Abstract
Background
Delayed gastric emptying (DGE) is a frequent complication after pancreaticoduodenectomy (PD). The diagnosis of DGE is based on International Study Group for Pancreatic Surgery (ISGPS) clinical criteria and objective assessments of DGE are infrequently used. The present literature review aimed to identify objective measures of DGE following PD and determine whether these measures correlate with the clinical definition of DGE.
Methods
A systematic search was performed using the MEDLINE Ovid, EMBASE, Google Scholar and CINAHL databases for studies including pancreatic surgery, delayed gastric emptying and gastric motility until June 2022. The primary outcome was modalities undertaken for the objective measurement of DGE following PD and correlation between objective measurements and clinical diagnosis of DGE. Relevant risk of bias analysis was performed.
Results
The search revealed 4881 records, of which 46 studies were included in the final analysis. There were four objective modalities of DGE assessment including gastric scintigraphy (n = 28), acetaminophen/paracetamol absorption test (n = 10), fluoroscopy (n = 6) and the 13C-acetate breath test (n = 3). Protocols were inconsistent, and reported correlations between clinical and objective measures of DGE were variable; however, amongst these measures, at least one study directly or indirectly inferred a correlation, with the greatest evidence accumulated for gastric scintigraphy.
Conclusion
Several objective modalities to assess DGE following PD have been identified and evaluated, however are infrequently used. Substantial variability exists in the literature regarding indications and interpretation of these tests, and there is a need for a real-time objective modality which correlates with ISGPS DGE definition after PD.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Delayed gastric emptying (DGE) is one of the most common complications following pancreaticoduodenectomy (PD), with postoperative DGE rates ranging between 10 and 45% [1, 2]. DGE can significantly increase postoperative morbidity, prolong hospital stay and increase healthcare costs [3, 4].
Historically, there have been several definitions of DGE, with studies using different definitions leading to significant challenges in interpreting findings across studies. In 2007, the International Study Group for Pancreatic Surgery (ISGPS) consensus statement standardized the definition of DGE [4]. While allowing for a standardized measure of DGE, this definition is reliant on subjective clinical judgement based on the duration of nasogastric (NG) tube intubation and reinsertion. The DGE grade can also only be established at the end of the patient's clinical course. Nevertheless, there have been several publications validating the ISGPS definition of DGE [1, 5, 6]. Furthermore, DGE can be classified into those relating to the surgical procedure itself (primary DGE) or to postoperative complications, e.g., pancreatic fistulas, hemorrhages or intra-abdominal abscess (secondary DGE) [7]. A more objective measure of DGE after PD may allow a real-time and impartial assessment to guide clinical management and develop strategies to prevent or treat DGE.
The aim of this systematic review was to identify the objective assessment modalities of DGE used in the literature following PD. This study also aimed to identify correlations between current clinical definitions of DGE and objective DGE assessments, along with correlations between postoperative symptoms and the objective assessment of DGE.
Materials and methods
This systematic review was completed in accordance with the PRISMA 2020 statement [8] and was prospectively registered with PROSPERO (ID: CRD42021260141).
Literature search
A systematic literature review of MEDLINE (OVID) (1946-June 2022), EMBASE (1980-June 2022), Google Scholar and CINAHL (1982-June 2022) databases was performed in June 2022.
In brief, the search was conducted using the following Medical Subject Heading (MeSH) terms and text words: “pancreaticoduodenectomy”, “pancreatectomy”, “Whipples” AND “gastroparesis”, “postgastrectomy syndrome”, “gastric emptying”, “delayed gastric emptying”, “DGE”, “gastrointestinal motility”, “gastrointestinal transit”, limiting to human studies in English. Reference lists of relevant records were also manually searched for additional eligible publications.
Inclusion and exclusion criteria
This literature search included studies involving pancreaticoduodenectomy (classical, pylorus-preserving or other variations) and excluded other forms of pancreatic resections such as distal, total and central pancreatectomy. The search only included studies that assessed DGE using a non-clinical and objective measure. Meta-analyses, review articles, case reports (with n ≤ 5), letters to the editors, conference proceedings and abstracts were excluded.
