Abstract
Purpose
Prostate biparametric magnetic resonance imaging (bpMRI) including T2-weighted imaging (T2WI) and diffusion-weighted imaging (DWI) might be an alternative to multiparametric MRI (mpMRI, including dynamic contrast imaging, DCE) to detect and guide targeted biopsy in patients with suspected prostate cancer (PCa). However, there is no upgrading peripheral zone PI-RADS 3 to PI-RADS 4 without DCE in bpMRI. The aim of this study was to evaluate bpMRI against mpMRI in biopsy-naïve men with elevated prostate-specific antigen (PSA) scheduled for robot-assisted-transperineal fusion-prostate biopsy (RA-TB).
Methods
Retrospective single-center-study of 563 biopsy-naïve men (from 01/2015 to 09/2018, mean PSA 9.7 ± 6.5 ng/mL) with PI-RADSv2.1 conform mpMRI at 3 T before RA-TB. Clinically significant prostate cancer (csPCa) was defined as ISUP grade ≥ 2 in any core. Two experienced readers independently evaluated images according to PI-RADSv2.1 criteria (separate readings for bpMRI and mpMRI sequences, 6-month interval). Reference standard was histology from RA-TB.
Results
PI-RADS 2 was scored in 5.1% of cases (3.4% cancer/3.4% csPCa), PI-RADS 3 in 16.9% (32.6%/3.2%), PI-RADS 4 in 57.6% (66.1%/58.3%) and PI-RADS 5 in 20.4% of cases (79.1%/74.8%). For mpMRI/bpMRI test comparison, sensitivity was 99.0%/97.1% (p < 0.001), specificity 47.5%/61.2% (p < 0.001), PPV 69.5%/75.1% (p < 0.001) and NPV 97.6%/94.6% (n.s.). csPCa was considered gold standard. 35 cases without cancer were upgraded to PI-RADS 4 (mpMRI) and six PI-RADS 3 cases with csPCa were not upgraded (bpMRI).
Conclusion
In patients planned for RA-TB with elevated PSA and clinical suspicion for PCa, specificity was higher in bpMRI vs. mpMRI, which could solve constrains regarding time and contrast agent.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Prostate cancer (PCa) represents the most common non-cutaneous cancer among men with a lifetime risk of up to 37% [1]. Multiparametric magnetic resonance imaging (mpMRI) is regarded as the best imaging modality for the prostate and further standardization for describing and interpreting imaging results due to the introduction of the Prostate Imaging–Reporting and Data System (PI-RADS) and helps increase cancer detection based on the improved information on tissue characteristics [2]. Currently, PI-RADSv2.1 conform mpMRI demands T2-weighted sequences (T2w), diffusion weighted Imaging (DWI), and dynamic contrast-enhanced imaging (DCE). It is notable, however, that in PI-RADSv2.1 DCE-imaging is solely a secondary sequence for lesions in the peripheral zone (PZ) to further characterize lesions of the PI-RADS 3 category and not included in evaluation of lesions in the transition zone (TZ). The benefits of a biparametric MRI protocol (bpMRI) are signified by reduced costs, shorter acquisition time, and no contrast agent associated risks.
In general, the ability to differentiate PCa from focal benign processes like chronic prostatitis can be difficult. On MRI, both T2w and ADC derived from DWI can display decreased signal intensity in the PZ. The classic shape in form of a band or wedge is sometimes replaced by focal or irregular appearances. Additionally, also inflammatory processes can lead to increased DCE values by increased perfusion. Furthermore, PCa and chronic prostatitis are often accompanied by elevation of serum prostate-specific antigen (PSA) [3,4,5]. Especially in case of PI-RADS 3 lesions, histopathology reveals chronic prostatitis in about 50% [4, 6]. However, the value of DCE in the detection of prostate cancer is still controversial. Some studies have shown that combining DCE MRI with T2w and DWI does not significantly improve the diagnostic accuracy for prostate cancer [7,8,9].
