Abstract
Background/Purpose
To examine whether vertical retrocolic duodenojejunostomy is superior to antecolic duodenojejunostomy with respect to gastric emptying in a prospective, randomized, controlled study of patients undergoing pylorus-preserving pancreaticoduodenectomy (PpPD).
Methods
Thirty-five patients undergoing PpPD between March 2005 and July 2007 were enrolled in the study. All provided informed consent. During PpPD, the patients were randomly assigned to either the antecolic (antecolic group, n = 17) or vertical retrocolic route (vertical retrocolic group, n = 18) just before the reconstruction. Each patient ingested 13C-acetate in a liquid meal before surgery and on postoperative day (POD) 30. Gastric emptying variables (Tmax, T1/2) were determined and compared between groups.
Results
Clinical delayed gastric emptying, defined as an inability of patients to take in an appropriate amount of solid food orally by POD 14, was found in 1 of 17 patients (6%) in the antecolic group and in 4 of 18 patients (22%) in the vertical retrocolic group, but the difference was not significant (P = 0.34). Tmax and T1/2 on POD 30 were prolonged in both groups in comparison to preoperative levels, but no significant difference was found between the two groups. Follow-up examinations revealed that gastric emptying had recovered to the preoperative level by POD 30 in approximately 80% of the patients, regardless of the reconstruction route.
Conclusions
Vertical retrocolic duodenojejunostomy does not seem to offer an advantage with respect to gastric emptying.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pylorus-preserving pancreaticoduodenectomy (PpPD) is generally accepted as a standard operation for periampullary lesions. PpPD, in comparison to classic pancreaticoduodenectomy with hemigastrectomy, is reported to improve quality of life, nutritional status and weight gain without any difference in operative morbidity and mortality or in postoperative survival [1–4].
Delayed gastric emptying (DGE), however, is reported to be the most common and frustrating complication after PpPD. Despite the lack of a certain definition for DGE, the reported incidence varies from 20 to 60% [5–13]. DGE results in a prolonged hospital stay, which adds to hospital costs. Although DGE itself is not a fatal complication, minimizing DGE is important in patients undergoing PpPD.
Two reconstruction routes are used for duodenojejunostomy, the antecolic route and the retrocolic route. The reported incidence of DGE is >30% for the retrocolic route [12, 14, 15], whereas that for the antecolic route is <15% [16, 17]. A recent prospective randomized controlled trial showed that DGE occurred in 50% of patients in whom the retrocolic route was used but in only 5% in whom the antecolic route was used [18]. These data suggest that the antecolic route is better. However, the 50% incidence of DGE associated with the retrocolic route seems high. We have shown that vertical retrocolic duodenojejunostomy, by which the stomach and duodenum are brought down through the left side of the transverse mesocolon in a straight, vertical manner, reduces the incidence of DGE [19].
Thus, a prospective randomized controlled trial was conducted to compare the incidence of clinical DGE and gastric emptying variables assessed by the 13C-acetate breath test between patients who underwent antecolic duodenojejunostomy and those who underwent vertical retrocolic duodenojejunostomy. The aim of the study was to establish the superiority of the vertical retrocolic route with respect to gastric emptying after PpPD.
Patients and methods
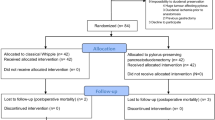
Of 50 patients underwent pancreaticoduodenectomy at Miyazaki University Hospital between March 2005 and July 2007, 46 patients were scheduled to undergo PpPD. Patients were recruited into the study before surgery, on the basis of whether PpPD was anticipated and informed consent was obtained. Specific exclusion criteria included tumor infiltration into the duodenal bulb or presence of lymph node metastasis of the prepylorus (n = 3), failure to provide informed consent including the 13C-acetate breath test (n = 4) were then excluded. Thus, 35 patients who underwent PpPD and consented to the protocol were enrolled in the study.
This prospective randomized controlled trial was approved by the ethical committee of our university hospital and informed consent was obtained from all patients. The randomization protocol involved assignment of patients to one of two reconstruction methods, the antecolic route and the vertical retrocolic route. Randomization took place during surgery before reconstruction. Gastric emptying was evaluated by means of the 13C-acetate breath test just before surgery and on postoperative day (POD) 30.