Data extraction
Two independent reviewers (THHW, AL) screened and assessed each article for inclusion and extracted data. A title and abstract screen were first performed, followed by a full-text review. Discrepancies were resolved by the senior author (SP). Data on the type of objective measure of DGE, how it was performed, whether there was any correlation between clinical and objective measures of DGE or between symptoms (not otherwise included in the clinical DGE definitions) and the objective measure of DGE were extracted. Data on routine exclusion of mechanical obstruction at the gastrojejunal anastomosis, whether primary and secondary DGE was clearly differentiated and whether the objective DGE results altered management were also extracted. Three risk of bias tools were used, including the revised Cochrane risk-of-bias tool for randomized trials (ROB 2), the Newcastle–Ottawa Scale (NOS) for cohort studies, and the Methodological Index for Non-Randomized Studies (MINORS) for case–control and case series [9,10,11].
Results
Included studies
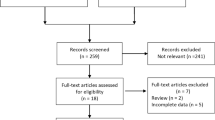
In total, 4881 articles were identified from the initial search, of which 46 articles met the inclusion criteria and formed the basis of the systematic review (PRISMA diagram shown in Fig. 1). There were 7 randomized control trials (RCT), 16 cohort studies, 8 case–control studies and 15 case series. These studies encompassed 4 different objective measures of DGE: 28 studies used gastric scintigraphy (Table 1), 10 studies used acetaminophen/paracetamol absorption test, 6 studies used fluoroscopy, and 3 studies used 13C-acetate breath test (Table 2). One study used both the acetaminophen/paracetamol absorption and fluoroscopy in the early and late postoperative phase, respectively [12]. Only one study specified the routine exclusion of mechanical obstruction as a cause of DGE [13], though several studies performed endoscopy or barium radiography but not specifically to assess for mechanical obstruction. No study clearly specified a subgroup analysis on primary or secondary DGE, however, several studies compared the rates of postoperative complications in the DGE and non-DGE cohorts [14,15,16,17,18,19,20]. No study used the results of the objective measure of DGE to alter patient management. Relevant results are presented in Tables 1 and 2.
Gastric scintigraphy
28 studies used gastric scintigraphy to diagnose DGE following PD [13,14,15, 21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45], involving serial imaging to track the transit of isotopes ingested with a meal. Heterogeneous protocols were identified, including the use of different isotopes (99mTc or 111mIn), test meals, serial imaging time intervals and definitions of DGE. Several studies also differentiated between liquid and solid phase gastric emptying [13, 23, 24, 27,28,29, 35, 40,41,42]. Additionally, one study used 170Er-labelled enteric-coated pancreatin microspheres along with 99mTc to assess gastric emptying [31]. More recent studies used the standardized technique of gastric scintigraphy based on consensus definition, using a 99mTc-labelled scrambled egg meal to assess solid gastric emptying, followed by serial imaging with a gamma camera at 1, 2 and 4 h following meal ingestion [58]. Residual gastric activity greater than 60% at 2 h was considered DGE [43, 44].
Acetaminophen/paracetamol absorption test
10 studies used the acetaminophen (also known as paracetamol) absorption test to define DGE following PD. This technique involves ingesting a standard dose of acetaminophen/paracetamol with regular serum acetaminophen/paracetamol concentration monitoring in the subsequent hours [59]. Any elevation in serum concentration indicates the passage of the ‘meal’ out of the stomach, indirectly assessing gastric emptying [53, 60]. Variable dosing was found between studies. Only one study by Strommer et al. [51] defined a numerical threshold for DGE, assessing maximal plasma concentration (< 25 μM) and time to reach this value (> 240 min). No other studies provided a quantitative definition for DGE, rather, they compared the results to different groups within their respective studies or to preoperative results. Additionally, one study used the acetaminophen/paracetamol absorption test for the liquid phase and sulphamethizole capsule for the solid phase [48].
Fluoroscopy
6 studies used fluoroscopy to define DGE following PD. Following the ingestion of barium or Gastrografin® (sodium amidotrizoate/amidotrizoate meglumine) contrast, serial radiographs were taken to determine the location and amount of contrast to assess gastric retention, emptying and therefore function. The 3 case series identified did not provide a fluoroscopic definition for DGE [17, 54, 55] while 2 studies defined DGE if contrast was present in the stomach after a defined time interval (1 and 4 h, respectively) [12, 18]. Furthermore, Nojiri et al. [19] described a classification system dividing gastric emptying into three grades depending on gastric distension and stasis appearances on fluoroscopic imaging.
