Abstract
The prokaryotic abundance and diversity in three cold, oligotrophic Patagonian lakes (Témpanos, Las Torres and Mercedes) in the northern region Aysén (Chile) were compared in winter and summer using 16S rRNA fluorescence in situ hybridization and PCR-denaturing gradient gel electrophoresis technique. Prokaryotic abundances, numerically dominated by Bacteria, were quite similar in the three lakes, but higher in sediments than in waters, and they were also higher in summer than in winter. The relative contribution of Archaea was greater in waters than in sediments, and in winter rather than in summer. Despite the phylogenetic analysis indicated that most sequences were affiliated to a few taxonomic groups, mainly referred to Proteobacteria (consisting of Beta-, Alpha- and Gammaproteobacteria) and Euryarchaeota (mainly related to uncultured methanogens), their relative abundances differed in each sample, resulting in different bacterial and archaeal assemblages. In winter, the abundance of the dominant bacterial phylotypes were mainly regulated by the increasing levels of total organic carbon in waters. Archaeal abundance and richness appeared mostly influenced by pH in winter and total nitrogen content in summer. The prokaryotic community composition at Témpanos lake, located most northerly and closer to a glacier, greatly differed in respect to the other two lakes. In this lake was detected the highest bacterial diversity, being Betaproteobacteria the most abundant group, whereas Alphaproteobacteria were distinctive of Mercedes. Archaeal community associated with sediments was mainly represent by members related to the order of Methanosarcinales at Mercedes and Las Torres lakes, and by Crenarchaeota at Témpanos lake. Our results indicate that the proximity to the glacier and the seasonality shape the composition of the prokaryotic communities in these remote lakes. These results may be used as baseline information to follow the microbial community responses to potential global changes and to anthropogenic impacts.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pristine, cold oligotrophic lakes like those in Chilean Patagonia are characterized by low temperature (values below 10 °C) and concentrations of nutrients and dissolved organic carbon, determining a low primary productivity and species diversity [3, 14, 31, 50]. Chilean Patagonian lakes are of glacial origin with different geomorphological characteristics and marked seasonal temperature variations (minimum of 4 °C in winter, and maximum of 20 °C in summer) [12, 47]. In winter, lacustrine surfaces are in part covered by a thin iced layer, which may affect all ecological conditions. As remote sites, the cold Chilean lakes are environments with little anthropogenic impact and therefore still harbor pristine biotopes [3, 31].
Cold environments are colonized by different microorganisms well adapted to the fluctuations in temperatures and nutrients availability, playing a relevant role in biogeochemical cycling and in functioning of aquatic environments [32, 35, 37, 40, 41, 48, 49]. Because only a minor fraction of microbial communities in natural environments is culturable [35, 50], different techniques including molecular, structural, genetic, and metabolic approaches have been applied to analyze the prokaryotic community structure and composition and to understand the role of microorganisms in different ecosystems. Among classical molecular analyses, based on 16S rRNA gene sequence analysis, the denaturing gradient gel electrophoresis (DGGE) has been commonly applied to investigate the prokaryotic community structure and composition in different aquatic environments [33, 43]. In addition, fluorescent in situ hybridization (FISH) using specific probes is commonly used to identify active members of the microbial community, obtaining a direct measure of their relative abundance [4, 51].
Prokaryotic communities in cold environments, frequently subjected to freezing periods, have to cope with low temperature, water, and nutrient availability [41]. Schiaffino et al. [44] reported that temperature is an important factor in shaping the prokaryotic abundances of Argentinean Patagonia lakes. The distribution patterns of major bacterial and archaeal groups in these lakes showed that Bacteria possessed similar abundances in late winter or in spring, whereas Archaea varied markedly in summer, spring, and winter, suggesting that the two prokaryotic domains could have different temporal ecological strategies in these lakes [44]. Similar bacterial communities were observed in three cold and nutrient-poor lakes in the Chilean Patagonia (Alto Reino, Las Dos Torres, and Venus), as a consequence of similar capability in responding to different environmental stressors [31].
The remote location of Patagonian lakes could be considered an important geographical factor in determining the characteristic diversity of microbial communities in each aquatic ecosystem [17, 31, 44]. In addition, these aquatic environments could be exposed to global climate changes and to human activities that, as main stressors, could modify the microbial community structure and composition. Therefore, Patagonian lakes represent suitable environments for evaluating changes in the prokaryotic community composition and diversity associated with pronounced environmental and anthropogenic perturbations.
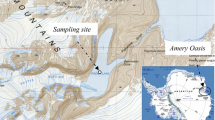
To obtain more information on the microbiota of remote, lacustrine ecosystems in Chilean Patagonia, in this study water and sediment samples were collected in winter and summer periods, from three cold, oligotrophic Patagonian lakes (Témpanos, Las Torres and Mercedes) located in the northern region of Aysén (Chile), along a longitudinal gradient (150 km) (Fig. 1).
The aim of this work is to evaluate the prokaryotic abundance, structure, and composition in these Patagonian lakes, using molecular approaches based on 16S rRNA FISH and PCR-DGGE techniques, and to test the importance of local environmental factors on the natural communities.
Materials and Methods
Study Sites and Sampling
Témpanos lake (44°27′39.5″S; 72°31′41.74″W) is located in the Queulat National Park and is close to a glacier, with an area of 1.34 km2 and an altitude of 115 m above the sea level (a.s.l.). Las Torres lake (45°43′56.96″S; 72°12′50.68″W) has an area of 3.3 km2 and an altitude of 267 m (a.s.l.). Mercedes lake (45°43′59.02″S, 72°12′50.35″W) has an area of 0.19 km2 and an altitude of 267 m (a.s.l.).
