurol 32:1037–1043
Chabal C, Jacobson L, Little J (1988) Effects of intrathecal fentanyl and lidocaine on somatosensory-evoked potentials, the H-reflex, and clinical responses. Anesth Analg 67:509–513
Chicoine MR, Park TS, Kaufman BA (1997) Selective dorsal rhizotomy and rates of orthopedic surgery in children with spastic cerebral palsy. J Neurosurg 86:34–39
Cohen AR, Webster HC (1991) How selective is selective posterior rhizotomy? Surg Neurol 35:267–272
Crichton JU, Mackinnon M, White CP (1995) The life-expectancy of persons with cerebral palsy. Dev Med Child Neurol 37:567–576
Deletis V, Vodusek DB, Abbott R, Epstein FJ, Turndorf H (1992) Intraoperative monitoring of the dorsal sacral roots: minimizing the risk of iatrogenic micturition disorders. Neurosurgery 30:72–75
du Plessis AJ, Volpe JJ (2002) Perinatal brain injury in the preterm and term newborn. Curr Opin Neurol 15:151–157
Dudgeon BJ, Libby AK, McLaughlin JF, Hays RM, Bjornson KF, Roberts TS (1994) Prospective measurement of functional changes after selective dorsal rhizotomy. Arch Phys Med Rehabil 75:46–53
Engsberg JR, Olree KS, Ross SA, Park TS (1998) Spasticity and strength changes as a function of selective dorsal rhizotomy. J Neurosurg 88:1020–1026
Engsberg JR, Ross SA, Collins DR, Park TS (2006) Effect of selective dorsal rhizotomy in the treatment of children with cerebral palsy. J Neurosurg 105:8–15
Evans PM, Evans SJ, Alberman E (1990) Cerebral palsy: why we must plan for survival. Arch Dis Child 65:1329–1333
Farmer JP, Mittal S (2005) Baclofen. In: Baltuch GH (ed) Surgical management of movement disorders. Taylor & Francis, Roca Raton, FL
Farmer JP, Mittal S (2005) Cerebral palsy. In: Baltuch GH (ed) Surgical management of movement disorders. Taylor & Francis, Roca Raton, FL, pp 245–256
Fasano VA, Barolat-Romana G, Ivaldi A, Sguazzi A (1976) Functional posterior radiculotomy, in the treatment of cerebral spasticity. Preoperative electric stimulation of posterior roots and its use in the choice of the roots to be sectioned. Neurochirurgie 22:23–34
Fasano VA, Broggi G, Zeme S (1988) Intraoperative electrical stimulation for functional posterior rhizotomy. Scand J Rehabil Med Suppl 17:149–154
Fasano VA, Broggi G, Zeme S, Lo RG, Sguazzi A (1980) Long-term results of posterior functional rhizotomy. Acta Neurochir Suppl (Wien) 30:435–439
Foerster O (1913) On the indications and results of the excision of posterior spinal nerve roots in men. Surg Gynecol Obstet 16:463–474
Folio MR, Fewell RR (2000) Peabody developmental motor scales. PRO-ED, Austin, TX
Fukuhara T, Najm IM, Levin KH, Luciano MG, Brant MSC (2000) Nerve rootlets to be sectioned for spasticity resolution in selective dorsal rhizotomy. Surg Neurol 54:126–132
Gros C, Ouaknine G, Vlahovitch B, Frerebeau P (1967) Selective posterior radicotomy in the neurosurgical treatment of pyramidal hypertension. Neurochirurgie 13:505–518
Grossi P, Arner S (1984) Effect of epidural morphine on the Hoffman-reflex in man. Acta Anaesthesiol Scand 28:152–154
Gul SM, Steinbok P, McLeod K (1999) Long-term outcome after selective posterior rhizotomy in children with spastic cerebral palsy. Pediatr Neurosurg 31:84–95
Heim RC, Park TS, Vogler GP, Kaufman BA, Noetzel MJ, Ortman MR (1995) Changes in hip migration after selective dorsal rhizotomy for spastic quadriplegia in cerebral palsy. J Neurosurg 82:567–571
Hodgkinson I, Berard C, Jindrich ML, Sindou M, Mertens P, Berard J (1997) Selective dorsal rhizotomy in children with cerebral palsy. Results in 18 cases at one year postoperatively. Stereotact Funct Neurosurg 69:259–267
Houle AM, Vernet O, Jednak R, Pippi Salle JL, Farmer JP (1998) Bladder function before and after selective dorsal rhizotomy in children with cerebral palsy. J Urol 160:1088–1091
Huang JC, Deletis V, Vodusek DB, Abbott R (1997) Preservation of pudendal afferents in sacral rhizotomies. Neurosurgery 41:411–415
