Abstract
Scorched mussels (Bivalvia: Mytilidae) are an important component of rocky shore assemblages around the world. The mussel beds of the mid-intertidal coasts of the south-western Atlantic are composed of two species of scorched mussels, which dominate the physiognomy and structure of the rocky shore communities. The present study investigates the variation in the shell-shape allometric trajectories of both species of mussels throughout their distributional range in the southwestern Atlantic coast, from latitude 34°S to 53°S. Shells of Brachidontes rodriguezii were collected at two Uruguayan localities and four Argentinean localities, while shells of Perumytilus purpuratus were collected at six Argentinean localities. Shell shapes of the specimens were studied by geometric morphometrics using landmark and semi-landmark methods. Different evolutionary histories and thermal regimes characterize these two species. Brachidontes rodriguezii seems to have a long history in the region and is present in a restricted area, the warm temperate region of the south-western Atlantic, while P. purpuratus seems to have a recent history and is present in a wide thermal range, involving the cold-temperate regions of the south-western Atlantic and south-eastern Pacific and the warm-temperate region of the south-eastern Pacific. Based on these considerations, we prompt the hypothesis that allometric trajectories of P. purpuratus shell shape is more variable and adapted to a specific habitat than B. rodriguezii. The impact of allometry was larger in P. purpuratus than in B. rodriguezii. Graphical evidence was gathered, showing that these two species differ in allometric growth forms and that the allometric shell morphology changes reflect environmental constraints and adaptation. While the intertidal mussel beds of the South American coasts are ecologically similar in appearance, we found that the allometric variation of the scorched mussels occurs in response to diverging evolutionary processes such as phenotypic plasticity in P. purpuratus and canalization in B. rodriguezii.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Comprehension about biological and physical factors triggering changes in phenotypic traits has been the main objective of evolutionary ecologists (Piersma and van Gils 2011). Evolutionary theory predicts a trade-off between environmental conditions and evolution generation, influencing the phenotypic and genotypic variations of populations and species (Wennersten and Forsman 2012). Changes in body form during growth are an important issue to study since many biomechanical, physiological, behavioral, and ecological variables are associated with allometry (that is, the proportion of shape changes that can be exclusively explained by variations in size). In general, the body grows allometrically through changes in character position as well as timing or rate of character´s development (Gould 1977; Zelditch and Fink 1996; Klingenberg and McIntyre 1998; Zelditch et al. 2000; Kimmel et al. 2008; Urdy et al. 2010a, b). Allometric growth within and among species is related to age-specific shifts in the variable environmental conditions, favouring attributes that optimize the individual survival (Gould 1977; among others). Phenotypic variability could be represented by several evolutionary components. However, phenotypic plasticity and canalization (Waddington 1942, 1957) are two opposed processes. Canalization refers to the buffering of development against environmental and genetic perturbations, while phenotypic plasticity is related to an adaptive change of the phenotype in reaction to the habitat´s particular heterogeneity (Pigliucci et al. 2006).
The shell is generally the most conspicuous part of the mollusk body and preserves the ontogenetic record of growth (Márquez et al. 2010; Urdy et al. 2010b). Therefore, the shell shape is a key morphological trait that reflects the phylogenetic history, function, and life-habit (Crampton and Maxwell 2000). Geometric morphometrics (GM) methods provide the most important tools that enable a clear mathematical separation between size and shape variation, whose variation can be analyzed separately, its relationships properly studied, and the geometry of the structures under study can be visualized along every step of the statistical analyses (Zelditch et al. 2012). Such visualizations provide information about direction and magnitude of shape change. There are several GM applications aimed to study different topics about marine bivalves shell shape variations. An extensive review of previous studies that applied GM methods (landmarks and outlines) was made by Rufino et al. (2013). As far as we know, there is only one highly detailed ontogenetic allometric study of the shell shape changes (Márquez et al. 2010), and inter-specific analyses are absent to date. Comparative studies of the ontogeny play a crucial role in the understanding of the processes of morphological diversification.