Contrarily, some studies have found that DCE MRI is highly sensitive in the diagnosis of PCa [10,11,12], especially in peripheral lesions, and combining DCE MRI with DWI can significantly improve the accuracy of cancer detection. With the introduction of PI-RADSv2.0, the role of DCE MRI was limited to upgrade lesions from PI-RADS 3 to PI-RADS 4 in the peripheral zone when contrast enhancement is observed in these lesions.
In view of the potential advantages, it may be questionable whether dynamic contrast enhancement should be mandatory in routine prostate MRI protocols or if bpMRI provides similar results to those of mpMRI for the detection and localization of PCa.
The aim of our study was to compare the detection rate of PCa and csPCa of bpMRI on Prostate Imaging Reporting and Data System (PI-RADS) v2.1 scoring in comparison to the mpMRI approach in biopsy-naïve men with elevated prostate-specific antigen (PSA). Histology of targeted and systemic biopsy of mpMRI guided robot-assisted transperineal fusion prostate biopsy (RA-TB) was defined as the reference standard.
Materials and methods
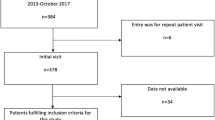
This retrospective study was approved by the institutional review board (359/2019BO2) and conducted in accordance with the Helsinki protocol. From January 2015 to September 2018, we included consecutive patients meeting the following inclusion criteria: rising and/or persistently elevated PSA, clinically indicated mpMRI of the prostate and planned for RA-TB without prior biopsy. Exclusion criteria were a PI-RADS score of 1 as they did not show a measurable or targetable lesion and/or a palpable tumor at digital rectal examination, as these were referred to biopsy without prior MRI. Digital rectal examination was performed by experienced urologist consultants (S.K., S.R., J.B.). Mean patient age was 66 ± 8 years (range 45–84 years).
MR imaging
All patients underwent mpMRI on a 3 T MRI system according to the European Society of Urogenital Radiology guidelines and adapting the ACR Prostate Imaging–Reporting and Data System (PI-RADS) v2.1 guidelines. The MRI acquisition protocol was in accordance with the technical requirements stated in the PI-RADS v2.1 update. No endorectal coil was used. All patients received body weight adapted gadolinium-based intravenous contrast agent followed by a saline flush. All Patients received 20 mg hyoscine butylbromide i.v. before the examination.
Two board-certified uroradiologists with 5 and 12 years of experience in prostate MRI reading analyzed the acquisitions for both mpMRI and bpMRI. Readers were blinded to the histopathologic diagnosis as well as the clinical procedures after the MRI. There were 6 months in between reading sessions for mpMRI and bpMRI.
An index lesion was defined before biopsy, this was considered as “target” in contrast to “off-target” biopsy, which is equivalently used for systematic, non-targeted biopsy of the prostate.
RI reader performance
MRI reader performance concordance analysis for PI-RADSv2.1 score was performed between two specialists for mpMRI. The agreement for both readers was substantial for both mpMRI (Kohen’s κ = 0.69, z = 8.05, p < 0.001) and bpMRI Kohen’s κ = 0.62, z = 9.75, p < 0.001). Importantly, for the PI-RADS 3 cases with upgrade to PI-RADS 4 in mpMRI, no reader differences were observed.
Robot-assisted mpMRI-TRUS fusion prostate biopsy (RA-TB)
Biopsy was performed using an iSRobot Mona LisaTM robot unit, an ultrasound machine (Pro Focus 2202, BK Medical, Peabody, MA) with multi-frequency ultrasound probe (BK 8848, BK Medical, Peabody, MA) and UroBiopsyTM 3D modelling software (both: Biobot Surgical, Singapore) as previously reported [13]. Four targeted biopsy samples and 14 off-target transperineal biopsy samples were obtained with focus on the peripheral zones. All procedures were performed by an experienced urologist consultant. Tissue samples were fixated with formalin solution and evaluated for histology. Clinically significant prostate cancer (csPCa) was defined was defined as ISUP grade ≥ 2 in any core.
