Abstract
Somitogenesis is the process for the development of somites in vertebrate embryos. This process is timely regulated by synchronous oscillatory expression of the segmentation clock genes. Mathematical models expressed by delay equations or ODEs have been proposed to depict the kinetics of these genes in interacting cells. Through mathematical analysis, we investigate the parameter regimes for synchronous oscillations and oscillation-arrested in an ODE model and a model with transcriptional and translational delays, both with Michaelis–Menten type degradations. Comparisons between these regimes for the two models are made. The delay model has larger capacity to accommodate synchronous oscillations. Based on the analysis and numerical computations extended from the analysis, we explore how the periods and amplitudes of the oscillations vary with the degradation rates, synthesis rates, and coupling strength. For typical parameter values, the period and amplitude increase as some synthesis rate or the coupling strength increases in the ODE model. Such variational properties of oscillations depend also on the magnitudes of time delays in delay model. We also illustrate the difference between the dynamics in systems modeled with linear degradation and the ones in systems with Michaelis–Menten type reactions for the degradation. The chief concerns are the connections between the dynamics in these models and the mechanism for the segmentation clocks, and the pertinence of mathematical modeling on somitogenesis in zebrafish.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Mathematical modeling for gene regulation can easily go up to multiple or high dimensions, if several genes, their mRNA and protein products are taken into account. Such models can be expressed by coupled nonlinear systems. Aside from the complicated biochemical details, there are time delays involved in the process, including the synthesis of mRNA and proteins, and the modification and transport of molecules. The amount of time lags in these processes may extend to tens of minutes. It is therefore reasonable to incorporate delays into modeling of such processes. Mathematical techniques for analyzing such coupled nonlinear systems with multiple delays become essential in understanding the dynamical properties of those models.
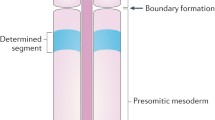
One interesting instance is the modeling of somitogenesis in vertebrate development. Somites are segmental structures arising one by one from the presomitic mesoderm (PSM) and laying along the antero-posterior axis of vertebrate embryos. They later develop, through further differentiation, into vertebrate, rib, and tail. The process during the development of somites is called somitogenesis. It involves gene regulation in both space and time. Somite segmentation depends on oscillatory gene expression for cells in the PSM, with neighboring cells oscillating in synchrony. In zebrafish, the involved oscillating genes include her1, her7, and cells interact through Delta–Notch signaling (Horikawa et al. 2006; Jiang et al. 2000; Mara et al. 2007; Özbudak and Lewis 2008; Riedel-Kruse et al. 2007). Mutations in the Notch cell–cell signaling pathway disrupt synchronization and somite formation and lead to defective somite boundary and irregular segmented body axis (Jiang et al. 2000; Lewis 2003). Therefore, both oscillation and synchronization of clock gene expression are necessary dynamics for normal segmentation. In addition, the oscillation period of clock genes in the tail bud of zebrafish is about 30 min which matches the period of somites made (Hanneman and Westerfield 1989; Holley 2007). This shows that the expressions of the clock genes play an important role in somitogenesis and mathematical model can help understand the processes during somitogenesis.
Autorepression of clock genes by its own protein product has been advocated as the main mechanism for the single cell oscillations in the zebrafish segmentation clock. A simple mathematical model for such regulation involving the mRNA and protein of either her1 or her7 gene was proposed in Lewis (2003). A key feature of this system is the inclusion of transcriptional and translational time delays. On the other hand, by considering the processes in the cell responsible for time delays, an ODE system was studied in Uriu et al. (2010). Therein, the transport of Her protein from cytoplasm to nucleus is taken into account, so that the protein concentrations in cytoplasm and nucleus are both state variables. Although many components of the segmentation clock in zebrafish have been identified in the past decade, further detailed regulatory interactions among these components, and the associated biochemical evidences were recently explored. That the core of the clock’s regulatory circuit consisting two distinct negative feedback loops, one with Her1 homodimers and the other with Her7:Hes6 heterodimers, was reported in Schröter et al. (2012). More complete models taking into account mRNA molecules of her1, her7, hes6, deltaC, and their monomer proteins, and dimer proteins have been investigated (Ay et al. 2013, 2014). Further understanding of the transcriptional regulation of each her/hes gene in the clock was achieved through experiments and computational modeling in Schwendinger-Schreck et al. (2014).
Normal segmentation relies on well-timed oscillation of clock genes and synchronization over neighboring cells. Cell–cell communication via the Delta–Notch signaling synchronizes adjacent cells so that their gene expressions oscillate in phase. However, as pointed out in Uriu et al. (2010), how synchronized oscillation is achieved is not obvious, as Delta protein in a cell stimulates the expression of her clock gene in neighboring cells, but the uprising expression of her in these cells in turn suppresses their delta genes. Mathematical justification of such a mechanism is therefore of interest. On the other hand, it was reported that when the Notch signaling is absent, the clock desynchronization is due to the stochastic dissociation of Her1/7 repressor proteins from the oscillating her1/7 autorepressed target genes (Jenkins et al. 2015).
The “clock and wave” mechanism for the formation of somites was first proposed in Cooke and Zeeman (1976). Based on the experimental evidences in Dubrulle et al. (2001), Dubrulle and Pourquié (2002), Pourquié (2004) and Baker et al. (2006) developed a “clock and wavefront” model to investigate pattern formation of somites, which depends on the gradient of FGF8 expression along the antero-posterior axis of vertebrate embryos. That when the boundaries of somites form was later found to be determined by the clock gene (Özbudak and Lewis 2008). To depict the posterior-to-anterior slowing of oscillation rate, Cinquin (2007) proposed a multicellular model for zebrafish somitogenesis that involves heterodimerization of clock proteins Her1 and Her7, with the control protein Hes6; the later interacts with clock proteins and controls the rate of oscillation (Kawamura et al. 2005; Sieger et al. 2006). Campanelli and Gedeon (2010) constructed a delay model extended from Lewis’s model to study how the transcription binding sites and decay rates for clock protein monomers and dimers affect the formation of the gene-expression wave. In Uriu et al. (2009), traveling wave solutions on a lattice system consisting of the ODEs with some of the reaction rates formulated in gradient along the lattice was investigated. Contrary to this result, a recent experimental and computational study indicates that Her7 protein degrades uniformly along the PSM (Ay et al. 2014). Instead, it was asserted that an increasing gradient of gene expression time delays from the posterior to the anterior leads to the traveling wave patterns (Ay et al. 2013, 2014). We note that in the mathematical models, linear degradation was adopted in Lewis (2003), Cinquin (2007), Schröter et al. (2012), Ay et al. (2013) and Jenkins et al. (2015), whereas the Michaelis–Menten reaction for degradation was employed in Uriu et al. (2009, 2010). The reason for adopting Michaelis–Menten reaction is that degradation of proteins generally involves enzymatic reactions. We shall compare these formulations in Sect. 2.
The above-mentioned kinetic models were regarded complicated and difficult to analyze mathematically, as commented in Baker and Schnell (2009). Researchers turned to seek other theoretical framework to encompass both temporal and spatial aspects of somitogenesis. By focussing on the collective behaviors in terms of the phase in a collection of cells clocks, rather than concentrating on the internal machinery of cells, a coupled phase oscillator model has been employed in Morelli et al. (2009) and Herrgen et al. (2010). Such system of phase equations is adapted from the Kuramoto’s model which is closely associated with the theory of weakly coupled system for neuronal dynamics (Kuramoto 1984; Ermentrout and Terman 2010). Kuramoto’s model was extended to incorporate time delay in Yeung and Strogatz (1999). From this model, under some assumptions, a collective frequency \(\varOmega \) of coupled oscillations was derived to satisfy
where \(\omega _L\) is the intrinsic frequency and \(\varepsilon \) is the coupling strength. Much of the theory and its fit to the experimental data are based on this relationship. While the kinetic models are formulated at the level of single cells, the phase model can be regarded as at the tissue level. However, the connection and correspondence between the kinetic models and phase model have remained elusive, both biologically and mathematically.
Systems modeled with time delays and systems without time delays can certainly exhibit disparate dynamics. In addition, systems employing linear degradation and the ones using Michaelis–Menten degradation can have completely different dynamics. Yet how the reaction rates, delay magnitudes, and coupling strength affect the collective behaviors for each of these systems is a complicated research task. In the context of biological oscillations, dynamical properties for periodic orbits, such as variation of periods and amplitudes with respect to parameters, are key targets. As these mathematical models are differential equations, further mathematical studies shall definitely help understand these models, and provide complements to purely numerical findings. However, developing effective mathematical approach to analyse these models is itself a challenge.
Theoretical analysis on the existence and stability of synchronous periodic solutions for the ODE system proposed in Uriu et al. (2010) is a nontrivial task and has not been reported. This is due to that as the parameters vary, the equilibrium, hence the linearization at the equilibrium, along with its eigenvalues, all change. Nevertheless, it is important to find conditions under which cells can achieve synchronization in a sufficiently short time and to compare models with delay and without delay to clarify which is more suitable to generate stable synchronous oscillations. Oscillation-arrested is another important dynamical mode in the clock gene models. Biologically, we regard this mode as the phase of formed somites in the gene expression. For a nonlinear system in multi-dimensional phase space, it is another task to justify that there does not exist an oscillation. Global convergence to an equilibrium provides such a scenario. However, concluding such a convergence for coupled nonlinear delayed systems requires a new idea, since for example, finding a global Lyapunov function for these systems does not seem possible.
In this paper, we shall perform mathematical analysis to investigate synchronous oscillations and oscillation-arrested for the ODE system proposed by Uriu et al. (2009, 2010) as well as a modified system with delays. We are interested in seeing the dynamics for the coupled cells as well as the parameter values corresponding to synchronous oscillations and global convergence to equilibrium. Hopf bifurcation theory is a natural mathematical tool to analyze the existence and stability of synchronous periodic orbits in the clock gene models. For the ODE system, we demonstrate that there are two parameters, one representing the activation rate (coupling strength), and the other expressing a synthesis rate, and each can serve as a bifurcation parameter in Hopf bifurcation. For the delay system, we use the sum of transcriptional delay and translational delay as the bifurcation parameter in delay Hopf bifurcation theorem, while holding the other parameters suitably fixed. The computation of Hopf bifurcation for delay system is rather involved, especially for the stability of bifurcating periodic orbits, via normal form theory and center manifold theorem. For both systems, under some conditions, we can assert that all solutions remain nonnegative and bounded in forward time. In addition, there exists a unique positive synchronous equilibrium, and a criterion for the global convergence to the synchronous equilibrium can be established by using the sequential-contracting argument developed in Shih and Tseng (2011). Moreover, extended from the mathematical analysis, we shall pursue further numerical findings and illustrate their connections with somitogenesis.
We shall also illustrate the dynamical differences between system modeled with linear degradations and the one with Michaelis–Menten type degradations. Although modeling with Hill-type functions which describe the repression and the Michaelis–Menten type reaction for the degradations result in some complications on the equations, we shall demonstrate that the mathematical analysis for such systems can still be performed. We note that the equations considered in this paper, while serving as basic mathematical models for segmentation clocks in zebrafish, carry typical forms for models describing the kinetics of gene regulation.
The paper is organized as follows. In Sect. 2, we introduce and compare the mathematical models on the segmentation clock gene in zebrafish. In Sect. 3, we show that the solutions remain nonnegative and bounded under some conditions. We then discuss the global convergence to the unique equilibrium. In Sects. 4.1 and 4.2, we discuss synchronous periodic solutions for the ODE system and the delay system, respectively. Several numerical examples which are based on our analytical results are illustrated. In Sect. 5, we compare the dynamics for systems with delays and without delays. We also compare the present results with some previous works. The paper then ends with a conclusion.
2 Cell-to-cell kinetic models
The prevailing models of segmentation clock for zebrafish are based on the mechanism of direct autorepression of the clock gene by its own product and interaction of neighboring cells through positive feedback via Delta–Notch signaling. The basic model contains only one cyclic gene, her1 or her7. Let \(x_{1}, x_{2}, x_{3}, x_{4}\) (resp., \(y_{1}, y_{2}, y_{3}, y_{4}\)) represent respectively, the concentrations of her mRNA, Her protein, delta mRNA, and Delta protein of the first cell (resp., the second cell). The cell-to-cell kinetic model for the segmentation clock is expressed by
Herein, \(a_{2}\) (resp., \(a_{4}\)) is the protein synthesis rate per mRNA molecule for her (resp., delta) gene; \(\tau _{1},~\tau _{2},~\tau _{3},~\tau _{4}\) are the time delays in the processes of her gene transcription, her gene translation, delta gene transcription, delta gene translation and delivery to cell membrane, respectively; function \(g_{H}\) relates the transcription initiation rate to the suppression from Her protein and the activation from the Delta protein of neighboring cells; function \(g_{D}\) relates the transcription initiation rate to the suppression from Her protein; \(-f_i, i=1, 2, 3,4, \) represent the degradations. In Lewis (2003) and Özbudak and Lewis (2008), linear degradations are adopted:
and \(g_{H}\) and \(g_{D}\) take the forms
where \(k_{H}\) (resp., \(k_{D}\)) is the maximal synthesis rate of her (resp., delta) mRNA, and \(P_{0}\) (resp., \(P_{D_{0}}\)) is the critical number of molecules of Her (resp., Delta) protein per cell for inhibition of transcription (resp., activation of Notch).
