Abstract
The productivity of poplar is associated with large nitrogen (N) requirements. Exogenous arbuscular mycorrhizal fungi (AMF) show potential for use as bio-fertilizers. Understanding the interaction between N and exogenous AMF has theoretical and practical significance for poplar plantation. A pot experiment was conducted to assess the effects of N and exogenous Rhizophagus irregularis on plant growth, nutrient uptake, photosynthesis, water status, and leaf anatomical properties of Populus × canadensis ‘Neva’ in natural soil. The results showed that N fertilization increased plant growth, net photosynthesis, water status and the conduit diameter of midribs. The concentrations of carbon (C) and N in leaves were increased, but the phosphorus (P) concentration was decreased by N fertilization. The effectiveness of exogenous R. irregularis varied under different N levels. Under low N levels, exogenous R. irregularis-inoculated plants grew faster and exhibited superior photosynthetic capacity, water status and leaf conduit diameters than non-inoculated plants. Under high N levels, C, N and P concentrations were enhanced by exogenous R. irregularis inoculation. Furthermore, the average conduit diameter of midribs presented a significant positive correlation with plant growth parameters, photosynthesis, relative water content (RWC) and leaf C and N concentrations. It was concluded that exogenous R. irregularis exerted the strongest positive effects under low N levels by promoting plant growth and photosynthesis, and the fungus promoted plant nutrition decoupled from the level of N fertilization. Moreover, the improvement of plant physiological traits due to N fertilization or exogenous R. irregularis inoculation was accompanied by changes in internal anatomical properties.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Populus species and their hybrids are cultivated worldwide due to their high economic value (Hacke and others 2010). Populus × canadensis (a hybrid of P. nigra × P. deltoides) ‘Neva’ is widely planted in China as an intensively managed woody plant species (Liu and others 2014). In many forest tree plantations, plant productivity is constrained by a lack of accessible nitrogen (N) (Hacke and others 2010). N fertilization is widely used to achieve high productivity (Cooke and others 2005). However, the fertilization strategy used by growers is often more heavily based on economic pressure than agronomic pressure (Chu and others 2007). Unfortunately, improper fertilization is widespread (Chu and others 2007). Excess N can generate serious environmental pollution and economic waste (Li and Korpelainen 2015).
Soil microorganisms can influence plant resource capture and can be used as substitutes for chemical fertilizers to maintain productivity (Dodd and Ruiz-Lozano 2012). Among rhizosphere microorganisms, arbuscular mycorrhizal fungi (AMF) form symbiotic relationships with most land plant species and play a vital role in the stress tolerance and water and nutrient uptake of host plants (Fellbaum and others 2012; Smith and Read 2008). AMF acquire carbohydrates from their host plants and provide plants with mineral nutrients in return, such as phosphorus (P) and N (Fellbaum and others 2012; Kiers and others 2011). Recently, considerable attention has been given to the use of exogenous AMF as biofertilizers due to their positive effect on plant nutrition (Pellegrino and others 2012). The positive effects of exogenous AMF on biomass production or nutrient uptake have been observed in several plant species, such as Zea mays (Li and others 2012), Medicago sativa (Pellegrino and others 2012), Manihot esculenta Crantz (Ceballos and others 2013), P. × canadensis (Liu and others 2014) and Hedysarum coronarium (Labidi and others 2015).
The effects of AMF on plant growth are not invariant and can be altered by soil nutrient conditions (Treseder 2004), and contradictory responses to AMF inoculation have been observed for plant growth after N application (Johnson 2010). Fonseca and others (2001) observed that the effects of inoculation on the plant and shoot dry mass of Sorghum bicolor were increased by supplying N. In contrast, Johansen and others (1994) found that the effects of inoculation on the plant dry weight of Cucumis sativus were strongest under low N status. Thus, fertilization might have crucial impacts on the effectiveness of AMF.
Plant physio-biochemical and anatomical traits can be altered by environmental changes and are considered good indicators for assessing plant health. The effects of N fertilization on physiological responses in poplars have been extensively studied and include improvements in growth (Cooke and others 2005), gas exchange parameters (Yu and others 2010), and water and nutrient uptake (Wu and others 2008). Previous studies involving a range of different fungal and host plant species (including poplars) have indicated that AMF have a positive effect on plant physiolocal processes (He and others 2016; Li and others 2015; Yang and others 2014). Anatomical traits were also affected by nutrient conditions, for example, wider conduits were observed in the stems of high N-fertilized poplar (Hacke and others 2010; Plavcová and others 2013). However, the xylem of the leaves is rarely studied despite the fact that leaf veins are responsible for the transport of water, mineral nutrients and hormones to leaves (Haberer and Kieber 2002; Ohashi-Ito and others 2014; Scarpella and Meijer 2004). To our knowledge, no data have been published regarding the effects of AMF on anatomical traits under different N levels.