Statistical analysis
Variables are presented as mean and standard deviation, confidence intervals (CI) are given when indicated. Group comparisons were calculated with Kruskal–Wallis ANOVA as parameters did not show normal distribution (Kolmogorov–Smirnov test) and were corrected for multiple comparison with Dunn's multiple comparisons test. GraphPad Prism version 9.0.0, GraphPad Software, San Diego, California, USA, was used for statistical analysis, significance was considered as p < 0.05. For calculation of the test accuracy of bpMRI and mpMRI and the statistical comparison between both tests, we used the “Compbdt” package for R (R Core Team (2021). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/) as described and published previously [14]. Results of sensitivity and specificity were compared using the Wald test statistic to an alpha error of 5% and two-tailed McNemar test with Holm correction. Results for positive and negative predictive value (PPV, NPV) were compared using the Wald test statistic to an alpha error of 5% and weighted generalized score (WGS) test statistic.
Results
Patient characteristics
A total of 563 patients with mpMRI before RA-TB were included in this study. PSA value at time of MRI was 9.8 ± 6.4 ng/mL (range 1.2–39.0 ng/mL, 63.2% below PSA 10 ng/mL). No significant differences were found between PI-RADS groups for PSA density, except between PI-RADS 5 and PI-RADS 3 (mean rank difference 66.3, H = 11.8, p = 0.02).
Biopsy results
337 of 563 patients (59.9%) showed cancer in at least 1 biopsy core from 18 cores. 321 of 337 patients were positive at the site of the target lesion. 16 carcinomas were found off-target only. 68 cases were positive only in the target biopsy but not in systemic biopsy. In 253 cases, both targeted biopsy and off-target biopsy were positive. In 230 of 337 tumors, the target biopsy showed the highest Gleason score compared to 107 cases with highest Gleason score in off-target biopsy, 14 of those 107 cases resulted in an upgrade to a csPCa while targeted biopsy showed a Gleason 6 score.
Seven cases of PI-RADS 3 findings (all TZ) and eight cases of PI-RADS 4 cases (2 TZ, 6 PZ, none of them with contrast enhancement) were positive off-target only. From those, three cases with PI-RADS 3 showed csPCa, while none of the PI-RADS 4 cases was a csPCa. An overview of the biopsy results is provided in Fig. 1.
No severe complications, defined as Clavien–Dindo > Grade I were observed after biopsy, in 65 cases mild complications with transient urinary retention were documented.
MRI results
The mean prostate volume (Table 1) derived from MRI measurements was 45 ± 22 ml, resulting in a PSA density of 0.25 ± 0.19, with 199 patients ≤ 0.15 and 364 patients ≥ 0.15. 255 of the later were diagnosed with cancer on RA-TB (243 with csPCa). Lesion size was 12 ± 5 mm (range 3 – 30 mm) with a lesion volume of 3.2 ± 2.5 cc (range 0.6–10.3 cc). Most lesions were found in the PZ (426) followed by the TZ (137). Time from MRI to biopsy was 33 ± 25 days (range 1 – 114).
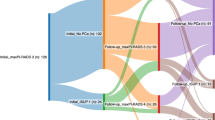
Flow charts for the results of mpMRI and bpMRI reading are summarized in Fig. 2. 29 out of 563 cases were classified as PI-RADS 2 (5.1%). Off-targeted biopsy was positive in one patient with non-suspicious mpMRI (PI-RADS 2, PSA 5.1 ng/ml, PSA density 0.2 ng/ml, 2/18 positive cores, Gleason score 4 + 3), resulting in a csPCa. A PI-RADS 5 score was given in 115 patients (20.4%). Of those, carcinomas were detected in 79.1% (91 out of 115). Here, 94.5% were classified as csPCa.