There are two scorched mussel species (Mytilidae: Brachidontinae) occurring in the southern south-western Atlantic: Brachidontes rodriguezii (d’Orbigny 1842), restricted to the warm-temperate region (32°–43°S; Scarabino et al. 2006; Trovant et al. 2013) known as the Argentine Biogeographic province, and Perumytilus purpuratus (Lamarck 1819), that spans the warm-temperate region of the south-eastern Pacific (south of 3°S) and the cold-temperate region of the southern South America (north to 41°S on the Atlantic), respectively known as the Chile-Peru and Magellanic Biogeographic Provinces(Briggs and Bowen 2013). Trovant et al. (2015) showed that Perumytilus includes two genetically distinct clades. The North clade confined to the warm-temperate Chile-Peru Province, and the South clade to the cold-temperate Magellanic Biogeographic Province, the latter being the only brachidontin restricted to cold-temperate waters. Moreover, Trovant et al. (2015) hypothesized that differentiation of the cold-temperate clade originated from budding-off from range edge populations of the warm-temperate clade, adaptation to a colder thermal regime, and gradual expansion along the coasts of Patagonia. During the Quaternary glacial maxima, populations from southern Chile were likely extirpated, and populations from central Chile and the southwest Atlantic remained separated by long periods (the last culminating in the LGM) from those inhabiting non-glaciated eastern Patagonia. After ice receded, the range of the South clade presumably expanded back, a secondary contact zone being established between the two clades within a relatively narrow latitudinal range between Valdivia (39°48°S) and Concepcion (36°49°S). There is some evidence for the incipient speciation of these two clades in their genetic and sperm structure (Trovant et al. op.cit.; Briones et al. 2012). However, no morphological differences have been found between shell shape of both North and South clades. Northwards expansion of P. purpuratus into the warm-temperate region of the south-western Atlantic may have been constrained by high-density blocking involving interaction with B. rodriguezii. These two species occur sympatrically between 41° and 43°S in the south-western Atlantic, either of them dominating extensive intertidal beds respectively north and south of that latitudinal range (Adami et al. 2013), having a high level of genetic divergence and no evidence of hybridization among them (Trovant et al. 2013). In this context, Van der Molen et al. (2013) suggest that adult individuals of B. rodriguezzi and P. purpuratus present different outline shell shapes in the transition zone and the use of geometric morphometrics provides an effective tool to identify species of little scorched mussels. As an extension of the latter study, and knowing that different evolutionary histories and thermal regimes characterize these two species, we consider that B. rodriguezii and P. purpuratus offer an excellent opportunity to study intra and inter-specific variations in response to different environmental conditions. They are interesting for a comparison of ontogenetic trajectories on the shell form as their uniform physiognomy (defined as the appearance of ecological communities; Whittaker 1962) consists of dense and extended monocultures, under different environmental conditions as salinity, temperature, substrate, pH, among other variables. The aim of this study is to compare the post- recruited ontogenetic processes of the interspecific phenotypic diversity of two species of scorched mussel distributed along the south-western Atlantic Ocean. Due to the fact that P. purpuratus is distributed along warm and cold temperate regions of the south-eastern Pacific Ocean, as well as in the cold temperate region of the south-western Atlantic, we suggest that allometric trajectories of the shell shape can be potentially seen as adaptations to specific habitats that might have originated part of the significant differences with B. rodriguezii, which is restricted to the warm-temperate region. All in all, we predict smaller amounts of shell shape variation along the ontogeny of B. rodriguezii than in P. purpuratus.
Materials and methods
Study area
The study region extends over the warm and cold temperate regions of the south-western Atlantic, respectively known as the Argentine and Magellanic Biogeographic Provinces (Balech and Ehrlich 2008; and references therein). This region exhibits large topographical changes and climatic heterogeneity. Ph and the tidal amplitude increase with latitude, from basic to more basic (Tyberghein et al. 2012) and from microtidal to macrotidal (Servicio de Hidrografía Naval 2014), there is a declining trend in sea surface temperature mean with latitude (Tyberghein et al. 2012). Additionally, air temperature changes seasonally in response to variation in solar radiation (Piola and Falabella 2009).
