Abstract
The utility of assessment of cardiovascular calcifications for predicting stroke incidence remains unclear. This study assessed the relationship between cardiovascular calcifications including coronary artery calcification (CAC), aortic valve (AVC), and aortic root (ARC) assessed by coronary computed tomography (CT) and stroke incidence in patients with suspected CAD. In this multicenter prospective cohort study, 1187 patients suspected of CAD who underwent coronary CT were enrolled. Cardiovascular events including stroke were documented. Hazard ratio (HR) and confidence interval (CI) were assessed by Cox proportional hazard model adjusted for the Framingham risk score. C statistics for stroke incidence were also examined by models including cardiovascular calcifications. A total of 980 patients (mean age, 65 ± 7 years; females, 45.8%) were assessed by the CAC, AVC, and ARC Agatston scores. During a median follow-up of 4.0 years, 19 patients developed stroke. Cox proportional hazard model showed severe CAC (Agatston score ≥ 90th percentile [580.0 value]) and presence of AVC and ARC were associated with stroke incidence (HR; 10.33 [95% CI; 2.08–51.26], 3.08 [1.19–7.98], and 2.75 [1.03–7.30], respectively). C statistic in the model with CAC and AVC severity for predicting stroke incidence was 0.841 (95% CI; 0.761–0.920), which was superior to the model with CAC alone (0.762 [95% CI; 0.665–0.859], P < 0.01). CAC, AVC, and ARC were associated with stroke incidence in patients suspected of CAD. Assessment of both CAC and AVC may be useful for prediction of stroke incidence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Stroke is the fourth leading cause of death in Japan; therefore, primary prevention is essential [1]. Since atherosclerosis is one of the important causes of stroke, the prognostication using atherosclerosis index, such as carotid intima-media thickness, has been previously investigated in cohort studies [2]. Coronary artery calcification (CAC), illustrated by chest computed tomography (CT) reflects the atherosclerotic stage and is one of the indicators for predicting coronary artery disease (CAD) [3,4,5]. This assessment has been recommended in few guidelines, whereby CAC is examined in clinical practice [6]. Prospectively, CAC may be also associated with stroke incidence; however, its evidence is scarce and not fully investigated especially in the Asian population [7,8,9,10]. The difference between the Western and Asian population is because in Japan, hemorrhagic and lacunar stroke is more prevalent than in Western countries [11]. Moreover, aortic valve and aortic root calcification can be assessed by coronary CT. Aortic valve calcification (AVC), primarily caused by aging, is considered to be an indicator of atherosclerosis. Although a study showed positive correlation of AVC with stroke incidence in the Western population [7], it still remains controversial [12,13,14]. In particular, it has been suggested that AVC was milder in Asians than in the Western population due to racial/ethnic differences via lipoprotein little A antigen particles or the associated factors, which could affect the impact of AVC on stroke incidence in the Asian population [15]. Although plaque in the aortic arch was considered as one of the embolic sources for ischemic stroke [16], the association between plaque in the aortic root and stroke has not been elucidated [17].
To clarify the relationship between these cardiovascular calcifications and incidence of stroke, we examined the Japanese patients suspected of CAD to prognosticate not only cardiac events but also stroke. Therefore, we performed a sub-study based on the Nationwide Gender-Specific Atherosclerosis Determinants Estimation and Ischemic Cardiovascular Disease Prospective Cohort (NADESICO) study, comprising a long-term follow-up study of patients suspected of CAD using coronary CT imaging, and attempted to prognosticate the major adverse cardiovascular events (MACEs) [18,19,20].
Materials and methods
Study design
The NADESICO study was a prospective, multicenter cohort study designed to evaluate the difference in impact of coronary atherosclerosis, including CAC, on MACEs according to sex [18]. Our study protocol was registered in the University Hospital Medical Information (UMIN) Clinical Trials Registry (UMIN-CTR ID: UMIN000001577) before data were released to the lead author. The protocol was approved by the institutional review board of each center including the National Cerebral and Cardiovascular Center (NCVC, M20-029-7), and written informed consent was obtained from each patient before participation. Author did not have access to information that could identified individual participants during or after data collection.