With the idea of considering more detailed components of the process and releasing the concern of time delays, an ODE model was proposed and studied in Uriu et al. (2009, 2010). Therein, the process of Her protein transport from cytoplasm to nucleus is taken into account so that Her protein is decomposed into two components: Her protein in cytoplasm and Her protein in nucleus. Let \(x_{1}, x_{2}, x_{3}, x_{4}, x_{5}\) (resp., \(y_{1}, y_{2}, y_{3}, y_{4}, y_{5}\)) be the concentrations of her mRNA, Her protein in cytoplasm, Her protein in nucleus, delta mRNA, and Delta protein of the first cell (resp., the second cell), respectively. The kinetics of gene regulation was modeled by
where \(\nu _{3}\) and \(\nu _{9}\) are the synthesis rates of Her protein in cytoplasm and Delta protein, respectively, and \(\nu _{5}\) is the transportation rate of Her protein from cytoplasm to nucleus. In addition, \(g_{H}\) and \(g_{D}\) are defined by
where h and n are the Hill coefficients related to the dimerization process of Her proteins and the number of Her protein binding sites on DNA (Zeiser et al. 2007); \(\nu _{1}\) is the Basal transcription rate of her mRNA, \(\nu _{c}\) is the activation rate of her mRNA transcription by Delta–Notch signal, \(\nu _{7}\) is the synthesis rate of delta mRNA, and \(k_{1}\) and \(k_{7}\) are the threshold constants for the suppression of her mRNA and delta mRNA transcriptions by Her protein in nucleus, respectively. As degradation of proteins generally involves enzymatic reactions, the Michaelis–Menten type reactions were adopted therein:
where \(\nu _{2},\nu _{4},\nu _{6},\nu _{8},\nu _{10}\) (resp., \(k_{2},k_{4},k_{6},k_{8},k_{10}\)) are the maximum degradation rates (resp., Michaelis constants for the degradation) of her mRNA, Her protein in cytoplasm, Her protein in nucleus, delta mRNA, and Delta protein, respectively. Note that \(f_{2}\) contains the degradation term \(\nu _{4}u/(k_{4}+u)\) and the translocation term \(\nu _{5}u\). More detailed explanations for employing Michaelis–Menten type degradation in the modeling can be found in Uriu et al. (2009).
A simplified model which combines the two equations for delta mRNA and Delta protein into merely one equation for Delta protein was also investigated in Uriu et al. (2010). This reduction led to a system of eight ODEs:
where \(g_{D}\) relates both the transcription and translation initiation rates to Delta protein concentration, and is still defined by (6); \(f_{4}\) is now the degradation for Delta protein, and is defined in (7). In addition, in this model, \(\nu _{7}\) is the synthesis rate of Delta protein, \(k_{7}\) is the threshold constant for the suppression of Delta protein synthesis by Her protein, \(\nu _{8}\) is the maximum degradation rate of Delta protein, and \(k_{8}\) is the Michaelis constant for Delta protein degradation in nucleus. It was reported therein that both of these two ODE models (5) and (8) exhibit the main feature of her gene regulation and the parameter regimes of stable synchronous periodic solution are much alike.
Models containing more components of the segmentation clock have been studied in Cinquin (2007), Schröter et al. (2012), Ay et al. (2013, 2014) and Jenkins et al. (2015). For example, there are 44 parameters and 14 equations in each cell in the model investigated in Ay et al. (2013, 2014), taking into account the mRNA molecules of her1, her7, hes6, deltaC, and their monomer proteins and dimer proteins. The degradations in these models were all formulated in the form of linear functions (3).
A chief goal in modeling the segmentation clock is to be able to generate oscillatory traveling wave patterns along the PSM. The wave starts from synchronous oscillation at posterior of PSM, and then the oscillation slows down near the anterior of PSM, and is finally arrested at the anterior of PSM. This can be achieved by considering a lattice of cells, with a pertinent gradient of reaction rates or time delays along PSM. Recently, it has been found that increasing effective time delays along the PSM is responsible for the generation of traveling segmentation clock waves (Ay et al. 2013, 2014). An experimental evidence therein runs against some of the results in Uriu et al. (2009) where the gradient of a degradation rate was formulated to generate traveling waves. On the one hand, periods and amplitudes of oscillations depend also on other parameters. On the other hand, the dynamics in systems with linear degradations can be very different from the ones in systems with Michaelis–Menten type degradations. We shall illustrate this by an example in Sect. 4. Mathematical analysis with numerical simulation extended from the analysis is our approach for exploring how periods of oscillations are influenced by parameters.
A formal linearization of system (8) around an equilibrium was performed and the characteristic equation was derived in Uriu et al. (2010). However, the bifurcation analysis was not completed therein, as there are too many variables and parameters in the system. Therefore, basically, the results therein were obtained by numerical simulations on the ODE systems (5) and (8). However, there are insufficiencies for using purely numerical computations to find periodic solutions for systems with more than ten parameters in phase space of high dimension, as mentioned in Uriu et al. (2010). In this paper, we shall illustrate that such analysis can be performed, even though the equilibrium of nonlinear system (8) can not be computed exactly. The analysis will then lead to a theoretical support of the existence and stability of the synchronous periodic solutions. We shall also demonstrate that the parameter range for synchronous oscillations adopted in Uriu et al. (2010) can be interpreted within the Hopf bifurcation framework.
Examining and comparing whether the model with time delay or without time delay is more suitable for generating stable synchronous oscillation for the segmentation clock are very interesting in mathematical modeling on somitogenesis. We echo this interest and investigate the following system obtained by adding time delays into (8):
Herein, \(\tau _{1},~\tau _{2},~\tau _{4}\) represent respectively the time delays in the processes of her gene transcription, her gene translation, and delta gene transcription, translation, and delivery to cell membrane. We neglect the time delay in the translocation process in the third equation, as the time scale of the translocation is much smaller than the transcription and translation (Görlich and Kutay 1999; Makarov 2009; Simon et al. 1992).
Let us compare the above-mentioned models:
-
(i)
The transcription initiation rate for her, \(g_{H}(u,v)\), which plays the role of coupling function, is bounded in (4), but unbounded in (6).
-
(ii)
The degradation terms in (3) are linear and unbounded, whereas the ones in (7) are bounded but in more complicated nonlinear form.
-
(iii)
Transcription and translation time delays are taken into account in system (2) and system (9).
-
(iv)
The translocation process is included in systems (5), (8), and (9), where Her protein is decomposed into Her protein in cytoplasm and Her protein in nucleus.
-
(v)
The Hill coefficients are formulated in \(g_H\) and \(g_D\) in (6). Their values are determined from dimerization process and DNA binding sites. Functions \(g_H\) and \(g_D\) in (4) only represent the inhibitory protein as a dimer.
An analytical study for delay system (2) with degradation (3) and transcription function (4) was reported in Liao et al. (2012). Designed from this delay model, a nonautonomous lattice system which can generate normal traveling wave pattern was presented in Liao and Shih (2012). In this work, we shall analyze ODE system (8) and delay system (9), with transcription functions (6) and Michaelis–Menten type degradation (7). Our analysis can be extended to treat system (9) modified by adding translocation time delay and system (5) modified with time delays incorporated.
3 Basic properties and global dynamics
In this section, we discuss the basic properties for systems (8) and (9), with transcription (6) and degradation (7), to ensure that they are proper in modeling gene regulation. In particular, we shall assure that the mRNA concentrations and their protein products remain nonnegative along time. We also want to confirm that their solutions remain bounded throughout the evolutions. In addition, we shall discuss the existence of synchronous equilibrium and derive globally convergence to this equilibrium. Such convergence excludes oscillation and provides a regime for oscillation-arrested. The properties discussed for system (9) in this section are also valid for system (8), as system (9) reduces to (8) when the delays are zero: \(\tau _1=\tau _2=\tau _4=0\).
The coupled system (9) has three time delays \(\tau _{1},\tau _{2}\), and \(\tau _{4}\). Fundamental theory for delay equations is established on the infinite-dimensional phase space \(\mathscr {C}([-\tau _{M},0],\mathbb R^8_{+})\), the space of continuous functions from \([-\tau _{M},0]\) to \(\mathbb R^8_{+}\), where
Let \(\varPsi (t, \phi )\) be the flow map of (9) which depicts the evolution of the system at time t from initial condition \(\phi =(\phi _1,\ldots ,\phi _{8}) \in \mathscr {C}([-\tau _{M},0],\mathbb R^8_{+})\) at initial time \(t_0=0\). Denote by \(\mathbb {X}(t; \phi )\) the solution induced from (9), i.e., \(\mathbb {X}(t+\theta ; \phi )=\varPsi (t, \phi )(\theta )\), for \(\theta \in [-\tau _M, 0]\), and \(t > 0\). We also denote \(\mathbb {X}(t)=(\mathbf{{x}}(t),\mathbf{{y}}(t))=\mathbb {X}(t; \phi )=(\mathbf{{x}}(t; \phi ), \mathbf{{y}}(t; \phi ))\), where \(\mathbf{{x}}=(x_1, x_2, x_3, x_4), ~ \mathbf{{y}}=(y_1, y_2, y_3, y_4)\), if \(\phi \) is not specified. When \(\tau _1=\tau _2=\tau _4=0\), system (9) reduces to ODEs (8), with phase space \(\mathbb R^8_{+}\).
The following proposition ensures that the mRNA concentrations and their protein products remain non-negative along evolution.
Proposition 1
\(\mathscr {C}([-\tau _{M},0],\mathbb {R}^{8}_{+})\) is positively invariant under the flow generated by system (9), provided that the Hill coefficients h and n are even integers.
Proof
Assume that \(h=2\tilde{h}\) and \(n=2\tilde{n}\) for \(\tilde{h}, \tilde{n} \in {\mathbb N}\). We shall show that \(x_i(t) \ge 0, y_i(t) \ge 0, i=1, \ldots , 4\), for solution \(\mathbb {X}(t)=\mathbb {X}(t; \phi )\) evolved from an arbitrary \(\phi \in \mathscr {C}([-\tau _{M},0],\mathbb {R}^{8}_{+})\). Let \(I=I(\phi )\) be the maximal interval of existence for \(\mathbb {X}(t; \phi )\).
First,
for all \(t\in I\), since \(g_{D}(u)> 0\), for any \(u\in \mathbb {R}\). The solution for \(\dot{u}(t)=-\frac{\nu _{8}u(t)}{k_{8}+u(t)}\) remains nonnegative for all \(t \ge 0\), if \(u(0)\ge 0\). Hence, \(y_{4}(t)\ge 0\) if \(y_4(0) \ge 0\), for all \(t\in I\), by comparison arguments. Similarly, we obtain \(x_{4}(t)\ge 0\), for all \(t \in I\), from symmetry of system (9). With \(x_{4}(t), y_{4}(t)\ge 0\) for all \(t \in I\), we use similar argument to derive \(x_{1}(t), y_{1}(t)\ge 0\), for all \(t \in I\). Hence, for \(x_{2}(t)\), we have \(\dot{x}_{2}(t)\ge -\frac{\nu _{4}x_{2}(t)}{k_{4}+x_{2}(t)}\). Thus, \(x_2(t) \ge 0\) for all \(t \in I\), if \(x_2(0)\ge 0\). Similarly and successively, we conclude \(y_{2}(t), x_{3}(t), y_{3}(t)\ge 0\), for all \(t\in I\), if \(x_2(0), x_3(0), y_3(0) \ge 0\). \(\square \)
Next we derive a condition for the existence of an attracting set for system (9) in the following proposition. Notice that the degradation functions \(f_{i}\) in (7) are bounded, whereas the transcription function \(g_{H}\) in (6) is unbounded. The following quantities will be used to estimate the globally attracting set:
Proposition 2
Assume that h and n are even integers. If
then there exists a closed and bounded set \(\mathscr {Q}:=\varPi _{i=1}^{4} Q_{i}\times \varPi _{i=1}^{4} Q_{i} \subset \mathbb {R}^{8}_{+}, \) such that \(\mathbb {X}(t; \phi )\) converges to \(\mathscr {Q}\) for any \(\phi \in \mathscr {C}([-\tau _{M},0],\mathbb {R}^{8}_{+})\), where \(Q_{i}:= [\check{q}_{i},\hat{q}_{i}]\), with \(\hat{q}_{i}\), \(\check{q}_{i}\), \(i=1,\ldots , 4\), defined in (10) and (11).
Proof
The idea is to perform component estimation sequentially. With \(x_{i}(t), y_{i}(t) \ge 0, i=1, \ldots ,4\), from Proposition 1, we have
for all \(t\in I\). It follows that \(y_{4}(t)\) exists on \([0,\infty )\) and converges to \([0,~k_{8}\nu _{7}/(\nu _{8}-\nu _{7})]=:[0,\hat{q}_{4}]=:\tilde{Q}_{4}\), as \(t \rightarrow \infty \), due to \(\nu _{8}>\nu _{7}\). Subsequently, for an \(\epsilon >0\), there exists a \(t_{1}^{\epsilon }>0\) such that
Therefore, \(x_{1}(t)\) exists on \([0,\infty )\) and converges to \([0,~k_{2}(\nu _{1}+\nu _{c}(\hat{q}_{4}+\epsilon ))/(\nu _{2}-(\nu _{1} +\nu _{c}(\hat{q}_{4}+\epsilon )))]\) for all \(\epsilon >0\), and hence converges to \([0,~k_{2}(\nu _{1}+\nu _{c}\hat{q}_{4})/(\nu _{2}-(\nu _{1}+\nu _{c}\hat{q}_{4}))] =:[0,\hat{q}_{1}]=:\tilde{Q}_{1}\), as \(t \rightarrow \infty \), due to \(\nu _{2}>\nu _{1}+\nu _{c}\hat{q}_{4}\). Similarly, we can confirm that \(x_{2}(t)\) and \(x_{3}(t)\) exist on \([0,\infty )\) and converge to
respectively, due to \(\nu _{6}>\nu _{5}\hat{q}_{2}\). In addition, by the symmetry of system (9), \(x_{4}(t),y_{1}(t)\), \(y_{2}(t),y_{4}(t)\) also exist on \([0,\infty )\) and converge to sets \(\tilde{Q}_{4},~\tilde{Q}_{1},~\tilde{Q}_{2},~\tilde{Q}_{3}\), respectively.