Rhizophagus irregularis is a model fungus in AMF research and is widely used in laboratory and field studies (Ceballos and others 2013; Labidi and others 2015; Sýkorová and others 2012). It is also a high-quality AMF species that can increase the transfer of nutrients per unit of carbon to the plant (Engelmoer and others 2014; Kiers and others 2011). Previous work has indicated an improvement in drought tolerance of P. × canadensis by R. irregularis (Liu and others 2015) and a positive effect of exogenous R. irregularis and Glomus versiforme on the biomass and bioenergy production of P. × canadensis in unsterilized soil (Liu and others 2014). Nevertheless, the influence of N enhancement on the effectiveness of exogenous AMF remains unknown. In this study, a pot experiment was conducted to test the effects of N and exogenous R. irregularis on gas exchange, water status, nutrient status and leaf anatomy traits of P. × canadensis in natural soil. We hypothesized that (a) N fertilization would promote plant growth, photosynthesis, water status, nutrient status and leaf anatomical traits; (b) the growth of P. × canadensis could be enhanced by exogenous R. irregularis but the enhancement effect would vary between different N levels; and (c) improvements in plant growth and physiological status should be coupled with alterations of anatomical traits.
Materials and Methods
Plant Material and AMF Inoculum
Cuttings (15 cm in length) of hybrid poplar (P. × canadensis) were used as the plant material in this experiment, and were obtained from a base of P. × canadensis seedlings in Rougu country, Yangling City, Shaanxi Province, China. Permission to conduct the study on this site was provided by the owner of the nursery. The cuttings were surface sterilized with 75% (v/v) ethanol for 15 s and then rinsed three times with sterile distilled water for 10 s. After surface sterilization, the cuttings were directly planted into the pots.
The R. irregularis (Blaszk, Wubet, Renker & Buscot) Walker & Schüßler (BGC B109) used in this experiment was provided by the Institute of Plant Nutrition and Resources, Beijing Academy of Agriculture and Forestry Sciences, China. The AMF inoculum consisted of spores (50 spores g−1), hyphae, soil and infected root fragments.
Growth Conditions and Experimental Design
The growth substrate consisted of soil collected from the top layer (0–20 cm) of a field planted with poplars. The soil type was cinnamon, and the characteristics of the soil were as follows: 36.62 mg kg−1 available N, 11.71 mg kg−1 available P, 143.26 mg kg−1 available K, 18.19 g kg−1 organic matter, and pH 7.6 (soil:water ratio of 1.0:2.5 w/v). The pots (22.5 × 22.5 cm) were randomly arranged and were each filled with 4 kg of air-dried and sieved soil (2-mm sieve).
The experiment was conducted in the greenhouse of Northwest A&F University under a temperature of 25–35 °C, a daylight cycle of 12 h, and relative air humidity of 55–78%. The experiment consisted of a two-factorial design: exogenous R. irregularis inoculation (exogenous R. irregularis-inoculated plants and non-inoculated plants) and N fertilization (5 NH4NO3 levels). Each treatment had 30 replicates. Half of the total pots were inoculated with 20 g of inoculum, and the remainder received 20 g of autoclaved inoculum (121 °C, 2 h) with a microbial wash (1-µm nylon mesh) from the non-autoclaved inoculum to provide a usual microbial population free of AM propagules. The poplar cuttings were planted in early April 2013. Symbiosis was established after 3 months of growth (the analytical method will be discussed in the “Mycorrhizal colonization” section), and each group was then randomly assigned into 5 subgroups that received 0, 1, 5, 10, or 15 mmol L−1 NH4NO3 (N0, N1, N2, N3, and N4, respectively) every other day. The fertilization treatment was applied for 28 days. The plants were maintained at field capacity throughout the experiment. One month after fertilization, the growth parameters of P. × canadensis were measured.
Mycorrhizal Colonization
Fresh roots were first stained with trypan blue according to the method of Phillips and Hayman (1970). Mycorrhizal colonization was then assessed under an Olympus BX43F light microscope (200 × magnification; Olympus, Tokyo, Japan) using the gridline intersection method (McGonigle and others 1990).
Plant Growth Parameters and Leaf Nutrient Analysis
The plant height and stem diameter were measured with tape (Swordfish, China) and vernier calipers (ECV150C, China), respectively (Yang and others 2014). At the end of the experiment, the leaves, stems and roots were harvested and immediately dried at 80 °C to a constant weight to determine dry biomass (Liu and others 2014).
Dry leaves were fully ground and homogenized for nutrient analysis. The leaf N concentration was measured from 0.2 g of dried, ground leaves by the Kjeldahl method on a Kjeltec™ 8400 Analyzer Unit (FOSS-Tecator, Hoganas, Sweden) (Kong and others 2015). The leaf carbon (C) concentration was measured from 4 mg of dried, ground leaves using a Liqui TOCII analyzer (Elementar, Germany) (Liu and others 2014). To measure the phosphorus (P) concentrations, 1 g of dried, ground leaves was digested with HNO3-HClO4, and the P concentration was measured by the vanadomolybdate method (Yang and others 2014).
Gas Exchange and Water Status
Each leaf of a plant was numbered according to a leaf plastochron index (LPI) number, with the most recently expanded leaf (approximately 2 cm long) being numbered LPI 0 (Larson and Isebrands 1971). Gas exchange was measured using the leaves of LPI 6 between 08:00 and 11:30 h in the greenhouse. The net photosynthetic rate (P N), stomatal conductance (g s), intercellular CO2 concentration (C i) and transpiration rate (E) were measured with a Li-Cor 6400 portable photosynthesis system (Li-Cor Inc., Lincoln, NE, USA) and red/blue LED. All of the measurements were conducted with the following parameters: photosynthetically active irradiation of 1000 μmol m−2 s−1, CO2 concentration of 400 cm3 m−3, and leaf temperature of 25 °C.
