Abstract
Morphometric studies are approached through different methods, among them geometric morphometry, such as contours and landmarks. These methodologies gathered with classification methods, such as linear discriminant analysis (LDA) and principal component analysis (PCA), extract information efficiently and capture inter or intraspecific grouping. Three keyhole limpet species—Fissurellidea megatrema, Fissurellidea patagonica, and Fissurella radiosa—were contrasted through morphometrics: shell shape with linear morphometrics and foramen shape through elliptic Fourier analysis, and landmarks. The broad geographic range of F. radiosa allowed additional intraspecific analysis: three sampling sites, along the Southwest Atlantic coast (42°19′S 64°19′W–54°48´S 68°19′W), comprising two subspecies (F. radiosa radiosa and F. radiosa tixierae). The aim of this study was to evaluate which measurements most contribute to the classification of the species and to determine if the subspecies reflected a geographic pattern in the classification methods (LDA and PCA). The LDA revealed two linear morphological variables to differentiate between the three species and between localities of F. radiosa. For the subspecies (F. radiosa radiosa and F. radiosa tixierae), the LDA and PCA showed a biogeographic pattern related to the distribution (Argentinean and Magellan provinces). We concluded that the morphometrics methods and both classification analysis (LDA and PCA) capture information at the species and subspecies level and the foramen is the principal variable that contributes as a taxonomic tool.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Argentinean coast enables the study of several marine biogeographic aspects considering its latitudinal extension and the variability in oceanographic conditions. Two biogeographic provinces are represented in the Argentinean coast: the Argentinean and Magellan (Balech and Ehrlich 2008). The Argentinean biogeographic province extends from 30°S to 41°S–44°S, where northern winds and alternation of warmer coastal with temperate-cold waters typify this province; and the Magellan biogeographic province which extends from the Peninsula Valdés to the southern boundary. Subantarctic cold waters and strong west winds are predominant in this region (Balech and Ehrlich 2008).
The wide latitudinal distribution of many species allows analyzing if abundance, size, and morphology are related to this distribution. In fact, studies in marine bivalves and gastropods have evidenced shell shape variability across latitudinal gradients showing high morphometric diversity in different coastal environments (Stanley 1970; Graus 1974; Vermeij 1978; Trussell and Etter 2001; Watson et al. 2012). This diversity was also associated with environmental factors such as intertidal height, wave exposure, and pollution (Tablado and López Gappa 2001; Nuñez et al. 2012; Soria et al. 2017).
Geometric morphometric (GM) methods allowed splitting the shell form in its two components: shape and size, and preserved the geometry all through the analysis (Rufino et al. 2006). Outline or contour methods have been considered more suitable in the study of round type objects, such as mussels (Krapivka et al. 2007). However, recent studies apply landmarks and semi-landmarks, which are considered a robust method (Rufino et al. 2006; Trovant et al. 2017; Márquez et al. 2018). In populations with a wide geographic range, GM methods have been successfully applied, detecting inter or intra-specific variations (Ferson et al. 1985; Innes and Bates 1999; Palmer et al. 2004; Costa et al. 2008; Van der Molen et al. 2012). Gastropods with hard shells are a perfect model in morphological analysis based on their contour and linear morphometrics (Costa et al. 2008).
The family Fissurellidae has a wide geographic distribution, from the subtidal (Rico and López Gappa 2006) to the intertidal zone (McLean 1984a). Fissurellidea d´Orbigny, 1841 includes 11 species. In the Southwest Atlantic Fissurellidea megatrema (d´Orbigny, 1841) is distributed from Río de Janeiro, Brazil to Puntas Ninfas, Argentina and Fissurellidea patagonica (Strebel, 1907), which shows a wider distribution along the Southwest Atlantic, from the Uruguayan coast to the south of Tierra del Fuego, Argentina (Pastorino 1995). The genus Fissurella Bruguiére, 1789 presents 214 species. Fissurella radiosa Lesson, 1831, inhabits from Golfo San Matías, Argentina to the south of Cabo de Hornos in Tierra del Fuego (McLean 1984a). Two geographic subspecies were described: Fissurella radiosa radiosa distributed in the Magellanic biogeographic province (south of Chile and Argentina) and Fissurella radiosa tixierae with distribution in Golfo San Matías and Península Valdés in the Argentinean biogeographic province (McLean 1984a).The shell of F. megatrema is characterized by an oval and thin shape with radial ribs, foramen elongated and broad, while F. patagonica shows an elongated, oval, and thin shell with radial ribs and elongate-oval foramen (McLean 1984b). The shell’s description of F. radiosa is associated with both subspecies: F. radiosa radiosa presents a small to a medium shell, low to mildly elevated, raised ribs, and an anteriorly displaced foramen with a tripartite shape (McLean 1984a). F. radiosa tixierae displays small shells with moderate to strong elevation and narrow ribs. Tripartite foramen, slightly anterior displace (McLean 1984a). The size and shape of the apical perforation, or foramen, is a relevant feature in the shell of fissurellids (McLean 1984a).