Next, according to the convergence of \(x_{3}(t)\) to \(\tilde{Q}_{3}\) and \(y_{4}(t)\) to \(\tilde{Q}_{4}\), for any given \(\epsilon >0\), there exists a \(t_{2}^{\epsilon }>t_{1}^{\epsilon }\) such that
Therefore,
Hence, \(x_{1}(t)\) converges to \([\check{q}_{1},\hat{q}_{1}]=:Q_{1}\), as \(t \rightarrow \infty \), with \(\check{q}_{1}:=k_{1}^{n}k_{2}\nu _{1}/(\hat{q}_{3}^{n}\nu _{2}+k_{1}^{n}(\nu _{2}-\nu _{1}))\). By similar arguments, we can see that \(x_{2}(t),~x_{3}(t),~x_{4}(t)\) converge to \(Q_{2}:=[\check{q}_{2},\hat{q}_{2}],~Q_{3}:=[\check{q}_{3},\hat{q}_{3}],~Q_{4}:=[\check{q}_{4},\hat{q}_{4}]\), respectively, with
In addition, \(y_{i}(t)\) converges to \(Q_{i}\), \(i=1, \ldots ,4\), via the symmetry of system (9). \(\square \)
Remark 1
-
(i)
According to Propositions 1 and 2, if Hill coefficients h and n are even, then under the conditions of Proposition 2, every solution of (9) exists on \([0, \infty )\), and is bounded with nonnegative components. There are examples where solutions blow up when the conditions of Proposition 2 are violated.
-
(ii)
We have performed only two iteration steps in the proof of Proposition 2 to obtain an estimate for the attracting region. By continuing this process, the asymptotic dynamics can be captured in even smaller regions. Therefore, we can assert that every solution \(\mathbb {X}(t)\) lies in \(\mathscr {Q}\) after large time.
We say that an equilibrium \((x_1^{*}, \ldots , x_4^{*}\), \(y_1^{*}, \ldots , y_4^{*})\) of (9) is synchronous if \(x_i^{*}=y_i^{*}\), \(i=1, \ldots , 4\). Note that delay system (9) and ODE system (8) share identical equilibrium points. Next, we discuss synchronous equilibrium point for system (9).
Proposition 3
Assume \(\nu _{8}>\nu _{7}\). For any fixed integers \(h \ge 1\) and \(n \ge 1\), there exists a unique positive synchronous equilibrium point
for system (9), where
and \(\bar{x}_{3}\) is the unique solution to the equation
with
Proof
\((\bar{x}_{1},\bar{x}_{2},\bar{x}_{3},\bar{x}_{4}, \bar{x}_{1},\bar{x}_{2},\bar{x}_{3},\bar{x}_{4})\) is a synchronous equilibrium for (9) if and only if \((x_1, x_2, x_3, x_4)=(\bar{x}_{1}, \bar{x}_{2}, \bar{x}_{3}, \bar{x}_{4})\) satisfies
Accordingly,
and \(\bar{x}_{3}\) satisfies (14). Observe that there exists exactly one positive solution to (14), due to \(\nu _{8}>\nu _{7}\), and
and
Note that every component of \(\bar{\mathbb {X}}\) is positive. The assertion thus follows. \(\square \)
Now, let us discuss the global convergence to the synchronous equilibrium point \(\bar{\mathbb {X}}\) for system (9). For simplicity, we change \(Q_{i}\) to \([0, \hat{q}_{i}]\), i.e., we take \(\check{q}_{i}=0\), for \(i=1, \ldots ,4\). First, we translate the equilibrium \(\bar{\mathbb {X}}\) to the origin, i.e., we let \(\tilde{\mathbf{x}}(t)=\mathbf{x}(t)-\bar{\mathbf{x}}\), \(\tilde{\mathbf{y}}(t)=\mathbf{y}(t)-\bar{\mathbf{x}}\), and still denote \(\tilde{\mathbf{x}}, \tilde{\mathbf{y}}\) by \(\mathbf{x}\), \(\mathbf{y}\), respectively. System (9) becomes
We shall derive a criterion for global convergence to the origin in system (17). Let \(\mathbb {X}(t)=(x_1(t), \ldots , x_4(t), y_1(t), \ldots , y_4(t))\) be an arbitrary solution of (17), which exists on \([0, \infty )\), according to Proposition 2. With \(x_i(t), y_i(t)\) satisfying (17), by mean value theorem, we obtain
where \(\zeta _{i}(t)\) (resp., \( \zeta _{i+4}(t)\)) is between \(x_i(t)+\bar{x}_i\) and \(\bar{x}_i\) (resp., \(y_i(t)+\bar{x}_i\) and \(\bar{x}_i\)), \(i=1, \ldots , 4\), for all t large enough and
where \(u_{1}(t-\tau _{1})\) (resp., \(v_{1}(t-\tau _{1})\), \(u_{4}(t-\tau _{4})\), \(\tilde{u}_{1}(t-\tau _{1})\), \(\tilde{v}_{1}(t-\tau _{1})\), \(\tilde{u}_{4}(t-\tau _{4})\)) is some quantity between \(x_{3}(t-\tau _{1})+\bar{x}_{3}\) and \(\bar{x}_{3}\) (resp., \(y_{4}(t-\tau _{1})+\bar{x}_{4}\) and \(\bar{x}_{4}\), \(x_{3}(t-\tau _{4})+\bar{x}_{3}\) and \(\bar{x}_{3}\), \(y_{3}(t-\tau _{1})+\bar{x}_{3}\) and \(\bar{x}_{3}\), \(x_{4}(t-\tau _{1})+\bar{x}_{4}\) and \(\bar{x}_{4}\), \(y_{3}(t-\tau _{4})+\bar{x}_{3}\) and \(\bar{x}_{3}\)). Note that \(\mathbb {X}(t)\) converges to \(\mathscr {Q}-\bar{\mathbb {X}}=\varPi _{i=1}^{4}Q_{i}^{*}\times \varPi _{i=1}^{4}Q_{i}^{*}\), where \(Q_{i}^{*}:=[\check{q}_{i}-\bar{x}_{i},\hat{q}_{i}-\bar{x}_{i}]\), \(i=1, \ldots ,4\). Obviously, every component of (18) takes the form
where \(\zeta (t)\) lies in a compact set \(\tilde{Q}\) for all t large enough and w(t) is a continuous scalar function. We denote
It is straightforward to derive the following lemma; cf. Shih and Tseng (2008).
Lemma 1
Every solution of (19) converges to an interval \([-\tilde{\delta },\tilde{\delta }]\) as \(t \rightarrow \infty \), where
From Lemma 1, there exist eight intervals \(I_i:=[-\delta _i,\delta _i], i=1,\ldots ,8\), to which the ith component of solution \(\mathbb {X}(t)\) of (18) converges respectively. Since \(\zeta _{i}(t),\zeta _{i+4}(t) \in Q_{i}\) for all t large enough, we obtain
where
Next, we shall estimate the value of \(\delta _i\) through an iterative process. First, we define
Proposition 4
For each \(i=1,\ldots ,8\), there exists a sequence of nonnegative numbers \(\{\delta _i^{(k)}\}^\infty _{k=1}\) with \(\delta _i^{(k)}\ge \delta _i\) such that for each k, the ith component for the solution \(\mathbb {X}(t)\) of system (17) converges to \(I_i^{(k)}:=[-\delta _i^{(k)},\delta _i^{(k)}]\), as \(t\rightarrow \infty \), and \(\delta _i^{(k)}\) satisfies
where \(k\ge 1\), \( \delta _{3}^{(0)}:=\max \{|\check{q}_3-\bar{x}_{3}|, |\hat{q}_3-\bar{x}_3| \}\), \(\delta _{4}^{(0)}:=\max \{|\check{q}_4-\bar{x}_{4}|, |\hat{q}_4-\bar{x}_4| \}\), and \(\rho _{i}\) is defined in (21).
The proof is similar to the one for Proposition 2.5 in Liao et al. (2012) and is omitted.
Theorem 1
Every solution of system (9) converges to the synchronous equilibrium point \(\bar{\mathbb {X}}\) as \(t \rightarrow \infty \), if
Proof
To justify the assertion, we shall prove that every solution of (18) converges to the origin, as \(t \rightarrow \infty \). It suffices to show that \(\delta _{i}^{(k)}\) introduced in Proposition 4 converges to zero as k tends to infinity, for all \(i=1,\ldots ,8\). From (22), we obtain
Thus \(\delta _{1}^{(k)}\rightarrow 0\), as \(k \rightarrow \infty \), since \(0<\mathscr {R}<1\), by (23). Subsequently, \(\delta _{i}^{(k)}\rightarrow 0\), as \(k\rightarrow \infty \), for \(i=2,\ldots ,8\), according to (22). \(\square \)
Remark 2
(i) In fact, the upper bounds for the derivatives of \(g_H\) and \(g_D\) can be computed as
Note that \(\hat{\rho }_{i}\) is computable and \(\hat{\rho }_{i}\ge \rho _{i}\), \(i=1,2,3\). Therefore, condition (23) can be replaced by an explicit inequality:
(ii) By Theorem 1, \(\bar{\mathbb {X}}\) is a unique equilibrium for system (9) under (23) or (25).
Since all solutions tend to a steady state, the parameter regime under (23) or (25) corresponds to the non-oscillatory or oscillation-arrested phase for model (9), which is associated with the state of formed somites. Moreover, under condition (23) or (25), the magnitude of each \(\bar{x}_{i}\) determines the ultimate behavior of the system. In the following proposition, we further discuss how the parameters affect the magnitude of \(\bar{x}_{i}\), for \(i=1, \ldots ,4\).
Proposition 5
\(\bar{x}_{4}\) increases and \(\bar{x}_{i}\), \(i=1,2,3\), decreases, as one of \(k_{1}, k_{2}, \nu _{1},\nu _{c}\) decreases or \(\nu _{2}\) increases.
Proof
From (16), we see that if \(k_{2}\) decreases or \(\nu _{2}\) increases, then \(P_{R}(x_{3})\) increases and \(P_{L}(x_{3})\) is unchanged, and thus \(\bar{x}_{3}\) decreases. Similarly, if \(k_{1}, \nu _{1}\), or \(\nu _{c}\) decreases, then \(P_{L}(x_{3})\) decreases and \(P_{R}(x_{3})\) is unchanged, and therefore \(\bar{x}_{3}\) decreases. The assertions follow from \(\frac{\partial }{\partial \bar{x}_{3}}\bar{x}_{1}>0,~\frac{\partial }{\partial \bar{x}_{3}}\bar{x}_{2}>0\), and \(\frac{\partial }{\partial \bar{x}_{3}}\bar{x}_{4}<0\). \(\square \)
Proposition 5 indicates that if the transcription rate of her mRNA decreases or the degradation rate of her mRNA increases, then the steady state of her mRNA, Her protein, and delta mRNA all decrease and the Delta protein increases.
All the results in this section hold for \(\tau _{1}=\tau _2=\tau _4=0\), i.e., the ODE system (8) proposed in Uriu et al. (2010). The discussions herein can be extended to transcription functions \(g_{H}\) and \(g_{D}\) in more general form with \(\frac{\partial }{\partial u}g_{H}(u,v)<0\), \(\frac{\partial }{\partial v}g_{H}(u,v)>0\), and \(\frac{d}{du}g_{D}(u)<0\), for all \(u, v \ge 0\).
4 Synchronous oscillations
To elucidate synchronous oscillations through cell–cell interaction in segmentation clocks, we study synchronous periodic solutions generated in systems (8) and (9). In Sect. 4.1, for the ODE system (8), we take one of the activation parameters as bifurcation parameter and employ Hopf bifurcation theory to analyze the existence and stability of periodic solutions. In Sect. 4.2, we fix suitable values of all parameters in the delay system (9), and use the sum of transcription delay and translation delay as bifurcation parameter to apply the delay Hopf bifurcation theory. It will be seen that synchronous periodic solutions with periods around 30 min exist in each of these two systems, at certain parameter values.
4.1 ODE model (8)
We study the ODE model (8) with transcription function (6) and degradations (7). We plan to investigate the periodic solutions bifurcating from the synchronous equilibrium \(\bar{\mathbb {X}}=(\bar{\mathbf{x}},\bar{\mathbf{x}})\), with \(\bar{\mathbf{x}}=(\bar{x}_1,\bar{x}_2,\bar{x}_3,\bar{x}_4)\), of this system via Hopf bifurcation theorem. Let \(\tilde{\mathbf{x}}(t)=\mathbf{x}(t)-\bar{\mathbf{x}}\), \(\tilde{\mathbf{y}}(t)=\mathbf{y}(t)-\bar{\mathbf{x}}\), and still denote \(\tilde{\mathbf{x}}\), \(\tilde{\mathbf{y}}\) by \(\mathbf{x}\), \(\mathbf{y}\) respectively. The system becomes
The linearization of system (26) at the origin is given by
The characteristic equation for (27) is \(\varDelta =0\), where
and
The characteristic equation \(\varDelta =0\) can be factored as
where
Then, by the Routh–Hurwitz criterion, all roots of (30) have negative real parts if and only if
where \(\alpha _1:=\beta _1, \alpha _2:=\beta _2, \alpha _3:=\beta _3+\nu _3 \nu _5 \gamma _1, \alpha _4^\pm :=\beta _4+\nu _3\nu _5 (d_4 \gamma _1 \pm \gamma _2\gamma _3)\). As \(\alpha _1, \alpha _3, \alpha _4^+>0\), and \(\alpha _4^+> \alpha _4^-\), condition (32) reduces to
Lemma 2
All roots of (30) have negative real parts if and only if (33) or (34) holds.