Operative technique
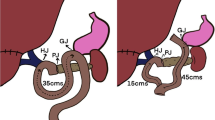
The area resected during PpPD included the gallbladder, common hepatic duct, head of the pancreas, duodenum (except for the first portion), and 10 cm of the proximal jejunum. A few arcades of the right gastric artery and right gastroepiploic artery to the stomach were divided along the wall of the antrum (approximately 2–3 cm from the pyloric ring) for dissection of the peripyloric lymph nodes. The duodenum was freed from the surrounding tissue and transected approximately 4–5 cm distal to the pyloric ring. The lymph nodes in the hepatoduodenal ligament, the para-aortic lymph nodes, and those along the common hepatic artery and the right side of the superior mesenteric artery were dissected. The right gastric artery was divided at its origin in all patients. The left gastric artery and vein were carefully preserved. The lesser omentum close to the liver was dissected to allow free movement of the stomach. The vagal nerve, with the exception of the hepatic and pyloric branches, was preserved. These procedures allowed the stomach and the duodenum to be mobilized to the left in a straight, vertical manner.
As the first step in reconstruction, the proximal jejunum was brought through the right side of the transverse mesocolon by the retrocolic route. An end-to-side pancreaticojejunostomy was performed with duct-to-mucosal anastomosis. The pancreatic duct was anastomosed to the whole layer of the small opening in the jejunum to approximate the duct to the jejunal mucosa with the use of eight interrupted 5-0 PDS-II sutures (polydioxanone, Johnson & Johnson Co.), regardless of the size of the pancreatic duct. A 5-Fr polyethylene pancreatic drainage tube with a small knob (Sumitomo Bakelite Co., Japan) was placed in the pancreatic duct and exteriorized through the jejunal limb. The cut surface of the pancreas was then anastomosed to the jejunal seromuscular layer, and the end-to-side pancreaticojejunostomy was completed. A one-layer end-to-side hepaticojejunostomy with interrupted 5-0 PDS sutures was then performed 5–10 cm distal to pancreaticojejunostomy.
The final step was randomized to either to the antecolic route or vertical retrocolic route. For vertical retrocolic duodenojejunostomy, the left side of the transverse mesocolon (left side of the middle colic vessels) was opened, and the duodenum was brought down together with the gastric antrum in a straight, vertical manner. A retrocolic end-to-side duodenojejunostomy was performed at the caudal side of the transverse mesocolon and the antrum was fixed to the transverse mesocolon with a few 4-0 silk sutures. For antecolic duodenojejunostomy, the stomach was brought down antecolically. Braun anastomosis was added in both groups. Finally, the opening of the old ligament of Treitz and the jejunum brought up for pancreaticojejunostomy and hepaticojejunostomy were fixed to the mesocolon, and two or three closed drains were placed around the pancreatic and biliary anastomosis. All patients were given prophylactic antibiotics and H2 blocker postoperatively; none were given prokinetic drugs such as erythromycin.
Data collection and study endpoints
Clinicopathological data were collected prospectively for all patients. Data included postoperative mortality and morbidity, including pancreatic fistula, intraabdominal bleeding, pancreaticojejunostomy or hepaticojejunostomy leakage, intraabdominal abscess, and wound infection. Pancreatic fistula was defined when an amylase level in the fluid from the closed drains was >10,000 IU/l.
The first endpoint was clinical DGE defined as (1) the need for nasogastric tube decompression for more than 10 days (DGE 10), (2) the need for reinsertion of the nasogastric tube, or (3) an inability to take in an appropriate amount solid food orally by POD 14 (DGE 14), as described elsewhere [18].