In Patagonian shores, the intertidal ecosystem is exposed from mid to high energy flows during high tides. The extreme desiccation is an important feature of these intertidal mussel-dominated beds. Strong dry winds (45–140 km/h), combined with low rainfall probably make these shores one of the most physically demanding rocky intertidal systems in the world (Bertness et al. 2006). Significant ecological barriers (extensive sandy beaches and La Plata River estuary) separate rocky shores along warm-temperate region. However, the cold-temperate region includes extensive rocky shores formed by stretches of cliffs and abrasion platforms with pebble beaches (Schellmann and Radtke 2000; Cavallotto 2008).
Fieldwork
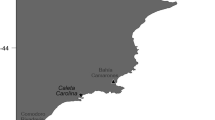
We collected specimens from 12 localities along a wide latitudinal gradient (19 south latitudinal grades, 2400 km straight-line), in the South Atlantic Ocean from Río Grande (53°44′S 67°43′W) in the south of Argentina to Punta del Diablo (34°02′S 53°32′W) in northern Uruguay (Fig. 1).
Map of the South Atlantic Ocean shelf showing the 12 sampling localities. From South to North are: RG Río Grande, PQ Punta Quilla, SJ Puerto San Julián, CO Caleta Olivia, BB Bahía Bustamante and PE Punta Escondida, and PCO Pehuencó, COO Claromecó, N Necochea, SC Santa Clara, LP La Paloma, PDI Punta del Diablo
At each site, ontogenetic series were collected from natural populations, and individuals ranging from 1.6 to 31.0 mm were analyzed. They were invariably obtained randomly from the mid-intertidal rocky zone, aiming at the centroid of patches with 100% mussel cover developed over gently sloping (“horizontal”) platforms. The density (ind 100 cm−2) of each locality were registered. The following environmental variables: concentration of silicate, salinity, concentration of nitrate, concentration of calcite, pH, sea surface temperature and concentration of chlorophyll-a were obtained at each locality, based on data downloaded from a 9.2 km spatial resolution satellite image database (Tyberghein et al. 2012, available at http://www.oracle.ugent.be/).
Geometric morphometric analysis
In all, 1249 specimens of the two species of mussels were analyzed. Around 100 individuals were randomly selected covering the ontogeny series from each site. Shell shape variables were generated from 4 anatomical landmarks and 40 semilandmarks to describe the overall outline form (for anatomical descriptions of the landmarks, see Fig. 2). Ten semilandmarks were pointed out on shell outline between landmarks 1, 2, 3 and 4. The exact location of semilandmark coordinates on the curve cannot be identified and hence it was mathematically estimated. Sliding was performed using the algorithm which optimizes the bending energy (Bookstein 1997; Gunz et al. 2005; Gunz and Mitteroecker 2013) to minimize the deformation between each specimen and the consensus shape (Procrustes average of all specimens) in the TPSRelw software (Rohlf 2004). After sliding the semilandmarks, all the 44-landmark configurations were superimposed by a Generalized Procrustes Analysis (Rohlf and Slice 1990; Slice et al. 1996). This procedure translates and rotates the landmark configurations to a common origin and scales them to unit centroid size. The Procrustes coordinates of the aligned individuals were then used as shape variables to perform the multivariate statistical analyses. Only the left shells were digitized to avoid redundant information in symmetric structures.
