Abstract
Background
The number of fluoroscopically guided interventions (FGI) has increased significantly over time. However, little attention has been paid to possible stochastic radiation effects. The aim of this retrospective study was to investigate the number of patients who received cumulative effective doses over 100 mSv during FGI procedures.
Material and Methods
Five thousand five hundred and fifty four classified FGI procedures were included. Radiation dose data, retrieved from an in-house-dose-management system, was analysed. Effective doses and cumulative effective doses (CED) were calculated. Patients who received a CED > 100 mSv were identified. Radiology reports, patient age, imaging and clinical data of these patients were used to identify reasons for CED ≥ 100 mSv.
Results
One Hundred and thirty two (41.1% female) of 3981 patients received a CED > 100 mSy, with a mean CED of 173.5 ± 84.5 mSv. Mean age at first intervention was 66.1 ± 11.7 years. 81 (61.4%) of 132 were older than 64 years, one patient was < 30 years. 110 patients received ≥ 100 mSv within one year (83.4%), through FGIs: EVAR, pelvic/mesenteric interventions (stent or embolization), hepatic interventions (chemoembolization, TIPSS), embolization of cerebral aneurysms or arterio-venous-malformations.
Conclusions
Substantial CED may occur in a small but not ignorable fraction of patients (~ 3%) undergoing FGIs. Approximately 2/3rd of patients may most likely not encounter radiation-related stochastic effects due to life-threatening diseases and age at first treatment > 65 years but 1/3rd may. Patients undergoing more than one FGI (77%) carry a higher risk of accumulating effective doses > 100 mSv. Remarkably, 23% received a mean CED 162.2 ± 72.3 mSv in a single procedure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, the number of fluoroscopically guided interventions (FGI) increased steadily over time. Technical improvements and growing experience of interventionists resulted in more and more indications for fluoroscopically guided endovascular procedures such as percutaneous endovascular interventions, transcatheter embolization and others. Above a certain threshold, ionizing radiation can cause deterministic effects such as tissue injuries. Exposure to ionizing radiation, especially above 100 mSv, may lead to organ doses > 100 mGy, a range at which a statistically significant excess of certain cancers like bone marrow, thyroid, bladder, breast, colon, and lung has been demonstrated in a number of studies, and there is a reasonable degree of agreement among official international organizations [1,2,3,4,5,6,7,8,9]. Thus, dose management is crucial in order to avoid these detrimental effects of radiation.
Many factors may contribute to a substantial radiation dose of patients during FGI. For example, patient weight and anatomy, anatomical location and complexity of treated pathologies, equipment settings and experience of the interventionist are all related to radiation dose [5]. Fluoroscopy using high frame rates, high dose mode, large field of view and a large number of angiographic series may also result in a substantial radiation dose of patients [6]. Several US and European centres reported average radiation exposure of patients in commonly performed FGIs expressed as kerma area product (KAP) and air kerma at reference point (AK) [2, 3, 7, 10,11,12,13,14,15,16,17,18,19]. However, cumulative effective dose (CED) of patients in FGI has not been reported so far from Europe, and there is only one study from USA [1].
The calculation of the effective dose (in this work) is based on the multiplication of dose conversion coefficients (DCC) by the kerma-air product specified by the fluoroscopic system. These DCCs differ depending on the body region examined and the FGI procedure and therefore the calculated effective dose will differ too. That means for example, if you undergo two different FGI’s at the same body region it may result in two different numbers of effective doses, even if the two single KAP would be the same. Therefore, the calculation of a cumulative effective dose has meaningfulness than just summing up the KAP.
Implementation of international basic safety standards increased awareness for potential radiation risks in patients and provided guidelines, how to minimize or even avoid these risks. Especially, dose monitoring systems proved to be a helpful tool to reduce radiation dose of patients [20, 21]. Nevertheless, due to more and more complex interventions, patients have increasing radiation risks, whether of radiation induced tissue reactions such as skin injuries or late stochastic effects, such as cancer. The aim of this study was to analyse how many patients are exposed to a substantial effective radiation dose above 100 mSv during commonly performed FGIs. In addition, procedures were identified, which are prone to deliver CED ≥ 100 mSv Since other studies did not deliver detailed information regarding the time intervals and timing of the applied FGIs, we analyzed among other things the time interval when a CED of 100 mSv was reached—it is important to know if 100 milisieverts cumulated within a year or 1 day.
