Abstract
In recent years, water purification by membrane desalination techniques has been growing drastically; after all, water scarcity is a significant issue to deal with in some parts of the world. To put one step forward toward resolving the issue of water scarcity, the best way is to upgrade the current desalination technique and membranes so that the output of clear water will be improved. In this review, we will focus on enhancing some crucial properties of the Polyamide (PA) and Polyimide (PI) membranes by incorporating some functional additives. Ag NPs (Silver nanoparticles), Cu NPs (Copper nanoparticles), GO (Graphene oxide), SWCNT (Single-walled carbon nanotube), and MWCNT (multi-walled carbon nanotube) are some of the additives which can be used with PA/PI active layer to improve some essential properties of membrane-like antifouling, biofouling, low water flux, selectivity, permeability, hydrophilicity, hydrophobicity, etc. The deposition of such additives onto the surface of the Polyimide/Polyamide coat or membrane can be done using interfacial polymerization or phase inversion. Membrane filtration can be done using reverse osmosis and electrodialysis techniques. A thin-film composite membrane comprising PA and MWCNTs, yielded a water flux of almost 25.9 L m−2 h−1, with a salt rejection of 98.1% exhibiting excellent hydrophilicity with a water contact angle of 59.6°.
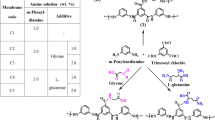
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Water scarcity is a significant issue in the upcoming days which means deficient availability of water resources to satisfy the amount of water usage within a particular country. This water scarcity results in more demand for water desalination [1, 2] as seawater is a significant source of salinated water [3]. So it is essential to have evolution in the current desalination processes as well as membranes employing upgrading technology or improving membrane properties by adding particular additives. The desalination process is a consumable energy process that utilizes non-conventional energy sources for operations. There are two main categories of desalination techniques: [4] membrane-based desalination and thermal desalination. The thermal desalination process has many sub-types like multistage flash (MSF) desalination, solar desalination, multi-effect distillation (MED), etc. Membrane desalination processes can be categorized into three major types, including forward osmosis, electrodialysis (ED), and reverse osmosis (RO) [5]. The desalination process involves the elimination of salts or unwanted minerals and impurities from water sources (which can be seawater, wastewater, etc.) [6], and it serves as an effective remedy for obtaining clean water for daily use for humans [5]. Membrane technology has brought about essential enhancements to the desalination sector, as it tends to be an energy-saving technique and is easily upgradable as compared to the thermal desalination process [7]. The reverse osmosis process is generally used for seawater desalination, while electrodialysis is widely used for brackish water [7]. Several examples of polymeric materials extensively used as desalination membranes, along with their chemical structures and desalination properties (salt rejection and flux), are enlisted in Table 1.
Membranes [4] made of polymeric materials have ruled the commercial market of desalination membranes since the initial stages of desalination [8]. A polymeric membrane comprising technical feasibility [9] enhances desalination membranes' performance in terms of permeability and selectivity [10]. An extensive array of materials capable of efficacious waste water treatment have been explored by researchers in this domain [4, 6, 11,12,13,14,15,16,17,18,19,20,21,22,23].
Desalination membranes can also be made of ceramics or inorganic substances, as well as mix matrix membranes (MMM). Mix matrix membrane (MMM) is a membrane consisting of both organic as well as inorganic elements in it, for example, nanoparticles/polymer (TiO2/Polyamide, etc.), carbon nanotubes/polymer (CNT/Polyimide) [8, 25], as illustrated in Fig. 1. Incorporating inorganic substances within organic desalination membranes significantly improves various membrane properties discriminatingly rendered by the individual component like exceptional biological, thermal and chemical stability of inorganic membranes combined with the favorable permselectivity, extended operation and greater packing density of polymeric membranes [10, 26,27,28].
This review emphasizes on specific polymers, including Polyamide (PA) and Polyimide (PI), and the mix matrix membrane (MMM) employing these polymers as desalination membranes [29]. The property alteration of these polymers through incorporation of some organic additives into the matrix material of the membrane to enhance various membrane properties [30,31,32] will be explored [1, 33] effectively without hindering the basic properties of the membrane. There can be many more permutations and combinations of these polymers with other additives [34] to achieve more enhanced desalination, on which further research can be done. A literature survey for this review article has been done in accordance with the SCOPUS data, Fig. 2 represents the number of yearly published articles based on Polyamide and Polyimide desalination membranes.
Polyamide-based system
Polyamide (PA) desalination membranes that are prepared through IP (interfacial polymerization) have played a significant role for the drastic growth of desalination techniques [35,36,37]. The inovation of a thin-film composite membrane made from Polyamides demonstrated excellent desalination performance, removing salt up to 99% from seawater by desalination through reverse osmosis. Cadotte and his coworkers, who discovered thin-film membrane in the late 70s, concluded that PA desalination membranes exhibited better performance compared to cellulosic desalination membranes [36, 38]. Interfacial condensation between m-phenylene diamide (MPD) and trimesoyl chloride (TMC) results in a wholly aromatic polyamide membrane [36, 37]. The interfacial polymerization technique consists of multiple condensation chemistries, involving the synthesis of Polyamide, Polyurethanes, Polycarbonate, etc. [37, 39, 40]. Polyamides also referred to as Nylons are primarily utilized for manufacturing synthetic fibers. Polyamides can be synthesized through self-condensation of amino acids or their cyclic lactum or via ring-opening polymerization and polycondensation of diamines and dibasic acids. Polyamides are a class of thermoplastic polymers. Recently many researches have been carried out on Polyamide, leading to the development of their nanocomposites. Newly developed Polyamides are filled with nanofibers resulting in the enhancement of various polymer properties [41].
Polyamide with metal-based additives
PA is compounded with different additives, resulting in the enhancement of performance [23] and efficiency of desalination membranes [42]. PA can be blended with an extensive array of additives while this review will be focusing on silver (Ag), copper (Cu), graphene oxide (GO), and carbon nanotubes (CNTs): Single-walled carbon nanotube (SWCNT) and multi-walled carbon nanotube (MWCNT) nanoparticles (NPs). Silver nanoparticles possessing very good bactericidal properties are employed in the manufacturing of anti-biofouling thin-film composite-membranes to enhance the antibactericidal capacities of the membrane material. It has been recorded that silver ions and silver nanoparticle compounds can hinder intracellular protein transportation and force the active bacteria to be inactive. Also, whenever the light is used as a catalyst in a reaction, Ag ions might form oxygen sensitive species, which helps in the application of membrane sterilization [3, 43].