Sampling was performed in winter (August 2010) and summer (January 2011) seasons. Surface water samples were collected manually approximately 50 m from the shore. Surface sediment samples (500 g) were collected by a Van Veen grab sampler at approximately 20-m depth in Témpanos and Las Torres, and at 10-m depth in Mercedes. Samples were immediately placed in sterile polyethylene bags (Whirlpak). After collection, water and sediment subsamples for microbial abundance estimation were fixed with formaldehyde 2% (v/v; final concentration), whereas subsamples for DNA extraction were directly stored at −20 °C until further processing.
Chemical Characterization of Water and Sediment Samples
Values of pH and temperature in water were recorded in situ by a portable multiparametric probe (Hanna HI 9812), and dissolved oxygen (DO) was measured using an Oxygen Meter (Cole Palmer, 5946-75). Total Organic Carbon (TOC) content of water samples was analyzed by a Shimadzu TOC analyzer Model 5050. Concentrations of total phosphorous (TP) and total nitrogen (TN) were determined according to standard methods [1].
Total organic matter in sediment samples was estimated by the loss-on-ignition technique, following the method described previously [11] and then converted to TOC according to Schumacher et al. [45]. In addition, the samples were independently analyzed for TP and TN using Kjeldahl digestion followed by colorimetric detection by molecular absorption spectrophotometry, according to standard methods [1].
In order to evaluate the input of contaminants from human activities, organochlorine and polychlorinated biphenyls pesticides were analyzed in samples collected from the three lakes, according to the methods reported by Barra et al. [6]. Briefly, sediment samples (2 g) were mixed with anhydrous sodium sulfate and then extracted by accelerated solvent extraction with acetone:cyclohexane (1:1) at 150 °C, 14 MPa for 5 min (three times). Extracts were concentrated to 0.2 ml and then cleaned up in a Florisil column (10-mm i.d., 3-g Florisil). The conditions of the performed GC/MS analysis were column from Machery Nagel (Optima d6, 60 m, 0.25 mm, 0.25 lm film thickness). The temperature program was 80 °C, then 3 min isothermal, 15 °C/min to 160 °C, and then 3 °C/min to 280 °C hold for 12 min. Quantification was performed with the external standard method with six calibration levels. Water samples (500 ml) were analyzed with liquid–liquid extraction followed by GC–MS for PCBs. The extracts were concentrated to 1 ml and analyzed by GC/MS as described above.
Prokaryotic Cell Abundance
The LIVE/DEAD® BacLight™ Bacterial Viability Kit (Molecular Probes, Eugene, OR), based on cell membrane integrity, was used to distinguish live from dead cells, and their sum was indicated as total counts (TC). Living cells (LC) with intact membranes stain green with SYTO®9, while dead cells with damaged membranes, which are considered to be dead, stain red with propidium iodide [23].
Aliquots of each water sample were concentrated onto black polycarbonate membrane filters (0.2-µm pore size, 25-mm diameter, Nuclepore Corporation, Pleasanton, USA). To detach cells from sediment samples, 1 g of each sample was diluted (1:10 w/v) in sterilized phosphate buffer solution (PBS) (130-mM NaCl; 7-mM Na2HPO4; 3-mM NaH2PO4, pH 7.4) containing Tween 20 (0.5%, v/v; Research Organics Inc.) and sodium pyrophosphate (0.1%, v/v; Sigma-Aldrich), and then sonicated in an ice-water bath three times for 10 s with 20 s interval between cycles using a Brandelin SonoPlus HD 200 (Electronic, Berlin) [26]. Cells from sediments were finally concentrated onto black polycarbonate membrane filters (0.2-µm pore size, 25-mm diameter). After staining, the filtered cells were counted using epifluorescence microscopy (Olympus BX-60M, at 1000× magnification) under ultraviolet light (HG 100 W). The microscope was equipped with a halogen (HG 100 W) light. A G 330–385 exciter filter, an FT 400 chromatic beam splitter, and an LP 420 barrier filter were used for direct counts.
Fluorescence in situ hybridization with rRNA-targeted oligonucleotide probes was used to estimate the abundance of microorganisms ascribed to Bacteria and Archaea domains, according to the protocol previously reported [39]. Cells were concentrated from samples on white polycarbonate filters (0.2-μm pore size, 25-mm diameter) and subsequently fixed for 30 min at room temperature by overlaying filters with a freshly prepared paraformaldehyde (final concentration, 4%) phosphate-buffered saline (PBS; 130-mM NaCl, 10-mM Na2HPO4, and 10-mM NaH2PO4, pH 7.4) solution (3 ml). The fixative was removed by applying vacuum, and filters were washed twice with 3 ml of PBS and, finally, with distilled water. Filters were air-dried and stored in a sterile Petri dish at −20 °C until processing. Filter sections were hybridized with Cyanine-labeled (Cy3) oligonucleotide probe EUB338 and ARCH915 (ThermoHybaid, Interactiva Division, Ulm, Germany) to enumerate bacterial and archaeal cells. Hybridization was performed at 46 °C for 2 h in a hybridization oven with continuous rotation. The filter sections were washed in 10 ml of hybridization buffer and incubated at 48 °C for 15 min. Subsequently, filter sections were mounted on glass slides with Citifluor AF1 (Citifluor Ltd., Canterbury, UK). For each sample, between 50 and 200 cells were then counted under epifluorescence, using Olympus BX60 microscope, equipped with an appropriate filter set for Cy3.
Genomic DNA Extraction and Denaturing Gradient Gel Electrophoresis Analysis
Each water sample (500 ml) was pre-filtered through 2.0-µm pore size membrane filters and concentrated onto 0.22-µm cellulose ester membranes (GSWP04700 Millipore), and genomic DNA was extracted using the Power Soil DNA isolation kit (MoBio Laboratories, Carlsbad, CA, USA), according to the manufacturer’s instructions.