Methods
Cohort and Data Sources
Data was collected at a tertiary care centre in Europe during January 2016–October 2020. For each procedure, the data consistency and completeness was checked, 10 data sets were excluded from further data processing due to missing or incomplete data. Our inclusion criteria were: Patients with age ≥ 18 years at first FGI, no difference in gender or other demographic criteria.
The exclusion criteria: Patients younger 18 years or data sets which were incomplete and useless for further analysis.
Medical information such as indication for FGI, underlying pathology etc. was retrieved from the electronic medical records stored in the hospital and radiology information system (HIS, RIS).
The study was conducted in accordance with the Declaration of Helsinki, and the study protocol was approved by the local Ethics Committee, Votum number 1259/2023.
At our center, 3 angiographic systems were used for (FGIs) namely Allura XPER and Allura XPER-Biplane (both Philips Medical Systems) as well as Axiom Artis (Siemens Healthineers).
After each procedure, all relevant parameters such as cumulative air kerma at reference point (CAK), kerma-area-product (KAP), fluoroscopy time, number of fluoroscopic images and angiographic series were automatically documented in the Structured Radiation Dose Report and stored in the Picture Archiving and Communication System (PACS). We used KAP to calculate effective dose/patient. Effective radiation dose data is automatically calculated and stored by our in-house developed dose management system “SumDose”. This database was used to identify patients who received a cumulative effective dose (CED) ≥ 100 mSv.
Dose conversion coefficients DCCE (Dose conversion coefficient from kerma-area product to effective dose expressed in mSv/Gy cm2) were taken from earlier publications [1, 22, 23]. Procedures which could not be correlated to any procedure which are described in previous publications [1, 22, 23] were excluded from the data analysis.
Statistical Analysis
Statistical software (Excel, Microsoft) was used to calculate mean value, five percentiles (10th, 25th, 50th, 75th, 95th), and interquartile range for age and dose parameters.
Results
Total Patients Cohort
In the time period of 57 month a total cohort of 3981 adult patients (mean age, 64.95 years; 41.1% female) underwent 5677 FGI procedures (1.43 procedures per person). After excluding 123 FGIs from this original dataset (since they could not be correlated to any other FGI published so far or were incomplete) finally 5554 datasets were included in this study of 3981 patients.
Cohort of Patients with CED ≥ 100 mSv
In the remaining cohort, we detected 132 patients (out of 3981 and thus 3.3%) with cumulated doses ≥ 100 mSv. These patients had undergone 359 FGI procedures.
Demographic Information
The mean age at first procedure in patients with CED ≥ 100 mSv was 66.1 (IQR 60–75) years. The mean number of FGI procedures per patient was 2.72 (IQR 2–4). 81 patients were 65 years or older at the first procedure (61.4% or nearly 2/3rd). The youngest patient with CED ≥ 100 mSv was 19 years old, the oldest 89 (Table 1). Median age of patients in this cohort was 68 years.
Thirteen patients younger than 50 years accumulated an effective dose ≥ 100 mSv which was documented after EVAR (Endovascular Aneurysm Repair) in 5 patients, after TIPSS (transjugular intrahepatic portosystemic stent shunt) in 4 patients, after thoracic-abdominal embolizations for treatment of life threatening bleedings in 3 patients and after treatment of a large cerebral AVM in 1 patient. 5 of 13 patients were exposed to a CED > 100 mSv in a single procedure (2 EVAR, 3 embolization procedures), 6 in 2 procedures (3 EVAR, 3 TIPSS) and 1 patient in 5 procedures (embolization of cerebral AVM), another patient in 6 TIPSS related procedures.