The relative water content (RWC) was measured by the method of Li and others (2015) and was calculated as follows: RWC = [(FW − DW) / (TW − DW)] × 100, where FW, DW and TW represent the sample fresh weight, dry weight and turgid weight, respectively. TW was determined by weighing the leaves after placement in distilled water for 24 h. The leaves were then dried at 70 °C until constant weight to determine DW. The intrinsic water use efficiency (WUEi) was calculated as follows: WUEi = P N/g s (Monclus and others 2006).
Leaf Anatomy
To analyze leaf anatomy, the midveins of the leaves of LPI 6 were fixed in FAA (formalin:glacial acetic acid:70% ethanol = 5:5:90) immediately after sampling. After washing in distilled water several times, transverse sections of six individual leaves per treatment were prepared with razor blades. The sections were stained with safranin and fast green according to the method of Dié and others (2012) with minor modifications. The sections were dehydrated through a gradient series of ethanol (30% ethanol for 3 min, 50% ethanol for 3 min). To allow anatomical observations to be performed, the sections were stained with 1% safranin (5 min) and 0.5% fast green (1 min). After staining, the sections were dehydrated through a gradient series of ethanol (95% ethanol for 1 min, 100% ethanol for 1 min) and cleared in xylene (3 min). Next, the sections were mounted on glass slides with gum. Photographs were taken with a digital camera attached to an Olympus BX43F microscope (Olympus, Tokyo, Japan) at a magnification of 200×. The conduit diameter was measured using a software tool provided in Image-Pro Plus (version 6.1, Media Cybernetics, Silver Spring, MD) and the mean diameter of conduit in the image was calculated. Thirty conduit diameters were observed for each leaf.
Data Analysis
Statistical analyses were performed using SPSS (version 17.0 for Windows). The data were tested for normality and homogeneity. A two-way ANOVA was employed to examine the effects of N fertilization and exogenous R. irregularis inoculation and their interactions on the measured parameters at p ≤ 0.05. Duncan’s multiple range test was performed to determine the group(s) that were different from the others at p ≤ 0.05. The correlation analyses between anatomic trait and morpho-physiological parameters were tested using Pearson’s correlation coefficients with a Bonferroni-corrected p value of 0.05/16 = 0.0031.
Results
AMF Colonization
According to the statistical results presented in Table 1, AMF colonization was significantly affected by N fertilization and exogenous R. irregularis inoculation (p ≤ 0.01, two-way ANOVA). N fertilization increased AMF colonization in both the exogenous R. irregularis-inoculated and non-inoculated plants (Table 2). AMF colonization was significantly higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants at each N level (p ≤ 0.05, Duncan’s test).
Plant Growth
Plant height and the stem diameter were significantly affected by N fertilization and exogenous R. irregularis inoculation (p ≤ 0.01, two-way ANOVA, Table 1). As the level of N fertilization increased, the plant height and stem diameter of both the exogenous R. irregularis-inoculated and non-inoculated plants increased and tended to be stable (Table 2). Plant height was significantly higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants under low (0, 1 mmol L−1 NH4NO3) and intermediate N levels (5 mmol L−1 NH4NO3) (p ≤ 0.05, Duncan’s test), and stem diameter was significantly higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants under low N levels (p ≤ 0.05, Duncan’s test). No significant differences in plant height or stem diameter were observed between the exogenous R. irregularis-inoculated and non-inoculated plants under the other tested N levels.
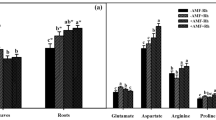
N fertilization had significant effects on leaf dry weight, stem dry weight, root dry weight and total dry weight (p ≤ 0.01, two-way ANOVA, Table 1). Exogenous R. irregularis had significant effects on stem dry weight, root dry weight and total dry weight (p ≤ 0.05, two-way ANOVA, Table 1). Leaf dry weight, stem dry weight and total dry weight increased with N fertilization, whereas root dry weight first increased and then decreased (Fig. 1). At low N levels, the total dry weight of the exogenous R. irregularis-inoculated plants was significantly higher than that of non-inoculated plants (p ≤ 0.05, Duncan’s test).
Effects of N fertilization and exogenous R. irregularis inoculation on dry weight of leaf (a), dry weight of stem (b), dry weight of root (c), total dry weight (d) of P. × canadensis Values are presented as means ± SD (n = 6). Means followed by the same letter do not differ significantly at p ≤ 0.05 by Duncan test
Nutrient Element Concentrations in the Leaves
N fertilization increased C and N concentrations but decreased the P concentration in both the exogenous R. irregularis-inoculated and non-inoculated plants (Table 3). No significant differences in the C, N or P concentrations were observed between the exogenous R. irregularis-inoculated and non-inoculated plants under low N levels (p > 0.05, Duncan’s test). However, higher C, N and P concentrations were observed in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants under high N concentrations (10, 15 mmol L−1 NH4NO3) (p ≤ 0.05, Duncan’s test). The results of two-way ANOVA indicated that the C concentration was significantly affected by N fertilization, exogenous R. irregularis inoculation and their interaction (all p ≤ 0.01, two-way ANOVA); the N concentration was significantly affected by N fertilization and exogenous R. irregularis inoculation (all p ≤ 0.01, two-way ANOVA); and the P concentration was significantly affected by N fertilization and the interaction between N fertilization and exogenous R. irregularis inoculation (all p ≤ 0.01, two-way ANOVA).