The secondary endpoint was recovery of gastric emptying as assessed by 13C-acetate breath test [20]. For at least 4 days before this test, all drugs, including H2 blocker, were withdrawn. All patients ingested a liquid meal (200 Kcal/200 ml, RACOL, Ohtsuka Pharmaceutical Co., Tokyo, Japan) labeled with 100 mg sodium 13C-acetate (Cambridge Isotope Laboratories, Inc., Andover, MA, USA) in the morning after an overnight fast before surgery and on POD 30. Breath samples were collected in the collection bag (1.3 l) before and after ingestion of the test meal, i.e., before and at 5, 10, 15, 20, 30, 40, 50, 60, 75, 90, 105, 120, and 180 min after ingestion of the 13C-acetate. The recovery of 13C in the breath samples was analyzed by isotope-selective infrared spectrometry (UBiT IR 300, Otsuka Electronics Co., Ltd, Osaka, Japan). The time when 13CO2 reached maximum excretion (Tmax) and half-emptying time (T1/2) were calculated by using analysis software (Microsoft Office Excel, Microsoft Japan, Tokyo, Japan).
Statistical analysis
All values are expressed as mean ± SD. Differences between groups were examined for statistical significance by chi-square test, unpaired or paired Student’s t-test, Wilcoxon signed-rank test, or Mann–Whitney U test. Statistical analysis was performed by the statistician who was blind to the study group.
Results
Patient characteristics
Clinical characteristics of the enrolled patients are shown in Table 1. There were no statistical differences between the two groups in age, sex ratio, type of disease, percentage of patients with malignant disease, preoperative laboratory data including N-benzoyl-l-tyrosyl-p-aminobenzoic acid (BT-PABA) test value, percentage of patients with diabetes mellitus, HbA1c, operation time, or length of the remaining duodenum.
Postoperative complications
As shown in Table 2, postoperative morbidity was observed in 9 of 17 patients (53%) in the antecolic group and 6 of 18 patients (33%) in the vertical retrocolic group. Intra-abdominal bleeding associated with pancreatic fistula and/or intra-abdominal abscess was observed in one patient in each group, and both patients were treated successfully by interventional transarterial embolization. Intra-abdominal abscess was the main complication and were treated successfully by drainage. No operative death or hospital death was observed.
Clinical DGE
DGE clinically defined as DGE10 or DGE14 and the length of postoperative hospital stay are shown in Table 2. The nasogastric tube was removed on POD 1.2 ± 0.4 in the antecolic group on POD 1.1 ± 0.3 in the vertical retrocolic group. No patient needed a nasogastric tube for more than 10 days (DGE10), and reinsertion of a nasogastric tube was not necessary in any patient. The number of days to the start of liquid diet was similar between the two groups (5.4 days in the antecolic group and 5.7 days in the vertical retrocolic group). With respect to DGE14, one patient in the antecolic group and four in the vertical retrocolic group failed unlimited solid food oral intake by POD 14. Thus, the incidence of DGE defined as DGE14 was 6% (1 of 17 patients) in the antecolic group and 22% (4 of 18 patients) in the vertical retrocolic group. Although the rate was higher in the vertical retrocolic group, the difference did not reach statistical significance (P = 0.34). The overall incidence of DGE after PpPD was 14% (5 of 35 patients).
13C-acetate gastric emptying test
Tmax did not differ between the vertical retrocolic group and the antecolic group before or on POD 30 (P = 0.56 before surgery and P = 0.31 on POD 30). Similarly, T1/2 did not differ between the two groups (P = 0.99 before surgery, P = 0.26 on POD 30) (Table 3). Neither reconstruction route had a significant effect on gastric emptying on POD 30 after PpPD.
The 13C-acetate gastric emptying test values before and on POD 30 were compared in each group. In the vertical retrocolic group, Tmax was significantly prolonged on POD 30 compared to that before surgery (2.12 ± 2.14 h versus 1.08 ± 0.29 h, P < 0.02), whereas no significant difference was found in the antecolic group (1.54 ± 1.22 h versus 1.11 ± 0.25 h, P = 0.29). However, T1/2 was significantly longer in each group on POD 30 compared to the corresponding preoperative value (P = 0.0023 in the antecolic group, P = 0.0002 in the vertical retrocolic group (Table 3). Gastric emptying was not completely restored to the preoperative level in either group by POD 30. Mean Tmax on POD 30 in the antecolic group was increased 1.39-fold, and that in the vertical retrocolic group was increased 1.96-fold. Similarly, T1/2 was increased 2.04-fold in the antecolic group and 3.23-fold in the vertical retrocolic group. Greater increases in Tmax and T1/2 were observed in the vertical retrocolic group than in the antecolic group.