DNA from each sediment sample (5 g) was extracted using the UltraClean Soils DNA kit (MoBio Laboratories, Carlsbad, CA, USA), according to the manufacturer’s instructions. DNA concentrations and purity were checked using a NanoDrop ND-1000, UV–vis spectrophotometer (NanoDrop Technologies, USA).
Bacterial 16S rRNA genes were amplified with eubacterial primer set, forward primer (Escherichia coli positions 9–27: 5′-GAGTTTGATCCTGGCTCAG-3′), and reverse primer (E. coli positions 1542-1525: 5′-AGAAAGGAGGTGATCCAGCC-3′). In order to avoid chimeric formation during the PCR amplification process, we chose to amplify the V3 region of bacterial 16S rRNA by a nested PCR using primers 341f-GCclamp (5′-CCTACGGGAGGCAGCAG-3′) and 534r (5′-ATTACCGCGGCTGCTGG-3′), as previously described [33].
Archaeal 16S rRNA genes were amplified using the Archaea-specific primers 21f (5′-TTCCGGTTGATCCYGCCGGA-3′) and 958r (5′-YCCGGCGTTGAMTCCAATT-3′), in PCR conditions described previously [18]. To amplify the V3 region of the Archaea 16S rRNA gene, a second nested PCR was performed using the Archaea-specific primers set, ARC344-GC (5′-ACGGGGCGCAGCAGGCGCGA-3′) and ARC517r (5′-ATTACCGCGGCTGCTGG-3′) [42].
Bacterial and archaeal PCR products were resolved in polyacrylamide gels according to Muyzer et al. [33] and Casamayor et al. [13], respectively, using a DGGE-2001 system (CBS Scientific Company, CA, USA). All visible bands in DGGE gels were excised, and the small blocks of gel were placed into 50 µl of HypureTM molecular biology grade water (nuclease-free) (Thermo scientific) at +4 °C overnight to allow diffusion of DNA out the gel blocks. The eluate (1 µl) was used as template DNA in a reamplification PCR, using primers and reaction conditions above described.
Sequencing of DGGE bands was performed with primers 341 f for Bacteria and ARC344f for Archaea at Macrogen laboratories (Macrogen Inc., Seoul, Korea). A nucleotide BLAST search (http://www.ncbi.nlm.nih.gov/BLAST) was performed to obtain sequences with the greatest significant alignment.
The partial 16S rRNA gene sequences were deposited in the GenBank database under accession numbers KU912443–KU942475.
Statistical Analysis
DGGE band position and intensity were determined using GELCOMPAR II software package (Applied Maths) and were manually modified. To compare bacterial and archaeal community across all samples, Sørensen similarity coefficient (S s) was applied to the banding patterns and Nonmetric Multi-Dimensional Scaling diagrams were obtained using the PRIMER 6.1.12 (Primer-E, Ltd). The total number of bands in each line was expressed as phylotype richness (R). The relative intensity was used to calculate the Shannon diversity (H’) and the evenness (E) indices, in order to describe possible changes in the DGGE profiles among samples from the same lake (alpha diversity), and among lakes (beta diversity) in the two seasons.
Pearson’s correlation analysis was applied to determine the links between prokaryotic abundances (Bacteria and Archaea), diversity indices (R, H’ and E), and the relative abundance of the dominant bacterial and archaeal groups, as derived by DGGE analysis, with the environmental parameters retrieved from water (pH, TOC, TP, and TN) and sediment (TOC, TP, and TN) samples. Principal Component Analyses (PCAs) were also performed on data from environmental factors and microbial community properties, using PRIMER 6.1.12. A (natural) logarithmic was employed to equalize variances and normalize the data prior to these analyses.
Results
Physical and Chemical Characteristics of the Lakes
Physical and chemical parameters recorded in water and sediment samples collected from the three Chilean Patagonia lakes are reported in Table 1. Values of pH (4.0–5.0) were constantly acidic in all samples, with a slight increase in winter. Temperature recorded in winter was 4 °C in all samples, while in summer ranged from 8.3 (in Témpanos) to 16.3 (in Mercedes). Dissolved oxygen ranged from 8.0 to 9.0 mg l− 1. TOC content in water was higher in winter, when abundant rains provided terrestrial inputs of organic matter, than in summer. Minimum and maximum TOC values were both recorded from Témpanos. TP concentrations were high in Témpanos in both seasons, and TN levels were greater in summer than in winter. The highest TN concentration was recorded in samples from Mercedes.
Sediments from the three lakes were characterized by muddy textures (mud and sand). TOC values were higher in sediments from Las Torres, in both seasons, than those recorded at Mercedes and Témpanos lakes (Table 1). The highest TP was observed in samples from Témpanos in summer. High TN abundances were recorded from Las Torres collected in both seasons, and the lowest was from Témpanos in summer.
Polychlorinated biphenyl and organochlorine pesticides were not detected in any water and sediment samples.
Prokaryotic Cell Abundance
Total counts (TC) from samples, obtained after live/dead staining, were one order of magnitude higher in sediment than in water samples and also in summer when compared to winter (Table 2). The percentage of living cells (LC) with respect to TC was always higher in sediment than in water samples. The lowest contribution of LC to TC was observed in waters collected from Las Torres and Témpanos in winter.
As resulted by FISH, Bacteria always numerically dominated over Archaea (Table 2). Archaeal contribution (ranging from 0.01 to 1.1%) was higher in water than in sediment, and also higher in winter than in summer.