Gas Exchange and Water Status
To test the physiological status of the P. × canadensis plants, gas exchange and water status were measured. N fertilization enhanced P N, g s, E, WUEi and RWC but decreased C i in both the exogenous R. irregularis-inoculated and non-inoculated plants (Table 4). Under low N levels P N was significantly higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants (p ≤ 0.05, Duncan’s test). At each N level, E was significantly higher and C i significantly lower in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants (p ≤ 0.05, Duncan’s test). Inoculation with exogenous R. irregularis led to increases in WUEi (29.3, 29.3, 13.8, 10.0, and 3.8%) and RWC (4.8, 4.6, 4.0, 2.7, and 1.0%) at each N level (0, 1, 5, 10, and 15 mmol L−1 NH4NO3, respectively). The results of two-way ANOVA results indicated that P N, C i, g s, E, RWC and WUEi were significantly affected by N fertilization and exogenous R. irregularis inoculation (all p < 0.01, two-way ANOVA).
Leaf Anatomy
The leaf conduit diameter was measured to determine whether anatomical alterations occurred in the plant leaves in response to N fertilization and exogenous R. irregularis inoculation. The average conduit diameter increased with N fertilization from 9.1 to 14.1 µm and from 10.2 to 14.3 µm in the non-inoculated plants and exogenous R. irregularis-inoculated plants, respectively (Figs. 2, 3). The average conduit diameter was significantly greater in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants under low N levels (p ≤ 0.05, Duncan’s test). The relative frequency of the conduit diameter was measured to identify any trends in the changes in conduit size under the experimental treatment. Both N fertilization and exogenous R. irregularis inoculation reduced the number of smaller conduits (0–8 μm in diameter) and increased the number of larger conduits (16–32 μm in diameter) (Fig. 4). The results of two-way ANOVA indicated that the average conduit diameter was significantly affected by N fertilization and exogenous R. irregularis inoculation (all p ≤ 0.01, two-way ANOVA).
The average conduit diameter was significantly and positively correlated with AMF colonization, the growth of plant height, the growth of stem diameter, leaf dry weight, stem dry weight, total dry weight, leaf C concentration, leaf N concentration, P N, C i, E and RWC (Table 5; p ≤ 0.0031, Pearson’s correlation coefficients with Bonferroni correction). A significant negative correlation of the average conduit diameter with leaf P concentration and g s was observed (Table 5; p ≤ 0.0031, Pearson’s correlation coefficients with a Bonferroni correction). The correlations of the average conduit diameter with root dry weight and WUEi were not significant (Table 5; p > 0.0031, Pearson’s correlation coefficients with Bonferroni correction).
Discussion
The results of this study showed that N fertilization significantly promoted the growth of P. × canadensis and influenced the effectiveness of exogenous R. irregularis. The effects of exogenous R. irregularis on plant growth, photosynthetic capacity, water status and leaf anatomical properties were strongest under low N levels. Under high N levels, exogenous R. irregularis inoculation increased C, N and P concentrations in the leaves, although there was no significant difference in plant growth and photosynthetic capacity between the exogenous R. irregularis-inoculated and non-inoculated plants. The results also showed that alterations of plant physiological processes were paralleled by anatomical changes, which indicated that the growth performance of P. × canadensis was closely related to its internal anatomical properties.
The results of this study showed that exogenous R. irregularis promoted the AMF root colonization rate at each N level and that the AMF root colonization rate was increased by N fertilization in both the exogenous R. irregularis-inoculated and non-inoculated plants. Previous studies showed that the AMF colonization rate could be altered by N fertilization, but the effect of N fertilization was highly variable (Treseder 2004; Zhen and others 2014). Olsson and others (2005) observed that N applications could reduce AMF colonization, whereas Treseder and Allen (2002) obtained the opposite result. The results of the present study were consistent with the latter finding and suggested that N fertilization supplies AMF with abundant nutrients for growth, in which N is a necessary component of chitin (Bago and others 2004). Johansen and others (1994) indicated that N fertilization has the potential to increase the hyphal length of AMF by 20–50%. Moreover, N-fertilized plants exhibited high rates of photosynthesis and could provide sufficient fixed carbon to sustain high rates of mycorrhizal colonization.