Dose Values and Analysis of Risk Factors for CED > 100mSv
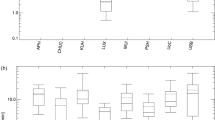
In a total of 5554 procedures, the mean effective dose for a single FGI was 14.3 mSv (IQR: 1.22–17.0 mSv) and the mean KAP 64.5 Gy m2 (IQR: 7.9–82.2 Gy m2) (Fig. 1).
132 (3.3%) patients (12.8% females; 87.2% male) received a CED above 100 mSv. In this group, the median and mean value of CED was 145.9 and 173.5 mSv respectively (IQR: 122–187 mSv) (Fig. 2a, b). Figure 2c shows the distribution of patients with a CED in the range of minimum 101 to maximum 591 mSv. The mean cumulative Air Kerma at reference point was 3.88 Gy (IQR: 2.4–4.7 Gy).
a Boxplots for cumulated effective dose (CED) per patient for the cohort of 132 patients who received CED ≥ 100 mSv from one or more FGIs and one single FGI (5 patients, right figure). b Numbers of procedures in patients who received a CED ≥ 100 mSv. Most left column: 30 patients (23%) of 132 had one single FGI and received therefore a mean CED of 162.2 ± 72.3 mSv. c CED histogram of patients, who received one or more fluoroscopically guided interventions (FGI) with a cumulative effective dose CED ≥ 100 mSv
A large number of patients with CED ≥ 100 mSv suffered from either malignant tumors (49 patients, 37.1%) or aortic aneurysms (32, 24.2%). 6 (4.5%) patients suffered from cerebral aneurysms or AVMs, 34 (25.7%) patients from abdominal bleeding, 3 (2.3%) patients from severe atherosclerosis and metabolic diseases. Hemorrhage originating from trauma was rare in our group of patients (6, equaling 4.5%). Type and number of the related FGI procedures with CED ≥ 100 mSv are given in detail in Table 2.
In the group of patients with a substantial effective dose exceeding 100 mSv, 30 (22.7%) patients underwent one single FGI (mean CED of 162.2 mSv). 76 (57.6%) patients received a CED ≥ 100 mSv from one or two interventions, 125 patients (95%) had five or less interventions (average 2.5) (Fig. 2b).
One hundred and two patients (77.2%) received a CED less than 200 mSv, 21 patients or 15.9% accumulated a CED from 200 to 300 mSv and 9 patients (6.8%) more than 300 mSv (Fig. 2c).
The maximum CED was 547.4 mSv in a patient who had two FGIs (thoracoabdominal embolization) on 2 consecutive days with 159.2 and 388.2 mSv with a cumulative KAP of 2119 Gy.cm2.
Analysis of the timeline demonstrated, that 54 (40.6%) patients accumulated a CED ≥ 100 mSv within 1 day. and additional 22% within 1 month (Table 4, Fig. 3). Procedures with a CED ≥ 100 mSv were performed within one year in 83.4% of patients, 69.9% were treated within 180 days (Tables 3 and 4).
Furthermore, the 6 types of fluoroscopic guided interventions contributing to CED of ≥ 100 mSv were analysed in detail in respect to the cumulated air kerma at the reference point (CAK) and CED. The endovascular aneurysm repair (EVAR) procedure with a mean CED of 184.3 mSv had the highest radiation exposure. Another potentially high dose procedure, pelvic stenting, demonstrated a mean CED of 132.3 mSv. However, whereas pelvic stenting contributed to the total CED 1.7%, contribution to total CAK was high (13%). In comparison, thoracoabdominal embolization contributed 27.3% to total CED, but 12.8% to total CAK (Fig. 4a and Table 2).