Bacterial DGGE Analyses
Bacterial DGGE profiles and the analysis of the bacterial community structure are reported in Fig. 2. The DGGE banding patterns of Bacteria are presented for water and sediment samples, in winter (w) and summer (s) periods (Fig. 2a). The total number of bands identified in each profile, ranging from 12 to 22 per sample, was lower in water (101) than in sediment (109) samples. When comparing per season, the number of bands was slightly higher in summer (106) than in winter (104). Based on the presence and absence of bands in each lane, S s were calculated from the DGGE profiles. Overall, bacterial populations occurring in water samples were more similar to each other (30.3–72.7% S s) than those in sediments (13.3–57.1% S s). The water samples from Témpanos (Tw and Ts), as well as the sediment samples from Mercedes (Ms) and Las Torres (LTw), were clearly different from the remaining samples, which clustered together to varying degrees (Fig. 2b).
Bacterial DGGE profiles obtained from water and sediment samples (a), and nonmetric multidimensional scaling diagrams (b) representing the bacterial community structure in Las Torres (LT), Mercedes (M), and Témpanos (T) lakes, in winter (w) and summer (s) periods. Sequenced bands are indicated by numbers
Bacterial Phylogeny
A total of 21 excised bands from bacterial DGGE profiles were sequenced to allow their phylogenetic affiliation. The levels of 16S rRNA sequence identity to the most closely related Bacteria are reported in Table 3. None of the bacterial bands was unique to one of the three studied lakes, indicating that common phylotypes were present in all of them, even if different arrangements in bacterial aggregation were observed. After BLAST search, only sequences from five bands (2, 5, 6, 7 and 12) showed high similarity (≥97%) with sequences deposited in the database. Almost all sequences were related to Proteobacteria, mainly consisting of the Beta- (bands 1–5, 7–9, 11–13, 17, 20, and 21), Alpha- (bands 6, 16, 18, and 19), and Gamma- (bands 14 and 15) proteobacterial subclasses. The only sequence related to Firmicutes (band 10) was dominant in all sediments, but occurred also in waters from Témpanos and Las Torres. Sequences belonging to Betaproteobacteria dominated in all fingerprints, but differed in their relative occurrence in sediment and in water samples. Janthinobacterium sp. (band 2) was exclusively retrieved in sediments. Some sequences (bands 1, 3, 9, 17, and 21) were more frequently found in sediments, while others (bands 4, 5, and 13) in waters, and still others (bands 7, 8, 9, 11, 12, and 20) were common to water and sediment samples. The sequence related to Sphingopyxis alaskensis (band 6) (Alphaproteobacteria) was retrieved more frequently in water than in sediment samples, while sequences referred to the two different species of Methylobacterium (bands 16 and 19) were more frequent in sediments. Sequences from bands 14 and 15 were assigned to two different species of Pseudomonas (Gammaproteobacteria), which were found more frequently in sediments than in waters.
Archaeal DGGE Analyses
Archaeal DGGE profiles in water and sediment samples collected in winter and summer and the analysis of the archaeal community structure are reported in Fig. 3. A total number of 90 archaeal phylotypes was observed, ranging from 4 to 14 per sample (Fig. 3a). Archaeal richness was slightly higher in sediments (49) than in waters (41), and in summer (53) rather than in winter (37). Based on the presence and absence of bands in each lane, S s were calculated for the DGGE profiles. Archaeal populations from sediments were more similar to each other (12.5–80% S s) than those from waters (14.3–61.5% S s). Sediments collected from Mercedes and Las Torres in winter were more similar to each other (Fig. 3b).
Archaeal DGGE profiles obtained from water and sediment samples (a), and nonmetric multidimensional scaling diagrams (b) representing the archaeal community structure in Las Torres (LT), Mercedes (M), and Témpanos (T) lakes, in winter (w) and summer (s) periods. Sequenced bands are indicated by numbers
Archaeal Phylogeny
A total of 16 DGGE bands were sequenced. Most sequences (12/16) were assigned to uncultured deposited clones and the remaining four could not be affiliated with any archaeal phyla (Table 4). From the twelve sequences, nine of them were related to Euryarchaeota and the remaining three to Crenarchaeota. Euryarchaeotal sequences were mainly referred to methanogens within the order of Methanosarcinales, including members of the families Methanosaetaceae (bands 2 and 6) and Methanosarcinaceae (bands 5 and 13). Sequences obtained from bands 2 and 6 were affiliated with two different Methanosaeta clones. Band 2 was mainly retrieved in sediments from Las Torres and Mercedes, in both seasonal periods. Band 6 was present only in waters from Las Torres and Mercedes in winter, while in Témpanos was present in both water and sediment samples, only in summer. The sequence from band 5, affiliated with a Methanosarcina clone, was retrieved in water samples from Las Torres and Mercedes in both seasons, while that from band 13, related to a Methanosarcina clone, was only found in sediments from Las Torres in winter. Band 14, referred to Thermoplasmata, was retrieved only from sediments collected from Las Torres in summer. Among Crenarchaeota, band 1 was common to all samples, while bands 10 and 11 were retrieved in sediments from all lakes, but in different periods.
Comparison of Prokaryotic Communities
Diversity indices (R, H’, and, E), as derived by DGGE profiles, from the same lake (alpha diversity) and among lakes (beta diversity) in water and sediments, are reported in Fig. 4. Bacterial H’ slightly varied among samples (from 2.3 to 2.9) and was higher in samples collected in summer than in winter. Bacteria from Las Torres showed the smallest R and H’ values, while the highest were observed in samples collected from Témpanos. Bacterial R and H’ were positively correlated (P < 0.01) in waters, while no correlation was observed in sediments. Overall, bacterial E slightly differed among samples (ranging from 0.80 to 0.87), indicating that bacterial populations were homogenously distributed in each sample.
Conversely to Bacteria, the smallest archaeal R index was present in water samples collected in winter at Témpanos, and the highest at Las Torres, and Mercedes in summer. H’ greatly varied among samples (from 1.35 to 2.52), being higher in sediments collected in summer. Differently to Bacteria, archaeal R and H’ were positively correlated (P < 0.05) in sediments, but not in waters. Archaeal evenness (E) showed greater variations in water (from 0.77 to 0.97) than in sediment (from 0.82 to 0.91) samples, and it was positively correlated to richness (R) (P < 0.05) in sediments but not in waters.