In this study, the basic N concentration in the soil was not enough for continuous growth of poplar during the entire experimental period. Leaf N concentration was widely used to assess N status of poplar, although optimum leaf N concentration varied depending on poplar clones and growth stage (Coleman and others 2006; Hansen and others 1988; van den Driessche 1999). In general, when soil N availability is not sufficient to support plant growth, N fertilization increases leaf N concentration and plant growth is positively correlated with leaf N concentrations (van den Driessche 1999). However, there is a threshold concentration of leaf N beyond which no further increases in plant growth occur (van den Driessche 1999). This threshold concentration is considered the optimum leaf N concentration. Leaf N concentration is often compared with the threshold concentration to assess whether the growth of poplar is N limited or not (Hansen and others 1988). In this study, 13 mg kg−1 of leaf N concentration seemed to be the threshold concentration, as indicated by our finding that plant height and total dry weight was not affected by N fertilization when the concentration of NH4NO3 was higher than 5 mmol L−1 (Table 2; Fig. 1). Leaf N concentrations were lower than 13 mg kg−1 under low N levels (Table 2), suggesting that poplar growth was limited by N supply. Such kind of soil was used to better understand the effects of N fertilization and exogenous R. irregularis inoculation on plant growth performance. The results of this study were consistent with previous studies (Ceballos and others 2013; Cooke and others 2005; Labidi and others 2015; Sýkorová and others 2012; Yan and others 2015) showing that N fertilization facilitates plant growth and exogenous R. irregularis has a significant influence on plant growth (growth-promoting effects). However, the impact of exogenous R. irregularis on plant growth was influenced by N fertilization. The growth-promoting effects of exogenous R. irregularis were observed only under low N condition, with significantly higher values of plant height, stem diameter, and total dry weights being observed in the exogenous R. irregularis-inoculated plants than the non-inoculated plants. Rhizophagus irregularis is considered a high quality fungal species and can survive and persist in the field in the presence of a high diversity of native AMF in natural soil (Sýkorová and others 2012). A potential mechanism underlying the benefits of inoculation under low N levels may be the greater reliance of plants on AMF for nutrient uptake under nutrient-limited conditions (Smith and Read 2008). Such an interpretation was supported by our finding that there was no significant difference in plant growth between the exogenous R. irregularis-inoculated and non-inoculated plants when plant growth was not limited by soil N nutrition under high N levels. Under high N conditions, other environmental factors, such as the atmospheric CO2 concentration and temperature, might be the limiting factors for poplar growth (Tjoelker and others 1998).
In this study, the trend of the changes in leaf C concentrations was similar to that for N concentrations, which were increased with N fertilization and were significantly higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants under high N levels. These results were consistent with previous reports showing that N fertilization enhanced C and N concentrations in the leaves (Peuke 2009). Yasumura and others (2007) suggested that the majority of leaf N is located in proteins involved in photosynthesis; therefore, the carbon assimilation capacity of a plant is positively related to the N content in the leaves (Yan and others 2015). In agreement with previous findings (Peuke 2009), leaf P concentrations was observed to decrease with N application but decreased less in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants. The difference in the magnitude of the decrease likely occurred because of the role of exogenous R. irregularis inoculation in plant P uptake (Johnson 2010). Rhizophagus irregularis is high-quality AMF and can promote plant nutrient uptake (Engelmoer and others 2014; Kiers and others 2011). In this study, no significant differences in C, N and P concentrations were observed between the exogenous R. irregularis-inoculated and non-inoculated plants under low N levels, probably due to a dilution effect that the exogenous R. irregularis-inoculated plants were taller and displayed a larger total dry weight than the non-inoculated plants (Marulanda and others 2003). In addition, exogenous R. irregularis increased nutrient concentrations under high N levels, although growth-promoting effects were not observed. According to Smith and Smith (2011), the mycorrhizal pathway for nutrient uptake contributed to plant nutrition regardless of mycorrhizal growth responses. Smith and others (2004) performed an experiment using compartmented pots and 32P and suggested that R. irregularis contributes to plant P uptake regardless of mycorrhizal growth responses. Similar to P uptake, the mycorrhizal pathway was operational in N uptake from the soil to plants (Smith and Smith 2011). Therefore, the results of this study suggested that exogenous R. irregularis promoted plant nutrition decoupled from the level of N fertilization.
The results of the present study showed that exogenous R. irregularis promoted P N under low N levels. Positive effects of R. irregularis on plant photosynthetic capacity have also been observed in previous studies (He and others 2016, Liu and others 2014, 2015). An explanation for the present results might be that poplars rely more heavily on exogenous R. irregularis to absorb the finite-resources available under nutrient-poor conditions (Smith and Read 2008), such that P N and dry mass accumulation were higher in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants at low N levels. N fertilization provided the poplars with more available N for growth and might have reduced their dependence on exogenous R. irregularis, as P N and plant growth were not influenced by exogenous R. irregularis under high N levels. Nitrogen fertilization enhanced P N and g s but decreased C i, which was consistent with previous studies and suggested that the increase in P N due to N application was frequently related to increases in g s or the carboxylation capacity (Yu and others 2010). In addition, the lower C i observed in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants suggests that exogenous R. irregularis inoculation improved the ability of plants to assimilate CO2 (Zhu and others 2011).
In this study, WUEi, RWC and E were increased by N fertilization, and exogenous R. irregularis inoculation strengthened these increases under low N levels. This was in agreement with previous reports of a positive effect of N fertilization on plant water status (Feng and others 2012, Wu and others 2008). We speculated that exogenous R. irregularis improved the water status of poplars and thereby increased their ability to adapt to low N conditions. The enhancement of WUEi and RWC suggested that the plants’ water absorption capacity was improved, thus providing additional water for transpiration and plant growth (Wu and others 2008).