13C-acetate breath test
3 studies used the 13C-acetate breath test to define DGE following PD. Following ingestion of a 13C-labelled triglyceride meal, serial breath samples were obtained to determine the concentration of exhaled 13CO2 or other metabolites using spectrometry, and the time to peak 13CO2 was determined. No quantitative definition for DGE was used, rather, results were compared between different groups within the studies or to preoperative results. All studies were performed preoperatively and at least 1 month after surgery, but not immediately after surgery [61, 62].
Correlation between clinical and objective DGE
All objective measures of DGE identified in this review had implied or explicit correlations between clinical and objective measures of DGE. These results are summarized in Table 3. Results were too heterogeneous to allow meta-analysis.
8 studies correlated clinical DGE with gastric scintigraphy. Of these, 2 studies by Patti et al. and Hunt and Maclean, found no correlation between clinical DGE and scintigraphy [22, 23]. One study by Shan et al. [41] compared rates of clinical (subjective) DGE (‘sDGE’) and objective DGE (‘oDGE’). In the pylorus-preserving PD group, 42% had sDGE and 42% had oDGE, while in the pylorus-resecting PD group, 15% had sDGE and 88% had oDGE. However, no conclusion was drawn on the correlation between the clinical and objective DGE. Eshuis et al. [44] found a strong association between scintigraphy results and clinically relevant DGE (grade B or C) and concluded that gastric scintigraphy performed on day 7 following surgery predicted the severity of clinical DGE. Similarly, van Samkar et al. [43] found that scintigraphy performed at day 10 and day 21 had 100% positive predictive value and 100% specificity for clinical DGE.
For the acetaminophen/paracetamol group, 2 studies implied a correlation between the clinical and objective measure of DGE. Although no statistical analysis was performed, both studies found that patients with low serum acetaminophen/paracetamol also had concurrent clinical DGE, suggesting correlation [16, 51].
Krishna et al. [18] was the only study using fluoroscopy which implied an association with clinical DGE, with all 3 patients requiring reinsertion of NG tube demonstrating prolonged gastric Gastrografin® retention.
Only 1 study by Chijiiwa et al. [56] implied a correlation between clinical DGE and the 13C-acetate breath test. No statistical difference in both the incidence of clinical and objective DGE were found in the subgroups.
Correlations between symptoms and objective DGE
3 objective measures of DGE had implied or explicit correlations between symptoms (including abdominal pain, early satiety, nausea and vomiting and/or loss of appetite) and objective DGE, with 1 study each (Table 4). No studies were identified for fluoroscopy.
For gastric scintigraphy, Pastorino et al. [30] demonstrated a reduced gastric emptying time (49.3 min compared to 82.3 min) with patients who had better clinical outcomes (score 1 and 2 according to the questionnaire used) compared to those with a poorer clinical outcome.
For the acetaminophen/paracetamol absorption test, Takeda et al. [49] found that the improvement of test results coincided with the recovery of symptoms. Specifically, the area under the curve at 90 min following administration was 48.1% at 1 month postoperatively which returned to preoperative baseline values at 6 months, coinciding with the improvement in symptoms.
For 13C-acetate breath test, only 1 study by Kawai et al. [20] demonstrated symptoms in the immediate postoperative period was associated with DGE on the 13C-acetate breath test months after the surgery, suggesting association.
Discussion
Delayed gastric emptying is a common complication following PD and is associated with increased morbidity, prolonged hospitalization and increased costs to healthcare [3, 4]. While the ISGPS definition for DGE is the most widely used, it relies on subjective clinical judgement and only diagnoses DGE at the end of the clinical course. This literature review identified 4 objective modalities to assess the presence of DGE following PD. These include gastric scintigraphy, acetaminophen/paracetamol absorption test, fluoroscopy and 13C-acetate breath test. All modalities had at least 1 study that explicitly or implied a correlation with clinical DGE definition.
Historically, DGE assessments and definitions have been heterogeneous, making meaningful comparisons between different studies difficult. However, in 2007, the ISGPS developed a consensus definition for DGE following pancreatic surgery [4]. This definition classified the severity of DGE (A, B or C) based on the duration of NG intubation or reinsertion, with the condition of no underlying mechanical obstruction to cause symptoms. Only 1 study in this review clearly included this condition in their methodology [13]. Moreover, DGE can be further subclassed into primary or secondary, dependent on the presumed cause being attributed to the surgical procedure or postoperative complications, respectively [7]. While secondary DGE is expected to resolve following the treatment of postoperative complications, this is not necessarily true for primary DGE, and is therefore the focus of this review.