Ming Qiu and his coworkers have studied the Zwitterion-silver nanocomposite used for forward osmosis technique in order to enhance the water flux as well as biofouling resistance properties of desalination membranes, in which they firstly synthesized a Polyethersulfone membrane by a non-solvent induced phase separation methodology [44]. The thickness of casted membrane was kept up to 150 µm. They synthesized a PA coat onto the exterior face of the support membrane via interfacial polymerization [45] with subsequent deposition of silver nanoparticles onto the Polyamide active layers via in-situ formation [46]. As a result of this experiment, the water flux was improved from 4.92 to 7.26 L m−2 h−1 while the hydrophilicity and water bounding properties of membrane were also ameliorated. Both membranes mean normal TFC membrane, and the Zwitterion-silver thin-film composite-membrane was suspended into bacteria for 2 h. The pristine thin-film membrane showed 58% E.coli retention, whereas the zwitterion-silver thin-film membrane showed E.coli retention of about only 4% and the antimicrobial efficiency was found to be greater than 96% [46]. Similar to this work, one group of researchers studied the incorporation of hydrophilic silver NPs onto the TFC Polyamide membrane which included the primary manufacturing of Polysulfone substrate by conventional phase inversion technique [47]. Silver nanoparticles were deposited onto the Polysulfone substrate via in-situ formation [48], leading to the generation of Polysulfone-silver nanoparticle substrates. The thin-film composite was prepared by interfacial polymerization [49] involving reactions on normal Polysulfone membrane as well as the Polysulfone-silver substrate. According to the concentration of AgNO3, they categorized membrane as TFCAg1, TFCAg5, TFCAg20, TFCAg50 and TFCAg100. TFC-Ag20 showed a water flux of 50 ± 4.2 l/m2h, 170% more than a thin-film membrane without an additive, silver. TFC-Ag20 demonstrated NaCl rejection up to 99.1 ± 0.1%, while membranes without any additives showed rejection of 97.4 ± 0.5%. The size of the nano-channel produced was about 2.5 nm. Overloading silver nanoparticles (TFC-Ag100) resulted in lower water permeability as well as poor salt rejection [50]. Also, graphene oxide (GO), silver (Ag), and titanium dioxide (TiO2) NPs can also be combined to form a nanocomposite of Ag@rGO@TiO2 and desalination membrane having an active Polyamide layer.. Hamidreza Abadikhah and his colleagues performed research on the similar topic in which graphene oxide was synthesized via the Hummer's method [51]. They prepared a suspension of TiO2 and AgNO3 in ethylene glycol solution followed by the subsequent incorporation of the GO susupension (prepared via Hummer’s method) to the suspension containing TiO2 and AgNO3 nanoparticles. Following the homogenization of these two suspensions, the mixture of these suspensions was subjected to microwave irradiation at 600 W for 5 min. Figure 3 depicts various stages involved in the synthesis of Ag@rGO@TiO2 composite via microwave irradiation technique.
The synthesis of Si3N4/Polyethersulfone substrate via phase inversion technique was reported [52]. In order to fabricate the TFC membrane, interfacial polymerization between m-Phenylenediamine (MPD) and Trimesoyl chloride (TMC) onto the deposited active layer of Polyamide on the PES/Si3N4 surface layer was performed resulting in the generation of a multi-functional thin-film membrane. Membrane nanofiltration experiments revealed that the flux of the multi-functional thin-film membrane was 1.7 times higher than the new TFC membrane. The as-prepared membrane exhibited an Na2SO3 salt rejection capacity of upto 96% [53]. Table 2 enlists the characteristics of MTFN membranes comprising 0.05–0.4 wt% Ag@rGO@TiO2.
Incorporation of metal–organic frameworks (MOFs) [54] into the membrane comprising of Polyamide can also improve the membrane properties. Alireza Zirehpour and his co-workers focused on studying the performance of desalination membranes along with their structural properties. They fabricated nano-scaled metal–organic framework particles by combining 1,3,5,-benzene tricarboxylic acid and silver to enhance forward osmosis technique. Firstly, the substrate membrane was synthesized using Polyvinylpyrrolidone (PVP) and Polyethersulfone. Then thin-film membrane of Polyamide was manufactured onto the surface of the Polyethersulfone layer via IP technique followed by the subsequent deposition of MOFs onto the surface of the Polyamide active layer through interfacial polymerization. It was ascertained that the water permeability was improved by 126% when the membrane consisted of 0.04% MOF (metal–organic frameworks). Modified TFN membranes showed enhanced desalination through forward osmosis. Table 3 enlists the water flux of TFN membranes incorporating MOF in different proportions.
Also, the surface of the Polyamide layer has become more hydrophilic in nature, thereby improving the fouling resistance of the membrane [55]. Silver nanoparticles can also be combined with some carbon nitrides to enhance the forward osmosis technique; Dong wang and his coworkers tried to improve the forward osmosis performance by using a triple-layered TFN (thin-film nanocomposite) membrane. They prepared carbon nanotube dispersion according to the following method [56, 57]: aCN/AP (Silver phosphate-loaded acidified graphitic carbon nitride) was synthesized by mixing carbon nitride with AP solution [3] followed by the deposition of carbon nanotubes (CNT) onto the Polyethersulfone substrate membrane by mixing distinct amount of CNT dispersion solution onto the substrate membrane. Polyamide active coat was formed on the upper surface of the support membrane, resulting in the generation of TFN membrane. With a similar process, i.e., interfacial polymerization, they prepared TFN-aCN/AP membrane. The hydrophilic nature of membrane was enhanced by this technique. The optimized TFN-aCN/AP membrane demonstrated greater water flux of 67.0 L m−2 h−1 while the backward salt flux was found to be lower than 3 g m−2 h−1. TFN-aCN/AP membrane exhibited very good antimicrobial properties toward both E. coli as well as S. aureus with sterilization rates of 99% and 92%, respectively [3]. Graphene oxides’ quantum dots could also be used in conjunction with silver NPs, this is another viable way for altering the properties of TFC membrane, and work contingent with this combination has been reported in this domain. Shuya li and their team worked on improving the antibacterial properties of Polyamide TFN. They added GOQD (graphene oxide quantum dots) filled with silver phosphate (GOQD/AP) into a PA active coat. The AP/GOQD nanocomposite was fabricated facilely via an electrostatically driven technique [58, 59]. They prepared thin-film nanofiltration (TFN) membranes and thin-film nanocomposites comprising AP/GOQD nanocomposite membranes via interfacial polymerization carried on Polyethersulfone (PES) membrane. The final structure included a base layer of PES substrate membrane onto a Polyamide active layer with a subsequent PA active layer on its top, deposited with GOQD/AP nanocomposite through interfacial polymerization. TFN containing GOQD/AP exhibited a good water flux of 39.6 L m−2 h−1 at the pressure of 15 bar. Salt rejection was retained at 98.2%. TFN membrane consisting of 50 mg L−1 GOQD/AP membrane exhibited robust antibacterial properties toward E.coli when the rate of sterilization was 99.9%. Furthermore, TFN-GOQD/AP50 demonstrated excellent anti-biofouling performance while performing RO technique [60]. Addition of silver NPs on the surface of TFC membrane showed ameliorated antifouling properties [61]. In this work, Ulrike M. Hirsch and their colleagues focused on improving the antifouling properties of the TFC membrane with help of plasma-enhanced magnetron sputtering for easing the reverse osmosis technique. They coated an active layer of Polyamide onto TFC membrane followed by plasma-activation to incorporate functional groups onto the membrane surface. Employing radio frequency magnetron sputtering at 13.56 MHz, Ag NPs were incorporated from a spherical sputter target onto the TFC membrane. The average diameter size of Ag NPs was around 30 nm. As a result of this experiment, the formation of biofilm consisting of Pseudomonas sp. was decreased by 64.6% at an average after a cultivation period of 14 days [62].
Table 4 enlists the properties demonstrated by Polyamide-based membranes integrated with different forms of functionalized silver nanoparticles.