Participants
The NADESICO study is a prospective cohort study of outpatients who examined coronary multi-slice CT with suspicious of CAD in multicenter hospitals in Japan. Patients for this study were enrolled from the cardiology department of each of the 15 hospitals in Japan participating in the NADESICO study between December 2008 and April 2013. The inclusion and exclusion criteria for the NADESICO study have been described elsewhere [19, 20]. The inclusion criteria were: (1) patients aged 50–74 years suspected of CAD in a stable setting and adequate indications for plain CT and coronary CT angiography and (2) patients without a history of myocardial infarction or coronary artery revascularization at enrollment. In the NADESICO study, patients with rare diseases and patients who were not expected long-term survival were excluded. Therefore, the patients who met the following criteria were excluded: (1) with a history of Kawasaki disease, (2) diagnosis of coronary artery malformation, (3) diagnosis of familial hypercholesterolemia, (4) with poor prognosis due to malignant tumors, (5) undergoing dialysis, or (6) undergoing treatment for a serious mental or neurological disorder. Attending physicians at each hospital screened the outpatients suspected CAD with adequate indications for coronary CT angiography, and, when they met the inclusion criteria and did not meet the exclusion criteria, they were enrolled after the informed consent was given and consent was obtained. The patients had been enrolled until the target number of patients was reached.
The sample size in NADESICO study was determined based on the previous study [21]. When setting 10% event rate at 3 years with power = 0.80 and α = 0.05, sample size was 454. When considering 10% dropout rate, the sample size would be 500, which the patient’s number 980 in our dataset has satisfied.
Data collection
Clinical data for diagnosis and treatment were collected by investigators at each hospital and sent to the NCVC. The definitions of each baseline characteristic were described elsewhere [18,19,20].
CT was performed according to the guidelines of the Japanese Circulation Society and institutional protocol with electrocardiogram gating and at least 64 channels. CT images were digitally transferred to the NCVC and evaluated in an independent imaging core laboratory. CAC, AVC, and ARC scores were calculated using the Agatston method by SYNAPSE VINCENT® (FUJIFILM Medical IT Solutions Co., Ltd., Tokyo, Japan). CAC was measured as the total Agatston score in all coronary arteries, AVC was measured as the total Agatston score in the aortic valve leaflets and annular calcification, and ARC was measured as the total Agatston score at the site of ARC within 3 cm of the aortic annulus, including the sinus of Valsalva and sinotubular junction [22, 23]. The measurement was performed by an experienced radiologist or cardiologist who were blinded to all clinical data.
Follow-up
Patients were followed up annually and evaluated for the presence or absence of events by their attending physicians at each hospital. Stroke was defined when more than two items of the following three criteria according to the protocol in the NADESICO study were met: (1) neurological symptoms, including disturbance consciousness, and focal neurological symptoms such as paralysis or sensory disturbance, (2) lesions detected by brain CT or MRI, and (3) exclusion of other causes of neurological symptoms, such as hypoglycemia and hepatic encephalopathy. As a reference analysis, the association of CAC, AVC, and ARC with the incidence of cardiac events was also examined in this study, and a cardiac event was defined as the composite of cardiovascular death except for stroke, myocardial infarction, revascularization, and hospitalization for unstable angina, heart failure, or aortic disease [18,19,20]. Each attending physician annually followed up the patients by using medical examination, telephone, and mail as long as possible until March 2020. The follow-up period was originally set at 3 years based on JCAD study [21]; however, it finally took time to collect follow-up data which ended up at approximately 5 years.
Statistical analysis
Continuous data are presented as means ± standard deviations and categorical data are displayed as numbers (%). One-way analysis of variance and chi-square tests were used to analyze significant differences between three groups (stroke, cardiac event, and no-event) for continuous and categorical variables, respectively. The correlation was examined between the stroke or cardiac event group and no-event group. The staging of CAC, AVC, or ARC severity based on its 90th percentile was determined considering the distribution of the CAC, AVC, or ARC Agatston scores as described in previous studies (Agatston score = 0, 0 < Agatston score < 90th percentile, Agatston score ≥ 90th percentile) [19]. Hazard ratio (HR) was calculated for stroke and cardiac events by univariate analysis including baseline characteristics, blood examination, the Framingham risk score (FRS), and Agatston score.