Landmarks and semilandmark configurations on the outline of the scorched mussel shell Brachidontes rodriguezii. Landmarks (black dots) are: (1) umbo (anterior end), (2) tip of the posterior hinge ligament, (3) maximum posterior curvature, (4) projection at 90° of the vector formed between the landmark 1 and 2 on the outline of the shell. The semilandmarks (grey dots) (5–14) semi-landmarks along the boundary between the landmarks 1 and 2, (15–24) semi-landmarks between landmarks 2 and 3, (25–34) semi-landmarks between landmarks 3 and 4, and (35–44) semi-landmarks between landmarks 4 and 1. Scale bar = 1 cm
Ontogenies analyses
In the present study, we understand “allometry” as the pattern of covariation among morphological traits or the relationship between size and shape components (Gould 1966; Mosimann 1970). To estimate the between-species allometry variation, we use a pooled species multivariate regression of the independent aligned Procrustes coordinates (the shape variables) on centroid size (the size measure, Monteiro 1999; Klingenberg 2016). To evaluate the differences on intra and inter-specific allometric trajectories, we used a permutation MANCOVA test in R using Adonis function (Zelditch et al. 2012) to characterize how the ontogenies differ, testing the null hypothesis that the direction of ontogenetic change does no differ between species. After that, we study the direction of ontogenetic change throughout angle between allometric trajectory corporations. The aligned shape coordinates were used as dependent variables, species as the grouping variable, and centroid size as a covariate. If the interaction size x species is significant, then it follows that there are differences in the slopes. Moreover, we used angular comparisons of vector directions after the routine available in MorphoJ (Klingenberg 2011). This routine is used to compare the among-sites pairwise angular directions (both between and within-species), thus enabling the testing of the null hypothesis that the allometric vectors are no more similar than expected by chance. Angle between allometric trajectories is one of the most widespread methods to compare allometry trajectories among different groups (Klingenberg and Marugán-Lobón 2013). Mean angle refers to the (within-group) average trajectory. We used mean angle as a proxy to between-group variation in allometric trajectories. All morphometric analyses were conducted with the MorphoJ, version 1.06d software. Differences among inter and intraspecific mean angle were tested using ANOVA tests. Finally, we used the Mantel’s simple test (Mantel 1967) to measure the degree of association between the angles and a matrix of environmental distances for each species independently: the Euclidean distances between localities based on the mean of eight environmental variables presented as supplementary material. When the variation patterns can be represented in matrix format, the Mantel test is one of the most widely used to assess matrix proportionality. Mantel test was used originally to evaluate the relationship between phenetic distances among local populations and their geographic distances (Sokal 1979). Since then, Mantel test has been used in several disciplines such as ecology (Fortin and Gurevitch 2001; MacDougall-Shackleton and MacDougall-Shackleton 2001; Wright and Wilkinson 2001), morphology (van Schaik et al. 2003; Márquez and Van der Molen 2011), and behaviour (Cheverud 1989), among others. Here we formalized among-group environmental and growth differences by obtaining Euclidian distances from raw environmental variables (Table 1 in supplementary material), and using the among-localities angles corresponding to allometric trajectories, respectively. Statistical analysis was run using Infostat software (Di Rienzo et al. 2009).
Results
Ontogenetic allometry
The relation between pooled within-species shape and size accounts for 31.2% of the variation in shape (Fig. 3a). Also, the permutation test indicates that such allometry is highly significant (p < 0.0001). The interspecific allometry shape changes were associated with the umbo position and the amplitude of the shell (similar to the relationship between height/length). Smaller specimens exhibit a rounded, sub-terminal umbo, and an expansion in anterior-dorsal and ventral direction, generating a rounded shell with anterior-dorsal and posterior–ventral curves slightly outward. Conversely, larger shells present a compression in dorsal–ventral direction, generating a terminal umbo with an elongated shell with an anterior–ventral convex curve (Fig. 3b). All localities of both species presented allometric growth (Table 1; Fig. 3c). Between-species and among-localities allometric trajectories were significantly different in term of their slopes (Table 2). The directions of all pairwise allometric trajectories were similar (p < 0.001), but the mean of between-species pairwise angle comparisons was greater than the within-species average (Table 2 in supplementary material).
Allometric trajectories of the scorched mussel species of South American intertidal. a Pooled within-species regression. b Overlapped wireframes for shapes predicted by the multivariate regression of shape on size for Brachidontes rodriguezii (black vectors) and Perumytilus purpuratus (grey vectors) from the smallest (CS = 1) to largest specimens (CS = 70). c Pooled within-localities regression. Biggest circle indicates the centroid and the whiskers the standards deviations of two axes from each locality. Codes from South to North are: RG Río Grande, PQ Punta Quilla, SJ Puerto San Julián, CO Caleta Olivia, BB Bahía Bustamante and PE Punta Escondida, and PCO Pehuencó, COO Claromecó, N Necochea, SC Santa Clara, LP La Paloma, PDI Punta del Diablo
Within-species comparisons showed that localities of P. purpuratus present more among-site angle dispersion than B. rodriguezii sites (Fig. 4). Finally, Mantel tests showed a significant correlation between the allometric angles and environmental (Table 1 in supplementary material) distances (R = 0.55; p = 0.02) in B. rodriguezii, whereas the same test provided non-significant results in P. purpuratus (R = − 0.17; p = 0.62).