Flowchart for mpMRI reading (top) and bpMRI reading (bottom) with number and percentage of PI-RADS scores. Presence of cancer and number of clinically significant prostate cancer (csPCa) are outlined. PI-RADS 3 lesions located in the peripheral zone (PZ) that were positive in dynamic contrast-enhanced imaging (DCE +) are delineated
mpMRI and bpMRI results
95 lesions (16.9% of all lesions) were scored as PI-RADS 3 in mpMRI reading. 31 of those cases were cancer-positive after biopsy (32.6%) with 3 cases of csPCa (3.2%).
PI-RADS 4 was selected in 324 (57.6%) cases with biopsy-proven carcinoma in 214 occasions (66.1%), 189 (58.3%) of those with csPCa.
41 cases were upgraded from PI-RADS 3 to PI-RADS 4 due to contrast enhancement of PZ lesions in mpMRI. Of those, 6 cases were cancer-positive on biopsy, all of them with a csPCa. Thirty-five lesions resulted in benign histology, such as prostatitis.
In bpMRI reading, 136 lesions (24.2% of all lesions) were scored as PI-RADS 3. 37 of those cases were cancer-positive after biopsy (27.2%) with 9 cases of csPCa (6.6%). PIRADS 4 was selected in 283 (50.3%) cases, biopsy-proven carcinoma in 208 occasions (73.5%), csPCa in 183 (64.7%).
Performance of bpMRI and mpMRI
As no contrast agent was applied, six PI-RADS 3 lesions in the PZ were not upgraded to PI-RADS 4 in bpMRI. All of them were csPCa. With mpMRI, 35 cases were upgraded to PI-RADS 4 due to positive DCE with no proof of PCa in histology (10.8% of all PI-RADS 4 cases, Fig. 1).
Taken together, these findings resulted in a sensitivity of 99.0% (mpMRI) and 97.1% (bpMRI). McNemar’s chi-squared test of 4.2 revealed significant differences between mpMRI and bpMRI (p < 0.001). Specificity was 47.5% (mpMRI) and 61.2% (bpMRI), which was also significantly different between tests (McNemar’s chi-squared test of 33.0, p < 0.001). The presence of a csPCa in histology was considered the gold standard. Additional values for PPV and NPV are summarized in Table 2.
Discussion
Multiparametric MRI has undoubtedly gained momentum as a diagnostic tool to detect prostate cancer and is currently recognized as the best imaging method for assessing primary prostate cancer. While the current PI-RADSv2.1 protocol demands the use of contrast-enhanced sequences, several recent investigations have evaluated the use of bpMRI without DCE for several indications [15, 16].
Here, we challenged the need for mpMRI in a patient cohort with a clinical suspicion of prostate cancer that was scheduled for an MRI before histopathology from RA-TB was obtained. MRI examinations were independently read as bpMRI or mpMRI in separate sessions. In our study, all mpMRI scans were performed on 3 T scanners, adhering to the PI-RADSv2.1 protocol, undertaken by trained prostate-MRI technologists.
In this retrospective study in patients with high-risk for PCa comparing mpMRI including contrast agent and bpMRI, a sensitivity/specificity/PPV/NPV for mpMRI of 99.0%/47.5%/69.5%/97.6% and a sensitivity/specificity/PPV/NPV of 97.1%/61.2%/75.1%/94.6% for bpMRI were found.
The high imaging standards and experienced readers helped limit the proportion of “uncertain” (PI-RADS 3) diagnoses. PI-RADS 3 was present in 16.9% for mpMRI in our study, versus 28% and 21%, in the promis and precision trail [17, 18], respectively. These multicenter studies were performed on 1.5 T or 1.5 and 3 T scanners, respectively, resulting in more variability in acquisition parameters and multiple readers which might explain the slight difference in PI-RADS 3 findings.
The number of PI-RADS 3 lesions detected that were positive on biopsy was 32.6%, 9.7% of those had csPCa. This is in line with other studies [19,20,21] demonstrating the overall low rate of csPCa among the PI-RADS 3 lesions, even in a study collective with clinical high prevalence of PCa.