The leaf vascular system is responsible for the transport of water and nutrients to leaf cells (Price and Enquist 2007), and leaf veins play a vital role in the hydraulic and photosynthetic ability of leaves (Brodribb and others 2007). To our knowledge, little is known about the effect of exogenous AMF on anatomical traits of leaf xylem. In this study, poplar cuttings showed different anatomical responses to exogenous R. irregularis inoculation depending on N fertilization levels. Larger conduits of the leaf xylem of the exogenous R. irregularis-inoculated plants than those of the non-inoculated plants were observed only under low N conditions. N fertilization increased the average conduit diameter but decreased the effects of exogenous R. irregularis inoculation. N plays a vital role in the xylogenesis of hybrid poplar (Hacke and others 2010; Plavcová and others 2013), and wider conduits have been observed in poplars treated with high N fertilization rates (Plavcová and others 2013). The larger conduit diameters recorded in high N-fertilized or exogenous R. irregularis-inoculated plants suggested improved leaf vein systems. According to the Hagen-Poiseuille law, the hydraulic conductivity of a conduit should be in proportion to the lumen diameter to the fourth power (Tyree 2003). These anatomical differences in the leaves may be related to altered hydraulic properties that allow more efficient transport of water (Plavcová and others 2013) and soluble nutrients, thereby further promoting the photosynthesis of plants (Brodribb and others 2007). In this study, the larger conduit diameters and increased frequency of large conduits observed in high N-fertilized or exogenous R. irregularis-inoculated plants were likely the cause of higher transpiration rates and RWC.
Furthermore, the results showed that average conduit diameter exhibited a positive correlation with plant growth parameters (plant height, stem diameter, leaf dry weight, stem dry weight, total dry weight) and with physiological parameters such as RWC, leaf nutrients (C and N concentrations) and photosynthetic parameters (P N, C i, E), suggesting that the anatomical traits of leaves were closely associated with whole-plant functioning. Brodribb and others (2007) found that the leaf vein system influenced photosynthesis via its effect on leaf water flow. Woodruff and others (2008) observed that leaf anatomical traits were highly correlated with the height of Pseudotsuga menziesii. Studies on the anatomical traits of plants have indicated that anatomical traits may vary in response to the environment, and plants can adjust their phenotypes to preserve function under various conditions (Sultan 2000). In this study, the observed alterations of anatomical traits in response to N fertilization and exogenous R. irregularis inoculation were associated with changes in physiological and morphological traits. These adaptive adjustments allow plants to maintain fitness across a range of varied environments (Sack and Scoffoni 2013).
In conclusion, our results provide evidence supporting the hypothesis that the growth performance of P. × canadensis was promoted by N fertilization, and the application of exogenous AMF R. irregularis in unsterilized soil may further promote plant growth, the photosynthetic capacity and water status under low N levels. These alterations of the physiological traits of P. × canadensis were paralleled by changes in internal anatomical properties. Although changes in plant size were not observed after exogenous R. irregularis inoculation under high N levels, higher C, N and P concentrations were recorded in the exogenous R. irregularis-inoculated plants than in the non-inoculated plants, suggesting that exogenous R. irregularis increased plant nutrition in a manner decoupled from the level of N fertilization. Furthermore, this study demonstrated the efficacy of using exogenous R. irregularis to promote the growth of poplar in nutrient-limited soils and provides a starting point for the development of mycorrhiza bio-fertilizers for use in sustainable forest management.
References
Bago B, Cano C, Azcón-Aguilar C, Samson J, Coughlan AP, Piche Y (2004) Differential morphogenesis of the extraradical mycelium of an arbuscular mycorrhizal fungus grown monoxenically on spatially heterogeneous culture media. Mycologia 96:452–462
Brodribb TJ, Feild TS, Jordan GJ (2007) Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol 144:1890–1898
Ceballos I, Ruiz M, Fernández C, Peña R, Rodríguez A, Sanders IR (2013) The in vitro mass-produced model mycorrhizal fungus, Rhizophagus irregularis, significantly increases yields of the globally important food security crop cassava. PLOS ONE 8:e70633
Chu H, Lin X, Fujii T, Morimoto S, Yagi K, Hu J, Zhang J (2007) Soil microbial biomass, dehydrogenase activity, bacterial community structure in response to long-term fertilizer management. Soil Biol Biochem 39:2971–2976
Coleman M, Tolsted D, Nichols T, Johnson WD, Wene EG, Houghtaling T (2006) Post-establishment fertilization of Minnesota hybrid poplar plantations. Biomass Bioenerg 30:740–749