In fact, Routh–Hurwitz criterion leads to the condition under which a pair of purely imaginary eigenvalues exist, while the rest of eigenvalues still have negative real parts (Asada and Yoshida 2003). The following lemma can thus be derived.
Lemma 3
The equation \(\varDelta _{+}=0\) (resp., \(\varDelta _{-}=0\)) has a pair of purely imaginary roots, and its remaining roots have negative real parts if and only if
The following proposition follows from (32) and Lemma 3, and \(\alpha _1>0, \alpha _3>0, \alpha _4^{+} > 0\), and \(\alpha _4^{+} >\alpha _4^{-} \).
Proposition 6
The equation \(\varDelta =0\) has a pair of purely imaginary roots and the remaining roots have negative real parts if and only if \(\alpha _4^{-} > 0\) and
Notably, this pair of purely imaginary roots are roots of \(\varDelta _+=0\), and (35) is equivalent to
To apply the Hopf bifurcation theorem, one first need to find a set of parameter values at which a pair of eigenvalues cross the imaginary axis of complex plane transversally and real parts of the other eigenvalues remain negative. We denote by \(\mu \) one of the parameters in (8) and fix the other parameters, while allow \(\mu \) to vary. The synchronous equilibrium \(\bar{\mathbb {X}}\), and hence the characteristic roots of (30) then depend on \(\mu \). We thus obtain the following Hopf bifurcation theorem.
Theorem 2
Consider system (8) which has a synchronous equilibrium \(\bar{\mathbb {X}}\) at certain fixed parameters and \(\mu =\mu ^*\). Assume that the synchronous equilibrium is a function of \(\mu \) for \(\mu \) near \(\mu ^*\), i.e., \(\bar{\mathbb {X}}= \bar{\mathbb {X}}(\mu )\), and
where the coefficients \(\alpha _i=\alpha _i(\mu ), i=1,2,3\), and \(\alpha _4^{\pm }=\alpha _4^{\pm }(\mu )\) of \(\varDelta _{\pm }\) are functions of \(\mu \). Then the system undergoes a Hopf bifurcation at \(\mathbb {X}=\bar{\mathbb {X}}\) and \(\mu =\mu ^{*}\), and a small-amplitude synchronous periodic solution surrounding \(\bar{\mathbb {X}}\) emerges as \(\mu < \mu ^{*}\) or \(\mu > \mu ^{*}\) and \(\mu \) is close to \(\mu ^{*}\).
The proof of this theorem is sketched in “Appendix 1”. Notably, the assumption that the synchronous equilibrium is a function of \(\mu \) for \(\mu \) near \(\mu ^*\) can be realized by the implicit function theorem. System (8) consists of two identical cells under symmetric coupling, and thus \(S:=\{x_1=y_1, x_2=y_2, x_3=y_3, x_4=y_4\}\) is positively invariant under the solution flow. The characteristic equation restricted to S is exactly \(\varDelta _+=0\). Therefore, Theorem 2 leads to the existence of synchronous periodic solution. We can transform the system into normal form, and apply the center manifold theorem to analyze the stability of the bifurcating periodic solution. We summarize these formulations in “Appendix 2”, following the theory in Hassard et al. (1981). From the formulations, we obtain the following qualities
where \(\lambda (\mu )\) is the eigenvalue crossing the imaginary axis at \(\mu =\mu ^*\), \(\lambda (\mu ^*)=i \omega _0\), and \(g_{20}, g_{11}, g_{02}, g_{21}\) are defined in “Appendix 2”. These quantities can be computed numerically for the application of the following Hopf bifurcation theorem, recast from Hassard et al. (1981).
Theorem 3
Under the conditions of Theorem 2, the following hold for system (8):
-
(i)
The Hopf bifurcation is supercritical (resp., subcritical) and a bifurcating periodic solution exists for \(\mu >\mu ^{*}\) (resp., \(\mu <\mu ^{*}\)) with \(\mu \) near \(\mu ^{*}\), if \(p_{2}>0\) (resp., \(<0\)).
-
(ii)
The periodic solution is stable (resp., unstable), if \(\zeta _{2}<0\) (resp., \(>0\)).
-
(iii)
The period increases (resp., decreases) as \(\mu \) increases, if \(T_{2}>0\) (resp., \(<0\)) and \(p_{2}>0\); the period increases (resp., decreases) as \(\mu \) decreases, if \(T_{2}<0\) (resp., \(>0\)) and \(p_{2}<0\).
Performing bifurcation analysis in an ODE system with more than a dozen of parameters, such as (8), appears to be a difficult task, as commented in Uriu et al. (2010). One complication is that the equilibrium depends on the parameters, and thus the linearization at the equilibrium varies with parameter values in an implicit way. Nevertheless, our formulation above indicates that the Hopf bifurcation analysis still can be carried out, if we combine the Routh–Hurwitz criterion with numerical computation effectively. Let us demonstrate such analysis and computation in the following examples.
4.1.1 Numerical illustrations
In the following examples, we adopt the parameter values in Uriu et al. (2010) and illustrate that the numerically computed synchronous oscillation therein is generated by the Hopf bifurcation presented in Theorem 2. Zebrafish segmentation clock is around 30 min, and thus we focus on periodic solutions with periods within [25, 35] min. We take \(\mu =\nu _7\) and \(\mu =\nu _c\) as the bifurcation parameter in Examples 4.1 and 4.2, respectively. In Example 4.3, we show that the single-cell (decoupled-cell) system, i.e., system (8) with \(\nu _c = 0\), does not admit periodic solution when the coupled-cell system does. In Example 4.4, we illustrate the dynamical disparity between a system with linear degradations and a system with Michaelis–Menten degradations.
Example 4.1
We choose \(\mu =\nu _7\), the synthesis rate of Delta protein, as the bifurcation parameter. The values of the other parameters are adopted from Uriu et al. (2010), and shown in Tables 1 and 2. Note that \(\nu _7\) appears in \(\gamma _3\) in the characteristic equation (30). Thus, we single out \(\nu _7\) from \(\gamma _3\) by setting
so that \(\alpha _4^{+}= \beta _4 +\nu _3\nu _5\nu _7\gamma _2 \tilde{\gamma }_3\). We look for a value for \(\nu _7\) which satisfies
From this equality, \(\nu _7\) can be expressed by the other parameters and substituted into system (8) to solve the equilibrium. Here, the synchronous equilibrium of (8) with parameter values in Tables 1 and 2 can be computed as \(\bar{\mathbb {X}}=(\bar{\mathbf{x}},\bar{\mathbf{x}}),~\mathrm{with}~\bar{\mathbf{x}}=(\bar{x}_1,\bar{x}_2,\bar{x}_3,\bar{x}_4),\) and \(\bar{x}_1 \approx 1.040873, \bar{x}_2 \approx 0.745985, \bar{x}_3 \approx 0.189600, \bar{x}_4 \approx 0.469586\). With this value of \(\bar{\mathbf{x}}\), we then compute to obtain the potential bifurcation value \(\nu _7^{*}\approx 1.29729\). Next, we compute to find
Therefore, the conditions of Theorem 2 are met, and a small-amplitude periodic solution emerges, as the value of \(\nu _7\) passes through \(\nu _7^{*}\), according to Theorem 2. The solution evolved from initial value \(\phi =(0.1,0.1,0.1,0.1, 0.08,0.08, 0.08,0.08)\) is shown in Fig. 1. The periods and amplitudes of the oscillations corresponding to various values of \(\nu _7\) are listed in Fig. 2. We observe that the periods of oscillations increase as \(\nu _7\) increases. When \(\nu _7=1.912\), the parameter value used in Uriu et al. (2010), this system generates a synchronous oscillation with period about 35 min.
Example 4.2
We choose \(\mu =\nu _c\), the activation rate of her mRNA transcription by Delta–Notch signaling (the coupling strength), as the bifurcation parameter. The other parameter values are taken from Tables 1 and 2, except that \(\nu _7=1.912\) is fixed and \(\nu _c\) is varying. We single out \(\nu _c\) from \(\gamma _1\) and \(\gamma _2\) by setting
Then \(\alpha _3\), \(\alpha _4^{+}\), and \(\beta _4\) in the characteristic equation \(\varDelta _{+}\) become
We look for a value of \(\nu _c\) which satisfies
By expressing \(\nu _c\) in terms of the other parameters, we find the equilibrium \(\bar{\mathbb {X}}=(\bar{\mathbf{x}},\bar{\mathbf{x}}),~\mathrm{with}~\bar{\mathbf{x}}=(\bar{x}_1,\bar{x}_2,\bar{x}_3,\bar{x}_4),\) and \(\bar{x}_1 \approx 1.038105, \bar{x}_2 \approx 0.737551, \bar{x}_3 \approx 0.186135, \bar{x}_4 \approx 1.692328\), at which the bifurcation takes place. We then compute to find \(\nu _c^{*}\approx 0.189266\), and
Thus, as the value of \(\nu _c\) passes through \(\nu _c^{*}\), there emerges a small-amplitude periodic solution which surrounds the synchronous equilibrium \(\bar{\mathbb {X}}\). The periods and amplitudes of oscillations corresponding to various values of \(\nu _c\) are listed in Fig. 3, and it appears that the period increases as \(\nu _c\) increases. When \(\nu _c=0.708\), the system generates a synchronous oscillation, as in Example 4.1.
Example 4.3
Consider the single-cell or decoupled-cell system:
i.e., system (8) with \(\nu _c=0\). First, we are interested in seeing whether such single-cell system (37) also admits a periodic solution with the parameter values in Tables 1 and 2, and \(\nu _7=1.912\). We compute to find that, with these parameter values, the eigenvalues of the linearized system of (37) at the equilibrium \( \bar{\mathbf{x}}=(\bar{x}_1,\bar{x}_2,\bar{x}_3,\bar{x}_4)\) all have negative real parts, where \(\bar{x}_1 \approx 0.673668, \bar{x}_2 \approx 0.209108 , \bar{x}_3 \approx 0.036607 , \bar{x}_4 \approx 1.784716\). Numerical simulation shows that solutions originated from numerous initial values converge to the equilibrium \( \bar{\mathbf{x}}\), as shown in Fig. 4. Hence, it appears that the single-cell system (37) does not generate periodic solution with these parameter values, whereas there is a synchronous oscillation in the coupled-cell system with the same parameter values and \(\nu _c=0.708\).
One is curious about whether if the single-cell system (37) can ever generate oscillation. By the Hopf bifurcation analysis, we observe that, with the parameter values in Tables 1 and 2, except now taking \(\nu _1\) near \(\nu _1^{*} \approx 0.25209\), a small-amplitude periodic solution emerges, see Fig. 5. Note that \(\nu _1\) is considered in the range [0.001, 0.1] in Uriu et al. (2010), and \(\nu _1^{*} \approx 0.25209\) obviously lies out of this interval.
Example 4.4
We compare the dynamics for the system modeled with linear degradations and the system modeled with Michaelis–Menten degradation. The Michaelis–Menten type degradation for the her mRNA is given by \(f_1(x_1)=\nu _2 x_1/(k_2+x_1)\) in (7). We take two linear functions with larger slope \(d_1^l\) and smaller slope \(d_1^s\) to approximate the graph of \(f_1\) respectively. When \(\nu _2=0.963, k_2=9.916\), the graph of \(f_1\) is depicted in Fig. 6, where the graphs for linear functions with slopes \(d_1^l=0.095186\) and \(d_1^s=0.01067\) are also plotted. Similar approximations apply to \(f_2, f_3, f_4\) with larger linear rates \(d_i^l\) and smaller linear rates \(d_i^s, i=2, 3, 4\), respectively. When \(\nu _4 = 1.447, k_4 = 0.182\), \(\nu _6 = 0.147, k_6 = 0.302, \nu _8 = 2.315, k_8 = 0.377\), we take \( d_2^{l} = 4.892873, d_2^{s} = 0.188497, d_3^{l} = 0.274709, d_3^{s} = 0.026188, d_4^l= 3.835797, d_4^s= 0.460283\).
(i) We modify (8) by considering linear degradation in every component, i.e.,
for x-components, and similarly for the y-components. If we take \(d_i=d_i^l, i=1, 2, 3, 4\) or \(d_i=d_i^s, i=1, 2, 3, 4\), and the same values for the other parameters as in Example 4.1, numerical simulations indicate that all solutions converge to an equilibrium.
(ii) We employ linear degradations for \(x_1, x_4, y_1, y_4\), while keeping Michaelis–Menten degradations for \(x_2, x_3, y_2, y_3\), i.e.,
for x-components, and similarly for the y-components. Let us use the linear degradation with larger slopes \(d_1=d_1^l\), \(d_4=d_4^l\) to approximate the Michaelis–Menten degradation, and take the same values for the other parameters (except \(\nu _c\)) as in Example 4.1. Then synchronous periodic solutions emerge. Herein, \(\nu _c\) can serve as a bifurcation parameter, and the Hopf bifurcation occurs at \(\nu _c^{*}\approx 0.774146\). We fix \(\nu _c=0.78\) and vary only \(d_4\), the periods and amplitudes for the periodic solutions are plotted in Fig. 7. It appears that the periods and amplitudes decrease as \(d_4\) increases.
(iii) We use linear degradations with smaller rates \(d_1=d_1^s\) and \(d_4 =d_4^s\) in (39) to approximate the Michaelis–Menten degradations. Choosing \(\nu _c\) as the bifurcation parameter, it can be shown that Hopf bifurcation occurs at \(\nu _c^{*}\approx 0.072545\). We fix \(\nu _c=0.074\) and vary only \(d_4\). The periods and amplitudes of the periodic solutions appear to decrease as \(d_4\) increases, as shown in Fig. 8.