Hutton JL, Pharoah PO (2002) Effects of cognitive, motor, and sensory disabilities on survival in cerebral palsy. Arch Dis Child 86:84–89
Kinghorn J (1992) Upper extremity functional changes following selective posterior rhizotomy in children with cerebral palsy. Am J Occup Ther 46:502–507
Kuban KC, Leviton A (1994) Cerebral palsy. N Engl J Med 330:188–195
Lang FF, Deletis V, Cohen HW, Velasquez L, Abbott R (1994) Inclusion of the S2 dorsal rootlets in functional posterior rhizotomy for spasticity in children with cerebral palsy. Neurosurgery 34:847–853
Lazareff JA, Garcia-Mendez MA, De RR, Olmstead C (1999) Limited (L4–S1, L5–S1) selective dorsal rhizotomy for reducing spasticity in cerebral palsy. Acta Neurochir (Wien) 141:743–751
Lazareff JA, Mata-Acosta AM, Garcia-Mendez MA (1990) Limited selective posterior rhizotomy for the treatment of spasticity secondary to infantile cerebral palsy: a preliminary report. Neurosurgery 27:535–538
Lewin JE, Mix CM, Gaebler-Spira D (1993) Self-help and upper extremity changes in 36 children with cerebral palsy subsequent to selective posterior rhizotomy and intensive occupational and physical therapy. Phys Occup Ther Pediatr 13:25–42
Loewen P, Steinbok P, Holsti L, MacKay M (1998) Upper extremity performance and self-care skill changes in children with spastic cerebral palsy following selective posterior rhizotomy. Pediatr Neurosurg 29:191–198
Logigian EL, Shefner JM, Goumnerova L, Scott RM, Soriano SG, Madsen J (1996) The critical importance of stimulus intensity in intraoperative monitoring for partial dorsal rhizotomy. Muscle Nerve 19:415–422
Marbini A, Ferrari A, Cioni G, Bellanova MF, Fusco C, Gemignani F (2002) Immunohistochemical study of muscle biopsy in children with cerebral palsy. Brain Dev 24:63–66
Marty GR, Dias LS, Gaebler-Spira D (1995) Selective posterior rhizotomy and soft-tissue procedures for the treatment of cerebral diplegia. J Bone Joint Surg Am 77:713–718
McLaughlin JF, Bjornson K, Temkin N, Steinbok P, Wright FV, Reiner A, Roberts TS, Drake JM, O’Donnel MD, Rosenbaum PL, Barber J, Farrel A (2002) Selective dorsal rhizotomy: meta-analysis of the three randomized controlled trials. Dev Med Child Neurol 44:17–25
McLaughlin JF, Bjornson KF, Astley SJ, Graubert C, Hays RM, Roberts TS, Price R, Temkin N (1998) Selective dorsal rhizotomy: efficacy and safety in an investigator-masked randomized clinical trial. Dev Med Child Neurol 40:220–232
McLaughlin JF, Bjornson KF, Astley SJ, Hays RM, Hoffinger SA, Armantrout EA, Roberts TS (1994) The role of selective dorsal rhizotomy in cerebral palsy: critical evaluation of a prospective clinical series. Dev Med Child Neurol 36:755–769
McNeal DM, Hawtrey CE, Wolraich ML, Mapel JR (1983) Symptomatic neurogenic bladder in a cerebral-palsied population. Dev Med Child Neurol 25:612–616
Mittal S, Farmer JP, Al-Atassi B, Gibis J, Kennedy E, Galli C, Courchesnes G, Poulin C, Cantin MA, Benaroch TE (2002) Long-term functional outcome after selective posterior rhizotomy. J Neurosurg 97:315–325
Mittal S, Farmer JP, Al-Atassi B, Montpetit K, Gervais N, Poulin C, Benaroch TE, Cantin MA (2002) Functional performance following selective posterior rhizotomy: long-term results determined using a validated evaluative measure. J Neurosurg 97:510–518
Mittal S, Farmer JP, Al-Atassi B, Montpetit K, Gervais N, Poulin C, Cantin MA, Benaroch TE (2002) Impact of selective posterior rhizotomy on fine motor skills. Long-term results using a validated evaluative measure. Pediatr Neurosurg 36:133–141
Mittal S, Farmer JP, Poulin C, Silver K (2001) Reliability of intraoperative electrophysiological monitoring in selective posterior rhizotomy. J Neurosurg 95:67–75
Montgomery PC (1992) A clinical report of long term outcomes following selective posterior rhizotomy: implications for selection, follow-up, and research. Phys Occup Ther Pediatr 12:67–87