Cox proportional hazard models of the Agatston score for the incidence of stroke or cardiac events were developed along with HRs, and 95% confidence intervals (CIs) were calculated after adjustment for the FRS. Harrell’s concordance statistic (C-statistic) for predicting incidence of stroke or cardiac events was examined by various models, including the FRS calculated directly in each patient, CAC, AVC, and ARC staging [19, 24].The 95% CIs of C-statistics were estimated using 200 bootstrap samples. Kaplan–Meier analysis was performed to examine the incidence of stroke and cardiac events according to the category of CAC, AVC, or ARC. Statistical significance was defined as a two-tailed P-value of < 0.05. Statistical analyses were performed using Stata 17 (StataCorp, College Station, TX, USA).
Results
Cardiovascular risk factors and stroke incidence
A total of 980 patients (mean age; 65 ± 7 years, median follow-up period; 4.04 years) suspected of CAD with availability of the Agatston scores of CAC, AVC, and ARC were investigated. Among them, 19 patients developed stroke and 67 patients developed cardiac events (Supplementary Fig. 1). Patients who developed stroke were likely to have hypertension, dyslipidemia, and chronic kidney disease more frequently and higher hemoglobin A1c and serum triglyceride levels (Table 1). These factors were also associated with stroke incidence in the univariate analysis (Supplementary Table 1). More patients who developed cardiac events were male, who current or past smokers, had hypertension, dyslipidemia, chronic kidney disease, higher HbA1c and serum triglyceride levels, and lower levels of high-density lipoprotein cholesterol and estimated glomerular filtration rate. In the univariate analysis, these factors, except dyslipidemia, were associated with the incidence of cardiac events. FRS was not associated with stroke incidence (HR: 1.07 [95% CI: 0.92–1.23]); however, it was significantly associated with cardiac events in the univariate analysis (HR: 1.13 [95% CI: 1.05–1.22]).
Association of cardiovascular calcifications and stroke incidence
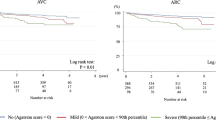
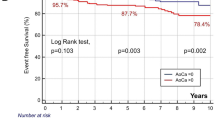
The median values (interquartile range) of the CAC, AVC, and ARC Agatston scores in this study were 23.0 (0–180.1), 0 (0–8.3), and 0 (0–25.8), respectively. Each patient with stroke or a cardiac event had significantly higher CAC, AVC, and ARC than patients without a cardiac event (Table 1). Figure 1 shows results of the incidence rates for stroke and cardiac events according to the category using the 90th percentile of the Agatston score (CAC: 580.0, AVC: 76.5, ARC: 174.8). Kaplan–Meier analysis according to the category using the 90th percentile value also showed that the three calcification indexes were associated with stroke events (Fig. 2). Cox proportional hazard model using the category with the 90th percentile value showed severe CAC (Agatston score ≥ 90th percentile), and presence of AVC and ARC (Agatston score > 0) was significantly associated with stroke incidence (Table 2). C-statistics in CAC, AVC, or ARC values alone were 0.768, 0.704, and 0.726, respectively, for stroke incidence (Table 3). In the model developed by adding AVC and CAC values, the C-statistic significantly increased compared with that in the model with CAC value alone, and no other models with the addition of the FRS were superior to the combined model comprising CAC and AVC values. In the Kaplan–Meier analysis, the patients with the CAC Agatston score ≥ 90th percentile and the AVC Agatston score > 0 showed the worst prognosis (Fig. 2D).