Several studies have now validated the ISGPS definition. These studies have found statistically significant differences in postoperative clinical outcomes, including further diagnostic evaluations (such as endoscopy or imaging), treatment, parenteral nutrition, ICU admission duration and overall hospitalization duration between the different DGE severities (including those with no DGE) [1, 5, 6]. Since the consensus statement, almost all studies examined in this literature review, including those exclusively using the clinical definition, used the ISGPS definition.
There are several advantages to the clinical ISGPS definition of DGE. In particular, it is non-invasive and requires minimal cost to the patient or health system. It allows a standardized definition for audit and research purposes and the development of risk stratification tools for DGE following PD [63]. However, there are some disadvantages. Firstly, this definition relies on clinician judgement on whether to maintain or re-insert the NG tube. This decision is based on symptoms (e.g., food intolerance, nausea, vomiting), which, albeit pragmatic, is an experience-based assessment of the patient’s underlying gastric physiological status. Secondly, medications, such as anti-emetics or prokinetics, may also influence patient symptoms and therefore clinician judgement [46]. Finally, the ISGPS definition is a retrospective assessment and while it is useful for audits and research, it does not allow a real-time diagnosis of DGE and thereby does not aid in guiding immediate inpatient management or predicting recovery. As such, several authors have proposed that a real-time and objective measure of DGE may provide a more accurate way of assessing the patient’s true gastric physiology, to potentially allow future research into this area to become more standardized and therefore guide postoperative management, such as decision for early parenteral nutrition support if stomach recovery is expected to be prolonged or other novel therapies [15, 39, 41, 43, 64]. Indeed, no studies identified in this review used the results of the objective assessment of DGE to guide inpatient management, thus would be a focus for future research. An ideal test would not only exclude mechanical obstruction but also assesses gastric motility, and there is currently a lack of such testing modality.
This literature review has identified several non-clinical or objective measures to assess and define DGE with the most common technique being gastric scintigraphy. First described by Griffith et al. [65] in 1966, this technique is still considered the standard for objectively assessing gastric emptying [58, 66], with a 2008 consensus statement developed by Abell et al. [58] to standardize protocols. Limitation to this measure include its relative cost, access to equipment, impracticality to apply in the immediate postoperative setting and concerns on the use of radioactive isotopes [46, 50]. Moreover, it may also not be feasible to administer the test meal in patients experiencing severe nausea or vomiting. Eight studies assessed the presence of associations between clinical DGE and gastric scintigraphy. Three studies, all published prior to 2006, either found no correlation between clinical DGE and gastric scintigraphy or did not conclude a correlation between the two measures. In contrast, the more recent studies have all found or implied a correlation between clinical DGE definitions and gastric scintigraphy [15, 40,41,42,43,44], which may be due to the standardization of DGE assessment protocols and definitions.
The other non-clinical or objective measures of DGE identified in the literature review all have advantages and disadvantages. In the acetaminophen/paracetamol absorption test, the advantages include its relative accessibility, the benefits of a bedside test, that it does not involve the nuclear medicine department and avoids radiation. However, it does require serial blood tests posing risks and requiring intensive input by both clinicians and laboratory. It is also not a direct measure of gastric emptying, albeit being correlated with clinical DGE following PD [58]. In the 13C-acetate breath test, the main advantage is its non-invasive nature. However, there are concerns of unreliable results following pancreatic surgery due to the possibility of altered physiology and intestinal absorption, thereby affecting test accuracy and reliability [67]. For fluoroscopy, the main advantages are that this technique is well-established in other fields of medicine, is readily available and allows a real-time assessment of gastric function. Limitations include its subjective nature, difficulty in quantifying results and radiation exposure.