Murphy KP, Molnar GE, Lankasky K (1995) Medical and functional status of adults with cerebral palsy. Dev Med Child Neurol 37:1075–1084
Murphy N, Such-Neibar T (2003) Cerebral palsy diagnosis and management: the state of the art. Curr Probl Pediatr Adolesc Health Care 33:146–169
Nishida T, Thatcher SW, Marty GR (1995) Selective posterior rhizotomy for children with cerebral palsy: a 7-year experience. Child’s Nerv Syst 11:374–380
Nordmark E, Jarnlo GB, Hagglund G (2000) Comparison of the gross motor function measure and paediatric evaluation of disability inventory in assessing motor function in children undergoing selective dorsal rhizotomy. Dev Med Child Neurol 42:245–252
O’Brien DF, Park TS (2006) A review of orthopedic surgeries after selective dorsal rhizotomy. Neurosurg Focus 21:e2
O’Brien DF, Park TS, Puglisi JA, Collins DR, Leuthardt EC (2004) Effect of selective dorsal rhizotomy on need for orthopedic surgery for spastic quadriplegic cerebral palsy: long-term outcome analysis in relation to age. J Neurosurg 101:59–63
O’Brien DF, Park TS, Puglisi JA, Collins DR, Leuthardt EC, Leonard JR (2005) Orthopedic surgery after selective dorsal rhizotomy for spastic diplegia in relation to ambulatory status and age. J Neurosurg 103:5–9
Park TS (1994) Selective dorsal rhizotomy for the spasticity of cerebral palsy. In: Rengachary SS, Wilkins RH (eds) Neurosurgical operative atlas. American Association of Neurological Surgeons, Park Ridge, IL, pp 183–190
Park TS, Johnston JM (2006) Surgical techniques of selective dorsal rhizotomy for spastic cerebral palsy. Technical note. Neurosurg Focus 21:e7
Park TS, Vogler GP, Phillips LH, Kaufman BA, Ortman MR, McClure SM, Gaffney PE (1994) Effects of selective dorsal rhizotomy for spastic diplegia on hip migration in cerebral palsy. Pediatr Neurosurg 20:43–49
Peacock WJ, Arens LJ (1982) Selective posterior rhizotomy for the relief of spasticity in cerebral palsy. S Afr Med J 62:119–124
Peacock WJ, Arens LJ, Berman B (1987) Cerebral palsy spasticity. Selective posterior rhizotomy. Pediatr Neurosci 13:61–66
Peacock WJ, Staudt LA (1991) Functional outcomes following selective posterior rhizotomy in children with cerebral palsy. J Neurosurg 74:380–385
Peter JC, Arens LJ (1993) Selective posterior lumbosacral rhizotomy for the management of cerebral palsy spasticity. A 10-year experience. S Afr Med J 83:745–747
Phillips LH, Park TS (1991) Electrophysiologic mapping of the segmental anatomy of the muscles of the lower extremity. Muscle Nerve 14:1213–1218
Reid CJ, Borzyskowski M (1993) Lower urinary tract dysfunction in cerebral palsy. Arch Dis Child 68:739–742
Rumeau-Rouquette C, Grandjean H, Cans C, du MC, Verrier A (1997) Prevalence and time trends of disabilities in school-age children. Int J Epidemiol 26:137–145
Russell DJ, Rosenbaum PL, Cadman DT, Gowland C, Hardy S, Jarvis S (1989) The gross motor function measure: a means to evaluate the effects of physical therapy. Dev Med Child Neurol 31:341–352
Sacco DJ, Tylkowski CM, Warf BC (2000) Nonselective partial dorsal rhizotomy: a clinical experience with 1-year follow-up. Pediatr Neurosurg 32:114–118
Saito N, Ebara S, Ohotsuka K, Kumeta H, Takaoka K (1998) Natural history of scoliosis in spastic cerebral palsy. Lancet 351:1687–1692
Schijman E, Erro MG, Meana NV (1993) Selective posterior rhizotomy: experience of 30 cases. Child’s Nerv Syst 9:474–477
Smyth MD, Peacock WJ (2000) The surgical treatment of spasticity. Muscle Nerve 23:153–163
Staudt LA, Nuwer MR, Peacock WJ (1995) Intraoperative monitoring during selective posterior rhizotomy: technique and patient outcome. Electroencephalogr Clin Neurophysiol 97:296–309
Steinbok P, Gustavsson B, Kestle JR, Reiner A, Cochrane DD (1995) Relationship of intraoperative electrophysiological criteria to outcome after selective functional posterior rhizotomy. J Neurosurg 83:18–26
Steinbok P, Hicdonmez T, Sawatzky B, Beauchamp R, Wickenheiser D (2005) Spinal deformities after selective dorsal rhizotomy for spastic cerebral palsy. J Neurosurg 102:363–373