Within and between-species angle variations. The central dot represents the mean; the median is showed as a central line; the limits of the box, the first and third quartiles, and the whiskers the 95% confidence interval. Different lowercase letters indicate significant differences (p < 0.05) in pairwise comparisons test. CO–SJ Caleta Olivia–San Julián, RG–BB Río Gallegos–Bahía Bustamante, SJ–CCO San Julián–Claromecó, PQ–SC Punta Quilla–Santa Clara, PCO–PDI Pehuencó–Punta del Diablo, N–SC Necochea–San Clemente
Discussion
This study used geometric morphometrics to compare the variation of the shell shape on the allometric ontogeny of B. rodriguezii and P. purpuratus, two small mussels from the south-western American coast. Also, we provide compelling graphical and analytical evidences that these species differ in allometric growth forms. We hypothesize that the allometries of the shell shape change reflect environmental constraints and adaptation. In fact, our results support the differentiation between the outline of B. rodriguezii and P. purpuratus shell shapes previously showed by Van der Molen et al. (2013) and Trovant et al. (2017), extending the difference to all the ontogeny. However, Aguirre et al. (2006) postulates no differences between B. rodriguezii and P. purpuratus shell shape, thus suggesting that B. rodriguezii could be considered a synonym of P. purpuratus. However, our results contradict this statement and suggest caution when assessing between-taxa relationships. The mytilids present morphological variations linked to geographical location and its position across the intertidal zone (Seed 1968; Briones and Guiñez 2005). Some environmental conditions, such as temperature, aerial exposure, and wave action have a major impact on the population dynamics of mussels, modifying their growth, reproduction, and shell morphology (Griffiths and Griffiths 1987; Hawkins et al. 1992; Seed and Suchanek 1992; Steffani and Branch 2003). Another potential modeler of the life-history pattern is the presence of predators (Reimer et al. 1995; Valladares et al. 2010). Here we show that the ontogenetic trajectories of B. rodriguezii and P. purpuratus deviate sharply from one another. During their ontogeny, B. rodriguezii follows a more conservative (more elliptical shape of the equal frequency ellipse) pattern through the morphospace than P. purpuratus, which present larger variation (larger minor axis of the ellipse, Fig. 3a). In the particular case of B. rodriguezii, the distribution of the variation on the allometric trajectory was restricted and presented a correspondence with the meso-scale environmental conditions. In spite of this, the ontogeny of the shell shape of B. rodriguezii presents a phenotype that arises as a balance between buffering development against perturbations and adaptive change in response to such perturbations or fluctuations. The biological robustness, defined here as the persistence of an organismal trait under perturbations (Félix and Wagner 2006), observed in the allometric trajectories exhibited by B. rodriguezii suggest environmental canalization (as defined by Wagner et al. 1997) of this species. Here we use canalization as defined by Willmore et al. (2007): a constraint on variability by restricting the scope of resultant variation expressed in the phenotype. On the other hand, the adaptation exhibited by P. purpuratus along its history of colonization of the South American coast can be the mechanism explaining the observed variation. Geographical expansion, from northern Chile to the Magellanic Biogeographic Province (Southern Chile and Southern Argentine), with a concurrent adaptation of marginal populations to a cold-temperate thermal regime, leading to incipient peripatric differentiation and the colonization of a new ecological niches (Trovant et al. 2015) is included in the adaptation P. purpuratus’s exhibited along the coast of South America. We found that P. purpuratus presents larger ontogenetic allometric variation than B. rodriguezii, thus being able to be more plastic in its phenotype. Therefore, phenotypic plastic responses to a new environmental variation along the ontogeny could help to expand the geographical distribution range via adaptation operating on a form to optimize the shell shape and buffering the environmental heterogeneity.