The main goal of this study was to evaluate which morphometric measurement was relevant in the classification of the species: F. megatrema, F. patagonica, and F. radiosa. We analyze the shell and foramen shape through linear and contour morphometrics. We also carried out a landmark analysis for F. radiosa and evaluated if there was a geographic pattern in the foramen shape, related with the subspecies (F. radiosa radiosa and F. radiosa tixierae). This allowed a comparison between methodologies. We hypothesize that, given the oceanographic variability along the Argentinean coast, the subspecies would present a clustering related to the biogeographic provinces and the environmental factors that characterized them.
Materials and methods
Sample sites
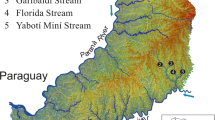
The samples of F. megatrema and F. patagonica were obtained from oceanographic campaigns on the scientific ship ARA Puerto Deseado. Individuals of F. megatrema were collected with a trawl net at the coast of Buenos Aires province (Mar del Plata, MDP) on August 2014 (38° 35′S 54° 54′W, n = 22) while F. patagonica was collected at the Patagonian coast (Rawson, R), on March 2013 (43° 22′S 64°55′W, n = 17) (Fig. 1). Shells of F. radiosa were obtained from the National Collection of Invertebrates of the Museo Argentino de Ciencias Naturales “Bernardino Rivadavia” (MACN-In). Three sites were selected along the Southwest Atlantic coast: Golfo San José (GSJ), Chubut (42° 19′S 64° 19′W, n = 29); Puerto Deseado (PD), Santa Cruz (47°45′S 65°53′W, n = 26) and Ushuaia (U), Tierra del Fuego (54°48′S 68° 19′W, n = 19) (Fig. 1). Environmental parameters are described in Table 1.
Geometric and linear morphometry of the shell
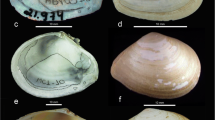
Individuals of F. megatrema and F. patagonica were dissected, the shell was removed with a scalpel, and preserved dry. Shells of F. radiosa, obtained from the museum, were preserved dry, categorized in bags according to the sampling site (Fig. 2).
The Geometric morphometry of the foramen of the three species (F. megatrema: n = 22, F. patagonica: n = 17 and F. radiosa n = 74) was analyzed by photographs (took with a Sony Cyber-Shot digital camera) of the dorsal side of the shell positioned in a black background to maximize the contrast (Fig. 3a).
a Fissurellidea megatrema. Measurements considered for linear discriminant analysis: total length (Tl), total width (Wi), anterior (X1) and posterior length (X2) of the shell. Foramen length (Fl), anterior (Aw) and posterior width (Pw). b Fissurella radiosa. Landmarks (black dots) configuration for the general Procrustes analysis
All the images were digitized with SHAPE–ChainCoder software package (Iwata and Ukai 2002), binarized to obtain a chain code, and the curves corresponded to the contour of the foramen for each specimen. The variation of the shell’s shape was studied by an EFA, which consists in decomposing a curve in a sum of ellipses related harmonically (Lestrel 1997). A total of 20 harmonics were used to characterize the contour. The shape of each shell was estimated to 77 coefficients of normalized elliptic Fourier descriptors (EFDs) (four coefficients for each harmonic, excluding the first three that were constant). The Principal components (PC) were obtained from this analysis.