The periods and amplitudes of the oscillations corresponding to various values of \(d_4\) for system (39) with larger degradation rates
The periods and amplitudes of the oscillations corresponding to various values of \(d_4\) for system (39) with smaller degradation rates
4.2 Delay model (9)
In this subsection, we discuss stable synchronous periodic orbits in delay model (9) with \(\tau _{i}>0\), \(i=1,2,4\), via delay Hopf bifurcation theory. We shall consider that all parameters and \(\tau _{4}\) are fixed and take \(r=\tau _{1}+\tau _{2}\) as the bifurcation parameter. One can also take \(\tau _{4}\) as a bifurcation parameter.
Assume that the synchronous equilibrium \(\bar{\mathbb {X}}\) of system (9) exists, as in Sect. 4.1. We consider system (17) which is a translation of (9) from the synchronous equilibrium \(\bar{\mathbb {X}}\) to the origin. The linearization of system (17) at the origin is given by
where \(d_1,~d_2,~d_3,~d_4,~\gamma _1,~\gamma _2\), and \(\gamma _3\) are defined as (28) and (29), and are positive.
The characteristic equation for (40) is \(\triangle (\lambda ,\tau _{1},\tau _{2},\tau _{4})= 0\), where \(\triangle (\lambda ,\tau _{1},\tau _{2},\tau _{4})\) is given by
By letting \(r:=\tau _{1}+\tau _{2}\), the characteristic equation can be factored as
where
and \(\beta _i,i=1,2, 3, 4\), are given in (31). From the structure of the characteristic equation, we employ \(r=\tau _{1}+\tau _{2}\) as the bifurcation parameter, while holding \(\tau _4\) fixed. We will analyze the existence of periodic solutions bifurcating from the origin of (17) for r near the bifurcation value.
Assume that all parameters and \(\tau _4 \) are fixed. We need to carry out the following steps to apply delay Hopf bifurcation theorem. Set \(\sigma =+\) or −.
-
(I)
Find a pair of purely imaginary characteristic values \(\lambda _{\sigma }=\pm i \omega _{\sigma }\) with \(\omega _{\sigma }>0\) and the corresponding bifurcation values \(\{r^{(k)}_{\sigma }(\omega _{\sigma })\}_{k \in \mathbb {Z}}\) such that \(\triangle _{\sigma }(\pm i \omega _{\sigma },r^{(k)}_{\sigma }(\omega _{\sigma }), \tau _4)=0\), for all \(k \in \mathbb {Z}\).
-
(II)
Examine that \( i \omega _{\sigma }\) is a simple purely imaginary characteristic value.
-
(III)
Examine the transversality: \(\mathrm{Re} \lambda '(r^{(k)}_{\sigma }(\omega _{\sigma })) \ne 0\), for some \(k \in \mathbb {Z}\).
For step (I), we substitute \(\lambda =i\omega \), into the characteristic equation (42), i.e., \(\triangle _{\pm }(i\omega ,r,\tau _4)=\tilde{R}_{\pm }(i\omega ,r,\tau _4)+i \tilde{I}_{\pm }(i\omega ,r,\tau _4)=0,\) where the real and imaginary parts are respectively
Through a manipulation by some trigonometric function properties (shown in “Appendix 3”), finding solution \(\omega \) to \(\triangle _{\pm }(i\omega ,r,\tau _4)=0\) becomes equivalent to solving
where
By considering the graphs of \( Q_{\pm }\), we can derive a condition for the existence of solution to (44):
Proposition 7
-
(i)
For any fixed \(\tau _4 \ge 0\), there exists a positive solution to \(Q_{+}(\omega )=\varGamma \) if \( Q_{+}(0)<\varGamma \), i.e.,
$$\begin{aligned} 0<\beta _{4}=d_1d_2d_3d_4< \nu _{3}\nu _{5}(d_{4}\gamma _{1}+\gamma _{2}\gamma _{3}). \end{aligned}$$(45) -
(ii)
For any fixed \(\tau _4 \ge 0\), each of \(Q_{-}(\omega )=\varGamma \) and \(Q_{+}(\omega )=\varGamma \) has a positive solution if \( Q_{-}(0)<\varGamma \), i.e.,
$$\begin{aligned} 0<\beta _{4}=d_1d_2d_3d_4 <\nu _{3}\nu _{5}|\gamma _{2}\gamma _{3}-d_{4}\gamma _{1}|. \end{aligned}$$(46)
Proposition 7 provides some conditions for the existence of purely imaginary characteristic values \(\pm i\omega _\sigma \). Notice that the inequalities in (45) and (46) actually express relations among parameters, rather than specifying the range for \(\beta _4\). Because of the trigonometric functions in \(\triangle _{\sigma }\), there is a sequence of values \(r= r_{\sigma }^{(k)}, k \in \mathbb {Z}\) at which the characteristic values \(\pm i\omega _\sigma \) exist. The process for executing (I)-(III) is sketched in “Appendix 3”, and the details are similar to those in Liao et al. (2012). With these executions, we obtain the following delay-induced Hopf bifurcation.
Theorem 4
Let all parameters and \(\tau _4 \ge 0\) be fixed. Assume that there exists a pair of purely imaginary characteristic values \(\pm i \omega _+\) (resp., \(\pm i\omega _-\)) satisfying conditions in (II) and (III) for a \(r_{+}^{(k)}(\omega _+)>0\) (resp., \(r_{-}^{(k)}(\omega _-)>0\)) at some \(k \in \mathbb {Z}\). Then Hopf bifurcation occurs at \(r=r_{+}^{(k)}(\omega _+)\) (resp., \(r=r_{-}^{(k)}(\omega _-)\)), and a periodic orbit bifurcates from the zero solution of system (17). Moreover, the periodic orbit bifurcating at \(r_{+}^{(k)}(\omega _+)>0\) is synchronous, while the one bifurcating at \(r_{-}^{(k)}(\omega _-)>0\) is asynchronous.
We have discussed periodic solutions generated by r-induced Hopf bifurcation, where the bifurcation values \(\{r^{(k)}_{\sigma }\}_{k \in \mathbb {Z}}\) form a sequence. This is typical in delay-induced Hopf bifurcations. Next, we discuss stability of the bifurcating periodic solution at the first bifurcation value.
If there exists a purely imaginary characteristic value \(i \omega _{\sigma }\) at its corresponding bifurcation values \(\{r^{(k)}_{\sigma }(\omega _{\sigma })\}_{k \in \mathbb {Z}}\) such that \(\triangle _{\sigma }(i \omega _{\sigma }, r^{(k)}_{\sigma }(\omega _{\sigma }),\tau _4)=0\), for all \(k \in \mathbb {Z}\) and \(\sigma =+\) or −, then we define the first bifurcation value, \(r_{c}\) of r as
Moreover, we denote by \(i \omega _c\) the purely imaginary characteristic root of \(\triangle (i \omega _c, r_c,\tau _4)=0\). In “Appendix 4”, we show that the \(r_c\) and \(\omega _c\) are well defined under some condition.
The stability of bifurcating periodic solution of system (17) induced by \(r=\tau _1+\tau _2\), can be analyzed through carrying out the following steps:
-
(IV)
Confirm that Hopf bifurcation occurs at \(r=r_{c}\) and \(\lambda =i \omega _{c}\).
-
(V)
Confirm the stability of the equilibrium for \(r<r_c\).
-
(VI)
Analyze the stability of bifurcating periodic orbit, by constructing local coordinates for the two-dimensional center manifold \(\mathcal{M}_0\) at \(r=r_{c}\), with the center manifold theorem and normal form method.
Locating all roots of \(\triangle (\cdot , \cdot ,\cdot )=0\) is highly nontrivial. Steps (IV) and (V) ensure that the first stability switch of the origin (from stability to instability) occurs at \(r=r_{c}\), for any \(\tau _4\ge 0\). It will be shown in “Appendix 4” that the origin of system (17) is asymptotically stable when delays are zero, by the Routh–Hurwitz criterion. Moreover, this stability continues to hold for \(r<r_c\) and any \(\tau _{4} \ge 0\), under some conditions, as shown in Proposition 8 (in “Appendix 4”). The detail of step (VI) is arranged in “Appendix 5”. In step (VI), there are certain terms \(g_{20},g_{11},g_{02}\), and \(g_{21}\) derived from the vector field on \(\mathcal{M}_0\). Subsequently, we obtain the following quantities which determine the direction and stability of the bifurcating periodic solution at the minimal bifurcation value \(r_{c}\):
where \(\lambda (r)\) is the eigenvalue crossing the imaginary axis at \(r=r_c\), and \(\lambda (r_c)=i \omega _c\). Computation of these terms and the following theorem can be found in Hassard et al. (1981).
Theorem 5
Let all parameters and \(\tau _4 \ge 0\) be fixed. Assume that system (17) undergoes a Hopf bifurcation at the origin at a minimal bifurcation value \(r=r_{c}\), and the conditions in (V) hold.
-
(i)
If \(p_{2}>0\) (resp., \(<0\)), then the bifurcation is supercritical (resp., subcritical) and a periodic solution bifurcates for \(r>r_{c}\) (resp., \(r<r_{c}\)) with r near \(r_{c}\).
-
(ii)
If \(\zeta _{2}<0\) (resp., \(>0\)), then the bifurcating periodic solution is stable (resp., unstable).
-
(iii)
If \(T_{2}>0\) (resp., \(<0\)) and \(p_{2}>0\), then the period increases (resp., decreases) as r increases. If \(T_{2}<0\) (resp., \(>0\)) and \(p_{2}<0\), then the period increases (resp., decreases) as r decreases.
According to Theorems 4 and 5, let us summarize the criteria for the occurrence of stable synchronous and asynchronous oscillations, respectively.
-
Condition (S): The minimal bifurcation value \(r_c\) and the corresponding purely imaginary characteristic value \(i \omega _c\) exist such that \(\triangle _+(i\omega _c, r_c, \tau _4)=0\); the equilibrium \(\bar{\mathbb {X}}\) is asymptotically stable for \(r<r_c\); the Hopf bifurcation occurs at \(r_c\), and \(\zeta _{2}<0\).
-
Condition (AS): The minimal bifurcation value \(r_c\) and the corresponding purely imaginary characteristic value \(i \omega _c\) exist such that \(\triangle _-(i\omega _c, r_c, \tau _4)=0\); the equilibrium \(\bar{\mathbb {X}}\) is asymptotically stable for \(r<r_c\); the Hopf bifurcation occurs at \(r_c\), and \(\zeta _{2}<0\).
Corollary 1
If condition (S) (resp., (AS)) holds for the parameters and \(\tau _4 \) at some fixed values, then there exists a stable synchronous (resp., asynchronous) periodic solution of system (17), for \(|r-r_{c}|\) small and \(r>r_{c}\) if \(p_{2}>0\), and \(r<r_{c}\) if \(p_{2}<0\).
Remark 3
-
(i)
For the delay model (9), while holding all parameters and \(\tau _4\) fixed, there could be a phase exchange between synchronous oscillation and asynchronous oscillation, as r varies. We shall illustrate this in the following subsection.
-
(ii)
The conditions in Theorems 4 and 5 can all be examined numerically. In addition, the analysis in this subsection can be extended to coupled-cell system with four time delays, \(\tau _{1},~\tau _{2},~\tau _{3}\), and \(\tau _{4}\), where \(\tau _{3}\) is the time lag for the process of translocation.
4.2.1 Numerical illustrations
In this subsection, we perform several numerical simulations to illustrate the dynamical scenarios for system (9), and connect the dynamics to the somitogenesis mechanism. We first illustrate Theorem 4 by Example 4.5, then pursue extended numerical findings to explore further dynamical scenarios and properties for system (9). In numerical simulations on the delay equations (9), when the conditions in Theorem 4 or Theorem 5 are met, periodic solution emerges if we choose r to be a little larger or smaller than the bifurcation value \(r_c\). Often the periodic solution persists if we further increase or decrease the bifurcation parameter r.
Example 4.5
We consider the following parameters
System (8) and system (9) both have a synchronous equilibrium at
If there is no delay: \( \tau _1=\tau _2=\tau _4=0\), i.e., considering system (8), then all eigenvalues of the linearized system at the equilibrium have negative real parts, as (33) is met. Thus, \( \bar{\mathbb {X}}\) is asymptotically stable in system (8). Numerical simulation further indicates that all solutions converge to \( \bar{\mathbb {X}}\) as \(t \rightarrow \infty \), as demonstrated in Fig. 9a. It appears that there is no oscillation for ODE system (8) with parameters in (48).
The solution evolved from initial value \(\phi =(0.06, 2.6, 17, 0.2, 0.07, 2.7, 18, 0.3)\), for a ODE system (8) with parameter values in (48), tends to the synchronous equilibrium \(\bar{\mathbb {X}}\), b system (9) with delays in (49) and the same parameter values, tends to a stable synchronous solution
On the other hand, if we consider delay system (9) with delays
then the solution evolved from the same initial value tends to a stable synchronous oscillation shown in Fig. 9b. The existence of this periodic solution follows from Theorem 4, by taking \(\tau _1+\tau _2\) to be near the minimal bifurcation value \(\tau _c= 6.66245\). This example illustrates that when the ODE model (8) does not have oscillation, by adding suitable delays into the system, synchronous oscillation could be generated.