Cooke JEK, Martin TA, Davis JM (2005) Short-term physiological and developmental responses to nitrogen availability in hybrid poplar. New Phytol 167:41–52
Dié A, Kitin P, Kouamé FNG, Van den Bulcke J, Van Acker J, Beeckman H (2012) Fluctuations of cambial activity in relation to precipitation result in annual rings and intra-annual growth zones of xylem and phloem in teak (Tectona grandis) in Ivory Coast. Ann Bot 110:861–873
Dodd IC, Ruiz-Lozano JM (2012) Microbial enhancement of crop resource use efficiency. Curr Opin Biotechnol 23:236–242
Engelmoer DJP, Behm JE, Kiers ET (2014) Intense competition between arbuscular mycorrhizal mutualists in an in vitro root microbiome negatively affects total fungal abundance. Mol Ecol 23:1584–1593
Fellbaum CR, Gachomo EW, Beesetty Y, Choudhari S, Strahan GD, Pfeffer PE, Kiers ET, Bücking H (2012) Carbon availability triggers fungal nitrogen uptake and transport in arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci USA 109:2666–2671
Feng XP, Chen Y, Qi YH, Yu CL, Zheng BS, Brancourt-Hulmel M, Jiang DA (2012) Nitrogen enhanced photosynthesis of Miscanthus by increasing stomatal conductance and phosphoenolpyruvate carboxylase concentration. Photosynthetica 50:577–586
Fonseca HM, Berbara RL, Daft MJ (2001) Shoot δ15N and δ13C values of non-host Brassica rapa change when exposed to ± Glomus etunicatum inoculum and three levels of phosphorus and nitrogen. Mycorrhiza 11:151–158
Haberer G, Kieber JJ (2002) Cytokinins. New insights into a classic phytohormone. Plant Physiol 128:354–362
Hacke UG, Plavcová L, Almeida-Rodriguez A, King-Jones S, Zhou W, Cooke JEK (2010) Influence of nitrogen fertilization on xylem traits and aquaporin expression in stems of hybrid poplar. Tree Physiol 30:1016–1025
Hansen EA, McLaughlin RA, Pope PE (1988) Biomass and nitrogen dynamics of hybrid poplar on two different soils: implications for fertilization strategy. Can J For Res 18:223–230
He F, Zhang H, Tang M (2016) Aquaporin gene expression and physiological responses of Robinia pseudoacacia L. to the mycorrhizal fungus Rhizophagus irregularis and drought stress. Mycorrhiza 26:311–323
Johansen A, Jakobsen I, Jensen ES (1994) Hyphal N transport by a vesicular-arbuscular mycorrhizal fungus associated with cucumber grown at three nitrogen levels. Plant Soil 160:1–9
Johnson NC (2010) Resource stoichiometry elucidates the structure and function of arbuscular mycorrhizas across scales. New Phytol 185:631–647
Kiers ET, Duhamel M, Beesetty Y, Mensah JA, Franken O, Verbruggen E, Fellbaum CR, Kowalchuk GA, Hart MM, Bago A, Palmer TM, West SA, Vandenkoornhuyse P, Jansa J, Bücking H (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333:880–882
Kong Z, Glick BR, Duan J, Ding S, Tian J, McConkey BJ, Wei G (2015) Effects of 1-aminocyclopropane-1-carboxylate (ACC) deaminase-overproducing Sinorhizobium meliloti on plant growth and copper tolerance of Medicago lupulina. Plant Soil 391:383–398
Labidi S, Jeddi FB, Tisserant B, Yousfi M, Sanaa M, Dalpé Y, Sahraoui ALH (2015) Field application of mycorrhizal bio-inoculants affects the mineral uptake of a forage legume (Hedysarum coronarium L.) on a highly calcareous soil. Mycorrhiza 25:297–309
Larson PR, Isebrands JG (1971) The plastochron index as applied to developmental studies of cottonwood. Can J Forest Res 1:1–11
Li C, Korpelainen H (2015) Transcriptomic regulatory network underlying morphological and physiological acclimation to nitrogen starvation and excess in poplar roots and leaves. Tree Physiol 35:1279–1282
Li H, Li X, Dou Z, Zhang J, Wang C (2012) Earthworm (Aporrectodea trapezoides)-mycorrhiza (Glomus intraradices) interaction and nitrogen and phosphorus uptake by maize. Biol Fertil Soils 48:75–85
Li Z, Wu N, Liu T, Chen H, Tang M (2015) Effect of arbuscular mycorrhizal inoculation on water status and photosynthesis of Populus cathayana males and females under water stress. Physiol Plant 155:192–204
Liu T, Wang C, Chen H, Fang F, Zhu X, Tang M (2014) Effects of arbuscular mycorrhizal colonization on the biomass and bioenergy production of Populus × canadensis ‘Neva’ in sterilized and unsterilized soil. Acta. Physiol Plant 36:871–880
Liu T, Sheng M, Wang CY, Chen H, Li Z, Tang M (2015) Impact of arbuscular mycorrhizal fungi on the growth, water status and photosynthesis of hybrid poplar under drought stress and recovery. Photosynthetica 53:250–258
Marulanda A, Azcón R, Ruiz-Lozano JM (2003) Contribution of six arbuscular mycorrhizal fungal isolates to water uptake by Lactuca sativa plants under drought stress. Physiol Plant 119:526–533
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Monclus R, Dreyer E, Villar M, Delmotte FM, Delay D, Petit JM, Barbaroux C, Le Thiec D, Bréchet C, Brignolas F (2006) Impact of drought on productivity and water use efficiency in 29 genotypes of Populus deltoides × Populus nigra. New Phytol 169:765–777
Ohashi-Ito K, Fukuda H (2014) Xylem. Curr Biol 24:R1149
Olsson PA, Burleigh SH, van Aarle IM (2005) The influence of external nitrogen on carbon allocation to Glomus intraradices in monoxenic arbuscular mycorrhiza. New Phytol 168:677–686