The linear morphometry was characterized by different measurements using photographs of the shell uploaded to the Adobe Photoshop CS6 software. A total of eight linear variables were studied, considering the shell and foramen: total length (Tl), total width (Wi), anterior (X1), and posterior length (X2), foramen length (Fl), anterior (Aw), and posterior width (Pw) (mm, ± 0.001) (Fig. 3a). The height (H) was added as a lateral view analysis and was measured with calipers (mm, ± 0.001). The variables obtained through the contour and linear analysis of the shell were studied with a Linear Discriminant Analysis (LDA). The Tl was excluded from the analysis, since it presented a high correlation with other variables (X1 and X2), eluding the probability of covering up relevant information.
The images of the shells of F. radiosa were landmarked using TpsDig2 (Rohlf 2010) and TpsUtil software (Rohlf 2016). In each shell, four landmarks were selected round the foramen: two in the maximum length (1,3) and two in the center (2,4) (Fig. 3b). To remove variation of size, position, and orientation, a least-squares Procrustes superimposition was carried out (Rohlf and Slice 1990). The variables obtained were analyzed with a Principal Component Analysis (PCA).
Multivariate statistical analysis (MANOVA) was carried out for each analysis. A test of Hotelling corrected by Bonferroni was carried out for the LDA. In both analyses (PCA and LDA), the assumption of multivariate normality was tested with Shapiro–Wilk’s test and homogeneity of the covariance matrix with a test of Box. The tests with p < 0.01 were considered significant.
All statistical analyses were performed using the software R (v. 3.4.3, R Core Team, 2017) and the packages MASS (Venables and Ripley 2002), geomorph (Adams et al. 2020), and Morpho (Schlager 2017).
Results
Morphological analysis between species
The analysis allowed separating individuals from the three species (Fig. 4a). The first two linear discriminants (LDs) explained the total variability for the three species. The LD1 accounted for 98.29% of the variability while the LD2 accounted for 1.71% of the total variability. The morphometric variables showed significant differences (MANOVA, Wilk’s λ = 0.01, F2,36 = 113.67, p < 0.001) among species. The most relevant variables were X1 and X2, both in LD1. X1 (− 1.81) positioned F. megatrema (red) and F. patagonica (green) to the left of the graphic, while X2 (1.48) located F. radiosa (blue) towards the right of the graphic.
a Linear discriminant analysis for the three species. Codes: red Fissurellidea megatrema, green Fissurellidea patagonica, blue Fissurella radiosa. b Linear discriminant analysis for the three distributions of Fissurella radiosa. Codes: red, Golfo San José; green, Puerto Deseado; blue, Ushuaia. Linear measurements: Total length (Tl), total width (Wi), anterior (X1) and posterior length (X2) of the shell. Foramen length (Fl), anterior (Aw) and posterior width (Pw). Ellipses represent 95% confidence level
Morphological analysis among localities
The analysis allowed the separation of the individuals from the three localities (Fig. 4b). Graphic exploration of the LDA (Fig. 4b) and PCA (Fig. 5), showed that both methods generated a similar analysis of variance for the data. The LDA showed that the LD1 explained 79.01% of the variability between localities and LD2 accounted for 20.99% of the total variability. The morphometric variables showed significant differences (MANOVA, Wilk’s λ = 0.13, F2,18 = 12.31, p < 0.001) between sites. The most significant variable was H, in LD1 (− 1.53), locating individuals to the left of the graphic (GSJ, red). Another variable with considerable importance was X1, as well as for LD1 (1.37), locating individuals with a posterior displaced foramen to the right of the graphic (PD, green and U, blue).
Principal component analysis of Procrustes analysis. The polygons indicate each population distribution of Fissurella radiosa. Codes: red, Golfo San José; green, Puerto Deseado; blue, Ushuaia. Morphological changes are included with the shape deformation along the principal component 1 and principal component 2. Codes: blue, positive extreme; red, negative extreme
The foramen shape variation was explained with the first two PC that accounted for 89.96% of the total variation (Fig. 5). The clustering in the morphospace showed significant differences for the localities (MANOVA, Wilk’s λ = 0.31, F2,16 = 6.35, p < 0.001). Individuals at the positive extreme of PC1 showed a narrower and longer foramen shape (Fig. 5). In contrast, individuals positioned at the negative extreme presented the opposite shape. Individuals located toward the positive extreme of PC2 had an anterior displacement of the foramen, differing from the individuals at the negative extreme, which had a posterior displacement (Fig. 5).