The incidence rate of stroke and cardiac events stratified by CAC, AVC, or ARC severity. Agatston score = 0, no calcification; 0 < Agatston score < 90th percentile, mild calcification; 90th percentile ≤ Agatston score, severe calcification. ARC, aortic root calcification; AVC, aortic valve calcification; CAC, coronary artery calcification
Kaplan–Meier analysis for stroke incidence based on cardiovascular calcifications severity. Grouping for stoke event: A–C Group 1, Agatston score = 0; Group 2, 0 < Agatston score < 90th percentile; Group 3, Agatston score ≥ 90th percentile; D Group 1, CAC Agatston score < 90th percentile and AVC Agatston score = 0; Group 2, CAC Agatston score < 90th percentile and AVC Agatston score > 0; Group 3, CAC Agatston score ≥ 90th percentile and AVC Agatston score > 0. ARC, aortic root calcification; AVC, aortic valve calcification; CAC, coronary artery calcification
Kaplan–Meier analysis according to the category based on the 90th percentile value also showed that all the three calcification indexes were associated with cardiac events (Supplementary Fig. 2). C-statistics in CAC, AVC, or ARC alone were 0.640, 0.598, and 0.604, respectively, for cardiac events (Table 3). C-statistic increased significantly by adding the FRS or FRS and AVC values to the CAC value compared with that when CAC (P = 0.03 or < 0.01, respectively) or the FRS was staged alone (P = 0.04 or 0.02, respectively).
Discussion
Cardiovascular system calcification and stroke incidence
We previously reported the association between calcifications in the cardiovascular system and MACEs in the NADESICO study [19]. However, because mechanisms of stroke and cardiac events are not entirely similar, the association may be different with respect to stroke and cardiac events [25]. With further detailed analysis, we aimed to explore the association between cardiovascular calcifications and stroke incidence in the primary prevention population. The present study is unique because the association of CAC, AVC, and ARC with the incidence of stroke and cardiac events were simultaneously examined in the Asian population. The findings of this study correspond to those of previous studies on the Western population concerning the association between CAC and stroke incidence [7,8,9,10]. Our results may be rational because no significant differences were reported in characteristics of CAC between the Asian and Western populations in the primary cohort [26]. Although the detailed mechanisms linking CAC and stroke incidence have not been clarified, CAC may be associated with cardiovascular events, including stroke, because CAC occurs during the inflammatory process of atherosclerosis and reflects its severity [27]. Especially, CAC also reflects atherosclerosis in muscular arteries, such as intracranial arteries; therefore, CAC may be associated with stroke incidence [28, 29]. Furthermore, it has been suggested that atrial fibrillation is associated with arteriosclerosis or inflammation; therefore, severe CAC may also be associated with stroke incidence and atrial fibrillation [28].
In this study, AVC was also associated with stroke incidence. For prognosticating stroke incidence, although the impact did not seem to be superior to that of CAC, additional assessment of AVC to CAC severity effectively predicted stroke incidence. AVC was one of the atherosclerotic indicators, and most patients with AVC had hypertension, diabetes mellitus, and atrial fibrillation [12]. These factors were also associated with stroke incidence, which contributed to the association between AVC and stroke incidence [12]. AVC has also been reported as a major cause of cardioembolism in bicuspid aortic valve patients [30]. However, since inflammation due to mechanical endothelial damage caused by local blood turbulence around the valve also affects AVC, the impact of AVC on stroke incidence may be attenuated compared with that of CAC [31, 32].
Although severe ARC was significantly associated with stroke incidence, the impact was not strong. ARC was also one of the atherosclerotic markers, which might be associated with stroke incidence to some degree [33]. There may be a difference in the atherosclerotic process between the aortic root with elastic artery and the intracerebral artery as muscular artery, and it may explain the differences in association with stroke incidence [34, 35]. In addition, a lower distribution of ARC may also attenuate the association with stroke incidence compared to that in the aortic arch [32].