Many researchers have concluded that incorporating metal nanoparticles like copper, etc., into polymeric membranes improved the antibacterial, fouling resistance, and anti-biofouling properties. Copper has been well known for its antibacterial activities for ages. Also, it is economically affordable. It is a perfect option as a biocidal agent [42]. Similar to silver, copper can also used in desalination membranes due to its various advantageous properties. In 2018, B. Rodrigues and his group members studied the incorporation of copper (Cu) nanoparticle and m-Phenylenediamine (MPD) onto the active coat of the TFC membrane to enhance their anti-biofouling properties, in which they firstly prepared a Polysulfone support membrane by using the phase inversion technique [63,64,65]. The TFC membrane was fabricated by interfacial polymerization of MPD and TMC onto the base membrane according to their methodology [65,66,67]. The modified membrane was fabricated by incorporating Cu-MPD by in-situ formation. Then the membrane was dipped into TMC solution, and interfacial polymerization was carried out, thereby curing the membrane. The modified membrane showed a decrease in hydrophilicity but had higher surface roughness. The membrane will possess copper toxicity because of its ability to release Cu2+ ions from the surface. Hence, while showing excellent antibacterial properties [68], it also possesses good anti-adhesion properties. Modified membrane showed salt removal of 97%. Modified membrane permeates the flux of 1.6 L m−2 h−1 bar−1, which is 1.3 times higher than that of the pristine TFC membrane [69]. In a similar way as silver, copper could also be used with GO to improve various particular properties of the fabricated membrane. E. A. Ali and his group of researchers focused on improving membrane performance via surface modification and chelation to improve desalination. Firstly, they prepared a thin-film membrane containing PA active layer by IP onto the commercial PS support membrane. Employing the similar process, i.e., IP, they incorporated GO onto the surface of Polyamide active layer. Then, this modified membrane was immersed in the aqueous solution of dehydrated copper chloride (CuCl2·2H2O). In conclusion, the PA-Cu2+-GO membrane possessed greater clean water permeability of 44.25 L m−2 h−1and solute water flux of 33.77 L m−2 h−1, whereas the unmodified membrane exhibited a clean water permeability of 21.36 L m−2 h−1. Modified membrane showed excellent salt rejection of ≥ 98.5% while improving the chlorine resistance as well as fouling resistance property of TFC membrane [70]. In 2014, Moshe Ben-Sasson and his coworkers tried to functionalize the surface of the TFC membrane with Cu nanoparticles to incorporate anti-microbial properties [71]. A thin-film composite membrane of Polyamide [72], was then impregnated into isopropanol and deionized water solution, followed immersion of the membrane into a Cu NPs dispersion solution in order to functionalize the membrane surface. Salt rejection of Cu-modified membrane was found to be 98.86 ± 0.27%. The water contact angle of the functionalized membrane was 45.6 ± 8.4°, while the pristine membrane showed a contact angle of 52.6 ± 5.3° thereby slightly improving its hydrophilicity. The surface roughness of the TFC membrane got enhanced from 95 ± 11 nm to 102 ± 17 nm [42]. Table 5 summarizes the antibacterial characteristics (% bacterial removal) of functionalized TFC membranes.
Polyamide with graphene oxide nanoparticles (GO NPs)
Shahrzad Shokrgozar Eslah and his team attempted to improve the forward osmosis water desalination technique by incorporating GO (graphene oxide) nanosheet into polyamide TFC membrane. Firstly, a TFC support substrate was prepared via the phase inversion technique. Then, a PA active coat was incorporated onto the surface of the base substrate via interfacial polymerization, followed by the integration of GO nanosheets onto the surface of the TFC membrane via the same process, i.e., IP. As per the loading of the GO nanosheet, the properties of the modified membrane were altered. As a result of this experiment, water permeability was improved upto 2.02 L m−2 h−1 by incorporating 0.1 wt% GO, but the addition of 0.2 wt% of GO into the membrane resulted in lower water flux values. The surface roughness of the TFC membrane increased with GO loading [73, 74]. Salt rejection was improved up to 88% with the GO loading of 0.1 wt%, and the salt reverse diffusion was decreased upto 39% [75, 76].
Research on "PA/ GO composite membrane for enhancing pervaporation desalination process" was performed by Xiaoying Zhao and his fellow researchers. GO in its nanoform was deposited onto the Polyacrylonitrile (PAN) substrate via pressure-assisted ultra-filtration with consequent interfacial polymerization. After deposition of GO on the membrane, the PA active layer was formed onto the GO composite membrane through IP. Figure 4 illustrates different layers present in the synthesized PA/GO/PAN-thin-film composite membrane.
Through experimental analysis it was ascertained that the hydrophilicity of the TFC membrane was ameliorated. The maximum water flux recorded was upto 37.1 L m−2 h−1. With a 50 nm layer of GO, the salt rejection was 99.80%, but the flux decreased to 28.1 L m−2 h−1 [77]. Coating of GO could be done on the support substrate in order to improve the hydrophilicity [78]. Saira Bano and her teammates were working on increasing the flux as well as the antifouling properties of the PA TFC membrane by the addition of GO NPs. GO was synthesized via Hummer's method and a coat of PA was incorporated via IP on the PSF ultra-filtration membrane [79]. Table 6 represents various characteristics of Polyamide-based composites incorporating functionalized copper nanoparticles.
Figure 5 illustrates the various stages involved in the fabrication of PA/GO TFC membranes. The blue-colored beads represented the GO particles depicting a color change of the membrane, while the IP shows the curing of the PA/GO TFC membrane.
The synthesized membrane with 0.3 wt% loading of GO showed a contact angle of 60 ± 1°, resulting in an enhancement of the hydrophilicity. Roughness (Ra) of the modified membrane was up to 16.38. Salt rejection of GO/PA TFC membrane was 87% and 97% for NaCl and MgSO4, respectively. The water flux of the modified TFC membrane was maintained above 80 L m−2 h−1 [80]. Polyamide can also be functionalized with bactericidal graphene quantum dots; S.Fatemeh Syedpour and their teammates focused on improving the shortcomings associated with the forward osmosis method like biofouling detrimentally impacting the operational expenses, the lifetime of membrane and flux efficiency through the incorporation of bactericidal graphene quantum dots (GQDs). GQD was synthesized by the direct pyrolysis of CA [81]. Polyethersulfone membrane was manufactured via the conventional phase inversion method, and an active Polyamide coat was formed onto the surface of the membrane via IP. As a result of incorporating GQD, the hydrophilic nature of the membrane was improved, and the contact angle decreased from 72.9° to 51° (for TFC with 0.5wt.% loading of GQD). The antibacterial properties of the modified membrane is shown in Table 7. When GQD loading was 0.1 wt%, the water permeability was 150% higher than that of the pristine TFC membrane [82].
Table 7 enlists the bacterial inactivation (%) of the TFC membranes modified with GQD.
Table 8 depicts the characteristics of Polyamide-based membranes encompassing functionalized GO nanoparticles.