The number of PI-RADS 3 findings with positive DCE leading to an upgrade to PI-RADS 4 with subsequent diagnosis of a csPCa was limited in this cohort (n = 6) compared to 36 cases upgraded to PI-RADS 4 due to positive DCE with no evidence of cancer upon biopsy. This number is relatively low compared to other studies, which might be due to the highly selected cohort of patients scheduled for biopsy. A recent study by Sherrer et al. [22] also found a relative low number of DCE-positive findings that would have been missed on bpMRI.
Robot-assisted transperineal fusion prostate biopsy was used in all cases for biopsy resulting in a very standardized biopsy procedure that is not commonly used in comparable studies [23, 24]. The number of positive cores as reported by the pathologists was used. We did not include the core length or the percentage of infiltration in our analysis [25].
This paper makes contributions to existing literature with controversy regarding the use of a bpMRI approach in men with suspicion of PCa. While some publications demonstrate the necessity for DCE in line with current recommendations of the PI-RADS committee [26, 27], a recent meta-analysis concludes that bpMRI is feasible csPCa detection [15]. The need for defined indications, however, is emphasized in this evaluation. They conclude that the broad variability in sensitivity and specificity for detection of csPCa in the analyzed studies might primarily be influenced by reader experience and the disease prevalence in the patients included.
Our study provides evidence that the bpMRI pathway demonstrates similar sensitivity and better specificity compared to the mpMRT pathway in men with high clinical suspicion for PCa. Sensitivity/specificity was 99.0/47.5% for mpMRI and 97.1/60.2% for bpMRI in our study regarding detection of csPCa. A wide range in sensitivity (45–95%) and specificity (45–100%) for bpMRI are reported in a recent meta-analysis for the detection of PCa [28], as well as for csPCa [29] with sensitivity between 44 and 100%) and specificity (15–97%). With high sensitivity values and fair specificity values, our results reflect the preselected cohort with a disease prevalence of 59.9%.
Sensitivity and NPV are high in mpMRI in our study which can be explained by the high-risk population for PCa. Due to the high clinical suspicion for PCa, all patients underwent biopsy irrespective of the MRI findings as mentioned in the manuscript. Moreover, very high sensitivity and NPV are not uncommon for high-risk populations as demonstrated in several studies [29,30,31,32,33]. Thus, Cuocolo et al. emphasize the need for a standardized imaging protocol and prospective studies for validation of bpMRI as current studies are very heterogenous in design [29]. The PI-RADS steering committee has also recognized the increasing demand for prostate MRI in general and recently discussed possible applications for bpMRI considering high-quality imaging, expert interpretation quality and clinical risk stratification [16]. The need for prospective studies with biopsy decisions made according to MRI without DCE and definite clinical and operational benefits is again highlighted.
This study has several limitations. First, this is a retrospective single center study with a preselected clinical population scheduled for biopsy with no randomization. Second, RA-GB was planned due to clinical suspicious of PCa, regardless of mpMRI results, including PI-RADS 2 scores. This may be considered as potential bias inflating the PCa detection rates in off-target-biopsy. Another potential limitation of this study design is a possible change of the index lesion when bpMRI is applied compared to mpMRI as no update to PI-RADS 4 in PZ lesions with positive DCE is applicable. This was not the case in the cohort of investigation as no competing lesion was present in these cases and PI-RADS 3 lesions were also considered for biopsy. However, this will be of relevance in a prospective randomized trial with limitation of biopsy to ≥ PI-RADS 4 lesions. The examinations in this study were acquired before the PI-RADS v2.1 update in 2019, however, the MRI examination protocol fully met the technical requirements proposed there. We did not evaluate the family history on prostate cancer for this study which can limit further evaluation of the investigated population. Regarding histological evaluation of the biopsies, cancer core length and percentage of infiltration were not assessed in this study.