While the aforementioned objective measures of gastric emptying are valuable measures of gastric function, they all represent indirect functional measures of gastric transit in contributing to the patient’s symptoms. Rather, it may be more useful to assess the direct physiological status of gastric motility after pancreatoduodenectomy, particularly in patients with primary DGE to inform targeted treatments. By understanding the underlying pathophysiology of these patients, rather than functional status, clinicians may potentially be able to predict, assess and even aim to treat DGE with novel strategies, such as gastric pacing or ablation, which are currently under research [68]. Numerous studies have investigated gastric physiology or function by assessing either the peristaltic or electrophysiological activity of the stomach, termed gastric slow waves [69, 70]. Non-surgical and post-surgical gastric dysfunctions have been associated with gastric slow wave abnormalities [71,72,73]. Recent bioengineering developments now allow for more accurate quantification of these gastric slow waves to be possible, particularly with the recent development of non-invasive high-resolution electrogastrography devices to assess gastric electrical activity [68]. This validated technique has been correlated with patient symptom severity in other conditions, albeit never in PD patients [74]. Further research is now required to determine whether these novel techniques may better assess the pathophysiology of DGE post-PD [35, 75].
The strength of this review was the broad search strategy, which allowed a large number of studies to be included in this literature search. The main limitation of this review was the relatively few studies for each objective technique with heterogeneous protocols (including different interventions and time points after surgery), meaning that statistical comparisons between various techniques (e.g. network meta-analysis) could not be performed. A further limitation was the lack of high-quality studies and studies directly comparing objective assessment of DGE with clinical DGE or symptoms. This review now provides the foundations for future research into this area of clinical assessment and the development of an objective clinical tool to more accurately assess DGE following PD.
Conclusion
This literature review identified several techniques which objectively assess gastric function following surgery, with the most common being gastric scintigraphy. There is currently no consensus on the preferred objective measure of delayed gastric emptying following pancreatic surgery. Therefore, a consensus may be useful in defining or developing a more objective and standardized measure of delayed gastric emptying following pancreaticoduodenectomy.
References
Welsch T, Borm M, Degrate L et al (2010) Evaluation of the International Study Group of Pancreatic Surgery definition of delayed gastric emptying after pancreatoduodenectomy in a high-volume centre. BJS 97:1043–1050
Varghese C, Bhat S, Wang TH et al (2021) Impact of gastric resection and enteric anastomotic configuration on delayed gastric emptying after pancreaticoduodenectomy: a network meta-analysis of randomized trials. BJS Open 5(3):zrab035
Francken MFG, van Roessel S, Swijnenburg RJ et al (2021) Hospital costs of delayed gastric emptying following pancreatoduodenectomy and the financial headroom for novel prophylactic treatment strategies. HPB 23:1865–1872
Wente MN, Bassi C, Dervenis C et al (2007) Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 142:761–768
Malleo G, Crippa S, Butturini G et al (2010) Delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy: validation of International Study Group of Pancreatic Surgery classification and analysis of risk factors. HPB 12:610–618
Park JS, Hwang HK, Kim JK et al (2009) Clinical validation and risk factors for delayed gastric emptying based on the International Study Group of Pancreatic Surgery (ISGPS) Classification. Surgery 146:882–887
Courvoisier T, Donatini G, Faure JP et al (2015) Primary versus secondary delayed gastric emptying (DGE) grades B and C of the International Study Group of Pancreatic Surgery after pancreatoduodenectomy: a retrospective analysis on a group of 132 patients. Updates Surg 67:305–309