Pellegrino E, Turrini A, Gamper HA, Cafà G, Bonari E, Young JPW, Giovannetti M (2012) Establishment, persistence and effectiveness of arbuscular mycorrhizal fungi inoculants in the field revealed using molecular genetic tracing and measurement of yield components. New Phytol 194:810–822
Peuke AD (2009) Nutrient composition of leaves and fruit juice of grapevine as affected by soil and nitrogen fertilization. J Plant Nutr Soil Sc 172:557–564
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–161
Plavcová L, Hacke UG, Almeida-Rodriguez AM, Li E, Douglas CJ (2013) Gene expression patterns underlying changes in xylem structure and function in response to increased nitrogen availability in hybrid poplar. Plant Cell Environ 36:186–199
Price CA, Enquist BJ (2007) Scaling mass and morphology in leaves: an extension of the WBE model. Ecology 88:1132–1141
Sack L, Scoffoni C (2013) Leaf venation: structure, function, development, evolution, ecology and applications in the past, present and future. New Phytol 198:983–1000
Scarpella E, Meijer AH (2004) Pattern formation in the vascular system of monocot and dicot plant species. New Phytol 164:209–242
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. 3rd edn. Academic Press, New York
Smith SE, Smith FA (2011) Roles of arbuscular mycorrhizas in plant nutrition and growth: new paradigms from cellular to ecosystem scales. Annu Rev Plant Biol 62:227–250
Smith SE, Smith FA, Jakobsen I (2004) Functional diversity in arbuscular mycorrhizal (AM) symbioses: the contribution of the mycorrhizal P uptake pathway is not correlated with mycorrhizal responses in growth or total P uptake. New Phytol 162:511–524
Sultan SE (2000) Phenotypic plasticity for plant development, function and life history. Trends Plant Sci 5:537–542
Sýkorová Z, Börstler B, Zvolenská S, Fehrer J, Gryndler M, Vosátka M, Redecker D (2012) Long-term tracing of Rhizophagus irregularis isolate BEG140 inoculated on Phalaris arundinacea in a coal mine spoil bank, using mitochondrial large subunit rDNA markers. Mycorrhiza 22:69–80
Tjoelker MG, Oleksyn J, Reich PB (1998) Seedlings of five boreal tree species differ in acclimation of net photosynthesis to elevated CO2 and temperature. Tree Physiol 18:715–726
Treseder KK (2004) A meta-analysis of mycorrhizal responses to nitrogen, phosphorus, and atmospheric CO2 in field studies. New Phytol 164:347–355
Treseder KK, Allen MF (2002) Direct nitrogen and phosphorus limitation of arbuscular mycorrhizal fungi: a model and field test. New Phytol 155:507–515
Tyree MT (2003) Plant hydraulics: the ascent of water. Nature 423:923–923
van den Driessche R (1999) First-year growth response of four Populus trichocarpa × Populus deltoides clones to fertilizer placement and level. Can J Forest Res 29:554–562
Woodruff DR, Meinzer FC, Lachenbruch B (2008) Height-related trends in leaf xylem anatomy and shoot hydraulic characteristics in a tall conifer: safety versus efficiency in water transport. New Phytol 180:90–99
Wu F, Bao W, Li F, Wu N (2008) Effects of drought stress and N supply on the growth, biomass partitioning and water-use efficiency of Sophora davidii seedlings. Environ Exp Bot 63:248–255
Yan Z, Kim N, Han W, Guo Y, Han T, Du E, Fang J (2015) Effects of nitrogen and phosphorus supply on growth rate, leaf stoichiometry, and nutrient resorption of Arabidopsis thaliana. Plant Soil 388:147–155
Yang Y, Tang M, Sulpice R, Chen H, Tian S, Ban Y (2014) Arbuscular mycorrhizal fungi alter fractal dimension characteristics of Robinia pseudoacacia L. seedlings through regulating plant growth, leaf water status, photosynthesis, and nutrient concentration under drought stress. J Plant Growth Regul 33:612–625
Yasumura Y, Hikosaka K, Hirose T (2007) Nitrogen resorption and protein degradation during leaf senescence in Chenopodium album grown in different light and nitrogen conditions. Funct Plant Biol 34:409–417
Yu C, Lv DG, Qin SJ, Yang L, Ma HY, Liu GC (2010) Changes in photosynthesis, fluorescence, and nitrogen metabolism of hawthorn (Crataegus pinnatifida) in response to exogenous glutamic acid. Photosynthetica 48:339–347
Zhen L, Yang G, Yang H, Chen Y, Liu N, Zhang Y (2014) Arbuscular mycorrhizal fungi affect seedling recruitment: a potential mechanism by which N deposition favors the dominance of grasses over forbs. Plant Soil 375:127–136
Zhu XC, Song FB, Liu SQ, Liu TD (2011) Effects of arbuscular mycorrhizal fungus on photosynthesis and water status of maize under high temperature stress. Plant Soil 346:189–199
Acknowledgements
This study was supported by the National Natural Science Foundation of China (41671268 and 31270639, http://www.nsfc.gov.cn/), and the Shaanxi Science and Technology Innovation Project Plan (2016KTCL02-07).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Fei Wu and Haoqiang Zhang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wu, F., Zhang, H., Fang, F. et al. Effects of Nitrogen and Exogenous Rhizophagus irregularis on the Nutrient Status, Photosynthesis and Leaf Anatomy of Populus × canadensis ‘Neva’. J Plant Growth Regul 36, 824–835 (2017). https://doi.org/10.1007/s00344-017-9686-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-017-9686-6