In summary, bpMRI demonstrated better specificity and positive predictive value while mpMRI showed slightly better sensitivity and negative predictive value in a real-world population with high risk for prostate cancer scheduled for RA-TB.
Conclusion
In patients with a suspected PCa and elevated PSA scheduled for biopsy, mpMRI demonstrated slightly better sensitivity while specificity was superior in bpMRI for the detection of csPCa in a cohort of high-risk patients for PCa. We conclude that bpMRI is sufficient for planning and performance of targeted biopsy in patients with suspected PCa in biopsy naïve patients undergoing first RA-TB biopsy. These findings need to be confirmed in prospective, randomized studies before mpMRI can be recommended in selected cases.
Availability of data and material
All relevant data are included in the manuscript.
Code availability
Not applicable.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer Statistics, 2021. CA Cancer J Clin 71(1):7–33. https://doi.org/10.3322/caac.21654
Lee DJ, Recabal P, Sjoberg DD, Thong A, Lee JK, Eastham JA et al (2016) Comparative effectiveness of targeted prostate biopsy using magnetic resonance imaging ultrasound fusion software and visual targeting: a prospective study. J Urol 196(3):697–702. https://doi.org/10.1016/j.juro.2016.03.149
Bergamini S, Bellei E, Reggiani Bonetti L, Monari E, Cuoghi A, Borelli F et al (2014) Inflammation: an important parameter in the search of prostate cancer biomarkers. Proteome Sci 12:32. https://doi.org/10.1186/1477-5956-12-32
Jyoti R, Jina NH, Haxhimolla HZ (2016) In-gantry MRI guided prostate biopsy diagnosis of prostatitis and its relationship with PIRADS vol 2 based score. J Med Imaging Radiat Oncol 61(2):212–215. https://doi.org/10.1111/1754-9485.12555
Srigley JR (2004) Benign mimickers of prostatic adenocarcinoma. Mod Pathol 17(3):328–348. https://doi.org/10.1038/modpathol.3800055
Kaufmann S, Bedke J, Gatidis S, Hennenlotter J, Kramer U, Notohamiprodjo M et al (2015) Prostate cancer gene 3 (PCA3) is of additional predictive value in patients with PI-RADS grade III (intermediate) lesions in the MR-guided re-biopsy setting for prostate cancer. World J Urol 34(4):509–515. https://doi.org/10.1007/s00345-015-1655-8
Stanzione A, Imbriaco M, Cocozza S, Fusco F, Rusconi G, Nappi C et al (2016) Biparametric 3T magnetic resonance imaging for prostatic cancer detection in a biopsy-naïve patient population: a further improvement of PI-RADS v2? Eur J Radiol 85(12):2269–2274. https://doi.org/10.1016/j.ejrad.2016.10.009
Hansford BG, Peng Y, Jiang Y, Vannier MW, Antic T, Thomas S et al (2015) Dynamic contrast-enhanced MR Imaging curve-type analysis: is it Helpful in the differentiation of prostate cancer from healthy peripheral zone? Radiology 275(2):448–457. https://doi.org/10.1148/radiol.14140847
Scialpi M, Prosperi E, D’Andrea A, Martorana E, Malaspina C, Palumbo B et al (2017) Biparametric versus multiparametric MRI with Non-endorectal Coil at 3T in the detection and localization of prostate cancer. Anticancer Res 37(3):1263–1271. https://doi.org/10.21873/anticanres.11443
Girouin N, Mège-Lechevallier F, Tonina Senes A, Bissery A, Rabilloud M, Maréchal JM et al (2007) Prostate dynamic contrast-enhanced MRI with simple visual diagnostic criteria: is it reasonable? Eur Radiol 17(6):1498–1509. https://doi.org/10.1007/s00330-006-0478-9
Kim CK, Park BK, Lee HM, Kwon GY (2007) Value of diffusion-weighted imaging for the prediction of prostate cancer location at 3T using a phased-array coil: preliminary results. Invest Radiol 42(12):842–847. https://doi.org/10.1097/RLI.0b013e3181461d21