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71
Sterne JAC, Savovic J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Wells GA, Shea B, O’Connell D et al (2014) T Newcastle-Ottawa quality assessment scale cohort studies. University of Ottawa
Slim K, Nini E, Forestier D et al (2003) Methodological index for non-randomized studies (MINORS): development and validation of a new instrument. ANZ J Surg 73:712–716
Watanabe Y, Tsumura H, Sakurai H et al (1992) Gastric emptying after pancreatoduodenectomy with total stomach preservation and selective proximal vagotomy. Surg Today 22:426–431
Fink AS, DeSouza LR, Mayer EA et al (1988) Long-term evaluation of pylorus preservation during pancreaticoduodenectomy. World J Surg 12:663–670. https://doi.org/10.1007/BF01655880
Kollmar O, Moussavian MR, Richter S et al (2008) Prophylactic octreotide and delayed gastric emptying after pancreaticoduodenectomy: results of a prospective randomized double-blinded placebo-controlled trial. Eur J Surg Oncol 34:868–875
Samaddar A, Kaman L, Dahiya D et al (2017) Objective assessment of delayed gastric emptying using gastric scintigraphy in post pancreaticoduodenectomy patients. ANZ J Surg 87:E80–E84
Tamandl D, Sahora K, Prucker J et al (2014) Impact of the reconstruction method on delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy: a prospective randomized study. World J Surg 38:465–475. https://doi.org/10.1007/s00268-013-2274-4
Abdel-Wahab M, Sultan A, ElGwalby N et al (2001) Modified pancreaticoduodenectomy: experience with 81 cases, Wahab modification. Hepatogastroenterology 48:1572–1576
Krishna A, Bansal VK, Kumar S et al (2015) Preventing delayed gastric emptying after Whipple’s procedure—isolated Roux loop reconstruction with pancreaticogastrostomy. Indian J Surg 77:703–707
Nojiri M, Yokoyama Y, Maeda T et al (2018) Impact of the gastrojejunal anatomic position as the mechanism of delayed gastric emptying after pancreatoduodenectomy. Surgery 163:1063–1070
Kawai M, Tani M, Hirono S et al (2014) Pylorus-resecting pancreaticoduodenectomy offers long-term outcomes similar to those of pylorus-preserving pancreaticoduodenectomy: results of a prospective study. World J Surg 38:1476–1483. https://doi.org/10.1007/s00268-013-2420-z
Braasch JW, Deziel DJ, Rossi RL (1986) Pyloric and gastric preserving pancreatic resection. Experience with 87 patients. Ann Surg 204:411–418
Patti MG, Pellegrini CA, Way LW (1987) Gastric emptying and small bowel transit of solid food after pylorus-preserving pancreaticoduodenectomy. Arch Surg 122:528–532
Hunt DR, McLean R (1989) Pylorus-preserving pancreatectomy: functional results. BJS 76:173–176
Lerut J, Luder PJ, Krahenbuhl L et al (1991) Pylorus-preserving pancreatoduodenectomy. Experience in 20 patients. HPB Surg 4:109–119
De Bernardinis G, Agnifili A, Gola P et al (1993) An original reconstructive method after pylorus-preserving pancreatoduodenectomy. Surg Today 23:481–485
Kingsnorth AN, Gray MR, Berg JD (1993) A novel reconstructive technique for pylorus-preserving pancreaticoduodenectomy: avoidance of early postoperative gastric stasis. Ann R Coll Surg Engl 75:38–42
Williamson RCN, Bliouras N, Cooper MJ et al (1993) Gastric emptying and enterogastric reflux after conservative and conventional pancreatoduodenectomy. Surgery 114:82–86
Yeo CJ, Barry MK, Sauter PK et al (1993) Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. A prospective, randomized, placebo-controlled trial. Ann Surg 218:229–237
Yung BC, Sostre S, Yeo CJ et al (1993) Comparison of left anterior oblique, anterior, and geometric mean methods in gastric emptying assessment of postpancreaticoduodenectomy patients. Clin Nucl Med 18:776–781
Pastorino G, Ermili F, Zappatore F et al (1995) Multiparametric evaluation of functional outcome after pylorus-preserving duodenopancreatectomy. Hepatogastroenterology 42:62–67
Bruno MJ, Borm JJJ, Hoek FJ et al (1997) Comparative effects of enteric-coated pancreatin microsphere therapy after conventional and pylorus-preserving pancreatoduodenectomy. BJS 84:952–956
Lupo LG, Pannarale OC, Altomare DF et al (1998) Is pyloric function preserved in pylorus-preserving pancreaticoduodenectomy? Eur J Surg 164:127–132
Hishinuma S, Ogata Y, Matsui J et al (1999) Evaluation of pylorus-preserving pancreatoduodenectomy with the Imanaga reconstruction by hepatobiliary and gastrointestinal dual scintigraphy. BJS 86:1306–1311