Polyamide with carbon nanotube (CNT)
Hao Sun and his team of researchers were concentrated on improving the permeability of the TFC membrane by adding a Polyamide matrix to the MWCNT framework. They initially prepared PVDF [88] (Polyvinylidene fluoride) via a non-solvent-induced phase separation technique, followed by the production of a TFC membrane by synthesizing an active layer of PA onto the MWCNT framework. A framework of MWCNTs was deposited onto the substrate via vacuum filtration of the MWCNT suspension, with subsequent precipitation of the PA layer onto the support layer through IP. After analyzing the test results, they found out that the modified membrane demonstrated an excellent salt rejection of ≥ 99%. The contact angle property shown by the PA/MWCNT-TFC was 55.5°, whereas the pristine membrane showed a contact angle of 91.2° and the surface roughness was increased upto 53.11 nm [89]. A similar combination of materials could be used to improve the chlorine resistance and other important properties of the membrane. Junwo park and their teammates performed research on improving chlorine immunity of reverse osmosis membranes by incorporation of CNTs and they fabricated a TFC membrane encompassing MWCNT/PA via the interfacial polymerization technique.
The experiment resulted in a membrane permeate flux of 13.4 L m−2 h−1 and salt rejection of upto 92.5%. The chlorine resistance was determined by the decrease in the rejection after immersing the membrane in NaOCl (3000 ppm) solution for 4 h, after the test, the salt rejection had decreased from 92.5 to 76.5% [90]. CNTs could also be used in conjunction with titania nanotubes. I.wan Azelee and their coworkers focussed on improving the desalination of the Polyamide-TFC membrane by incorporating acid-treated carbon nanotubes-titania nanotubes (TNT). Initially, they had prepared the MWCNT and TNT hybrid composite via the hydrothermal method, after which, these hybrid composites were incorporated onto the surface of the support membrane simultaneously during IP of the polyamide coat. As a result of this experiment, researchers found NaCl salt rejection to be 97.9%. The contact angle property shown by the membrane was 69.66 ± 4.79° [91].
Table 9 represents various characteristics of Polyamide-based membranes encompassing functionalized CNTs.
In 2017, Javad Farahbaksh and his colleagues studied the effect of incorporating pristine and oxidized MWCNT onto a PA membrane to enhance the antifouling properties and performance efficiency. Pristine and oxidized MWCNTs were embedded within Polyamide reverse osmosis membranes via IP technique. Membranes with pristine MWCNT exhibited a water flux of 25.9 L m−2 h−1, while the membranes modified with oxidized MWCNT demonstrated a water flux of 28.9 L m−2 h−1. 98.1% and 97.8% were the salt rejection of membrane when modified with pristine and oxidized MWCNT, respectively. The membrane modified with oxidized MWCNT showed a water contact angle of 59.6° [92].
Polyimide (PI)-based system
Bo Feny and his team of researchers synthesized a mixed matrix membrane (MMM) of graphene oxide/Polyimide for desalination. An aqueous suspension solution of GO was prepared, and it was synthesized by the modified Hummer's method [97] by utilizing graphite as the starting material. PI composite membranes integrating asymmetric GO/PI MMMs were fabricated using the water bath technique. Modifying the as-prepared membrane with GO/PI, rendered a water contact angle of 59°. At 90 °C, water flux becomes 36.1 L m−2 h−1, and the salt rejection stays unchanged at 99% [98]. Additionally, Chaoyi Ba and co-workers modified P84 co-Polyimide membrane with Polyethyleneimine (PEI) to enhance nanofiltration. The P84 membrane was synthesized by the solution casting technique, followed by phase inversion. The chemical modification of P84 membrane within PEI solution engendered the formation of TFC membrane. As a result of this trial, the optimized membrane demonstrated salt rejection of 50.9 ± 5.1% and flux of 54.16 L m−2 h−1 at 13.88 bar pressure [99]. Apart from pristine Polyimide system, modified Polyimides were also studied, for instance, Geetanjali Shukla and her teammates focused on enhancing the desalination performance of the membrane by adding phosphorylated GO-sulfonated Polyimide (SPI) composite. Phosphorylated GO was synthesized by treating oxygenated GO with 3-aminopropyltrimethoxysilane [100]. Sulfonated Polyimide (SPI) was manufactured by sulfonation of 4,4’-bis(4-aminophenoxy)biphenyl as per their technique[101]. Finally, SPI/PGO -TFC membrane was fabricated via solution casting. Encompassing PGO with the membrane improved the membrane's oxidative, mechanical, and thermal resistant characteristics. The optimum SPI/PGO membrane possessed an ionic conductivity of 6.8 × 10–2 S/cm, which was most suitable for electrodialytic water desalination [102]. Chengyu Yan and their coworkers researched on improving the desalination of nanofiltration membranes by crosslinking PI with PA (MPD). Nanofiltration TFN membranes were synthesized via IP of Piperazine (PIP) and MPD [38, 103,104,105]. The as-prepared modified membranes demonstrated Na2SO4 and NaCl rejection upto 99.13% and 97.45%, respectively. The modified membranes also exhibited a water contact angle of 54.28° indicating hydrophilicity of the prepared membranes. After soaking the membrane insolvent for several weeks, the membrane showed more than 90% rejection of Na2SO4, indicating excellent solvent resistance [106]. Table 10 represents the characteristics of Polyimide-based composite desalination membranes incorporating different additives.
Conclusion and future scope
In upcoming days, the central issue of shortage of drinking and usable water will arise someday since there are minimal resources of usable water. So, in the future, there will be a significant need to improve desalination technology and apply desalination techniques on a large scale to meet the necessary water supply for daily use in the world. The heart of the desalination technique is at its membrane use since the actual salt removal is caused by a membrane only. Polymeric membranes are predominantly employed for desalination applications. Being lightweight, polymeric membranes provide better strength-to-weight ratio, and most importantly, they are corrosion resistant and readily available. In order to enhance the membrane characteristics, many researchers have combined various polymeric materials to synthesize a TFC membrane, for example, PSF/PA, PSF/PI, PAN/PA, etc. Another way to alter the membrane properties is to incorporate various functional additives into the membrane like silver, copper, CNTs, GO nanoparticles etc. These additives can be added to the surface of the membrane via a particular process to improve a particular property/set of properties of TFC membranes.
In this review, we have deeply emphasized on desalination membranes comprising Polyamide and Polyimide. Polyamide and Polyimide are used in combination with certain additives that are deposited on the surface of the membrane to improve a diverse array of membrane properties like water flux, salt rejection, hydrophilicity, fouling resistance, anti-bactericidal properties, etc. The following conclusions can be drawn from this review:
-
Anti-bacterial capabilities of silver NPs and copper NPs are the best, resulting in improved anti-biofouling properties.
-
The addition of graphene oxide to the TFC membrane enhances mechanical strength and increases hydrophilicity.
-
Incorporating CNT onto the membrane improved desalination at slightly higher temperatures without any membrane distortion.
-
Even a little excess loading of GO and CNTs in the TFC membrane can result in a significant reduction in permeability.
-
A life cycle evaluation of any modified TFC membrane may be performed to determine the membrane's effective life duration.
-
There can be desalination consisting of two different mixed matrix membranes, for example, the system containing PA/GO/PSF TFC membrane and A/PI/PSF TFC membrane arranged in an alternating manner to achieve maximum rejection or other requirements through a single system.
-
In most cases, Polysulfone, Polyethersulfone, Polyacrylonitrile and Polyvinylidene fluoride have been used as membrane support materials.
Availability of data and materials
All data generated or analyzed during this study are included in the submitted manuscript.
Code availability
Not applicable.