The individuals of PD and U (right of LDA and PCA graphics) could represent to F. radiosa radiosa (Magellanic region), while GSJ (left of LDA and PCA graphics) could define to F. radiosa tixierae (Argentinean region).
Discussion
In fissurellids, the foramen is a taxonomic tool (size, position, shape) and its shape could also be related to different environmental conditions. Our results revealed a distinction between Fissurella and Fissurellidea associated with two morphometric measurements (X1 and X2) related to the displacement (or position) of the foramen, in the shell. F. megatrema presents a large and oval foramen that covers more than one third of the shell length (McLean 1984b). The foramen in F. patagonica is smaller and occupies less than a quarter of the shell length (McLean 1984b), while F. radiosa presents an elliptic foramen with a central widening and a slight anterior displacement (McLean 1984a). Although these species are taxonomically classified, our results (obtained by linear and contour morphometrics) allowed us to define these two measures as the most important taxonomically.
The great variability in shell shape of F. radiosa could be associated with the wide distribution of F. radiosa along the southwest Atlantic coast, as well as with the classification of the subspecies -embracing the biogeographic provinces- as it was described by McLean (1984a). The LDA showed that the most relevant measurements were the displacement of the foramen (X1) and the height (H) of the shell, while the PCA determined that the displacement and the width of the foramen were relevant. Intraspecific morphological differences may be associated with the transitional environmental conditions of the provincial limits. Individuals located in Golfo San José could be associated with wider foramen shapes, and as the temperature decreases, this structure becomes narrower (Puerto Deseado and Ushuaia).
Several studies revealed spatial differences in shell morphology of certain species coexisting in a transition zone, or species that present a latitudinal range distribution (Marquez and Van der Molen 2011; Teso et al. 2011; Van der Molen et al. 2012; Avaca et al. 2013; Rufino et al. 2013). Malvé et al. (2018) analyzed differences considering Argentinean and Magellan provinces in the gastropod Trophon geversianus (Pallas, 1774). The variability observed in the shell slightly increased in higher latitudes and might be associated with seawater pH. Sepúlveda and Ibáñez (2012) stated a clinal variation in shell morphology of Acanthina monodon (Pallas, 1774) along the southeastern Pacific coast showing high biogeographical and latitudinal variation. Gordillo et al. (2011) used Elliptic Fourier Analysis for the study of morphological variation of the venerid Tawera gayi (Hupé, 1854) showing that contours allowed to differentiate the three species of Tawera which exhibited ecophenotypic plasticity. We concluded that the pattern observed (through the contour and linear analysis) in F. radiosa is seemingly related to the biogeographical province classification and environmental conditions, suggesting shell morphologies connected to the transition zone.
The diversity of morphometric methods used in this study (linear morphometrics, contour, and landmarks) enable us to approach the same objective and compare or complement them. The foramen results to be the most relevant variable and we reinforce its contribution as a taxonomic tool. Future comparative studies, incorporating localities of interest and environmental data, could contribute to further morphometric analysis.
Data availability
The R script used for the analysis of landmarks is available on GitHub, https://github.com/millacarmona/gmorphometrics.