Sumida K, Nimura Y, Yasui A et al (1999) Influence of vagal pyloric branches on gastric acid secretion and gastrointestinal motility in patients following a pylorus preserving pancreatoduodenectomy. Hepatogastroenterology 46:336–342
Thor P, Matyja A, Popiela T et al (1999) Early effects of standard and pylorus-preserving pancreatectomy on myoelectric activity and gastric emptying. Hepatogastroenterology 46:1963–1967
Sato T, Konishi K, Yabushita K et al (2000) A new examination for both biliary and gastrointestinal function after pancreatobiliary surgery—single-isotope two-day method. Hepatogastroenterology 47:140–142
Sato T, Konishi K, Yabushita K et al (2000) Long-term postoperative functional evaluation of pylorus preservation in Imanaga pancreatoduodenectomy. Dig Dis Sci 45:1907–1912
Caronna R, Cardi M, Sammartino P et al (2003) Functional results of a personal technique of reconstruction after pancreaticoduodenectomy. J Exp Clin Cancer Res 22:187–189
Kim DK, Hindenburg AA, Sharma SK et al (2005) Is pylorospasm a cause of delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy? Ann Surg Oncol 12:222–227
Shan YS, Sy ED, Tsai ML et al (2005) Effects of somatostatin prophylaxis after pylorus-preserving pancreaticoduodenectomy: increased delayed gastric emptying and reduced plasma motilin. World J Surg 29:1319–1324. https://doi.org/10.1007/s00268-005-7943-5
Shan YS, Tsai ML, Chiu NT et al (2005) Reconsideration of delayed gastric emptying in pancreaticoduodenectomy. World J Surg 29:873–879. https://doi.org/10.1007/s00268-005-7473-1
Shan YS, Hsieh YH, Yao WJ et al (2007) Impaired emptying of the retained distal stomach causes delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy. World J Surg 31:1606–1615. https://doi.org/10.1007/s00268-007-9100-9
van Samkar G, Eshuis WJ, Lemmers M et al (2013) Value of scintigraphy for assessing delayed gastric emptying after pancreatic surgery. World J Surg 37:2911–2917. https://doi.org/10.1007/s00268-013-2219-y
Eshuis WJ, De Bree K, Sprangers MAG et al (2015) Gastric emptying and quality of life after pancreatoduodenectomy with retrocolic or antecolic gastroenteric anastomosis. BJS 102:1123–1132
Shahbazov R, Naziruddin B, Yadav K et al (2018) Risk factors for early readmission after total pancreatectomy and islet auto transplantation. HPB 20:166–174
Ueno T, Tanaka A, Hamanaka Y et al (1995) A proposal mechanism of early delayed gastric emptying after pylorus preserving pancreatoduodenectomy. Hepatogastroenterology 42:269–274
Muller MW, Friess H, Beger HG et al (1997) Gastric emptying following pylorus-preserving Whipple and duodenum-preserving pancreatic head resection in patients with chronic pancreatitis. Am J Surg 173:257–263
Kobayashi I, Miyachi M, Kanai M et al (1998) Different gastric emptying of solid and liquid meals after pylorus-preserving pancreatoduodenectomy. BJS 85:927–930
Takeda T, Yoshida J, Tanaka M et al (1999) Delayed gastric emptying after Billroth I pylorus-preserving pancreatoduodenectomy: effect of postoperative time and cisapride. Ann Surg 229:223–229
Ohtsuka T, Takahata S, Ohuchida J et al (2002) Gastric phase 3 motility after pylorus-preserving pancreatoduodenectomy. Ann Surg 235:417–423
Strommer L, Raty S, Hennig R et al (2005) Delayed gastric emptying and intestinal hormones following pancreatoduodenectomy. Pancreatology 5:537–544
Ohuchida J, Chijiiwa K, Ohtsuka T et al (2007) Pylorus-preserving pancreatoduodenectomy: preoperative pancreatic function and outcome. Hepatogastroenterology 54:913–916
Harmuth S, Wewalka M, Holst JJ et al (2014) Distal gastrectomy in pancreaticoduodenectomy is associated with accelerated gastric emptying, enhanced postprandial release of GLP-1, and improved insulin sensitivity. J Gastrointest Surg 18:52–59
Traverso LW, Longmire WP Jr (1980) Preservation of the pylorus in pancreaticoduodenectomy a follow-up evaluation. Ann Surg 192:306–310
Krishna A, Bansal VK, Kumar S et al (2020) Pancreaticogastrostomy after Whipple’s surgery avoids pancreatic fistula—a large case series analysis. Indian J Surg 82:415–420
Chijiiwa K, Imamura N, Ohuchida J et al (2009) Prospective randomized controlled study of gastric emptying assessed by (13)C-acetate breath test after pylorus-preserving pancreaticoduodenectomy: comparison between antecolic and vertical retrocolic duodenojejunostomy. J Hepatobiliary Pancreat Surg 16:49–55