References
Adami ML, Pastorino G, Orensanz JM (2013) On the conchological differences between Brachidontes rodriguezii and B. purpuratus (Mollusca: Mytilidae) from the intertidal of the Argentine littoral. Malacologia 56:1–9
Aguirre ML, Perez IS, Negro Sirch Y (2006) Morphological variability of Brachidontes Swainson (Bivalvia, Mytilidae) in the marine Quaternary of Argentina (SW Atlantic). Palaeogeogr Palaeoclimatol Palaeoecol 239:100–125
Balech E, Ehrlich MD (2008) Esquema biogeográfico del mar argentino. Rev Inv y Des Pes (Mar del Plata) 19:45–75
Bertness MD, Mullan C, Silliman BR, Bazterrica MC, Reyna MV, Hidalgo F, Farin JK (2006) The community structure of western Atlantic Patagonian rocky shores. Ecol Monogr 76:429–460
Bookstein FL (1997) Morphometric tools for landmark data: geometry and biology. Cambridge University Press, Cambridge
Briggs JC, Bowen BW (2013) Marine shelf habitat: biogeography and evolution. J Biogeogr 40(6):1023–1035
Briones C, Guiñez R (2005) Bilateral asymmetry of shell shape and spatial position in matrices of the mussel Perumytilus purpuratus (Lamarck, 1819) (Bivalvia: Mytilidae). Rev Chil Hist Nat 78:3–14
Briones C, Guinez R, Garrido O, Oyarzun PA, Toro JE, Perez M (2012) Sperm polymorphism and genetic divergence in the mussel Perumytilus purpuratus. Mar Biol 159:1865–1870
Cavallotto JL (2008) Geología y geomorfología de los ambientes costeros y marinos. In: Boltovskoy D (ed) Atlas de Sensibilidad Ambiental del Mar y la costa Patagónica. Proyecto ARG 02/018 ‘Conservación de la Diversidad Biológica y Prevención de la Contaminación Marina en Patagonia’, donación GEF N°28385 (1994–1997). Gestión: Secretaría de Ambiente y Desarrollo Sustentable de la Nación, PNUD, Servicio de Hidrografía Naval, Fundación Funprecit
Cheverud JM (1989) A comparative analysis of morphological variation patterns in the Papionins. Evolution 43:1737–1747
Crampton JS, Maxwell PA (2000) Size: all it’s shaped up to be? Evolution of shape through the lifespan of the Cenozoic bivalve Spissatella (Crassatellidae). In: Harper EM, Taylor JD, Crame JA (eds) The evolutionary biology of the Bivalvia. Geological Society London, Special Publications, London, pp 399–423
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2009) InfoStat versión 2009. InfoStat Group, FCA, Universidad Nacional de Córdoba, Córdoba
Félix MA, Wagner A (2006) Robustness and evolution: concepts, insights and challenges from a developmental model system. Heredity 100(2):132–140
Fortin M-J, Gurevitch J (2001) Mantel tests: spatial structure in field experiments. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments, 2nd edn. Oxford University Press, New York, pp 308–326
Gould SJ (1966) Allometry and size in ontogeny and phylogeny. Biol Rev 41(4):587–638
Gould SJ (1977) Ontogeny and phylogeny. Harvard University Press, Cambridge
Griffiths CL, Griffiths RJ (1987) Bivalvia. In: Pandian TJ, Vernberg FJ (eds) Animal energetics. Academic Press, New York, pp 1–88
Gunz P, Mitteroecker P (2013) Semilandmarks: a method for quantifying curves and surfaces. Hystrix 24(1):103–109
Gunz P, Mitteroecker P, Bookstein FL (2005) Semilandmarks in three dimensions. In: Slice DE (ed) Modern morphometrics in physical anthropology. Klewer Academic/Plenum, New York, pp 73–98
Hawkins AJS, Bayne BL, Gosling E (1992) Physiological interrelations, and the regulation of production [in Mytilus]. Dev Aquac Fish Sci 25:171–222
Kimmel CB, Aguirre WE, Ullmann B, Currey M, Cresko WA (2008) Allometric change accompanies opercular shape evolution in Alaskan threespine sticklebacks. Behaviour 145(4):669–691
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Res 11:353–357
Klingenberg CP (2016) Size, shape, and form: concepts of allometry in geometric morphometrics. Dev Gen Evol 226(3):113–137