Discussion
In our cohort of 3981 patients, 3.3% received a CED ≥ 100 mSv which is comparable to 4% reported in a previous study from a tertiary center in the United States [1, 22]. Different to other publications [1, 22], our cohort included significantly more male than female patients (87.2 vs. 69.6%). In our patient population, the number of procedures for treatment of cerebrovascular diseases was higher (4.6% in our cohort compared to 0.69% in patients reported in [1]). Furthermore, in our cohort, we had a higher number of endovascular aortic aneurysm repairs (EVAR) (24.2%). Aortic aneurysms are more frequent in male than female patients. Dose data of EVAR were not included in the previous studies [1]. The number of patients with malignant tumors and trauma was similar compared to the patient cohort of Li et al. (37.12 vs. 36.69% and 4.51 vs. 4.64% respectively). The mean age at first procedure was 66.1 (IQR 60–75) years in our cohort which is slightly higher than stated in other publications.
The most important side effects of radiation exposure are tissue injuries, and stochastic effects, like cancer. The risk of tissue injuries is well documented in the literature [4, 24, 25]. However, stochastic effects in FGI were not addressed until lately [18, 26, 27]. Other studies and study as well indicate that the risk of stochastic effects is not negligible. The risk of stochastic effects increases at effective dose of > 100 mSv [28]. However, even below a CED of100 mSv, stochastic effects may occur, but there is a higher degree of certainty [8, 9]. A recent paper shows that there is growing evidence of development of cancers in patients receiving CED < 100 mSv over a protracted period of time [29]. Since the number of FGIs and the number of patients undergoing more than 1 procedure within a few years or during lifetime is steadily increasing worldwide, operators have to be aware that in complex endovascular procedures or obese patients a critical effective dose level of more than 100 mSv may be reached in a considerable number of patients [18]. In our cohort of patients with CED > 100 mSv, patients underwent an average of 2.72 procedures which indicates that a significant number of patients will be exposed to radiation in the same part of the body during multiple FGI sessions and imaging procedures. In our cohort, we observed a CED ≥ 100 mSv mainly in patients suffering from life threatening diseases (malignancies, hemorrhage, neurovascular disorders, portal hypertension) and an average age above 65 years. Unlike in other study cohorts, we did not encounter a CED ≥ 100 mSv in patients with an age below 18 years. Radiation exposures above 100 mSv is especially critical in children and young adults. Thus, in children and adolescents the risk of long term effects of radiation has to be carefully weighed against the risk of the disease to be treated.
Another important finding in our study was that a rather large number of patients received a CED ≥ 100 mSv within a rather short period of time (40.9% within 1 day, another 22% within 30 days).
The highest CED originated from thoracoabdominal embolization, EVAR and TIPS procedures. This is probably related to the complexity of procedures. Patients undergoing multiple chemoembolizations demonstrated a CED ≥ 100 mSv within 30–990 days. In chemoembolization, the main reason for higher CED is rarely related to the complexity of the procedure, but rather to the number of treatment sessions within a few months.
A limitation of this study is that patient’s weight and height or the body mass index was not considered for calculation of CED. This may result in considerable inaccuracies in calculations of CED. Furthermore, the use of monoplane or biplane systems will also affect these estimations of CED. Thus, in future studies body weight and type of angiographic imaging (mono- or biplane) should, therefore, be considered in calculations of CED.
Conclusions
In our study 3.3% of all patients treated by FGIs received a cumulative effective dose ≥ 100 mSv. The vast majority of patients were older than 65 years at the first FGI. 9.9% were below 50 years of age. We conclude, that high CED may occur in a small group of patients treated by FGI, but most patients will not encounter late stochastic effects of radiations since they suffer from life threatening diseases and are older than 65 years at the time of treatment.
References
Li X, Hirsch JA, Rehani MM, Ganguli S, Yang K, Liu B. Radiation effective dose above 100 mSv from fluoroscopically guided intervention: frequency and patient medical condition. AJR Am J Roentgenol. 2020;215(2):433–40. https://doi.org/10.2214/AJR.19.22227.
Miller DL, Balter S, Cole PE, Lu HT, Berenstein A, Albert R, et al. Radiation doses in interventional radiology procedures: the RAD-IR study: part II: skin dose. J Vasc Interv Radiol. 2003;14(8):977–90. https://doi.org/10.1097/01.rvi.0000084601.43811.cb.