Hiyoshi M, Chijiiwa K, Ohuchida J et al (2012) Comparative study of gastric emptying and nutritional status after pylorus-preserving vs. subtotal stomach-preserving pancreaticoduodenectomy. Hepatogastroenterology 59:1018–1022
Abell TL, Camilleri M, Donohoe K et al (2008) Consensus recommendations for gastric emptying scintigraphy: a joint report of the American neurogastroenterology and motility society and the society of nuclear medicine. J Nucl Med Technol 36:44–54
Heading R, Nimmo J, Prescott L et al (1973) The dependence of paracetamol absorption on the rate of gastric emptying. Br J Pharmacol 47:415
Willems M, Quartero AO, Numans ME (2001) How useful is paracetamol absorption as a marker of gastric emptying? A systematic literature study. Dig Dis Sci 46:2256–2262
Braden B, Adams S, Duan L-P et al (1995) The [13C] acetate breath test accurately reflects gastric emptying of liquids in both liquid and semisolid test meals. Gastroenterology 108:1048–1055
Nakamura H, Morifuji M, Murakami Y et al (2009) Usefulness of a 13C-labeled mixed triglyceride breath test for assessing pancreatic exocrine function after pancreatic surgery. Surgery 145:168–175
Werba G, Sparks AD, Lin PP et al (2022) The PrEDICT-DGE score as a simple preoperative screening tool identifies patients at increased risk for delayed gastric emptying after pancreaticoduodenectomy. HPB 24:30–39
Reber HA (2007) Delayed gastric emptying—what should be required for diagnosis? Surgery 142:769–770
Griffith G, Owen G, Kirkman S et al (1966) Measurement of rate of gastric emptying using chromium-51. Lancet 1:1244–1245
Fried M (1994) Methods to study gastric emptying. Dig Dis Sci 39:114S-115S
Nguyen NQ, Bryant LK, Burgstad CM et al (2013) Gastric emptying measurement of liquid nutrients using the (13)C-octanoate breath test in critically ill patients: a comparison with scintigraphy. Intensive Care Med 39:1238–1246
Carson DA, O’Grady G, Du P et al (2021) Body surface mapping of the stomach: new directions for clinically evaluating gastric electrical activity. Neurogastroenterol Motil 33:e14048
Farrugia G (2008) Interstitial cells of Cajal in health and disease. Neurogastroenterol Motil 20:54–63
Cheng LK (2015) Slow wave conduction patterns in the stomach: from Waller’s foundations to current challenges. Acta Physiol 213:384–393
Angeli TR, Cheng LK, Du P et al (2015) Loss of interstitial cells of Cajal and patterns of gastric dysrhythmia in patients with chronic unexplained nausea and vomiting. Gastroenterology 149(56–66):e55
Wang TH, Angeli TR, Beban G et al (2019) Slow-wave coupling across a gastroduodenal anastomosis as a mechanism for postsurgical gastric dysfunction: evidence for a “gastrointestinal aberrant pathway.” Am J Physiol Gastrointest Liver Physiol 317:G141-g146
O’Grady G, Wang TH, Du P et al (2014) Recent progress in gastric arrhythmia: pathophysiology, clinical significance and future horizons. Clin Exp Pharmacol Physiol 41:854–862
Gharibans AA, Calder S, Varghese C et al (2022) Gastric dysfunction in patients with chronic nausea and vomiting syndromes defined by a novel non-invasive gastric mapping device. medRxiv
Dua MM, Navalgund A, Axelrod S et al (2018) Monitoring gastric myoelectric activity after pancreaticoduodenectomy for diet “readiness.” Am J Physiol Gastrointest Liver Physiol 315:G743–G751
Funding
T H.-H. Wang was supported by the Auckland Medical Research Foundation Douglas Goodfellow Medical Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
GOG is the director and shareholder of Alimetry. All authors agree that GOG’s conflict of interest has not impacted on the results of this study (GOG was not involved in the data collection nor data analyses). There are no other conflicts of interest.
Ethical approval
All authors comply with WJS’ ethical policies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, T.HH., Lin, A.Y., Mentor, K. et al. Delayed Gastric Emptying and Gastric Remnant Function After Pancreaticoduodenectomy: A Systematic Review of Objective Assessment Modalities. World J Surg 47, 236–259 (2023). https://doi.org/10.1007/s00268-022-06784-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-022-06784-7