Example 4.6
Table 3 lists the ranges of all parameters considered in Uriu et al. (2010). We like to see how likely system (9) with delays is able to generate synchronous oscillation and asynchronous oscillation in these parameter ranges. We assume \(\nu _{8}>\nu _{7}\) so that the synchronous equilibrium exists, by Proposition 3. We take \(\tau _{2}=2.8\), and respectively, \(h=n=1, 2, 3, 4\), \(\tau _{4}=25,35,45,55,65,75\), and then randomly choose 10,000 parameter sets of values from Table 3. We examine condition (S) and condition (AS) to determine the existence of stable synchronous and asynchronous periodic solutions when r near \(r_{c}\), respectively. The numbers of parameter sets that satisfy each of these two conditions are recorded in Table 4.
In Table 4a, each denominator (resp., numerator) accounts for the number of parameter sets with which system (9) admits stable synchronous periodic solutions (resp., stable synchronous periodic solutions with period within [25, 35] min), when r is near the minimal bifurcation value \(r_{c}\) (i.e., \(\tau _1\) near \(r_{c}-\tau _2=r_c-2.8) \). It appears in Table 4a that the most stable synchronous oscillations with period within [25, 35] min generated is when \(n=h=1\) and \(\tau _4=35\). We also record in Table 4b the number of cases which generate stable asynchronous periodic solution, when r is near \(r_{c}\).
We then perform a statistics to the data in Table 4 and collect the parameter values with which system (9) satisfies condition (S) and hence generates synchronous oscillation. As an illustration, we display the frequency histograms for the case \(\tau _4=35\) and \(n=h=1\). Figure 10 indicates that parameters \(\nu _{6}, \nu _8, k_6\) with \(\nu _{6}<4\), \(\nu _{8}>5\), and \(k_{6}>5\) together tend to satisfy condition (S). Figure 11 indicates that \(\nu _{3}>25\) or \(\nu _{4}<5\) or \(\nu _{5}>0.5\), or \(\nu _{7}<5\) tends to satisfy condition (S). Hence, if the degradation rate \(\nu _6\) of Her protein in nucleus is small, and the degradation rate \(\nu _8\) of Delta protein is large, then the system tends to generate stable periodic solutions. In addition, we found that system with different Hill coefficients and \(\tau _4\) has similar distribution for synchronous and asynchronous oscillation. That is, the Hill coefficients do not affect significantly the parameter distribution for oscillation, and this is consistent with the observation in Uriu et al. (2010) for the ODE system (8).
Example 4.7
From our numerical simulations on system (9) with parameters satisfying condition (S) or condition (AS), we further observe the following properties:
-
(i)
Phase exchange: We choose two sets of parameter values from the computation in Table 4 to demonstrate the phase exchange between synchronous and asynchronous oscillations as bifurcation parameter r varies:
$$\begin{aligned} (k_1,k_2,k_4,k_6,k_7,k_8)= & {} (1.65443,1.39621,8.06972,8.61674,\nonumber \\&9.71803,3.87536),\nonumber \\ (\nu _1,\nu _2,\nu _3,\nu _4,\nu _5,\nu _6,\nu _7,\nu _8,\nu _c)= & {} (0.075602,2.07115,10.9502,5.59773,\nonumber \\&0.1928248,2.02374,5.26644,7.09196,\nonumber \\&0.591048), \end{aligned}$$(50)$$\begin{aligned} (k_1,k_2,k_4,k_6,k_7,k_8)= & {} (0.847943,1.4226,4.90274,1.35171,\nonumber \\&5.89576,3.91132),\nonumber \\ \quad (\nu _1,\nu _2,\nu _3,\nu _4,\nu _5,\nu _6,\nu _7,\nu _8,\nu _c)= & {} (0.011547,3.30484,30.2745,0.835863,\nonumber \\&0.685058,1.34702,0.362676,5.74838,\nonumber \\&0.587321). \end{aligned}$$(51)System (9) with parameters (50) (resp., (51)) satisfies condition (S) (resp., (AS)) with minimal bifurcation value \(r_{c}^s\) (resp., \(r_{c}^a\) ). When r is near \(r_{c}^s\) (resp., \(r_{c}^a\)), there exists a stable synchronous (resp., asynchronous) periodic solution bifurcating from the equilibrium \(\bar{\mathbb {X}}\), by Corollary 1. If we take r near \(r_{c}^s\) (resp., \(r_{c}^a\)), and then increase it to another bifurcation value larger than \(r_c^s\) (resp., \(r_{c}^a\)), then the system undergoes a phase exchange from synchronous (resp., asynchronous) oscillation to asynchronous (resp., synchronous) oscillation, as shown in Fig. 12 (resp., Fig. 13). In addition, if we set delays to zeros, then these periodic solutions disappear, as shown in Fig. 14. Restated, holding a suitable \(\tau _4\) and varying r could generate a sequence of phase exchanges between synchronous and asynchronous oscillations, and the oscillation disappears when delays are all zeros. In Figs. 12, 13 and 14, we plot components \(x_{1}(t)\) and \(y_{1}(t)\) of the solution evolved from the initial value \(\phi =(0.1,0.1,0.1,0.1,0.2,0.2,0.2,0.2)\).
-
(ii)
Existence of stable synchronous or asynchronous periodic solution depends sensitively on the magnitudes of delays r and \(\tau _4\). One may not be able to find a periodic solution when the value of r is not close enough to a bifurcation value.
-
(iii)
We observe that the system tends to generate stable synchronous oscillations with a period about 30 min, if the system satisfies condition (S) and \(r_c < 10\).
-
(iv)
For a set of fixed parameter values, the period of bifurcating periodic solution is larger at larger bifurcation value. On the other hand, for system (9) with parameters which yield larger minimal bifurcation value, the periodic solution bifurcating at such value has larger period.
With \(\tau _{4}=35\), the parameter values in (50), and \(r=3.401106\) near \(r_{c}^s\), in a \(t \in [0, 600]\), b \(t \in [2600, 3000]\), and \(r=6.04389\) near certain bifurcation value larger than \(r_{c}^s\) in c \(t \in [0, 600]\), d \(t \in [2600, 3000]\), the system undergoes a phase exchange from a stable synchronous oscillation to a stable asynchronous oscillation
With \(\tau _{4}=35\), the parameter values in (51), and \(r=7.15282\) near \(r_{c}^a\) in a \(t \in [0, 600]\), b \(t \in [2600, 3000]\), and \(r=9.14428\) near certain bifurcation value larger than \(r_{c}^a\) in c \(t \in [0, 600]\), d \(t \in [2600, 3000]\), the system undergoes a phase exchange from a stable asynchronous oscillation to a stable synchronous oscillation
5 Comparison between models
In this section, we summarize the dynamical properties according to our theories in Sects. 3 and 4 and subsequent numerical computations, and make a comparison between ODE model (8) and delay model (9). Such a comparison and the study on whether the ODE model (8) or delay model (9) is more suitable for modeling the segmentation clock have been an appealing research interest, as mentioned in the Introduction.
Let us classify the basic dynamics for (9) with respect to parameters. Recall \(\beta _{4}=d_1d_2d_3d_4\) defined in (31) and \(\check{\beta }_{4}:=\check{d}_{1}\check{d}_{2}\check{d}_{3}\check{d}_{4}\) defined in (20). We introduce intervals
shown in Fig. 15a. According to Proposition 8 (in “Appendix 4”), and (25) in Remark 2(i), we summarize:
-
(i)
If \(\beta _{4} <\nu _{3}\nu _{5}|\gamma _{2}\gamma _{3}-d_{4}\gamma _{1}|\), i.e., \(\beta _{4} \in I_1\), then there exist \(i \omega _+\), \(i \omega _-\), \(\{r^{(k)}_+(\omega _+)\}_{k\in \mathbb {Z}}\), \(\{r^{(k)}_-(\omega _-)\}_{k\in \mathbb {Z}}\) such that \(\triangle _+(i \omega _+, r^{(k)}_+(\omega _+),\tau _4)=0\) and \(\triangle _-(i \omega _-, r^{(k)}_-(\omega _-),\tau _4)=0\), for all \(k \in \mathbb {Z}\).
-
(ii)
If \(\nu _{3}\nu _{5}|\gamma _{2}\gamma _{3}-d_{4}\gamma _{1}|< \beta _4 <\nu _{3}\nu _{5}(\gamma _{2}\gamma _{3}+d_{4}\gamma _{1})\), i.e., \(\beta _{4}\in I_2\), then there exist \(i \omega _+\) and \(\{r^{(k)}_+(\omega _+)\}_{k\in \mathbb {Z}}\) such that \(\triangle _+(i \omega _+, r^{(k)}_+(\omega _+),\tau _4)=0\), for all \(k \in \mathbb {Z}\).
-
(iii)
If \(\check{\beta }_4 > \nu _{3}\nu _{5}(\hat{\rho }_{1}\check{d}_{4}+\hat{\rho }_{2}\hat{\rho }_{3})\), then there does not exist oscillation for both ODE system (8) and delay system (9).
a Partition for the range of \(\beta _4\): \(I_{1}=(0, \nu _{3}\nu _{5}|\gamma _{2}\gamma _{3}-d_{4}\gamma _{1}|)\), \(I_{2}=(\nu _{3}\nu _{5}|\gamma _{2}\gamma _{3}-d_{4}\gamma _{1}|, \nu _{3}\nu _{5}(\gamma _{2}\gamma _{3}+d_{4}\gamma _{1}))\). b Partition for the range of \(\check{\beta }_4\): \(I_{4}=(\nu _{3}\nu _{5}(\hat{\rho }_{1}\check{d}_{4}+\hat{\rho }_{2}\hat{\rho }_{3}), \infty )\). \(\hat{\rho }_{i}\) is defined in (24), \(i=1,2,3\)
Notably, \( \check{\beta }_{4} \le \beta _4\), as \(\check{d}_{i}\le d_i\) for \(i=1, 2, 3, 4\), and \(\gamma _i \le \hat{\rho }_{i}, i=1, 2, 3\), where \(\hat{\rho }_{i}\) is defined in (24). In addition, it has been shown in Lemma 2 that all eigenvalues of the linearized system of (8) at the equilibrium have negative real parts if and only if
Therefore, if \(\gamma _2 \gamma _3-d_4 \gamma _1>0\), \(\beta _4 \in I_2\), and \(\beta _4\) lies in the range in (52), there does not exist synchronous periodic solution for the ODE system (8), through Hopf bifurcation. However, with such parameter values, synchronous periodic solution can exist for the delay system (9), according to delay Hopf bifurcation. In fact, Example 4.5 is such an instance. The Hopf bifurcation occurs for ODE system (8), when the right-hand inequality in (52) becomes equality, as seen in Proposition 6. Let us summarize the dynamical properties for systems (8) and (9):
-
Synchronous oscillation: From our analytical and numerical results, it appears that the delay system (9) has wider parameter regime for synchronous periodic solution than the ODE system (8).
-
Period with respect to coupling strength: In ODE system (8), the period of synchronous oscillation increases as the coupling strength increases, as shown in Fig. 3 in Example 4.2. This is in contrast to the finding in Herrgen et al. (2010), which was based on some experiment and the theory of coupled phase oscillator. On the other hand, it is possible that the period of synchronous oscillation increases as the coupling strength decreases in the delay system (9).
-
Oscillation-arrested: For parameters satisfying (iii), both systems (8) and (9) admit global convergence to the synchronous equilibrium and there is no oscillation.
-
Gradient structure: The gradient structure was adopted to generate traveling waves in N-cell systems extended from the cell–cell ODE system (8) in Uriu et al. (2009), and Lewis’s delay system (2) in Liao and Shih (2012). Such wave patterns depict spatial-temporal oscillatory gene expression in the PSM of zebrafish. These oscillatory waves have larger periods at the anterior and smaller periods at the posterior of the PSM. For the delay system (9), construction of such traveling waves is also feasible. According to the analytical results in Sects. 3 and 4, the degradation rates should be relatively large (resp., small) and the synthesis rates relatively small (resp., large) at the anterior (resp., posterior) of PSM to generate oscillation-arrested (resp., oscillation). We thus summarize the following distribution.
Distribution (D1): One of \(k_1,~k_2,~k_4,~k_6,~k_7,~k_8, \nu _1,~\nu _3,~\nu _c\) is relatively small (resp., large), or one of \(\nu _{2},~\nu _{4},~\nu _{6},~\nu _8\) is relatively large (resp., small) to yield relatively large (resp., small) degradation rates and small (resp., large) synthesis rates for the cells located at the anterior (resp., posterior) of PSM.
In addition, from our numerical computation and Table 4, we see that relatively large (resp., small) \(\nu _4\) and \(\nu _6\) and relatively small (resp., large) \(k_6\) and \(\nu _3\), which are consistent with the distribution (D1), tend to generate oscillation-arrested (resp., oscillation). On the other hand, the following distribution (D2) was adopted for the ODE system (8) to generate normal traveling wave pattern in Uriu et al. (2009).
Distribution (D2): One of \(k_4,~k_6,~\nu _2,~\nu _3,~\nu _5,~\nu _8\) is relatively small (resp., large), or one of \(k_1,~k_2,~k_7,~k_8,~\nu _1,~\nu _c\) is relatively large (resp., small) for the parameters corresponding to cells located at the anterior (resp., posterior) of PSM.
The distributions (D1) for delay system (9) and (D2) for ODE system (8) are disparate except \(k_4,~k_6\), and \(\nu _3\), and so the regimes for generating normal traveling wave patterns are distinct for these two systems.