Greer MD, Shih JH, Lay N, Barrett T, Kayat Bittencourt L, Borofsky S et al (2017) Validation of the dominant sequence paradigm and role of dynamic contrast-enhanced imaging in PI-RADS version 2. Radiology 285(3):859–869. https://doi.org/10.1148/radiol.2017161316
Kaufmann S, Mischinger J, Amend B, Rausch S, Adam M, Scharpf M et al (2017) First report of robot-assisted transperineal fusion versus off-target biopsy in patients undergoing repeat prostate biopsy. World J Urol 35(7):1023–1029. https://doi.org/10.1007/s00345-016-1970-8
Roldán-Nofuentes JA (2020) Compbdt: an R program to compare two binary diagnostic tests subject to a paired design. BMC Med Res Methodol 20(1):143. https://doi.org/10.1186/s12874-020-00988-y
Cuocolo R, Verde F, Ponsiglione A, Romeo V, Petretta M, Imbriaco M et al (2021) Clinically significant prostate cancer detection with biparametric MRI: a systematic review and meta-analysis. AJR Am J Roentgenol 216(3):608–621. https://doi.org/10.2214/ajr.20.23219
Schoots IG, Barentsz JO, Bittencourt LK, Haider MA, Macura KJ, Margolis DJA et al (2021) PI-RADS committee position on mri without contrast medium in biopsy-naive men with suspected prostate cancer: narrative review. AJR Am J Roentgenol 216(1):3–19. https://doi.org/10.2214/ajr.20.24268
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389(10071):815–822. https://doi.org/10.1016/s0140-6736(16)32401-1
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH et al (2018) MRI-Targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 378(19):1767–1777. https://doi.org/10.1056/NEJMoa1801993
Hermie I, Van Besien J, De Visschere P, Lumen N, Decaestecker K (2019) Which clinical and radiological characteristics can predict clinically significant prostate cancer in PI-RADS 3 lesions? A retrospective study in a high-volume academic center. Eur J Radiol 114:92–98. https://doi.org/10.1016/j.ejrad.2019.02.031
Yang S, Zhao W, Tan S, Zhang Y, Wei C, Chen T et al (2020) Combining clinical and MRI data to manage PI-RADS 3 lesions and reduce excessive biopsy. Transl Androl Urol 9(3):1252–1261. https://doi.org/10.21037/tau-19-755
Stabile A, Dell’Oglio P, De Cobelli F, Esposito A, Gandaglia G, Fossati N et al (2018) Association between prostate imaging Reporting and Data System (PI-RADS) score for the index lesion and multifocal, clinically significant prostate cancer. Eur Urol Oncol 1(1):29–36. https://doi.org/10.1016/j.euo.2018.01.002
Sherrer RL, Glaser ZA, Gordetsky JB, Nix JW, Porter KK, Rais-Bahrami S (2019) Comparison of biparametric MRI to full multiparametric MRI for detection of clinically significant prostate cancer. Prostate Cancer Prostatic Dis 22(2):331–336. https://doi.org/10.1038/s41391-018-0107-0
Tamada T, Kido A, Yamamoto A, Takeuchi M, Miyaji Y, Moriya T et al (2021) Comparison of biparametric and multiparametric MRI for clinically significant prostate cancer detection with PI-RADS version 2.1. J Magn Reson Imaging 53(1):283–291. https://doi.org/10.1002/jmri.27283
Xu L, Zhang G, Shi B, Liu Y, Zou T, Yan W et al (2019) Comparison of biparametric and multiparametric MRI in the diagnosis of prostate cancer. Cancer Imaging 19(1):90. https://doi.org/10.1186/s40644-019-0274-9
Grignon DJ (2018) Prostate cancer reporting and staging: needle biopsy and radical prostatectomy specimens. Mod Pathol 31(S1):S96-109. https://doi.org/10.1038/modpathol.2017.167
Ullrich T, Quentin M, Arsov C, Laqua N, Abrar D, Hiester A et al (2020) Value of Dynamic Contrast-Enhanced (DCE) MR imaging in peripheral lesions in PI-RADS-4 Patients. Rofo 192(5):441–447. https://doi.org/10.1055/a-1020-4026