Steinbok P, Kestle JR (1996) Variation between centers in electrophysiologic techniques used in lumbosacral selective dorsal rhizotomy for spastic cerebral palsy. Pediatr Neurosurg 25:233–239
Steinbok P, Langill L, Cochrane DD, Keyes R (1992) Observations on electrical stimulation of lumbosacral nerve roots in children with and without lower limb spasticity. Child’s Nerv Syst 8:376–382
Steinbok P, Reiner A, Beauchamp RD, Cochrane DD, Keyes R (1992) Selective functional posterior rhizotomy for treatment of spastic cerebral palsy in children. Review of 50 consecutive cases. Pediatr Neurosurg 18:34–42
Steinbok P, Reiner AM, Beauchamp R, Armstrong RW, Cochrane DD, Kestle J (1997) A randomized clinical trial to compare selective posterior rhizotomy plus physiotherapy with physiotherapy alone in children with spastic diplegic cerebral palsy. Dev Med Child Neurol 39:178–184
Steinbok P, Schrag C (1998) Complications after selective posterior rhizotomy for spasticity in children with cerebral palsy. Pediatr Neurosurg 28:300–313
Storrs BB, Nishida T (1988) Use of the ‘H’ reflex recovery curve in selective posterior rhizotomy. Pediatr Neurosci 14:120–123
Subramanian N, Vaughan CL, Peter JC, Arens LJ (1998) Gait before and 10 years after rhizotomy in children with cerebral palsy spasticity. J Neurosurg 88:1014–1019
Swaminathan S, Quinn J, Stabile MW, Bader D, Platzker AC, Keens TG (1989) Long-term pulmonary sequelae of meconium aspiration syndrome. J Pediatr 114:356–361
Sweetser PM, Badell A, Schneider S, Badlani GH (1995) Effects of sacral dorsal rhizotomy on bladder function in patients with spastic cerebral palsy. Neurourol Urodyn 14:57–64
Thomas SS, Aiona MD, Pierce R, Piatt JH (1996) Gait changes in children with spastic diplegia after selective dorsal rhizotomy. J Pediatr Orthop 16:747–752
Van de Wiele BM, Staudt LA, Rubinstien EH, Nuwer M, Peacock WJ (1996) Perioperative complications in children undergoing selective posterior rhizotomy: a review of 105 cases. Paediatr Anaesth 6:479–486
Vaughan CL, Berman B, Staudt LA, Peacock WJ (1988) Gait analysis of cerebral palsy children before and after rhizotomy. Pediatr Neurosci 14:297–300
von Koch CS, Park TS, Steinbok P, Smyth M, Peacock WJ (2001) Selective posterior rhizotomy and intrathecal baclofen for the treatment of spasticity. Pediatr Neurosurg 35:57–65
Warf BC, Nelson KR (1996) The electromyographic responses to dorsal rootlet stimulation during partial dorsal rhizotomy are inconsistent. Pediatr Neurosurg 25:13–19
Weiss IP, Schiff SJ (1993) Reflex variability in selective dorsal rhizotomy. J Neurosurg 79:346–353
Wong AM, Chen CL, Hong WH, Tang FT, Lui TN, Chou SW (2000) Motor control assessment for rhizotomy in cerebral palsy. Am J Phys Med Rehabil 79:441–450
Wright FV, Sheil EM, Drake JM, Wedge JH, Naumann S (1998) Evaluation of selective dorsal rhizotomy for the reduction of spasticity in cerebral palsy: a randomized controlled trial. Dev Med Child Neurol 40:239–247
Yang TF, Chan RC, Wong TT, Bair WN, Kao CC, Chuang TY, Hsu TC (1996) Quantitative measurement of improvement in sitting balance in children with spastic cerebral palsy after selective posterior rhizotomy. Am J Phys Med Rehabil 75:348–352
Acknowledgments
We would like to acknowledge the help of Drs. Jeffery Golan and Jeffery Hall for their input with regards to spinal changes post rhizotomy. We also like to thank the Spasticity teams of the Montreal Children’s and Shriners Hospitals for their clinical input and Mrs. Lili Orsini and Alaa Arafah for their help in preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farmer, JP., Sabbagh, A.J. Selective dorsal rhizotomies in the treatment of spasticity related to cerebral palsy. Childs Nerv Syst 23, 991–1002 (2007). https://doi.org/10.1007/s00381-007-0398-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-007-0398-2