Miller DL, Balter S, Cole PE, Lu HT, Schueler BA, Geisinger M, et al. Radiation doses in interventional radiology procedures: the RAD-IR study: part I: overall measures of dose. J Vasc Interv Radiol. 2003;14(6):711–27. https://doi.org/10.1097/01.rvi.0000079980.80153.4b.
Rehani MM, Srimahachota S. Skin injuries in interventional procedures. Radiat Prot Dosimetry. 2011;147(1–2):8–12. https://doi.org/10.1093/rpd/ncr257.
Rosenkrantz AB, Friedberg EB, Prologo JD, Everett C, Duszak R Jr. Generalist versus subspecialist workforce characteristics of invasive procedures performed by radiologists. Radiology. 2018;289(1):140–7. https://doi.org/10.1148/radiol.2018180761.
Pitton MB, Kloeckner R, Schneider J, Ruckes C, Bersch A, Duber C. Radiation exposure in vascular angiographic procedures. J Vasc Interv Radiol. 2012;23(11):1487–95. https://doi.org/10.1016/j.jvir.2012.05.048.
Alexander MD, Oliff MC, Olorunsola OG, Brus-Ramer M, Nickoloff EL, Meyers PM. Patient radiation exposure during diagnostic and therapeutic interventional neuroradiology procedures. J Neurointerv Surg. 2010;2(1):6–10. https://doi.org/10.1136/jnis.2009.000802.
United Nations Scientific Committee on the Effects of Atomic Radiation (2018) Sources, effects and risks of ionizing radiation UNSCEAR 2017 Report New York, NY: UNSCEAR. https://www.unscear.org/unscear/uploads/documents/publications/UNSCEAR_2017_Annex-B.pdf Published March 2018.
National Council of Radiation Protection and Measurements (2018) NCRP Commentary No. 27: Implications of recent epidemiologic studies for the linear-non threshold model and radiation protection. Bethesda, Maryland. Accessed August 7, 2018
D’Ercole L, Thyrion FZ, Bocchiola M, Mantovani L, Klersy C. Proposed local diagnostic reference levels in angiography and interventional neuroradiology and a preliminary analysis according to the complexity of the procedures. Phys Med. 2012;28(1):61–70. https://doi.org/10.1016/j.ejmp.2010.10.008.
Heilmaier C, Zuber N, Berthold C, Kara L, Weishaupt D. Establishing local diagnostic reference levels in IR procedures with dose management software. J Vasc Interv Radiol. 2017;28(3):429–41. https://doi.org/10.1016/j.jvir.2016.10.006.
Miller DL, Kwon D, Bonavia GH. Reference levels for patient radiation doses in interventional radiology: proposed initial values for U.S. practice. Radiology. 2009;253(3):753–64. https://doi.org/10.1148/radiol.2533090354.
Ruiz-Cruces R, Vano E, Carrera-Magarino F, Moreno-Rodriguez F, Soler-Cantos MM, Canis-Lopez M, et al. Diagnostic reference levels and complexity indices in interventional radiology: a national programme. Eur Radiol. 2016;26(12):4268–76. https://doi.org/10.1007/s00330-016-4334-2.
Schegerer AA, Frija G, Paulo G, Jaschke W, Tsapaki V, Repussard J, et al. Radiation dose and diagnostic reference levels for four interventional radiology procedures: results of the prospective European multicenter survey EUCLID. Eur Radiol. 2021;31(12):9346–60. https://doi.org/10.1007/s00330-021-08029-y.
Tsapaki V, Ahmed NA, AlSuwaidi JS, Beganovic A, Benider A, BenOmrane L, et al. Radiation exposure to patients during interventional procedures in 20 countries: initial IAEA project results. AJR Am J Roentgenol. 2009;193(2):559–69. https://doi.org/10.2214/AJR.08.2115.
Tuthill E, O’Hora L, O’Donohoe M, Panci S, Gilligan P, Campion D, et al. Investigation of reference levels and radiation dose associated with abdominal EVAR (endovascular aneurysm repair) procedures across several European Centres. Eur Radiol. 2017;27(11):4846–56. https://doi.org/10.1007/s00330-017-4791-2.