Padhani AR, Weinreb J, Rosenkrantz AB, Villeirs G, Turkbey B, Barentsz J (2019) Prostate imaging-reporting and data system steering committee: PI-RADS v2 status update and future directions. Eur Urol 75(3):385–396. https://doi.org/10.1016/j.eururo.2018.05.035
Woo S, Suh CH, Kim SY, Cho JY, Kim SH, Moon MH (2018) Head-to-head comparison between biparametric and multiparametric MRI for the diagnosis of prostate cancer: a systematic review and meta-analysis. AJR Am J Roentgenol 211(5):W226–W241. https://doi.org/10.2214/ajr.18.19880
Cuocolo R, Stanzione A, Rusconi G, Petretta M, Ponsiglione A, Fusco F et al (2018) PSA-density does not improve bi-parametric prostate MR detection of prostate cancer in a biopsy naïve patient population. Eur J Radiol 104:64–70. https://doi.org/10.1016/j.ejrad.2018.05.004
Fascelli M, Rais-Bahrami S, Sankineni S, Brown AM, George AK, Ho R et al (2016) Combined biparametric prostate magnetic resonance imaging and prostate-specific antigen in the detection of prostate cancer: a validation study in a biopsy-naive patient population. Urology 88:125–134. https://doi.org/10.1016/j.urology.2015.09.035
Merisaari H, Jambor I, Ettala O, Boström PJ, Montoya Perez I, Verho J et al (2019) IMPROD biparametric MRI in men with a clinical suspicion of prostate cancer (IMPROD Trial): Sensitivity for prostate cancer detection in correlation with whole-mount prostatectomy sections and implications for focal therapy. J Magn Reson Imaging 50(5):1641–1650. https://doi.org/10.1002/jmri.26727
van der Leest M, Israël B, Cornel EB, Zámecnik P, Schoots IG, van der Lelij H et al (2019) High diagnostic performance of short magnetic resonance imaging protocols for prostate cancer detection in biopsy-naïve men: the next step in magnetic resonance imaging accessibility. Eur Urol 76(5):574–581. https://doi.org/10.1016/j.eururo.2019.05.029
Van Nieuwenhove S, Saussez TP, Thiry S, Trefois P, Annet L, Michoux N et al (2019) Prospective comparison of a fast 1.5-T biparametric with the 3.0-T multiparametric ESUR magnetic resonance imaging protocol as a triage test for men at risk of prostate cancer. BJU Int 123(3):411–420. https://doi.org/10.1111/bju.14538
Funding
Open Access funding enabled and organized by Projekt DEAL. No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
WT: protocol/project development, data collection or management, data analysis, manuscript writing, and manuscript editing. SM: data collection or management, data analysis, and manuscript writing. SR: data collection or management, and manuscript editing. MS: data collection or management and manuscript editing. KN: protocol/project development and manuscript editing. AS: protocol/project development and manuscript editing. SKr: protocol/project development and manuscript editing. JB: protocol/project development, data analysis, manuscript writing, and manuscript editing. SKa: protocol/project development, data collection or management, data analysis, manuscript writing, and manuscript editing. This work is part of the MD thesis of Simone Moser.
Corresponding author
Ethics declarations
Conflict of interest
The authors state that they have no conflicts of interest.
Ethics approval
This retrospective study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The local ethics committee approved this study.
Consent for publication
No patient images are included in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thaiss, W.M., Moser, S., Hepp, T. et al. Head-to-head comparison of biparametric versus multiparametric MRI of the prostate before robot-assisted transperineal fusion prostate biopsy. World J Urol 40, 2431–2438 (2022). https://doi.org/10.1007/s00345-022-04120-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-022-04120-1