Models including the FRS model
Model with CAC alone was useful for prediction of stroke incidence, and additional impact of the FRS, which consisted of atherosclerotic risk factors, was not observed. One of the possibilities was that the FRS did not include atrial fibrillation, which was a trigger for cardiogenic cerebral embolism accounting for about 20–30% of stroke cases [36].Additionally, although age, hypertension, and diabetes were particularly associated with the development of atrial fibrillation as shown in the CHADS2 score, the distribution of scores for these factors in the FRS model might be insufficient for predicting stroke incidence [37]. Moreover, congestive heart failure and history of stroke were not included in the FRS model, which might have lowered the C-statistic of the FRS. Regarding cardiac events, although all three calcification indicators were associated with incidence of cardiac events, the combination model of CAC/AVC and FRS was the most useful in predicting stroke incidence [3].
Limitations
First, although this study was planned to include the consecutive patients who met inclusion and did not meet exclusion criteria, the difference in the number of registered patients among institutions occurred due to the variability of patients who agreed to informed consent or differences in the way physicians thought about study entry between institutions. It may lead to selection bias and there may be limitations to the generalization of this study. Second, some detailed information associated with stroke incidence was not available in this study. It included comorbidities such as atrial fibrillation, heart failure, previous stroke, or antithrombotic agents as reported in previous studies [38,39,40,41]. Echocardiographic findings including aortic valve stenosis were not also available, which may be associated with stroke incidence. Moreover, we could not clarify etiologies of stroke, such as acute ischemic stroke, intracerebral hemorrhage, and subarachnoid hemorrhage in the present study. Third, since the number of stroke events was small, the relationship between calcifications and other clinical markers for incidence prediction were not fully investigated. Especially, we were unable to perform the multivariate analysis adjusted for various clinical atherosclerotic factors. These limitations may be caused by the plan that the NADESICO study primarily included patients with suspected coronary artery disease. However, the need for collaboration between cardiology and neurology has been reported to prevent incidence of stroke in recent years [42], our study is significant in the point of view. Future studies with a larger sample size including both perspectives are expected. Fourth, this was a cohort study including patients suspected of having CAD; therefore, caution should be exercised when extrapolating our results to primary prevention in general.
Conclusions
Cardiovascular calcifications including CAC, AVC and ARC were associated with the future incidence of stroke in patients suspected of having CAD. Further evaluation of cardiovascular calcifications may be necessary to determine the incidence of cardiovascular events, including stroke, in patients at a mild to moderate cardiovascular risk.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ARC:
-
Aortic root calcification
- AVC:
-
Aortic valve calcification
- CAC:
-
Coronary artery calcification
- CAD:
-
Coronary artery disease
- CAS:
-
Coronary artery stenosis
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- CVD:
-
Cardiovascular disease
- FRS:
-
Framingham risk score
- HR:
-
Hazard ratio
- MACE:
-
Major adverse cardiovascular events
- OR:
-
Odds ratio
References