Vano E, Miller DL, Martin CJ, Rehani MM, Kang K, Rosenstein M, et al. ICRP Publication 135: diagnostic reference levels in medical imaging. Ann ICRP. 2017;46(1):1–144. https://doi.org/10.1177/0146645317717209.
Brower C, Rehani MM. Radiation risk issues in recurrent imaging. Br J Radiol. 2021;94(1126):20210389. https://doi.org/10.1259/bjr.20210389.
Li X, Hirsch JA, Rehani MM, Yang K, Marschall TA, Liu B. Radiation exposure in 101 non-coronary fluoroscopically guided interventional procedures: reference levels of air kerma at the reference point and air kerma area product. Br J Radiol. 2022;95(1130):20211108. https://doi.org/10.1259/bjr.20211108.
Liu B, Hirsch JA, Li X, Sheridan RM, Rehani MM, Zheng H, et al. Radiation dose monitoring for fluoroscopically guided interventional procedures: effect on patient radiation exposure. Radiology. 2019;290(3):744–9. https://doi.org/10.1148/radiol.2019180799.
Loose RW, Vano E, Mildenberger P, Tsapaki V, Caramella D, Sjoberg J, et al. Radiation dose management systems-requirements and recommendations for users from the ESR EuroSafe Imaging initiative. Eur Radiol. 2021;31(4):2106–14. https://doi.org/10.1007/s00330-020-07290-x.
Li X, Hirsch JA, Rehani MM, Yang K, Liu B. Effective dose assessment for patients undergoing contemporary fluoroscopically guided interventional procedures. AJR Am J Roentgenol. 2020;214(1):158–70. https://doi.org/10.2214/AJR.19.21804.
National Council on Radiation Protection and Measurements. Ionizing radiation exposure of the population of the United States. Bethesda, MD: National Council on Radiation Protection and Measurements, 2009: Report 160
Jaschke W, Bartal G, Martin CJ, Vano E. Unintended and accidental exposures, significant dose events and trigger levels in interventional radiology. Cardiovasc Intervent Radiol. 2020;43(8):1114–21. https://doi.org/10.1007/s00270-020-02517-2.
Sanchez RM, Vano E, Fernandez JM, Rosati S, Lopez-Ibor L. Radiation doses in patient eye lenses during interventional neuroradiology procedures. AJNR Am J Neuroradiol. 2016;37(3):402–7. https://doi.org/10.3174/ajnr.A4549.
Cousins C, Miller DL, Bernardi G, Rehani MM, Schofield P, Vano E, et al. ICRP PUBLICATION 120: radiological protection in cardiology. Ann ICRP. 2013;42(1):1–125. https://doi.org/10.1016/j.icrp.2012.09.001.
Rehani MM, Miller DL, Baliyan V. High-dose fluoroscopically guided procedures in patients: radiation management recommendations for interventionalists. Cardiovasc Intervent Radiol. 2021;44(6):849–56. https://doi.org/10.1007/s00270-020-02703-2.
Zewde N, Ria F, Rehani MM. Organ doses and cancer risk assessment in patients exposed to high doses from recurrent CT exams. Eur J Radiol. 2022;149: 110224. https://doi.org/10.1016/j.ejrad.2022.110224.
Ruhm W, Laurier D, Wakeford R. Cancer risk following low doses of ionising radiation—current epidemiological evidence and implications for radiological protection. Mutat Res Genet Toxicol Environ Mutagen. 2022;873: 503436. https://doi.org/10.1016/j.mrgentox.2021.503436.
Funding
This study was not supported by any funding. The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for Publication
For this type of study consent for publication is not required.
Informed Consent
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gizewski, E.R., Verius, M., Rehani, M.M. et al. Cumulative Effective Dose During Fluoroscopically Guided Interventions (FGI): Analysis of More Than 5000 FGIs in a Single European Center. Cardiovasc Intervent Radiol 47, 101–108 (2024). https://doi.org/10.1007/s00270-023-03604-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-023-03604-w