Klingenberg CP, Marugán-Lobón J (2013) Evolutionary covariation in geometric morphometric data: analyzing integration, modularity and allometry in a phylogenetic context. Syst Biol 6(4):591–610
Klingenberg CP, McIntyre GS (1998) Geometric morphometrics of developmental instability: analyzing patterns of fluctuating asymmetry with Procrustes methods. Evolution 5:363–1375
MacDougall-Shackleton EA, MacDougall-Shackleton SA (2001) Cultural and genetic evolution in mountain white-crowned sparrows: song dialects are associated with population structure. Evolution 55:2568–2575
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Márquez F, Van der Molen S (2011) Intraspecific shell-shape variation in the razor clam Ensis macha along the Patagonian coast. J Mollus Stud 77(2):123–128
Márquez F, Amoroso R, Gowland Sainz MF, Van der Molen S (2010) Shell morphology changes in the scallop Aequipecten tehuelchus during its life span: a geometric morphometric approach. Aquat Biol 11:149–155
Monteiro LR (1999) Multivariate regression models and geometric morphometrics: the search for causal factors in the analysis of shape. Syst Biol 48:192–199
Mosimann JE (1970) Size allometry: size and shape variables with characterization of the lognormal and generalized gamma distributions. J Am Stat Assoc 65:930–945
Piersma T, Van Gils JA (2011) The flexible phenotype: a body-centred integration of ecology, physiology, and behaviour. Oxford University Press, Oxford
Pigliucci M, Murren CJ, Schlichting CD (2006) Phenotypic plasticity and evolution by genetic assimilation. J Exp Biol 209(12):2362–2367
Piola AR, Falabella V (2009) El mar patagónico. In: Fallabela V, Campaña C, Croxall J (eds) Atlas del mar patagónico. Especies y espacios. Conservation Society and BirdLife Internacional, Buenos Aires, pp 55–75
Reimer O, Olsson B, Tedengren M (1995) Growth, physiological rates and behavior of Mytilus edulis exposed to the predator Asterias rubens. Mar Behav Physiol 25(5):233–244
Rohlf FJ (2004) TpsRelw, relative warps analysis, 1.35. Department of Ecology and Evolution, State University of New York, Stony Brook
Rohlf FJ, Slice D (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Biol 39(1):40–59
Rufino MM, Vasconcelos P, Pereira F, Fernández-Tajes J, Darriba S, Méndez J, Gaspar MB (2013) Geographical variation in shell shape of the pod razor shell Ensis siliqua (Bivalvia: Pharidae). Helgol Mar Res 67(1):49–58
Scarabino F, Zaffaroni JC, Clavijo C, Carranza A, Nin M (2006) Bivalvos marinos y estuarinos de la costa uruguaya: faunística, distribución, taxonomía y conservación. In: Menafra R, Rodríguez-Gallego L, Scarabino F, Conde D (eds) Bases para la Conservación y el Manejo de la Costa Uruguaya. Vida Silvestre Publicaciones, Montevideo, pp 157–170
Schellmann G, Radtke U (2000) ESR dating stratigraphically well-constrained marine terraces along the Patagonian Atlantic coast (Argentina). Quat Int 68:261–273
Seed R (1968) Factors influencing shell shape in the mussel Mytilus edulis. J Mar Biol Ass UK 48(3):561–584
Seed R, Suchanek TH (1992) Population and community ecology of Mytilus. In: Gossling EM (ed) The mussel Mytilus: ecology, physiology, genetics and culture. Developments in Aquaculture and Fisheries Science, Elsevier, Amsterdam, pp 87–170
Slice DE, Bookstein FL, Marcus LE, Rohlf FJ (1996) Appendix I: a glossary for geometric morphometrics. In: Marcus LE, Corti M, Loy A, Naylor GJP, Slice D (eds) Advances in morphometrics. Plenum Press, New York, pp 531–551
Sokal RR (1979) Testing statistical significance of geographic variation patterns. Syst Zool 28:227–232
Steffani CN, Branch GM (2003) Growth rate, condition, and shell shape of Mytilus galloprovincialis: responses to wave exposure. Mar Ecol Prog Ser 246:197–209
Trovant B, Ruzzante DE, Basso NG, Orensanz JM (2013) Distinctness, phylogenetic relations and biogeography of intertidal mussels (Brachidontes, Mytilidae) from the south-western Atlantic. J Mar Biol Ass UK 93:1843–1855