6 Discussions and conclusions
In this paper, we analyzed the dynamics for two mathematical models on the kinetics of zebrafish segmentation clock genes. The ODE system (8) takes into account the translocation process in gene regulation to replace the consideration of time lags in transcription and translation. The delay system (9) includes these time delays into system (8). Mathematical analysis were performed to analyze the chief dynamics and locate the associated parameter regimes for both the delay and ODE systems. For oscillation-arrested, we derived conditions for global convergence to the synchronous equilibrium. For synchronous oscillations, we computed the characteristic equations of the linearized systems at the synchronous equilibrium for system (8) and system (9) respectively, and analyzed the characteristic values for each of these systems. With such computation, the Hopf bifurcation theory was applied to study the existence of synchronous periodic solutions for each of these systems, and asynchronous periodic solutions in delay system. For the ODE system, we took the synthesis rate of Delta protein or the activation rate of her mRNA transcription by Delta–Notch signal (coupling strength) as the bifurcation parameter. For the delay system, the sum of transcription delay and translation delay for her gene was used as the bifurcation parameter. Criterion for the existence of stable synchronous oscillations for each system was then established subsequently. Both systems admit the basic dynamical phases for segmentation clock genes and can accommodate the gradient structure associated with the traveling wave patterns.
It has been an interesting issue to investigate and distinguish what kind of mathematical models is more suitable to model gene regulation in somitogenesis. This is what we have pursued in this study. From our analysis and computation, it appears that the delay model (9) has wider parameter range for generating synchronous oscillations. However, the synchrony of such oscillation depends on the magnitude of the delay tightly, and may yield to asynchronous oscillation at larger or smaller delay magnitudes. On the other hand, for the ODE system (8), we have seen that the period of the periodic solution increases with increasing coupling strength \(\nu _c\) in Example 4.2. This is in contrast to the finding in the literature through experiments and the theory of coupled phase oscillators. In this regard, delay models appear to be more pertinent for modeling segmentation clock genes.
A mathematical reason for Lewis to consider delays in his model, (2) with (3), (4), is that, without adding delay, the dynamics for the decoupled single-cell system is determined by a two-dimensional ODE which can not generate oscillation, see Lewis (2003). On the other hand, for the decoupled single-cell system corresponding to (8), i.e., taking into account the translocation process and without adopting time delay, the dynamics is determined by a three-dimensional ODE, and thus can accommodate oscillations. However, the parameter values for such oscillation may not lie within the suitable range, as shown in Example 4.3.
Another issue is about the degradations. Mathematical models on segmentation clocks in the literature largely employ linear degradations. On the other hand, Michaelis–Menten type reactions for the degradation were adopted in Goldbeter and Pourquié (2008) and Uriu et al. (2009, 2010). From the view point of mathematical modeling, Michaelis–Menten reaction can be approximated by linear decay when the protein level is low (Murray 2002). However, some of the simulated protein levels in the literature are quite large (as large as hundreds). Certainly, the units for the variables and parameters will be important with this concern. We have illustrated in Example 4.4 that the dynamics in a system with linear degradations can be quite different from the ones in system with Michaelis–Menten degradations. Although some degradation rates were estimated from halflives of mRNA and proteins, experimental measurement of other reaction rates and quantifying some of the parameters will require further efforts, as mentioned in Ay et al. (2014) and Schwendinger-Schreck et al. (2014). These quantifications are also associated with what function forms the degradations are to be modeled with.
Systems (2), (8) and (9) actually represent typical forms of equations for modeling the kinetics of gene regulation in coupled cells. To investigate such somehow complicated equations, one certainly relies on numerical simulations. Nevertheless, assertions established from numerical simulations can certainly be strengthened by analytical supports. The analysis and techniques in this study are expected to provide a paradigm for mathematical investigation in other gene regulation models, in addition to the zebrafish somitogenesis.
References
Asada T, Yoshida H (2003) Coefficient criterion for four-dimensional Hopf bifurcations: a complete mathematical characterization and applications to economic dynamics. Chaos Solitons Fractals 18:525–536
Ay A, Knierer S, Sperlea A, Holland J, Özbudak EM (2013) Short-lived Her proteins drive robust synchronized oscillations in the zebrafish segmentation clock. Development 140(15):3244–3253
Ay A, Holland J, Sperlea A, Devakanmalai GS, Knierer S, Sangervasi S, Stevenson A, Özbudak EM (2014) Spatial gradients of protein-level time delays set the pace of the traveling segmentation clock waves. Development 141(21):4158–4167
Baker RE, Schnell S, Maini PK (2006) A mathematical investigation of a clock and wavefornt model for somitogenesis. J Math Biol 52(4):458–482
Baker RE, Schnell S (2009) How can mathematics help us explore vertebrate segmentation? HFSP 3(1):1–5
Campanelli M, Gedeon T (2010) Somitogenesis clock-wave initiation requires differential decay and multiple binding sites for clock protein. PLoS Comput Biol 6(4):e1000728
Cinquin O (2007) Repressor dimerization in the zebrafish somitogenesis clock. PLoS Comput Biol 3(2):e32
Cooke J, Zeeman EC (1976) A clock and wavefront model for control of the number of repeated structures during animal morphogenesis. J Theor Biol 58(2):455–476
Dubrulle J, McGrew MJ, Pourquié O (2001) FGF signalling controls somite boundary position and regulates segmentation clock control of spatiotemporal Hox gene activation. Cell 106(2):219–232
Dubrulle J, Pourquié O (2002) From head to tail: links between the segmentation clock and antero-posterior patterning of the embryo. Curr Opin Genet Dev 12(5):519–523
Ermentrout GB, Terman DH (2010) Mathematical foundations of neuroscience. Springer, New York
Goldbeter A, Pourquié O (2008) Modeling the segmentation clock as a network of coupled oscillations in the Notch, Wnt and FGF signaling pathways. J Theor Biol 252(3):574–585
Görlich D, Kutay U (1999) Transport between the cell nucleus and the cytoplasm. Annu Rev Cell Dev Biol 15:607–660
Hanneman E, Westerfield M (1989) Early expression of acetyl-choline-sterase activity in functionally distinct neurons of the zebrafish. J Comp Neurol 284(3):350–361
Hassard BD, Kazarinoff ND, Wan YH (1981) Theory and applications of Hopf bifurcation. Cambridge University, Cambridge, New York
Herrgen L, Ares S, Morelli LG, Schröter C, Jülicher F, Oates AC (2010) Intercellular coupling regulates the period of the segmentation clock. Curr Biol 20(14):1244–1253
Holley SA (2007) The genetics and embryology of zebrafish metamerism. Dev Dyn 236(6):1422–1449
Horikawa K, Ishimatsu K, Yoshimoto E, Kondo S, Takeda H (2006) Noise-resistant and synchronized oscillation of the segmentation clock. Nature 441(7094):719–723
Jenkins RP, Hanisch A, Soza-Ried C, Sahai E, Lewis J (2015) Stochastic regulation of her1/7 gene expression is the source of noise in the zebrafish somite clock counteracted by Notch signalling. PLoS Comput Biol 11(11):e1004459
Jiang YJ, Aerne BL, Smithers L, Haddon C, Ish-Horowicz D, Lewis J (2000) Notch signaling and the synchronization of the somite segmentation clock. Nature 408(6811):475–479
Kawamura A, Koshida S, Hijikata H, Sakaguchi T, Kondoh H, Takada S (2005) Zebrafish hairy/enhancer of split protein links FGF signaling to cyclic gene expression in the periodic segmentation of somites. Genes Dev 19(10):1156–1161
Kuramoto Y (1984) Chemical oscillations, waves, and turbulence. Springer, Berlin
Lewis J (2003) Autoinhibition with transcriptional delay: a simple mechanism for the zebrafish somitogenesis oscillator. Curr Biol 13(16):1398–1408
Liao KL (2012) Analysis on mathematical models of somitogenesis in zebrafish. Dissertation, National Chiao Tung University, Taiwan
Liao KL, Shih CW (2012) A lattice model on somitogenesis of zebrafish. Discrete Contin Dyn Syst B 17(8):2789–2814
Liao KL, Shih CW, Tseng JP (2012) Synchronized oscillations in mathematical model of segmentation in zebrafish. Nonlinearity 25:869–904
Makarov DE (2009) Computer simulations and theory of protein translocation. Acc Chem Res 42(2):281–289
Mara A, Schroeder J, Chalouni C, Holley SA (2007) Priming, initiation and synchronization of the segmentation clock by deltaD and deltaC. Nat Cell Biol 9(5):523–530
Morelli LG, Ares S, Herrgen L, Schröter C, Jölicher F, Oates AC (2009) Delayed coupling theory of vertebrate segmentation. HFSP 3(1):55–66
Murray JD (2002) Mathematical biology. Springer, New York
Özbudak EM, Lewis J (2008) Notch signalling synchronizes the zebrafish segmentation clock but is not needed to create somite boundaries. PLoS Genet 4(2):e15
Pourquié O (2004) The chick embryo: a leading model in somitogenesis studies. Mech Dev 121(9):1069–1079
Riedel-Kruse IH, Müller C, Oates AC (2007) Synchrony dynamics during initiation, failure, and rescue of the segmentation clock. Science 317(5846):1911–1915
Schröter C, Ares S, Morelli LG, Isakova A, Hens K, Soroldoni D, Gajewski M, Jülicher F, Maerkl SJ, Deplancke B, Oates AC (2012) Topology and dynamics of the zebrafish segmentation clock core circuit. PLoS Biol 10(7):e1001364
Schwendinger-Schreck J, Kang Y, Holley SA (2014) Modeling the zebrafish segmentation clock’s gene regulatory network constrained by expression data suggests evolutionary transitions between oscillating and nonoscillating transcription. Genetics 197(2):725–738
Simon SM, Peskin CS, Oster GF (1992) What drives the translocation of proteins? PNAS 89(9):3770–3774
Shih CW, Tseng JP (2008) Convergent dynamics for multistable delayed neural networks. Nonlinearity 21:2361–2389
Shih CW, Tseng JP (2011) Global synchronization and asymptotic phases for a ring of identical cells with delayed coupling. SIAM J Math Anal 43(4):1667–1697
Sieger D, Ackermann B, Winkler C, Tautz D, Gajewski M (2006) her1 and her13.2 are jointly required for somitic border specification along the entire axis of the fish embryo. Dev Biol 293:242–251
Uriu K, Morishita Y, Iwasa Y (2009) Traveling wave formation in vertebrate segmentation. J Theor Biol 257(3):385–396
Uriu K, Morishita Y, Iwasa Y (2010) Synchronized oscillation of the segmentation clock gene in vertebrate development. J Math Biol 61:207–229
Yeung HKS, Strogatz SH (1999) Time delay in the Kuramoto model of coupled oscillators. Phys Rev Lett 82:648–651
Yu W, Cao J (2006) Stability and Hopf bifurcation analysis on a four-neuron BAM neural network with time delays. Phys Lett A 351:64–78
Zeiser S, Müller J, Liebscher V (2007) Modeling the Hes1 oscillator. J Comput Biolo 14(7):984–1000
Acknowledgements
The authors are grateful to Chia-Chieh Jay Chu for his advice on computing the bifurcation value in ODEs. The authors were supported, in part, by the Ministry of Science and Technology, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Proof of Theorem 2
Under the conditions of the theorem, the characteristic equation (30) has a pair of complex roots \(R(\mu )\pm iI(\mu )\) for \(\mu \) near \(\mu ^*\). At \(\mu = \mu ^*\), this pair becomes purely imaginary, and the other roots have negative real parts, i.e.,
according to Proposition 6. Substituting \(\lambda (\mu )=R(\mu )+iI(\mu )\) into \(\varDelta _{+}\), and collecting the real and imaginary parts, we have
where \(R=R(\mu )\), \(I=I(\mu )\). If (54) has a solution with \(I(\mu )\ne 0\), then
Thus,
Substituting (55) into (53), using \(\alpha _1(\mu ^{*})\alpha _2(\mu ^{*})\alpha _3(\mu ^{*})-\alpha _3^2(\mu ^{*})-\alpha _1^2(\mu ^{*})\alpha _4^{+}(\mu ^{*})=0\), differentiating with respect to \(\mu \), and utilizing \(R(\mu ^{*})=0\) and \(I(\mu ^{*})\ne 0\), we obtain
by the assumption of the theorem. Thus the transversality condition is met. We conclude that system (8) undergoes a Hopf bifurcation at \(\bar{\mathbb {X}}\) and \(\mu =\mu ^{*}\). \(\square \)
Appendix 2: Stability of bifurcating periodic solution, ODE case
We express system (26) into the form:
where \(\mathbb {X}=(x_1,\ldots ,x_4,y_1,\ldots ,y_4)\), \(A(\mu )\) is the linear part, and \(\mathbf{f}=(f_1,\ldots ,f_8)\) is the nonlinear term. At \(\mu =\mu ^{*}\), \(A(\mu ^*)\) has a pair of purely imaginary eigenvalues \(\lambda (\mu ^{*})=\pm i\omega _0\). Let us make a transformation so that the linear part is in normal form. We consider the change of variables \(\mathbb {X}=P\mathbb {Z}\), where
and \(\mathbf{u}\in \mathbb {R}^8\) is the eigenvector of \(A(\mu ^{*})\) corresponding to the eigenvalue \(i\omega _0\) and \(\mathbf{v}_3,\ldots ,\mathbf{v}_8\) are the generalized eigenvectors for the remaining eigenvalues. Thus
where D is an \(6\times 6\) matrix. Then the transformed system becomes
where \(\mathbb {Z}=(z_1,\ldots , z_8)\) and \(\mathbf{F}(\mathbb {Z},\mu )=P^{-1}{} \mathbf{f}(P\mathbb {Z},\mu )\) with \(\mathbf{F}=(F_1,\ldots ,F_8)\). At \(\mu =\mu ^{*}\), \(\mathbb {Z}=\mathbf{0}\), we define
Next, for \(k=3,\ldots ,8\), we set
and let \(w^{k-2}_{11}\), \(w^{k-2}_{20} \in {\mathbb C}^{6}\) be the solutions of
where D is defined in (56). For \(k=3,\ldots , 8\), let
Then we define
Appendix 3: Sketch of steps (I)–(III) in Sect. 4.2
Step (I): For any fixed \(\tau _4 \ge 0\), we substitute \(\lambda =i\omega \) with \(\omega >0\) into the characteristic equation (42), i.e., \( \triangle _{\pm }(i\omega ,r,\tau _4)=\tilde{R}_{\pm }(i\omega ,r,\tau _4)+i \tilde{I}_{\pm }(i\omega ,r,\tau _4)=0,\) where the real and imaginary parts are respectively
By the properties of trigonometric functions, (57) can be written as
where \(L_{\pm }(\omega ):=[\nu _{3}\nu _{5}(d_{4}\gamma _{1}\pm \gamma _{2}\gamma _{3} \cos {(\tau _4 \omega )})]^2+[\nu _{3}\nu _{5}(\gamma _{1} \omega \mp \gamma _{2}\gamma _{3} \sin {(\tau _4 \omega )})]^{2} >0 \), if \(\omega \) is a solution of (58), and \(\phi _{\pm }\in [0, 2\pi )\) with
In order to find solution \(\omega \) to (58), we sum up the square of equations (58) and obtain
where
Since \(Q_{\pm }(0)=\beta _{4}^{2}\mp 2 d_{4}\nu _{3}^{2}\nu _{5}^{2}\gamma _{1}\gamma _{2}\gamma _{3}\) and both \(Q_{+}(\omega )\) and \(Q_{-}(\omega )\) are strictly increasing eventually and blow up as \(\omega \rightarrow \infty \), we thus establish Proposition 7.