Abbreviations
- aCN/AP:
-
Silver phosphate-loaded acidified graphitic carbon nitride
- Ag:
-
Silver
- Ag NPs:
-
Silver nanoparticles
- AgNO3 :
-
Silver nitrate
- CNT:
-
Carbon nanotube
- Cu:
-
Copper
- CuCl2·2H2O:
-
Dehydrated copper chloride
- CuNPs:
-
Copper nanoparticles
- DAPPC:
-
1,4-Bis(3-aminopropyl)-piperazine propane carboxylate
- ED:
-
Electrodialysis
- GO:
-
Graphene oxide
- GO NPs:
-
Graphene oxide nanoparticles
- GOQD:
-
Graphene oxide quantum dots
- IP:
-
Interfacial polymerization
- MED:
-
Multi-effect desalination
- MOF:
-
Metal organic framework
- MSF:
-
Multistage flash desalination
- MTFN:
-
Multifunctional thin-film nanocomposite
- MMM:
-
Mix matrix membrane
- MPD:
-
M-phenylene diamine
- MWCNT:
-
Muti-walled carbon nanotube
- Na2SO3 :
-
Sodium sulfite
- Na2SO4 :
-
Sodium sulfate
- NaCl:
-
Sodium chloride
- PA:
-
Polyamide
- PAN:
-
Polyacrylonitrile
- PEI:
-
Polyethyleneimine
- PES:
-
Polyethersulfone
- PI:
-
Polyimide
- PIP:
-
Piperazine
- PSF:
-
Polysulfone
- PVDF:
-
Polyvinylidene fluoride
- PVP:
-
Polyvinyl pyrrolidone
- RO:
-
Reverse osmosis
- SPI:
-
Sulfonated polyimide
- SWCNT:
-
Single-walled carbon nanotube
- T (\(^\circ{\rm C} )\) :
-
Temperature
- TFC:
-
Thin-film nanocomposite
- TiO2 :
-
Titanium dioxide
- TMC:
-
Trimesoyl chloride
- TNT:
-
Titania nanotubes
References
Dixit F, Zimmermann K, Dutta R et al (2022) Application of MXenes for water treatment and energy-efficient desalination: a review. J Hazard Mater 423:127050. https://doi.org/10.1016/j.jhazmat.2021.127050
Kavitha VU, Kandasubramanian B (2020) Tannins for wastewater treatment. SN Appl Sci 2:1081. https://doi.org/10.1007/s42452-020-2879-9
Wang D, Li J, Gao B et al (2021) Triple-layered thin film nanocomposite membrane toward enhanced forward osmosis performance. J Memb Sci 620:118879. https://doi.org/10.1016/j.memsci.2020.118879
Gupta P, Lapalikar V, Kundu R, Balasubramanian K (2016) Recent Advances in membrane based waste water treatment technology: a review. Energy Environ Focus 5:241–267. https://doi.org/10.1166/eef.2016.1227
Gude VG (2018) Energy storage for desalination. Renew Energy Powered Desalin Handb Appl Thermodyn. https://doi.org/10.1016/B978-0-12-815244-7.00010-6
Rastogi S, Kandasubramanian B (2020) Application of electrospun materials in water treatment. In: Electrospun materials and their allied applications. Wiley, pp 151–183
Turek M, Mitko K, Piotrowski K et al (2017) Prospects for high water recovery membrane desalination. Desalination 401:180–189. https://doi.org/10.1016/j.desal.2016.07.047
Sunada K, Kikuchi Y, Hashimoto K, Fujishima A (1998) Bactericidal and detoxification effects of TiO2 thin film photocatalysts. Environ Sci Technol 32:726–728. https://doi.org/10.1021/es970860o
Issac MN, Kandasubramanian B (2021) Effect of microplastics in water and aquatic systems. Environ Sci Pollut Res 28:19544–19562. https://doi.org/10.1007/s11356-021-13184-2
Lee KP, Arnot TC, Mattia D (2011) A review of reverse osmosis membrane materials for desalination: development to date and future potential. J Memb Sci 370:1–22. https://doi.org/10.1016/j.memsci.2010.12.036
Gore PM, Purushothaman A, Naebe M, et al (2019) Nanotechnology for oil-water separation, pp 299–339
Pillai A, Kandasubramanian B (2020) Carbon xerogels for effluent treatment. J Chem Eng Data 65:2255–2270. https://doi.org/10.1021/acs.jced.0c00092
Simon S, Balasubramanian K (2018) Facile immobilization of camphor soot on electrospun hydrophobic membrane for oil-water separation. Mater Focus 7:295–303. https://doi.org/10.1166/mat.2018.1511
Mayilswamy N, Kandasubramanian B (2022) Green composites prepared from soy protein, polylactic acid (PLA), starch, cellulose, chitin: a review. Emergent Mater 5:727–753. https://doi.org/10.1007/s42247-022-00354-2
Subash A, Naebe M, Wang X, Kandasubramanian B (2023) Biopolymer: a sustainable and efficacious material system for effluent removal. J Hazard Mater 443:130168. https://doi.org/10.1016/j.jhazmat.2022.130168
Mayilswamy N, Boney N, Kandasubramanian B (2022) Fabrication and molecular dynamics studies of layer-by-layer polyelectrolytic films. Eur Polym J 163:110945. https://doi.org/10.1016/j.eurpolymj.2021.110945
Sharma S, Balasubramanian K (2015) Molecularly imprinted and nanoengineered camphor soot functionalized PAN-nanofibers for effluent treatment. RSC Adv 5:31732–31741. https://doi.org/10.1039/c5ra02861b
Rule P, Balasubramanian K, Gonte RR (2014) Uranium(VI) remediation from aqueous environment using impregnated cellulose beads. J Environ Radioact 136:22–29. https://doi.org/10.1016/j.jenvrad.2014.05.004
Gore PM, Naebe M, Wang X, Kandasubramanian B (2020) Silk fibres exhibiting biodegradability and superhydrophobicity for recovery of petroleum oils from oily wastewater. J Hazard Mater 389:121823. https://doi.org/10.1016/j.jhazmat.2019.121823
Nitesh Singh NS, Balasubramanian K (2014) An effective technique for removal and recovery of uranium (VI) from aqueous solution using cellulose–camphor soot nanofibers. RSC Adv 4:27691–27701. https://doi.org/10.1039/C4RA01751J
Mishra P, Balasubramanian K (2014) Nanostructured microporous polymer composite imprinted with superhydrophobic camphor soot, for emphatic oil–water separation. RSC Adv 4:53291–53296. https://doi.org/10.1039/C4RA07410F
Gore P, Khraisheh M, Kandasubramanian B (2018) Nanofibers of resorcinol–formaldehyde for effective adsorption of As (III) ions from mimicked effluents. Environ Sci Pollut Res 25:11729–11745. https://doi.org/10.1007/s11356-018-1304-z
Gonte RR, Shelar G, Balasubramanian K (2014) Polymer–agro-waste composites for removal of Congo red dye from wastewater: adsorption isotherms and kinetics. Desalin Water Treat 52:7797–7811. https://doi.org/10.1080/19443994.2013.833876
Zaman NK, Rohani R, Mohammad AW, Isloor AM (2017) Polyimide-graphene oxide nanofiltration membrane: characterizations and application in enhanced high concentration salt removal. Chem Eng Sci. https://doi.org/10.1016/j.ces.2017.11.034
Noy A, Park HG, Fornasiero F et al (2007) Nanofluidics in carbon nanotubes. Nano Today 2:22–29. https://doi.org/10.1016/S1748-0132(07)70170-6
Okumus E, Gurkan T, Yilmaz L (1994) Development of a mixed-matrix membrane for pervaporation. Sep Sci Technol 29:2451–2473. https://doi.org/10.1080/01496399408002203
Ismail AF, Goh PS, Sanip SM, Aziz M (2009) Transport and separation properties of carbon nanotube-mixed matrix membrane. Sep Purif Technol 70:12–26. https://doi.org/10.1016/j.seppur.2009.09.002