When the relative abundances of dominant bacterial phylotypes were analyzed (Fig. 5), water samples from Las Torres and Témpanos lakes collected in winter showed the highest diversities. Alphaproteobacteria were more abundant in Mercedes. Phylotypes of Euryarchaeota were more abundant in water of Las Torres in winter, whereas those of Crenarchaeota were prevalent in sediment samples from Témpanos lake.
PCA analyses, based on the physical and chemical properties of water and sediment samples, the cell abundance of Bacteria and Archaea, their diversity indices, and main phylotypes, were performed to identify groups of samples with similar community composition and to find their relationships with environmental variables (Fig. 6). The two main components explained 66.4% of the total variance in water samples (Fig. 6a), and 69.1% in sediment samples (Fig. 6b).
Principal component analyses, based on physico-chemical variables (pH, TOC, TP, and TN), the relative cell abundance of Bacteria (Bac) and Archaea (Arch), their diversity indices (R, H’ and E), and most significant phylotypes in water (a) and sediment (b) samples collected from the three Chilean Patagonia lakes, recorded in winter (w, in blue) and summer (s, in red). Las Torres (LT), Mercedes (M), Témpanos (T), Bacterial phyla/classes: Alphaproteobacteria (Alpha), Betaproteobacteria (Beta), Gammaproteobacteria (Gamma), Firmicutes (Firmi). Archaeal phyla/genera: Euryarchaeota (Eury), Crenarchaeota (Cren), Methanosacina (Methsarcina), and Methanosaeta (Methsaeta)
In water samples, Axis 1 (explaining 34.7% of the variance) was strongly associated in the positive side with pH, Euryarchaeota and Methanosaeta, whereas it was related to all bacterial diversity indices (H’, R, and E) in the negative side. Axis 2 (31.7% of the variance) was mainly associated with archaeal richness (RA) and diversity (H’A) in the positive side, while with TOC, Alphaproteobacteria and Firmicutes in the negative side.
In sediments (Fig. 6b), Axis 1 (36.6% of the variance) was strongly associated with archaeal abundance (Arch) and richness (RA) in the positive side, whereas with bacterial abundance (Bac) in the negative side. Instead, Axis 2 (33.1%) was strongly related to a combination of all environmental variables (TOC, TN, and TP) in the positive quadrant and to Firmicutes and Betaproteobacteria in the negative side. Sediments collected in winter from the three lakes appeared completely separated from those collected in summer by Axis 2, suggesting a strong influence of seasonality on sediment rather than on water samples.
Témpanos and Mercedes lakes, with the highest negative scores for Axis 1, appeared more closely clustered in summer than in winter, mainly due to the bacterial community features in waters (Fig. 6a), and to those of archaeal community in sediments (Fig. 6b). Although distantly, in winter Las Torres and Mercedes lakes appeared clearly separated from Témpanos lake by Axis 2 (Fig. 6a) in waters, and Axis 1 (Fig. 6b) in sediments.
PCA clusters appeared quite different to those occurred in the NMDS plots (Figs. 2, 3), indicating that a combination of environmental factors and limnological characteristics shapes the prokaryotic abundance and diversity in the three lakes.
Discussion
The cold, oligotrophic Chilean Patagonia lakes are located in land-protected areas and some of them were previously described as pristine environments [3, 8, 31]. The absence of polychlorinated biphenyls and organochlorine pesticides in water and sediment samples collected from the studied lakes suggests low human activity [3, 6, 31, 50]. Although the low amounts of nutrients were indicative of oligotrophic environments, all lakes showed an increase in TOC in winter, as consequence of abundant rainfalls providing terrestrial inputs of organic matter [47]. However, anthropogenic activities in this area could expose them to the risk of altering their natural conditions.
Due to the different lake locations, and surrounding vegetation, the limnological properties appeared different in the three lakes. Lake Témpanos, located most northerly and closer to a glacier, showed less variation in temperatures recorded in winter and in summer than those from the other two lakes. Similarly to Témpanos, Mercedes lake, the smallest of the three studied, is partially covered by ice in winter. As a consequence of volcanic activity and the geologic composition of the area, pH values registered in the three lakes were always acidic [3]. Chaiten and Hudson are active volcanos separated by approximately 340 km, and the three lakes considered in this study are located between both volcanos. Besoain et al. [7] indicated that the eruptive activity of Hudson volcano in 1991 deposited 2500 kg/ha of sulfured volcanic ashes and the pH of affected waters decreased. Gammons et al. [20] reported that the hyper-acidic (pH 2.68) conditions of Caviahue lake, located in the northern Argentinean Patagonia (37.5°S, 71°W), are due to the presence of ions and rare earth elements incorporated by the eruptive activity of the Copahue volcano.
Prokaryotic abundances, dominated numerically by Bacteria, were quite similar in the three studied lakes, being higher in sediment than in water samples, and in summer than in winter. These results were similar to those of other cold oligotrophic lakes, also including those from Patagonia [2, 3, 25, 31, 44]. Although Archaea represent a minor component, their percentage contribution to the total prokaryotic community was highest in water samples collected in winter, when lacustrine surface were partially covered by a thin layer of ice. Recent investigations in Argentinean Patagonia lakes also reported that relative abundances of Bacteria and Archaea showed a marked seasonality, with peaks of Bacteria in summer and those of Archaea in winter [44].