Proposition 7 guarantees the existence of the minimal bifurcation value \(r_c\) and its corresponding purely imaginary eigenvalue \(i \omega _c\). In the sequel, we denote by \(\omega _+\) (resp., \(\omega _-\)) a positive solution to \(Q_{+}(\cdot )=\varGamma \) (resp., \(Q_{-}(\cdot )=\varGamma \)). By using (58), we have
Let \(\sigma =+\) or −. For each solution \(\omega _{\sigma }\) to \(Q_{\sigma }\), there exists a sequence \(\{r_{\sigma }^{(k)}(\omega _{\sigma })\}_{k \in \mathbb {Z}}\):
such that \(\triangle _{\sigma }( i \omega _{\sigma },r_{\sigma }^{(k)}(\omega _{\sigma }),\tau _4)=0\). To simplify the notation, we denote \(r_{\sigma }^{(k)}:= r_{\sigma }^{(k)}(\omega _{\sigma })\) as the bifurcation value. In particular, we shall take into account the case that \(r_{\sigma }^{(k)}>0\) in the following discussions. Accordingly, a positive solution \(\omega _{+}\) (resp., \(\omega _{-}\)) of \(Q_{+}(\cdot )=\varGamma \) (resp., \(Q_{-}(\cdot )=\varGamma \)) corresponds to a pair of purely imaginary roots \(\pm i \omega _{+}\) (resp., \(\pm i \omega _{-}\)) of \(\triangle _{+}(\cdot ,r_{+}^{(k)},\tau _4)=0\) (resp., \(\triangle _{-}(\cdot ,r_-^{(k)},\tau _4)=0\)). This completes the first step.
Step (II): Since every solution of (58) is also a solution to \(Q_+\) or \(Q_-\), if \(Q^{'}_{\sigma }(\omega _{\sigma })\ne 0\), and all other positive solutions to \(Q_{+}(\cdot )=\varGamma \) and \(Q_{-}(\cdot )=\varGamma \) are not integer multiples of \(\omega _{\sigma }\), then \( i \omega _{\sigma }\) is a simple purely imaginary eigenvalue, with \(\sigma =+\) or −.
Step (III): The following conditions guarantee the transversality property:
where
Appendix 4: Sketch of steps (IV) and (V) in Sect. 4.2
Step (IV): Let us consider the characteristic equation (41) with \(r=0\):
Our goal is to derive the condition for parameters under which all roots of (60) have negative real parts for all \(\tau _4\ge 0\). If so, then the stability switch will not occur for any \(\tau _4 \ge 0\) and \(r=0\). Moreover, for any fixed \(\tau _4 \ge 0\), there is also no stability switch when \(r<r_{c}\). Combining these properties, we can then confirm the asymptotical stability of the origin for system (17) for any \(\tau _4 \ge 0\) and \(r <r_{c}\).
We have seen that all characteristic values of (60) have negative real parts when \(\tau _4=0\), under condition (33) or (34). Therefore, it suffices to show that all characteristic values of (60) remain to have negative real parts for any \(\tau _4 \ge 0\) and \(r=0\).
Proposition 8
For any \(\tau _4 \ge 0\), all characteristic values of (60) have negative real parts if
where
Proof
We substitute \(\lambda =i\nu \) with \(\nu \ge 0\) into \(\triangle _{\pm }(\lambda ,0,\tau _4)=0\) and collect the real and imaginary parts to obtain
Next, we sum up the square of equations (64) to get
where
Then \(E'(\nu )=8\nu ^7+E_5 \nu ^5+E_3 \nu ^3+E_1 \nu \), where \(E_1,~E_3,~E_5\) are defined in (63). Thus \(E(\nu )\) is strictly increasing on \([0, \infty )\), under condition (61). Moreover, it is straightforward to verify that \(E(0)=(d_{4}\nu _{3}\nu _{5}\gamma _{1}+\beta _{4})^{2}> (\nu _{3}\nu _{5}\gamma _{2}\gamma _{3})^{2}\) if (62) holds. Consequently, \(E(\nu )>(\nu _{3}\nu _{5}\gamma _{2}\gamma _{3})^{2}\), for all \(\nu \ge 0\), and hence for all \(\nu \in {\mathbb R}\), as E is an even function. Therefore, there does not exist any real solution to (65) under conditions (61) and (62). The assertion thus follows from continuity of characteristic values. This completes the proof.
Step (V): Setting \(\sigma =+\) (resp., \(\sigma =-\)) if \(Q_+(\omega _c)=\varGamma \) (resp., \(Q_-(\omega _c)=\varGamma )\), the following conditions ensure that the Hopf bifurcation occurs at \(r=r_c\) and \(\lambda =i \omega _c\):
-
Condition (B1): \(Q^{'}_\sigma (\omega _c)\ne 0\), and all other positive solutions to \(Q_{+}(\cdot )=\varGamma \) and \(Q_{-}(\cdot )=\varGamma \) satisfy \(\omega \ne m \omega _c\) for any integer m;
-
Condition (B2): \([R_\sigma (\omega _c,r_c)]^{2}+[I_\sigma (\omega _c,r_c)]^{2}\ne 0\), \(W^{2}_{1,\sigma }(\omega _c,r_c)+W^{2}_{2,\sigma }(\omega _c,r_c)\ne 0\), and \(Q_{1,\sigma }(\omega _c,r_c)W_{1,\sigma }(\omega _c,r_c)+Q_{2,\sigma }(\omega _c,r_c)W_{2,\sigma }(\omega _c,r_c) \ne 0\).
Appendix 5: Stability of bifurcating periodic solution, delay case
Step (VI): The stability of the bifurcating periodic solution is determined by the nonlinear terms of the equations. The condition for the stability can be expressed after the equations are put in a suitable form, under the theory of center manifold and normal form. Let us outline the main process for this step, following Hassard et al. (1981), see also Liao et al. (2012) and Yu and Cao (2006).
We write system (17) into the form:
where \({\mathbb X}(t)= (x_{1}(t), \ldots , x_{4}(t),y_{1}(t), \ldots , y_{4}(t))^T\), \({\mathbb X}_{t}(\theta )={\mathbb X}(t+\theta ), \theta \in [-\tau _M, 0]\), \(L_{\mu }:\mathscr {C}\rightarrow \mathbb {R}^{8}\) is a linear operator, and \(G_{\mu }:\mathscr {C}\rightarrow \mathbb {R}^{8}\) is a nonlinear operator, and the phase space \(\mathscr {C}:=\mathscr {C}([-\tau _{M},0],\mathbb {R}_+^{8})\) is a Banach space under the supremum norm \(\Vert \phi \Vert =\sup \{|\phi (\theta )|: {-\tau _{M}\le \theta \le 0}\}\). We label these operators by \(\mu :=r-r_{c}=\tau _{1}+\tau _{2}-r_{c}\), for a fixed minimal bifurcation value \(r_c\). Herein, \(L_{\mu }\) is defined by
where
as expressed in (40), with \(d_{1},d_{2},d_{3},d_{4}\) and \(\gamma _1, \gamma _2, \gamma _3\) defined in (28) and (29), respectively. In fact, according to the Riesz representation theorem, by choosing
we see that
where \(\delta (\cdot )\) is the Dirac delta function. For operator \(G_{\mu }:\mathscr {C}\rightarrow \mathbb {R}^{8}\), its eight components are
successively, where \(\phi =(\phi _1, \ldots , \phi _8)\), and
To put (66) in a suitable form, we define two operators on \(\mathscr {C}^1 := \mathscr {C}^1([-\tau _{M},0],\mathbb {R}^{8})\):
Then (66) can be recast into
The adjoint operator \(A_\mu ^{*}\) of \(A_\mu \) can be computed as
where \(\psi \in \mathscr {C}^1([0,\tau _{M}],\mathbb {R}^{8})\). In the following computation, for convenience, we allow functions to take values in \(\mathbb {C}^{8}\). We use the bilinear form
for \(\phi \in \mathscr {C}([-\tau _{M},0],\mathbb {C}^{8})\), \(\psi \in \mathscr {C}([0,\tau _{M}],\mathbb {C}^{8})\), to determine the coordinates of the center manifold near the origin of (66), where \(\eta (\theta ):=\eta (\theta ,0)\).
Next, denote by \(q(\theta )\) the eigenvector of \(A:=A_{0}\), and \(q^{*}(\theta ^{*})\) of \(A^{*}:=A^{*}_{0}\) corresponding to purely imaginary eigenvalues \(i \omega _{c}\) and \(-i\omega _{c} \), respectively, namely,
We also impose the normalized condition \(\langle q^{*},q \rangle =1\) and \(\langle q^{*},\bar{q} \rangle =0\). To this end, we assume that
for \(\theta \in [- \tau _{M},0)\), \(\theta ^{*}\in (0, \tau _{M}]\), and \(q(0)=(q_{1}, \ldots , q_{8})^{T},~ q^{*}(0)=\frac{1}{\rho }(q_{1}^{*}, \ldots , q^{*}_{8})^{T}\), where \(\rho \), \(q_{i}\) and \(q^{*}_{i}\), \(i=1,\ldots ,8\), are to be determined. Substituting (69) into (68) and evaluating at \(\theta =0\), we obtain
where \(U_{j}=e^{-i\omega _{c}\tau _{j}}\), \(j=1, 2, 4\), and \(V_{j}=i\omega _{c}+d_{j}\), for \(j=1,\ldots , 4\). Notice that \(\langle q^{*},q\rangle =1\) and \(\langle q^{*},\bar{q}\rangle =0\), if we set
where \(J_{1}:=-\gamma _1(q_{3}\overline{q}_{1}^{*}+q_{7}\overline{q}_{5}^{*}) +\gamma _2(q_{8}\overline{q}_{1}^{*}+q_{4}\overline{q}_{5}^{*}) \), \(J_{2}:= \nu _{3}(q_{1}\overline{q}_{2}^{*}+q_{5}\overline{q}_{6}^{*})\), and \(J_{4}:=-\gamma _3(q_{3}\overline{q}_{4}^{*}+q_{7}\overline{q}_{8}^{*})\).
Now, we use q and \(q^{*}\) to construct a coordinate on the center manifold \(\mathscr {M}_{0}\) at \(\mu =0\). For each \(\phi \in \mathscr {C}([-\tau _{M},0],\mathbb {C}^{8})\), we associate a pair (z, w), with \(z=\langle q^{*}, \phi \rangle \), \(w=\phi -z q -\overline{z} \overline{q}= \phi -2 \mathrm{Re}(zq)\). Let \({\mathbb X}_{t}={\mathbb X}_{t}(\theta )=(x_{1,t}(\theta ), \ldots ,x_{4,t}(\theta ),y_{1,t}(\theta ), \ldots , y_{4,t}(\theta ))^T\) be a solution of (67), and let
On the center manifold \(\mathscr {M}_{0}\), \(W(t,\theta )= w(z(t),\bar{z}(t),\theta )\), where
Herein, z and \(\bar{z}\) are the local coordinates of the center manifold \(\mathscr {M}_{0}\) in directions \(q^{*}\) and \(\overline{{q}^{*}}\), respectively.
Hence, the solution \({\mathbb X}_{t}\in \mathscr {M}_{0}\) of (67), at \(\mu =0\), satisfies
where
and
where \(w_{20}^{(k)}(\theta )\) and \(w_{11}^{(k)}(\theta )\) are the kth components of \(w_{20}(\theta )\) and \(w_{11}(\theta )\) respectively. And for \(-\tau _{M}\le \theta <0\), it can be computed that
where
With \(g_{20},~g_{11},~g_{02}\), and \(g_{21}\), we can compute \(C_{1}(r_c),~p_{2},~\zeta _{2}\), and \(T_{2}\) for the stability and the other properties for the bifurcating periodic solution.
Rights and permissions
About this article
Cite this article
Chen, KW., Liao, KL. & Shih, CW. The kinetics in mathematical models on segmentation clock genes in zebrafish. J. Math. Biol. 76, 97–150 (2018). https://doi.org/10.1007/s00285-017-1138-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-017-1138-1