Trovant B, Orensanz JL, Ruzzante DE, Stotz W, Basso NG (2015) Scorched mussels (Bivalvia: Mytilidae: Brachidontinae) from the temperate coasts of South America: phylogenetic relationships, trans-Pacific connections and the footprints of quaternary glaciations. Mol Phyl Evol 82:60–74
Trovant B, Márquez F, del Río C, Ruzzante DE, Martínez S, Orensanz JM (2017) Insights on the history of the scorched mussel Brachidontes rodriguezii (Bivalvia: Mytilidae) in the Southwest Atlantic: a geometric morphometrics perspective. Hist Biol 1–9. https://doi.org/10.1080/08912963.2017.1306699
Tyberghein L, Verbruggen H, Pauly K, Troupin C, Mineur F, De Clerck O (2012) Bio-ORACLE: a global environmental dataset for marine species distribution modelling. Glob Ecol Biogeogr 21:272–281
Urdy S, Goudemand N, Bucher H, Chirat R (2010a) Allometries and the morphogenesis of the molluscan shell: a quantitative and theoretical model. J Exp Zool B Mol Dev Evol 314(4):280–302
Urdy S, Goudemand N, Bucher H, Chirat R (2010b) Growth-dependent phenotypic variation of molluscan shells: implications for allometric data interpretation. J Exp Zool B Mol Dev Evol 314(4):303–326
Valladares A, Manríquez G, Suárez-Isla B (2010) Shell shape variation in populations of Mytilus chilensis (Hupe 1854) from southern Chile: a geometric morphometric approach. Mar Biol 157(12):2731–2738
Van der Molen S, Márquez F, Idaszkin YL, Adami M (2013) Use of Shell-shape to discrimate between Brachidontes rodriguezii and Brachidontes purpuratus species (Mytilidae) in the transition zone of their distributions (South-western Atlantic). J Mar Biol Ass UK 93:803–808
van Schaik CP, Ancrenaz M, Gwendolyn B, Galdikas B, Knott CD, Singeton I, Suzuki A, Utami SS, Merrill M (2003) Orangutan cultures and the evolution of material culture. Science 299:102–105
Waddington CH (1942) The canalization of development and the inheritance of acquired characters. Nature 150(3811):563–565
Waddington CH (1957) Strategy of the genes. MacMillan, New York
Wagner GP, Booth G, Bagheri-Chaichian H (1997) A population genetic theory of canalization. Evolution 51:329–347
Wennersten L, Forsman A (2012) Population level consequences of polymorphism, plasticity, and randomized phenotype switching: a review of predictions. Biol Rev 87(3):756–767
Whittaker RH (1962) Classification of natural communities. Bot Rev 28(1):1–239
Willmore KE, Young NM, Richtsmeier JT (2007) Phenotypic variability: its components, measurement, and underlying developmental processes. Evol Biol 34(3–4):99–120
Wright TF, Wilkinson GS (2001) Population genetic structure and vocal dialects in an Amazon parrot. Proc R Soc Lond B Biol Sci 268:609–616
Zelditch ML, Fink WL (1996) Heterochrony and heterotopy: stability and innovation in the evolution of form. Paleobiology 22(2):241–254
Zelditch ML, Sheets HD, Fink WL (2000) Spatiotemporal reorganization of growth rates in the evolution of ontogeny. Evolution 54(4):1363–1371
Zelditch ML, Swiderski DL, Sheets HD, Fink WL (2012) Geometric morphometrics for biologists: a primer. Academic Press, New York
Acknowledgements
We thank Evangelina Schwindt, Veronica Savoya and Facundo Saleme for their help in the field and laboratory. Financial support was provided by the projects PIP1122015-0100241CO and PICT2015-3696 (FM). We thank the anonymous reviewers and Editorial Board Member for their thoughtful comments that enhanced the original version of this paper. This is publication #98 of the Laboratorio de Reproducción y Biología Integrativa de Invertebrados Marinos (LARBIM).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Márquez, F., Adami, M.L., Trovant, B. et al. Allometric differences on the shell shape of two scorched mussel species along the Atlantic South American Coast. Evol Ecol 32, 43–56 (2018). https://doi.org/10.1007/s10682-018-9928-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-018-9928-5