To compare the prokaryotic communities from the three lakes, we also analyzed the intrinsic genetic diversity by the fingerprinting method based on PCR-DGGE of 16S rRNA, which reveals prominent populations associated with different samples and sampling dates. Moreover, sequencing analysis of the most prominent bands was used to assess their potential affiliation to other cultured and uncultured prokaryotes from other environments. Although there were not many differences in the DGGE number of bands between water and sediment samples from the three lakes, a higher richness (numbers of bands) was found in sediments than in waters, and in summer than in winter samples, for both Bacteria and Archaea. Bacterial communities consisted of more numerous and more diverse populations with respect to those of Archaea. As indicated by the index Shannon H’, bacterial diversity showed a large variation among the examined samples from the three lakes (Fig. 4), and it was higher in summer than in winter. Bacterial evenness slightly differed among samples, indicating that bacterial populations were homogenously distributed in each sample. Interestingly, evenness of Archaea was greater than that of Bacteria, with higher values in water rather than in sediment samples.
As resulted by phylogenetic analysis, the closest relatives associated with Proteobacteria, mainly consisting of Beta-, Alpha-, and Gammaproteobacteria, and Firmicutes, were chemoorganotrophs or aerobic heterotrophs. These putative metabolisms were highly dependent on organic matter, which explains the relationship between TOC with bacterial community composition (Fig. 6).
Even if Proteobacteria was the largest phylum, the major subclasses and their proportions varied among the different lakes. Βetaproteobacteria constituted the most prominent bacterial group in the prokaryotic communities of the three Patagonian lakes. Similar results have been reported in different cold oligotrophic polar environments [22, 38], as well as in small oligotrophic mountain lakes [24, 53]. Among Betaproteobacteria, the genus Herminiimonas was often found in cold aquatic ecosystems, including the species H. glaciei able to live in ice, as miniaturized cells for extended periods of time [30]. Since they may compete effectively in their habitat, the capacity of certain psychrophiles to grow at higher temperature might be ecologically relevant. This may be the case of sequences related to Janthinobacterium sp., often retrieved in different environments, even if several species have been isolated from cold Antarctic habitats [49]. Similarly, the eurypsychrophilic alphaproteobacterium Sphingopyxis alaskensis, isolated from cold waters (4–10 °C) at the North Pacific Ocean near Alaska and Japan, is able to grow faster at higher temperatures [15]. Also, Sphingopyxis chilensis, isolated from the Bio Bio river in the central Chile, is able to grow at 30 °C [21].
Members of the genus Methylobacterium (Alphaproteobacteria) are able to grow on single-carbon compounds, such as CH4 or CH3OH, as a sole carbon and energy source [34], and their presence in the studied lakes suggests the production of organic matter from simple carbon compounds. Although Actinobacteria are recognized as ubiquitous and dominant in oligotrophic lakes [36], sequences related to this phylum were not detected in the samples from any of the three Patagonian lakes here studied. In fact, Aguayo et al. [3] suggested that an increase of Actinobacteria in Patagonian lakes could be indicative of alteration in the lacustrine homeostasis.
Phylogenetic information on archaeal communities in cold freshwater lakes is still very limited. Most of the archaeal sequences were affiliated with Euryarchaeota and the remaining with Crenarchaeota, as also reported in samples obtained from high-altitude lakes [38]. A large number of sequences could not be attributed to the phylum level, suggesting the presence of new phylotypes in these lakes. Overall, diversity indices for the water archaeal community appeared influenced by pH and TN. The highest archaeal richness and diversity were found at Mercedes and Las Torres lakes, where high TN concentrations were recorded in both seasons. Seasonal correlation between nitrogen compounds and Archaea were previously reported in the surface waters of Spanish alpine lakes [5], suggesting therefore a pivotal role of Archaea also in the N biogeochemistry of Chilean oligotrophic lakes.
Euryarchaeotal sequences were mainly related to methanogens belonging to the order Methanosarcinales, including the genera Methanosarcina and Methanosaeta, which catalyze the terminal phase of the degradation of organic matter in anoxic environments [27], but also in oxic lake waters [9]. These methanogens, retrieved as predominant in sediment samples from Las Torres and Mercedes, have been frequently observed in superficial zone of freshwater sediments [10, 16, 28, 46, 52], and also from different cold habitats, such as basal ice layers of glaciers and Antarctic lakes [14, 19]. Thermoplasmata, usually found as a minor group in freshwater lake sediments [29], were retrieved in this study only from sediments of Las Torres, suggesting their potential role in the methanogenic activities.
The prokaryotic community from water and sediment samples from the three Chilean Patagonian lakes appeared different in winter and summer, demonstrating that a close relationship exists among microbial community composition and the limnology of the lakes. Bacteria and Archaea could play different biogeochemical roles in waters and sediments in response to seasonal changes.
The prokaryotic community composition from Témpanos lake, located most northerly and closer to a glacier, greatly differed with respect to the other two lakes. The highest bacterial diversity was detected in this lake, mainly related to betaproteobacterial phylotypes, whereas Alphaproteobacteria were distinctive of Mercedes lake. Archaeal diversity was mainly related to euryarchaeotal methanogens, affiliated with members of the order Methanosarcinales, in samples from Mercedes and Las Torres, and to Crenarchaeota from Témpanos. These findings indicate that the different site location, the proximity to the glacier, and the seasonal availability of nutrients shape the composition of the prokaryotic communities in the three cold, oligotrophic Chilean Patagonian lakes. The absence of pesticides was considered as indicative of low human influences. As micro-scale indicators, the prokaryotic community characteristics registered at the present conditions can be used to follow the microbial responses to potential global changes and anthropogenic impacts.