Udayakumar KV, Gore PM, Kandasubramanian B (2021) Foamed materials for oil-water separation. Chem Eng J Adv 5:100076. https://doi.org/10.1016/j.ceja.2020.100076
Deoray N, Kandasubramanian B (2018) Review on three-dimensionally emulated fiber-embedded lactic acid polymer composites: opportunities in engineering sector. Polym Plast Technol Eng 57:860–874. https://doi.org/10.1080/03602559.2017.1354226
Patadiya J, Gawande A, Joshi G, Kandasubramanian B (2021) Additive manufacturing of shape memory polymer composites for futuristic technology. Ind Eng Chem Res 60:15885–15912. https://doi.org/10.1021/acs.iecr.1c03083
Khatavkar N, Balasubramanian K (2016) Composite materials for supersonic aircraft radomes with ameliorated radio frequency transmission-a review. RSC Adv 6:6709–6718. https://doi.org/10.1039/C5RA18712E
Wan C, Zhao F, Bao X et al (2008) Surface characteristics of polyhedral oligomeric silsesquioxane modified clay and its application in polymerization of macrocyclic polyester oligomers. J Phys Chem B 112:11915–11922. https://doi.org/10.1021/jp805259q
Sahoo BN, Kandasubramanian B (2014) An experimental design for the investigation of water repellent property of candle soot particles. Mater Chem Phys 148:134–142. https://doi.org/10.1016/j.matchemphys.2014.07.022
Mayilswamy N, Jaya Prakash N, Kandasubramanian B (2022) Design and fabrication of biodegradable electrospun nanofibers loaded with biocidal agents. Int J Polym Mater Polym Biomater. https://doi.org/10.1080/00914037.2021.2021905
Cadotte JE, Petersen RJ, Larson RE, Erickson EE (1980) A new thin-film composite seawater reverse osmosis membrane. Desalination 32:25–31. https://doi.org/10.1016/S0011-9164(00)86003-8
Cadotte JE (1985) Evolution of composite reverse osmosis membranes, pp 273–294
Freger V, Ramon GZ (2021) Polyamide desalination membranes: Formation, structure, and properties. Prog Polym Sci 122:101451. https://doi.org/10.1016/j.progpolymsci.2021.101451
Petersen RJ (1993) Composite reverse osmosis and nanofiltration membranes. J Memb Sci 83:81–150. https://doi.org/10.1016/0376-7388(93)80014-O
Morgan PW (2011) Interfacial polymerization. In: Encyclopedia of polymer science and technology. Wiley, Hoboken
Raaijmakers MJT, Benes NE (2016) Current trends in interfacial polymerization chemistry. Prog Polym Sci 63:86–142. https://doi.org/10.1016/j.progpolymsci.2016.06.004
Feldman D (2017) Polyamide nanocomposites. J Macromol Sci Part A 54:255–262. https://doi.org/10.1080/10601325.2017.1282700
Ben-Sasson M, Zodrow KR, Genggeng Q et al (2014) Surface functionalization of thin-film composite membranes with copper nanoparticles for antimicrobial surface properties. Environ Sci Technol 48:384–393. https://doi.org/10.1021/es404232s
Yu L, Zhou W, Li Y et al (2019) Antibacterial thin-film nanocomposite membranes incorporated with graphene oxide quantum dot-mediated silver nanoparticles for reverse osmosis application. ACS Sustain Chem Eng 7:8724–8734. https://doi.org/10.1021/acssuschemeng.9b00598
He M, Fan X, Yang Z et al (2016) Antifouling high-flux membranes via surface segregation and phase separation controlled by the synergy of hydrophobic and hydrogen bond interactions. J Memb Sci 520:814–822. https://doi.org/10.1016/j.memsci.2016.08.044
Yip NY, Tiraferri A, Phillip WA et al (2010) High performance thin-film composite forward osmosis membrane. Environ Sci Technol 44:3812–3818. https://doi.org/10.1021/es1002555
Qiu M, He C (2018) Novel zwitterion-silver nanocomposite modified thin-film composite forward osmosis membrane with simultaneous improved water flux and biofouling resistance property. Appl Surf Sci 455:492–501. https://doi.org/10.1016/j.apsusc.2018.06.020
Ma X-H, Yang Z, Yao Z-K et al (2017) A facile preparation of novel positively charged MOF/chitosan nanofiltration membranes. J Memb Sci 525:269–276. https://doi.org/10.1016/j.memsci.2016.11.015
Ben-Sasson M, Lu X, Bar-Zeev E et al (2014) In situ formation of silver nanoparticles on thin-film composite reverse osmosis membranes for biofouling mitigation. Water Res 62:260–270. https://doi.org/10.1016/j.watres.2014.05.049
Sharma L, Ye L, Yong C et al (2022) Aquaporin-based membranes made by interfacial polymerization in hollow fibers: visualization and role of aquaporin in water permeability. J Memb Sci 654:120551. https://doi.org/10.1016/j.memsci.2022.120551
Yang Z, Guo H, Yao Z et al (2019) Hydrophilic silver nanoparticles induce selective nanochannels in thin film nanocomposite polyamide membranes. Environ Sci Technol 53:5301–5308. https://doi.org/10.1021/acs.est.9b00473
Dong G, Hou J, Wang J et al (2016) Enhanced CO2/N2 separation by porous reduced graphene oxide/Pebax mixed matrix membranes. J Memb Sci 520:860–868. https://doi.org/10.1016/j.memsci.2016.08.059
Abadikhah H, Kalali EN, Behzadi S et al (2018) Amino functionalized silica nanoparticles incorporated thin film nanocomposite membrane with suppressed aggregation and high desalination performance. Polymer (Guildf) 154:200–209. https://doi.org/10.1016/j.polymer.2018.09.007
Abadikhah H, Naderi Kalali E, Khodi S et al (2019) Multifunctional thin-film nanofiltration membrane incorporated with reduced graphene oxide@TiO2@Ag nanocomposites for high desalination performance, dye retention, and antibacterial properties. ACS Appl Mater Interfaces 11:23535–23545. https://doi.org/10.1021/acsami.9b03557
Gonte RR, Deb PC, Balasubramanian K (2013) Hydrogen sorption onto hypercrosslinked polymer decorated with metal-organic framework. J Polym 2013:1–8. https://doi.org/10.1155/2013/684584
Zirehpour A, Rahimpour A, Ulbricht M (2017) Nano-sized metal organic framework to improve the structural properties and desalination performance of thin film composite forward osmosis membrane. J Memb Sci 531:59–67. https://doi.org/10.1016/j.memsci.2017.02.049
Wang Z, Wang Z, Lin S et al (2018) Nanoparticle-templated nanofiltration membranes for ultrahigh performance desalination. Nat Commun 9:2004. https://doi.org/10.1038/s41467-018-04467-3
Zhu Y, Xie W, Gao S et al (2016) Single-walled carbon nanotube film supported nanofiltration membrane with a nearly 10 nm thick polyamide selective layer for high-flux and high-rejection desalination. Small 12:5034–5041. https://doi.org/10.1002/smll.201601253