Tai SY, Cheon S, Yamaoka Y, Chien Y, Lu T (2022) Changes in the rankings of leading causes of death in Japan, Korea, and Taiwan from 1998 to 2018: a comparison if three ranking lists. BMC Public Health 22:926
Kokubo Y, Watanabe M, Higashiyama A, Nakao YM, Nakamura F, Miyamoto Y (2018) Impact of intima-media thickness progression in the common carotid arteries on the risk of incident cardiovascular disease in the Suita study. J Am Heart Assoc 7:e007720
Erbel R, Mohlenkamp S, Moebus S, Schmermund A, Lehmann N, Stang A, Dragano N, Gronemeyer D, Seibel R, Kalsch H, Brocker-Preuss M, Mann K, Siegrist J, Jockel K, Heinz Nixdorf Recall Study Investigative Group (2010) Coronary risk stratification, discrimination, and reclassification improvement based in quantification of subclinical coronary atherosclerosis; the Heinz Nixdorf Recall Study. J Am Coll Cardiol 56:1397–1406
Yeboah J, McClelland RL, Polonsky TS, Burke GL, Sibley CT, O’Leary D, Carr JJ, Goff DC, Greenland P, Herrington DM (2012) Comparison of novel risk markers for improvement in cardiovascular risk assessment in intermediate risk individuals. JAMA 308:788–795
Yeboah J, Young R, McClelland RL, Delaney JC, Polonsky TS, Dawood FZ, Blaha MJ, Miedema MD, Sibley CT, Carr J, Burke GL, Goff DC Jr, Psaty BM, Greenland P, Herrington DM (2016) Utility of nontraditional risk markers in atherosclerotic cardiovascular disease risk assessment. J Am Coll Cardiol 67:139–147
Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, Himmelfarb CD, Khera A, Lloyd-Jones D, McEvoy JW, Michos ED, Miedema MD, Munoz D, Smith SC Jr, Virani SS, Williams KA Sr, Yeboah J, Ziaeian B (2019) 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 140:e596–e646
Wong ND, Gransar H, Shaw L, Polk D, Moon JH, Miranda-Peats R, Hayes SW, Thomson LE, Rozanski A, Friedman JD, Berman DS (2009) Thoracic aortic calcium versus coronary artery calcium for the prediction of coronary heart disease and cardiovascular disease events. JACC Cardiovasc Imaging 2:319–326
Gibson AO, Blaha MJ, Arnan MK, Sacco RL, Szklo M, Herrington DM, Yeboah J (2014) Coronary artery calcium and incident cerebrovascular events in an asymptomatic cohort. The MESA study. JACC Cardiovasc Imaging 7:1108–1115
Hermann DM, Gronewold J, Lehmann N, Moebus S, Jockel KH, Bauer M, Erbel R, Heinz Nixdorf Recall Study Investigative Group (2013) Coronary artery calcification is an independent stroke predictor in the general population. Stroke 44:1008–1013
Gronewold J, Bauer M, Lehmann N, Mahabadi AA, Kälsch H, Weimar C, Berger K, Moebus S, Jöckel KH, Erbel R, Hermann DM, Heinz Nixdorf Recall Study Investigative Group (2014) Coronary artery calcification, intima-media thickness, and ankle-brachial index are complementary stroke predictors. Stroke 45:2702–2709
Takashima N, Arima H, Kita Y, Fujii T, Miyamatsu N, Komori M, Sugimoto Y, Nagata S, Miura K, Nozaki K (2017) Incidence, management and short-term outcome of stroke in a general population of 1.4 million Japanese – Shiga Stroke Registry. Circ J 81:1636–1646
Zhang D, Dai X, Wang C, Han K, Wang J, Dong Y, Chen J, Hou L (2018) Aortic valve calcification and risk of stroke: a systematic review and meta-analysis. J Clin Neurosci 55:32–37
Otto CM, Kuusisto J, Reichenbach DD, Gown AM, O’Brien KD (1994) Characterization of the early lesion of “degenerative” valvular aortic stenosis. Histological and immunohistochemical studies. Circulation 90:844–853
Fashanu OE, Bizanti A, AI-Abdouh A, Zhao D, Budoff MJ, Thomas IC, Longstreth WT Jr, Michos ED (2020) Progression of valvular calcification and risk of incident stroke: the Multi-Ethic Study of Atherosclerosis (MESA). Atherosclerosis 307:32–38
Makshood M, Joshi PH, Kanaya AM, Ayers C, Budoff M, Tsai MY, Blaha M, Michos ED, Post WS (2020) Lipoprotein (a) and aortic valve calcium in South Asians compared to other race/ethic groups. Atherosclerosis 313:14–19