References
APHA-AWWA-WPCF (1985) Standard methods for the examination of water and wastewater, 16th edn. EUA, Washington
Aguayo J, Barra R, Becerra J, Martínez M (2009) Degradation of 2, 4, 6 tribromophenol and 2, 4, 6 trichlorophenol by aerobic heterotrophic bacteria present in psychrophilic lakes. World J Microbiol Biotechnol 25:553–560
Aguayo P, González C, Barra R, Becerra J, Martínez M (2014) Herbicides induce change in metabolic and genetic diversity of bacterial community from a cold oligotrophic lake. World J Microbiol Biotechnol 30:1101–1110
Amann RI, Ludwig W, Schleifer KH (1995) Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59:143–169
Auguet JC, Nomokonova N, Camarero L, Casamayor EO (2011) Seasonal changes of freshwater ammonia oxidizing archaeal assemblages and nitrogen species in oligotrophic alpine lakes. Appl Environ Microbiol 77:1937–1945
Barra R, Cisternas M, Urrutia R, Pozo K, Pacheco P, Parra O, Focardi S (2001) First report on chlorinated pesticide deposition in a sediment core from a small lake in central Chile. Chemosphere 45:749–757
Besoain EM, Ruiz RS, Hepp CK (1995) The Hudson volcano eruption and its effects on the agriculture of the XI Región. Agricultura Técnica (Chile) 55:204–219
Bertrand S, Boës X, Castiaux J, Charlet F, Urrutia R, Espinoza C, Charlier B, Lepoint G, Fagel N (2005) Temporal evolution of sediment supply in Lago Puyehue (Southern Chile) during the last 600 years and its climatic significance. Quarter Res 64:163–175
Bogard MJ, del Giorgio PA, Boutet L, Garcia-Chaves MC, Prairie YT, Merante A, Derry AM (2014) Oxic water column methanogenesis as a major component of aquatic CH4 fluxes. Nat Commun 5:5350. doi:10.1038/ncomms6350
Borrel G, Jézéquel D, Biderre-Petit C, Morel-Desrosiers N, Morel JP, Peyret P, Fonty G, Lehours AC (2011) Production and consumption of methane in freshwater lake ecosystems. Res Microbiol 162:832–847
Boyle J (2002) Mineralogical and geochemical indicator techniques. In: Last W, Smol JP (eds) Tracking environmental change using lake sediments. Springer, New York, pp 83–141
Campos H (1984) Limnological studies of Araucanian lakes, Chile. Verhandlungen der Internationale Vereinigung für Theoretische und Angewandte Limnologie 22:1319–1327
Casamayor EO, Schäfer H, Baňeras L, Pedrós-Alió C, Muyzer G (2000) Identification of and spatio-temporal differences between microbial assemblages from two neighboring sulphur lakes: comparison by microscopy and denaturing gradient gel electrophoresis. Appl Environ Microbiol 66:499–508
Cavicchioli R (2006) Cold-adapted Archaea. Nat Rev Microbiol 4:331–343
Cavicchioli R, Ostrowski M, Fegatela F, Goodchild A, Boixereus G (2003) Life under nutrient limitation in oligotrophic marine environments: an eco/physiological perspective of Sphingopyxis alaskiensis (formerly Sphingomonas alaskiensis). Microb Ecol 45:203–217
Chan OC, Claus P, Casper P, Ulrich A, Lueders T, Conrad R (2005) Vertical distribution of structure and function of the methanogenic archaeal community in Lake Dagow sediment. Environ Microbiol 7:1139–1149
Curtis TP, Sloan WT (2004) Prokaryotic diversity and its limits: microbial community structure in nature and implications for microbial ecology. Curr Opin Microbiol 7:221–226
Ferrari VC, Hollibaugh JT (1999) Distribution of microbial assemblages in the central Arctic Ocean basin studied by PCR/DGGE: analysis of a large data set. Hydrobiologia 401:55–68
Franzmann PD (1996) Examination of Antarctic prokaryotic diversity through molecular comparisons. Biodivers Conserv 5:1295–1305
Gammons CH, Wood SA, Pedrozo F, Varekamp JC, Nelson BJ, Shope CL, Baffico G (2005) Hydrogeochemistry and rare earth element behavior in a volcanically acidified watershed in Patagonia, Argentina. Chem Geol 222:249–267
Godoy F, Vancanney M, Martinez M, Steinbüchel A, Swings J, Rehm A (2003) Sphingopyxis chilensis sp. a chlorophenol-degrading bacterium which accumulates polyhydroalkanoates, and transfer of Sphingomonas alaskensis to Sphingopyxis alaskensis comb. nov. Int J Syst Evol Microbiol 53:473–477
Gugliandolo C, Michaud L, Lo Giudice A, Lentini V, Rochera C, Camacho A, Maugeri TL (2016) Prokaryotic community in lacustrine sediments of Byers Peninsula (Livingston Island, Maritime Antarctica). Microb Ecol 71:387–400
Haugland RP (2002) LIVE/DEAD BacLight bacterial viability kits. In: Gregory J (ed) Handbook of fluorescent probes and research products, ninth edition. Molecular Probes, Eugene, pp 626–628
Hiorns WD, Methe BA, Nierzwicki-Bauer SA, Zehr JP (1997) Bacterial diversity in Adirondack mountain lakes as revealed by 16S rRNA gene sequences. Appl Environ Microbiol 63:2957–2960
Jiang H, Dong H, Zhang G, Yu B, Chapman L, Matthew F (2006) Microbial diversity in water and sediment of Lake Chaka, an athalassohaline lake in Nortwestern China. Appl Environ Microbiol 72:3832–3845
Kallmeyer J, Smith DC, Spivack AJ, D’Hondt S (2008) New cell extraction procedure applied to deep subsurface sediments. Limnol Oceanogr Methods 6:236–245
Kendall MM, Boone DR (2006) The order Methanosarcinales. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes 3rd edn, vol 3, Springer, New York, pp 244–256