Chen X, Dai Y, Wang X et al (2015) Synthesis and characterization of Ag3PO4 immobilized with graphene oxide (GO) for enhanced photocatalytic activity and stability over 2,4-dichlorophenol under visible light irradiation. J Hazard Mater 292:9–18. https://doi.org/10.1016/j.jhazmat.2015.01.032
Yang X, Cai H, Bao M et al (2018) Insight into the highly efficient degradation of PAHs in water over graphene oxide/Ag3PO4 composites under visible light irradiation. Chem Eng J 334:355–376. https://doi.org/10.1016/j.cej.2017.09.104
Li S, Gao B, Wang Y et al (2019) Antibacterial thin film nanocomposite reverse osmosis membrane by doping silver phosphate loaded graphene oxide quantum dots in polyamide layer. Desalination 464:94–104. https://doi.org/10.1016/j.desal.2019.04.029
Ambekar RS, Kandasubramanian B (2020) Antimicrobial electrospun materials. In: Electrospun materials and their allied applications. Wiley, pp 483–514
Hirsch UM, Teuscher N, Rühl M, Heilmann A (2019) Plasma-enhanced magnetron sputtering of silver nanoparticles on reverse osmosis membranes for improved antifouling properties. Surf Interfaces 16:1–7. https://doi.org/10.1016/j.surfin.2019.04.003
Bagchi B, Dey S, Bhandary S et al (2012) Antimicrobial efficacy and biocompatibility study of copper nanoparticle adsorbed mullite aggregates. Mater Sci Eng C 32:1897–1905. https://doi.org/10.1016/j.msec.2012.05.011
Ren G, Hu D, Cheng EWC et al (2009) Characterisation of copper oxide nanoparticles for antimicrobial applications. Int J Antimicrob Agents 33:587–590. https://doi.org/10.1016/j.ijantimicag.2008.12.004
García A, Quintero Y, Vicencio N et al (2016) Influence of TiO 2 nanostructures on anti-adhesion and photoinduced bactericidal properties of thin film composite membranes. RSC Adv 6:82941–82948. https://doi.org/10.1039/C6RA17999A
García A, Rodríguez B, Oztürk D et al (2018) Incorporation of CuO nanoparticles into thin-film composite reverse osmosis membranes (TFC-RO) for antibiofouling properties. Polym Bull 75:2053–2069. https://doi.org/10.1007/s00289-017-2146-4
Zhang L, Wang H, Yu W et al (2012) Facile and large-scale synthesis of functional poly(m-phenylenediamine) nanoparticles by Cu2+-assisted method with superior ability for dye adsorption. J Mater Chem 22:18244. https://doi.org/10.1039/c2jm32859c
Balasubramanian K, Yadav R, Prajith P (2015) Antibacterial nanofibers of polyoxymethylene/gold for pro-hygiene applications. Int J Plast Technol 19:363–367. https://doi.org/10.1007/s12588-015-9127-y
Rodríguez B, Oztürk D, Rosales M et al (2018) Antibiofouling thin-film composite membranes (TFC) by in situ formation of Cu-(m-phenylenediamine) oligomer complex. J Mater Sci 53:6325–6338. https://doi.org/10.1007/s10853-018-2039-4
Ali MEA, Hassan FM, Feng X (2016) Improving the performance of TFC membranes via chelation and surface reaction: applications in water desalination. J Mater Chem A 4:6620–6629. https://doi.org/10.1039/C6TA01460G
Ramdayal BK (2014) Antibacterial application of polyvinylalcohol-nanogold composite membranes. Colloids Surf A Physicochem Eng Asp 455:174–178. https://doi.org/10.1016/j.colsurfa.2014.04.050
Issac MN, Kandasubramanian B (2020) Review of manufacturing three-dimensional-printed membranes for water treatment. Environ Sci Pollut Res 27:36091–36108. https://doi.org/10.1007/s11356-020-09452-2
Ma N, Wei J, Liao R, Tang CY (2012) Zeolite-polyamide thin film nanocomposite membranes: towards enhanced performance for forward osmosis. J Memb Sci 405–406:149–157. https://doi.org/10.1016/j.memsci.2012.03.002
Hu M, Mi B (2013) Enabling graphene oxide nanosheets as water separation membranes. Environ Sci Technol 47:3715–3723. https://doi.org/10.1021/es400571g
Eslah SS, Shokrollahzadeh S, Jazani OM (2017) Forward osmosis water desalination : Fabrication of graphene oxide-polyamide/polysulfone thin-film nanocomposite membrane with high water flux and low reverse salt diffusion. Sep Sci Technol. https://doi.org/10.1080/01496395.2017.1398261
Purabgola A, Mayilswamy N, Kandasubramanian B (2022) Graphene-based TiO2 composites for photocatalysis & environmental remediation: synthesis and progress. Environ Sci Pollut Res 29:32305–32325. https://doi.org/10.1007/s11356-022-18983-9
Zhao X, Tong Z, Liu X, et al (2020) Facile preparation of polyamide-graphene oxide composite membranes for upgrading pervaporation desalination performances of hypersaline solutions. https://doi.org/10.1021/acs.iecr.0c01417
Thakur K, Kandasubramanian B (2019) Graphene and graphene oxide-based composites for removal of organic pollutants: a review. J Chem Eng Data 64:833–867. https://doi.org/10.1021/acs.jced.8b01057
Kim I-C, Jeong B-R, Kim S-J, Lee K-H (2013) Preparation of high flux thin film composite polyamide membrane: the effect of alkyl phosphate additives during interfacial polymerization. Desalination 308:111–114. https://doi.org/10.1016/j.desal.2012.08.001
Bano S, Mahmood A, Kim S, Lee K (2014) membrane with improved fl ux and antifouling properties. https://doi.org/10.1039/C4TA03607G
Dong Y, Chen C, Zheng X et al (2012) One-step and high yield simultaneous preparation of single- and multi-layer graphene quantum dots from CX-72 carbon black. J Mater Chem 22:8764. https://doi.org/10.1039/c2jm30658a
Seyedpour SF, Rahimpour A, Shamsabadi AA, Soroush M (2018) Graphical abstract. Chem Eng Res Des. https://doi.org/10.1016/j.cherd.2018.09.041
Pang R, Zhang K (2017) RSC Advances A facile and viable approach to fabricate polyamide membranes functionalized with graphene oxide. RSC Adv 7:53463–53471. https://doi.org/10.1039/C7RA11358G
Shi J, Wu W, Xia Y et al (2018) Con fi ned interfacial polymerization of polyamide-graphene oxide composite membranes for water desalination Pure water MPD solution. Desalination 441:77–86. https://doi.org/10.1016/j.desal.2018.04.030
Inurria A, Cay-durgun P, Rice D et al (2018) Polyamide thin- fi lm nanocomposite membranes with graphene oxide nanosheets: balancing membrane performance and fouling propensity. Desalination. https://doi.org/10.1016/j.desal.2018.07.004
Abbaszadeh M, Krizak D, Kundu S (2019) Layer-by-layer assembly of graphene oxide nanoplatelets embedded desalination membranes with improved chlorine resistance. Desalination 470:114116. https://doi.org/10.1016/j.desal.2019.114116
Yi Z, Shao F, Yu L et al (2020) Chemical grafting N-GOQD of polyamide reverse osmosis membrane with improved chlorine resistance, water fl ux and NaCl rejection. Desalination 479:114341. https://doi.org/10.1016/j.desal.2020.114341