References
Adams DC, Collyer ML, Kaliontzopoulou A (2020) Geomorph: software for geometric morphometric analyses. R Package Version 3(2):1
Avaca MS, Narvarte M, Martín P, Van Der Molen S (2013) Shell shape variation in the Nassariid Buccinanops globulosus in northern Patagonia. Helgol Mar Res 67:567–577. https://doi.org/10.1007/s10152-013-0344-5
Baldoni A, Molinari GN, Reta R, Guerrero RA (2015) Atlas de temperatura y salinidad de la plataforma continental del Atlántico Sudoccidental: períodos cálido y frío. INIDEP, Mar del Plata
Balech E, Ehrlich MS (2008) Esquema biogeográfico del mar Argentino. Rev Invest Des Pesq 19:45–75
Costa C, Menesatti P, Aguzzi J, Dandrea S, Antonucci F, Rimatori V, Pallottino P, Mattoccia M (2008) External shape differences between sympatric populations of commercial clams Tapes decussatus and T. philippinarum. Food Bioprocess Tech 3:43–48. https://doi.org/10.1007/S11947-008-0068-8
Ferson S, Rohlf FJ, Koehn RK (1985) Measuring shape variation of two dimensional outlines. Syst Biol 34:9–68. https://doi.org/10.2307/2413345
Gordillo S, Márquez F, Cárdenas J, Zubimendi MÁ (2011) Shell variability in Tawera gayi (Veneridae) from southern South America: a morphometric approach based on contour analysis. J Mar Biol Assoc UK 91:815822. https://doi.org/10.1017/S0025315410000391
Graus RR (1974) Latitudinal trends in the shell characteristics of marine gastropods. Lethaia 7:303–314. https://doi.org/10.1111/j.1502-3931.1974.tb00906.x
Innes DJ, Bates JA (1999) Morphological variation of Mytilus edulis and Mytilus trossulus in eastern Newfoundland. Mar Biol 133:691–699. https://doi.org/10.1007/s002270050510
Iwata H, Ukai Y (2002) SHAPE: a computer program package for quantitative evaluation of biological shapes based on elliptical Fourier descriptors. J Hered 93:384–385. https://doi.org/10.1093/jhered/93.5.384
Krapivka S, Toro JE, Alcapán AC, Astorga M, Presa P, Pérez M, Guiñez R (2007) Shell-shape variation along the latitudinal range of the Chilean blue mussel Mytilus chilensis (Hupe 1854). Aquac Res 38:1770–1777. https://doi.org/10.1111/j.1365-2109.2007.01839.x
Lestrel PE (1997) Fourier descriptors and their applications in biology. Cambridge University Press, Cambridge
Malvé ME, Rivadeneira MM, Gordillo S (2018) Biogeographic shell shape variation in Trophon geversianus (Gastropoda: Muricidae) along the Southwestern Atlantic Coast. Palaios 33:498–507. https://doi.org/10.2110/palo.2018.060
Márquez F, Van Der Molen S (2011) Intraspecific shell-shape variation in the razor clam Ensis macha along the Patagonian coast. J Mollus Stud 77:123–128. https://doi.org/10.1093/mollus/eyq044
Márquez F, Adami ML, Trovant B, Nieto-Vilela RA, González-José R (2018) Allometric differences on the shell shape of two scorched mussel species along the Atlantic South American Coast. Evol Ecol 32(1):43–56. https://doi.org/10.1007/s10682-018-9928-5
McLean JH (1984a) Systematics of Fissurella in the Peruvian and Magellanic faunal provinces (Gastropoda: Prosobranchia). Contrib Sci 354:1–70
McLean JH (1984b) Shell reduction and loss in fissurellids: a review of genera and species in the Fissurellidea group. Am Malacol Bull 2:21–34
Nuñez JD, Laitano MV, Cledón M (2012) An intertidal limpet species as a bioindicator: pollution effects reflected by shell characteristics. Ecol Indic 14:178–183. https://doi.org/10.1016/j.ecolind.2011.07.015
Palmer M, Pons GX, Linde M (2004) Discriminating between geographical groups of a Mediterranean commercial clam (Chamelea gallina (L.): Veneridae) by shape analysis. Fish Res 67:93–98. https://doi.org/10.1016/j.fishres.2003.07.006
Pastorino G (1995) Moluscos costeros recientes de Puerto Pirámide, Chubut, Argentina. Academia Nacional de Ciencias. Córdoba Miscelánea 93:1–30
R Core Team (2017) R: a language and environment for statistical computing. R Core Team, Vienna
Rico A, López Gappa J (2006) Intertidal and subtidal fouling assemblages in a Patagonian harbour (Argentina, southwest Atlantic). Hydrobiologia 563:9–18. https://doi.org/10.1007/s10750-005-1337-4
Rohlf FJ (2010) TpsDig2, version 2.16. Department of Ecology and Evolution, State University of New York, Stony Brook
Rohlf FJ (2016) TpsUtil, version 1.70. Department of Ecology and Evolution, State University of New York, Stony Brook
Rohlf FJ, Slice D (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Biol 39:40–59. https://doi.org/10.2307/2992207