Bazan HA, Pradhan S, Mojibian H, Kyriakides T, Dardik A (2007) Increased aortic arch calcification in patients older than 75 years: implications for carotid artery stenting in elderly patients. J Vasc surg 46:841–845
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, Marsh EE 3rd (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in acute stroke treatment. Stroke 24:35–41
Nakao YM, Miyamoto Y, Higashi M, Noguchi T, Ohishi M, Kubota I, Tsutsui H, Kawasaki T, Furukawa Y, Yoshimura M, Morita H, Nishimura K, Kada A, Goto Y, Okamura T, Tei C, Tomoike H, Naito H, Yasuda S (2018) Sex difference in impact of coronary artery calcification to predict coronary artery disease. Heart 104:1118–1124
Wada S, Iwanaga Y, Nakai M, Miyamoto Y, Noguchi T (2023) Aortic valve and aortic root calcification for predicting major adverse cardiovascular events: NADESICO study. Heart Vessels 38:562–569
Wada S, Iwanaga Y, Nakai M, Nakao YM, Miyamoto Y, Noguchi T, NADESICO Study Investigators (2023) Combination of coronary CT angiography, FFRct, and risk factors in the prediction of major adverse cardiovascular events in patients suspected CAD. Clin Cardial 46:494–501
Japanese Coronary Artery Disease (JCAD) Study Investigators (2006) Current status of the background of patients with coronary artery disease in Japan. Circ J 70:1256–1262
Pawade T, Sheth T, Guzzetti E, Dweck MR, Clavel MA (2019) Why and how to measure aortic valve calcification in patients with aortic stenosis. JACC Cardiovasc Imaging 12:1835–1848
Nafakhi H, AI-Nafakh HA, AI-Mosawi AA (2016) Aortic root calcification: a possible imaging biomarkers of coronary atherosclerosis. Pulse (Basel) 3:167–171
Vandvik PO, Lincoff AM, Gore JM, Gutterman DD, Sonnenberg FA, Alonso-Coello P, Aki EA, Lansberg MG, Guyatt GH, Spencer FA (2012) Primary and secondary prevention of cardiovascular disease: antithrombotic therapy and prevention of thrombosis, 9th er: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141:e637S-e668S
Shibata T, Kawakami S, Noguchi T, Tanaka T, Asaumi Y, Kanaya T, Nagai T, Fujino M, Nagatsuka K, Ishibashi-Ueda H, Nishimura K, Miyamoto Y, Kusano K, Anzai T, Goto Y, Ogawa H, Yasuda S (2015) Prevalence, clinical features, and prognosis of acute myocardial infarction attributable to coronary artery embolism. Circulation 132:241–250
Kanaya AM, Kanduka NR, Ewing SK, Herrington D, Liu K, Blaha MJ, Srivastava S, Dave SS, Budoff MJ (2014) Comparing coronary artery calcium among U.S. South Asians with four racial/ethnic groups: the MASALA and MESA studies. Atherosclerosis 234:102–107
Mohan J, Bhatti K, Tawney A, Zeltser R (2023) Coronary artery calcification. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing
O’Neal WT, Efird JT, Dawood FZ, Yeboah J, Alonso A, Heckbert SR, Soliman EZ (2014) Coronary artery calcium and risk of atrial fibrillation (from the multi-ethic study of atherosclerosis). Am J Cardiol 114:1707–1712
Chen X, Fisher M (2016) Pathological characteristics. Front Neurol Neurosci 40:21–33
Huntley GD, Michelena HI, Thaden JJ, Alkurashi AK, Pislaru SV, Pochettino A, Crestanello JA, Maleszewski JJ, Brown RD Jr, Nkomo VT (2023) Cerebral and retinal infarction in bicuspid aortic valve. J Am Heart Assoc 12:e028789
Freeman RV, Otto CM (2005) Spectrum of calcific aortic valve diseases: pathogenesis, disease progression, and treatment strategies. Circulation 111:3316–3326
Olsen M, Watchtell K, Bella JN, Liu JE, Boman K, Gerdts E, Papademetriou V, Nieminen MS, Rokkedal J, Dahlof B, Devereux RB (2004) Effect of losartan versus atenolol on aortic valve sclerosis (a LIFE substudy). Am J Cardiol 94:1076–1080
Desai MY, Cremer PC, Schoenhagen P (2018) Thoracic aortic calcification: diagnostic, prognostic and management considerations. JACC Cardiovasc Imaging 11:1012–1026