Sahoo BN, Balasubramanian K (2015) A nanocellular PVDF–graphite water-repellent composite coating. RSC Adv 5:6743–6751. https://doi.org/10.1039/C4RA06704E
Sun H, Li D, Liu B, Yao J (2019) Enhancing the permeability of TFC membranes based on incorporating polyamide matrix into MWCNTs framework. Appl Surf Sci 496:143680. https://doi.org/10.1016/j.apsusc.2019.143680
Taylor P, Park J, Choi W, et al. Desalination and water treatment enhancement of chlorine resistance in carbon nanotube based nanocomposite reverse osmosis membranes enhancement of chlorine resistance in carbon nanotube-based nanocomposite reverse osmosis membranes. 37–41. https://doi.org/10.5004/dwt.2010.1686
Azelee IW, Goh PS, Lau WJ et al (2017) Enhanced desalination of polyamide thin fi lm nanocomposite incorporated with acid treated multiwalled carbon nanotube-titania nanotube hybrid. Desalination 409:163–170. https://doi.org/10.1016/j.desal.2017.01.029
Farahbaksh J, Delnavaz M, Vatanpour V (2017) Investigation of raw and oxidized multiwalled carbon nanotubes in fabrication of reverse osmosis polyamide membranes for improvement in desalination and antifouling properties. Desalination 410:1–9. https://doi.org/10.1016/j.desal.2017.01.031
Park J, Choi W, Kim SH et al (2010) Enhancement of chlorine resistance in carbon nanotube based nanocomposite reverse osmosis membranes. Desalin Water Treat 15:198–204. https://doi.org/10.5004/dwt.2010.1686
Mahdavi MR, Delnavaz M, Vatanpour V (2017) Fabrication and water desalination performance of piperazine–polyamide nanocomposite nanofiltration membranes embedded with raw and oxidized MWCNTs. J Taiwan Inst Chem Eng 75:189–198. https://doi.org/10.1016/j.jtice.2017.03.039
Rashed AO, Esawi AMK, Ramadan AR (2020) Novel polysulfone/carbon nanotube-polyamide thin film nanocomposite membranes with improved water flux for forward osmosis desalination. ACS Omega 5:14427–14436. https://doi.org/10.1021/acsomega.0c00973
Zhao M, Zhang HF, Huang H, Zhang YS (2017) Preparation and properties of nanocomposite MWCNTs/polyamide reverse osmosis membrane for desalination by interfacial polymerization. Key Eng Mater 727:1016–1025. https://doi.org/10.4028/www.scientific.net/KEM.727.1016
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339–1339. https://doi.org/10.1021/ja01539a017
Huang A, Feng B (2018) Synthesis of novel graphene oxide-polyimide hollow fi ber membranes for seawater desalination. J Memb Sci 548:59–65. https://doi.org/10.1016/j.memsci.2017.11.016
Ba C, Langer J, Economy J (2009) Chemical modification of P84 copolyimide membranes by polyethylenimine for nanofiltration. 327:49–58. https://doi.org/10.1016/j.memsci.2008.10.051
Pandey RP, Shahi VK (2014) A N-o-sulphonic acid benzyl chitosan (NSBC) and N,N-dimethylene phosphonic acid propylsilane graphene oxide (NMPSGO) based multi-functional polymer electrolyte membrane with enhanced water retention and conductivity. RSC Adv 4:57200–57209. https://doi.org/10.1039/C4RA09581B
Tripathi BP, Chakrabarty T, Shahi VK (2010) Highly charged and stable cross-linked 4,4′-bis(4-aminophenoxy)biphenyl-3,3′-disulfonic acid (BAPBDS)-sulfonated poly(ether sulfone) polymer electrolyte membranes impervious to methanol. J Mater Chem 20:8036. https://doi.org/10.1039/c0jm01183e
Shukla G, Pandey RP, Shahi VK (2016) Author’s accepted manuscript improved performance. Elsevier
Wang S, Yi Z, Zhao X et al (2017) Aggregation suppressed thin film nanocomposite (TFN) membranes prepared with an in situ generation of TiO 2 nanoadditives. RSC Adv 7:26136–26144. https://doi.org/10.1039/C7RA02374J
Jeong B-H, Hoek EMV, Yan Y et al (2007) Interfacial polymerization of thin film nanocomposites: a new concept for reverse osmosis membranes. J Memb Sci 294:1–7. https://doi.org/10.1016/j.memsci.2007.02.025
Lee HS, Im SJ, Kim JH et al (2008) Polyamide thin-film nanofiltration membranes containing TiO2 nanoparticles. Desalination 219:48–56. https://doi.org/10.1016/j.desal.2007.06.003
Yang C, Xu W, Nan Y, Wang Y (2020) Novel solvent-resistant nanofiltration membranes using MPD co-crosslinked polyimide for efficient desalination. J Memb Sci 616:118603. https://doi.org/10.1016/j.memsci.2020.118603
Feng B, Xu K, Huang A (2016) RSC advances matrix membranes for desalination†. RSC Adv 7:2211–2217. https://doi.org/10.1039/C6RA24974D
Hamdy G, Taher A (2020) Enhanced chlorine-resistant and low biofouling reverse osmosis polyimide-graphene oxide thin film nanocomposite membranes for water desalination. Polym Eng Sci 60:2567–2580. https://doi.org/10.1002/pen.25495
Acknowledgements
The author would like to acknowledge Dr. S. P. Bhosle, Principal & Head, Maharashtra Institute of Technology, Aurangabad, Dr. Aniruddha Chatterjee, Head of Department, Plastic and polymer engineering, Maharashtra Institute of Technology, Aurangabad and Mrs. Suranjana Mandal, Associate Professor, Maharashtra Institute of Technology, Aurangabad and Dr. CP Ramanarayanan, Vice Chancellor of DIAT (DU), Pune, for their continuous encouragement and support. Authors wish to extend special thanks to Dr. Amrita Nighojkar, Miss. Niranjana Jaya Prakash, Mr. Jigar Patadiya, and Miss. Alsha Subash for their unwavering and continuous technical support throughout the review writing. The authors are thankful to the editors and anonymous reviewers who has helped in enhancing the quality of the manuscript.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
Y A Ghodke contributed in Material preparation, data collection analysis and writing of the article. N Mayilswamy contributed to material preparation and writing of the article. B Kandasubramanian made substantial contribution to conceptualization, discussion and reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent to participate
Not applicable.
Consent for publication
The authors consent to publish the article on acceptance.
Ethics approval
The submitted article complies with the ethical guidelines of the journal and does not contain the results of studies involving humans and/or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghodke, Y.A., Mayilswamy, N. & Kandasubramanian, B. Polyamide (PA)- and Polyimide (PI)-based membranes for desalination application. Polym. Bull. 80, 10661–10695 (2023). https://doi.org/10.1007/s00289-022-04559-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04559-7