Koizumi Y, Takii S, Fukui M (2004) Depth-related change in archaeal community structure in a freshwater lake sediment as determined with denaturing gradient gel electrophoresis of amplified 16S rRNA genes and reversely transcribed rRNA fragments. FEMS Microbiol Ecol 48:285–292
Liu FH, Lin GH, Gao G, Qin BQ, Zhang JS, Zhao GP, Zhou ZH, Shen JH (2009) Bacterial and archaeal assemblages in sediments of a large shallow freshwater lake, Lake Taihu, as revealed by denaturing gradient gel electrophoresis. J Appl Microbiol 106:1022–1032
Loveland-Curtze J, Miteva VI, Brenchley JE (2009) Herminiimonas glaciei sp. nov., a novel ultramicrobacterium from 3042 m deep Greenland glacial ice. Int J Syst Evol Microbiol 59:1272–1277
Mackenzie R, Barros J, Martínez M (2011) Metabolic and molecular analysis of aerobic heterotrophic bacteria in cold-oligotrophic freshwater ecosystems. World J Microbiol Biotechnol 27:2499–2504
Miteva VI, Sheridan PP, Brenchley JE (2004) Phylogenetic and physiological diversity of microorganisms isolated from a deep greenland glacier ice core. Appl Environ Microbiol 70:202–213
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nayak DD, Marx CJ (2014) Genetic and phenotypic comparison of facultative methylotrophy between Methylobacterium extorquens strains PA1 and AM1. PLoS ONE 9:e107887
Nocker A, Burr M, Camper AK (2007) Genotypic microbial community profiling: a critical technical review. Microb Ecol 54:276–289
Newton RJ, Jones SE, Eiler A, McMahon KD, Bertilsson S (2011) A guide to the natural history of freshwater lake. Microbiol Mol Biol Rev 75:14–49
Pearce DA (2003) Bacterioplankton community structure in a Maritime Antarctic oligotrophic lake during a period of holomixis, as determined by DGGE and FISH. Microb Ecol 46:92–105
Pearce DA, Galand PE (2008) Microbial biodiversity and biogeography. In: Vincent W, Laybourn-Parry J (eds) Polar lakes and rivers, limnology of Arctic and Antarctic aquatic ecosystems. Oxford University Press, New York, pp 213–230
Pernthaler J, Glöckner FO, Schönhuber W, Amann R (2001) Fluorescence in situ hybridization with rRNA-targeted oligonucleotide probes. Methods Microbiol 30:207–226
Price B, Sowers T (2004) Temperature dependence of metabolic rates for microbial growth, maintenance, and survival. PNAS 10:4631–4636
Priscu J, Christner B (2003) Earth’s icy biosphere. In: Bull AT (ed) Microbial diversity and bioprospecting. ASM press, Washington, pp 130–145
Raskin L, Stromley JM, Rittmann BE, Stahl DA (1994) Group specific 16S rRNA hybridization probes to describe natural communities of methanogens. Appl Environ Microbiol 60:1232–1240
Schäfer H, Muyzer G (2001) Denaturing gradient gel electrophoresis in marine microbial ecology. Methods Microbiol 30:425–468
Schiaffino MR, Sánchez ML, Gerea M, Unrein F, Balagué V, Gasol JM, Izaguirre I (2016) Distribution patterns of the abundance of major bacterial and archaeal groups in Patagonian lakes. J Plankton Res 38:64–82
Schumacher BA (2002) Methods for the determination of total organic carbon (TOC) in soils and sediments. Ecological risk assessment support center office of research and development US. Environmental Protection Agency, Washington DC, pp 1–25
Schwarz JIK, Eckert W, Conrad R (2007) Community structure of Archaea and Bacteria in a profundal lake sediment Lake Kinneret (Israel). Syst Appl Microbiol 30:239–254
Soto D (2002) Oligotrophic patterns in southern Chilean lakes: the relevance of nutrients and mixing depth. Rev Chil Hist Nat 75:377–393
Steven B, Briggs G, McKay CP, Pollard WH, Greer CW, Whyte LG (2007) Characterization of the microbial diversity in a permafrost sample from the Canadian high Arctic using culture-dependent and culture-independent methods. FEMS Microbiol Ecol 59:513–523
Tindall BJ, Brambilla E, Steffen M, Neumann R, Pukall R, Kroppenstedt RM, Stackebrandt E (2000) Cultivatable microbial biodiversity: gnawing at the Gordian knot. Environ Microbiol 72:310–318
Woelfl S, Villalobos L, Parra O (2003) Trophic parameters and method validation in Lake Riñihue (North Patagonia: Chile). Rev Chil Hist Nat 76:459–474
Wilhartitz IC, Mach RL, Teira E, Reinthaler T, Herndl GJ, Farnleitner AH (2007) Prokaryotic community analysis with CARD-FISH in comparison to FISH in ultra-oligotrophic ground- and drinking water. J Appl Microbiol 103:871–881
Ye WJ, Liu XL, Lin SQ, Tan J, Pan JL, Li DT (2009) The vertical distribution of bacterial and archaeal communities in the water and sediment of Lake Taihu. FEMS Microbiol Ecol 70:263–276
Zwart G, Crump BC, Agterveld MP, Hagen F, Han SK (2002) Typical freshwater bacteria: an analysis of available 16S rRNA gene sequences from plankton of lakes and rivers. Aquat Microb Ecol 28:141–155
Acknowledgements
This work was supported by grants Fondecyt no 1100462 and Enlace VRID no 214.036.041-1.0.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
This article does not contain any studies with human or animal subjects.
Rights and permissions
About this article
Cite this article
Aguayo, P., González, P., Campos, V. et al. Comparison of Prokaryotic Diversity in Cold, Oligotrophic Remote Lakes of Chilean Patagonia. Curr Microbiol 74, 598–613 (2017). https://doi.org/10.1007/s00284-017-1209-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-017-1209-y