Feng Y, Wang X, Zhao Y, Li L, Niu P, Huang Y, Han Y, Tan W, Huo Y (2021) A comparison of passive and active wall mechanics between elastic and muscular arteries of juvenile and adult rats. J Biomech 126:110642
Miyata M (2021) Basic research sheds light on the aspect of cardio-ankle vascular index (CAVI) including elastic and muscular arteries. J Atheroscler Thromb 28:588–589
Lip GYH, Nieuwlaat R, Pisters R, Lane DA, Crijns H (2010) Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest 137:263–272
Mehilli J, Winhard M (2022) The CHADS-P2A2RC score: ready to guide the selection of treatment. Eur Heart J 43:1005–1007
Choi SE, Sagris D, Hill A, Lip GYH, Abdul-Rahim AH (2023) Atrial fibrillation and stroke. Expert Rev Cardiovasc Ther 21:35–56
Haeusler KG, Laufs U, Endres M (2011) Chronic heart failure and ischemic stroke. Stroke 42:2977–2982
Schwartz NE, Diener HC, Albers GW (2009) Antithrombotic agents for stroke prevention. Handb Clin Neurol 94:1277–1294
Komatsu T, Tachibana H, Sato Y, Ozawa M, Kunugida F, Orii M, Nakamura M (2010) Relationship between CHADS2 score and ischemic stroke during rhythm control therapy for paroxysmal atrial fibrillation. Int Heart J 51:24–29
Kim JS, Thijs V, Yudi M, Toyoda K, Shiozawa M, Zening J, Clapp B, Albers B, Diener HC (2022) Establishment of the heart and brain team for patent foramen ovale closure in stroke patients: an expert opinion. J Stroke 24:345–351
Acknowledgements
We would like to thank Drs. Masanao Naya (Hokkaido University Hospital, Sapporo, Japan), Masafumi Watanabe (Yamagata University Hospital, Yamagata, Japan), Tetsu Watanabe (Yamagata University Hospital, Yamagata, Japan), Satoshi Yasuda (Tohoku University Hospital, Sendai, Japan), Michihiro Yoshimura (Jikei University School of Medicine, Tokyo, Japan), Makoto Kawai (Jikei University School of Medicine, Tokyo, Japan), Tomonori Okamura (Keio University School of Meidicine, Tokyo, Japan), Hiroaki Naito (Nissei Hospital, Osaka, Japan), Masahiro Higashi (National Hospital Organization Osaka National Hospital, Osaka Japan), Hideaki Morita (Osaka medical College, Takatsuki, Japan), Kunihiro Nishimura (National Cerebral and Cardiovascular center, Suita, Japan), Yoko Nakao (National Cerebral and Cardiovascular center, Suita, Japan), Yasuhide Asaumi (National Cerebral and Cardiovascular center, Suita, Japan), Hiroyuki Miura (National Cerebral and Cardiovascular center, Suita, Japan), Yutaka Furukawa (Kobe City Medical Center General Hospital, Tokyo, Japan), Hiroyuki Tsutsui (Kyusyu University Hospital Tomohiro Kawasaki Shin Koga Hospital), Mitsuru Oishi (Kagoshima University Hospital), Masaaki Miyata (Kagoshima University Hospital), Hitonobu Tomoike (NTT reserch, Inc.MEI Lab), Ms. Junko Masue (National Cerebral and Cardiovascular center, Suita, Japan), Ms. Yoko Sumita (National Cerebral and Cardiovascular center, Suita, Japan), and Mr. Yusuke Sasahara (National Cerebral and Cardiovascular center, Suita, Japan) for supporting the NADESICO study.
Funding
This research was supported by Japan Agency for Medical Research and Development under Grant Number 20gk0210026h001.
Author information
Authors and Affiliations
Contributions
Study concept and design: S.W., Y.I., and T.N.; Data curation: S.W. and M.N.; Analysis and interpretation of data: S.W., M.N., and Y.I.; Contribution to the interpretation of results: Y.M. and T.N.; Supervision: Y. M. and T. N.; Drafting the manuscript (original draft): S.W.; Drafting, reviewing, and editing: Y.I. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wada, S., Iwanaga, Y., Nakai, M. et al. Clinical impact of cardiovascular calcifications on stroke incidence in primary prevention: analysis in NADESICO study. Heart Vessels 39, 754–762 (2024). https://doi.org/10.1007/s00380-024-02394-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-024-02394-6