Rufino MM, Gaspar MB, Pereira AM, Vasconcelos P (2006) The use of shape to distinguish Chamelea gallina and Chamelea striatula (Bivalvia: Veneridae): linear and geometric morphometric methods. J Morphol 267:1433–1440. https://doi.org/10.1002/jmor.10489
Rufino MM, Vasconcelos P, Pereira F, Fernández-Tajes J, Darriba S, Méndez J, Gaspar MB (2013) Geographical variation in shell shape of the pod razor shell Ensis siliqua (Bivalvia: Pharidae). Helgol Mar Res 67:49–58. https://doi.org/10.1007/s10152-012-0303-6
Schlager S (2017) Morpho and Rvcg—shape analysis in R. In: Zheng G, Li S, Szekely G (eds) Statistical shape and deformation analysis. Academic Press, Cambridge, pp 217–256
Sepúlveda RD, Ibáñez CM (2012) Clinal variation in the shell morphology of intertidal snail Acanthina monodon in the Southeastern Pacific Ocean. Mar Biol Res 8:363–372. https://doi.org/10.1080/17451000.2011.634813
Soria SA, Teso V, Gutiérrez JL, Arribas LP, Scarabino F, Palomo MG (2017) Variation in density, size, and morphology of the pulmonate limpet Siphonaria lessonii along the Southwestern Atlantic. J Sea Res 129:29–35. https://doi.org/10.1016/j.seares.2017.08.014
Stanley SM (1970) Relation of shell form to life habitats in the Bivalvia (Mollusca). Mem Geol Soc Am 125:1–296
Tablado A, López-Gappa J (2001) Morphometric diversity of the pulmonate limpet Siphonaria lessoni in different coastal environments. Sci Mar 65:33–41. https://doi.org/10.3989/scimar.2001.65n133
Teso V, Signorelli JH, Pastorino G (2011) Shell phenotypic variation in the southwestern Atlantic gastropod Olivancillaria carcellesi (Mollusca: Olividae). J Mar Biol Assoc UK 91:1089–1094. https://doi.org/10.1017/S0025315410001475
Trovant B, Márquez F, Del Río C, Ruzzante DE, Martínez S, Orensanz JM (2017) Insights on the history of the scorched mussel Brachidontes rodriguezii (Bivalvia: Mytilidae) in the Southwest Atlantic: a geometric morphometrics perspective. Hist Biol 30:564–572. https://doi.org/10.1080/08912963.2017.1306699
Trussell GC, Etter RJ (2001) Integrating genetic and environmental forces that shape the evolution of geographic variation in a marine snail. Genetica 112:321–337. https://doi.org/10.1007/978-94-010-0585-2_20
Van Der Molen S, Márquez F, Idaszkin YL, Adami M (2012) Use of shell-shape to discriminate between Brachidontes rodriguezii and Brachidontes purpuratus species (Mytilidae) in the transition zone of their distributions (south-western Atlantic). J Mar Biol Assoc UK 93:803–808. https://doi.org/10.1017/S0025315412001221
Venables WN, Ripley BD (2002) Modern Applied Statistics with S. Springer, New York
Vermeij GJ (1978) Biogeography and adaptation: patterns of marine life. Harvard University Press, Cambridge
Watson SA, Peck LS, Tyler PA, Southgate PC, Tan KS, Day RW, Morley SA (2012) Marine invertebrate skeleton size varies with latitude, temperature and carbonate saturation: implications for global change and ocean acidification. Glob Chang Biol 18:3026–3038. https://doi.org/10.1111/j.1365-2486.2012.02755.x
Acknowledgements
We are grateful to the Invertebrate Division at the Museo Argentino de Ciencias Naturales B. Rivadavia. We thank Dr. Juan López Gappa for the accurate revision of this manuscript. We are also grateful to Alejandro Tablado, Mariela Romanelli, Sofía Giménez Ghirimoldi, Brenda Doti, María E. Torroglosa and Ignacio Soto for suggestions made during this work.
Funding
This research was partially supported by Grants: PICT 1159 and UBACyT X086.
Author information
Authors and Affiliations
Contributions
MCY and JG conceived and designed research. JG participated in the research cruises to take the samples. MCY conducted the measurements and contributed new analytical tools. This study was the grade thesis of MCY supervised by JG. MCY and JG analyzed data. MCY wrote most of the manuscript. Both authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
The authors declare consent to participate.
Consent for publication
The authors declare consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yuvero, M.C., Giménez, J. Morphological shell characterization of Fissurellidea and Fissurella (Vetigastropoda: Fissurellidae) along the Argentinean coast, from temperate to subantarctic waters. Polar Biol 44, 1903–1909 (2021). https://doi.org/10.1007/s00300-021-02927-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02927-z