Tmax before and after surgery in each patient is shown in Fig. 1. Individual changes in Tmax before and after surgery were similar to individual changes in T1/2. A greater than twofold increase in Tmax was observed in 3 (18%) of 17 patients in the antecolic group, and in 4 (22%) of 18 patients in the vertical retrocolic group. Tmax of all patients before surgery (n = 35) was 1.09 ± 0.26 h, ranging from 0.7 to 1.5 h. Tmax greater than 1.5 h on POD 30 was found in four patients in each group. Tmax on POD 30 remained similar to the preoperative level in most patients (approximately 80%) in both groups.
Discussion
The present study showed that the incidence of clinical DGE was lower with the antecolic route than with the vertical retrocolic route, but the difference was not significant (6% with the antecolic route versus 22% with the vertical retrocolic route, P = 0.34). Moreover, gastric emptying (Tmax, T1/2) as assessed by the 13C-acetate breath test did not differ significantly between the antecolic route and the vertical retrocolic route before or on day 30 after PpPD. T1/2 was significantly prolonged in both groups after PpPD, indicating that gastric emptying remained impaired on POD 30, regardless of the reconstruction route. The degree of impairment was greater in patients in whom vertical retrocolic reconstruction was performed. An analysis of individual patients revealed that on POD 30, gastric emptying was similar to the preoperative level in approximately 80% of patients, regardless of the reconstruction route.
Since Traverso and Longmire [1] first reported PpPD in 1978, the procedure has been accepted as a standard procedure for periampullary diseases. This is because it yields better quality of life, nutritional status, and weight gain without any difference in postoperative survival than the Whipple procedure [1–4, 21]. The postoperative mortality rate has fallen recently, but complications associated with pancreaticoduodenectomy remain, the most troublesome of which are pancreatic fistula, intra-abdominal infection, intra-abdominal bleeding, wound infection, and DGE. DGE was first reported by Warshaw and Torchiana [5]. Postoperative DGE decreases patient comfort, increases the risk of aspiration pneumonia, prolongs hospital stay, and increases medical costs.
DGE is considered a specific complication of PpPD, because it is specifically attributed to pylorus-sparing resection of the pancreatic head [5–7, 10]. Several underlying mechanisms have been proposed: (1) gastric atony or gastroparesis caused by vagotomy, resection of the duodenal pacemaker, or disruption of the gastroduodenal neural connections [11], (2) local ischemic injury of the antrum and pylorus [7], (3) gastric atony in response to a reduced circulating levels of motilin [12], (4) torsion or angulation of the reconstructed alimentary tract [7], (5) gastric dysrhythmia or gastroparesis secondary to an intraabdominal complication such as anastomotic leakage, abscess, or local inflammation [15, 21]. Recent studies have shown that DGE does not occur as a result of pylorus preservation but rather as a consequence of postoperative complications [17, 22, 23]. Although the exact mechanism underlying DGE is not clear, our results suggest that DGE is related to clinical or even subclinical local inflammation caused by postoperative complications; three of our five patients with DGE (DGE14) had abscess or pancreatic fistula.
DGE has been generally defined as DGE10 (need for a nasogastric tube for more than 10 days) and DGE14 (failure to tolerate solid food by POD 14). The reported incidence of DGE ranges from 20 to 60% [5–13]. In the present study, no patient needed nasogastric decompression for more than 3 days. The nasogastric tube was removed on POD 1 in 30 (86%) of the 35 patients and on POD 2 in the remaining five. None required reinsertion of a nasogastric tube. With respect to DGE14, failure to tolerate solid food was observed in 5 of 35 patients, for an overall incidence of 14%.
A difference in DGE with respect to the reconstruction route, whether antecolic or retrocolic duodenojejunostomy, has been reported. In a retrospective study, Park et al. [23]. found that the incidence of DGE was 31.7% in the retrocolic group, but only 6.5% in the antecolic group. Hartel et al. [24]. reported an incidence of 24% with the retrocolic route and 5% with the antecolic route. Sugiyama et al. [25]. reported that DGE occurred in 1 of 12 patients (8%) in the antecolic group, but in 13 of 18 patients (72%) in the retrocolic group. These retrospective studies have suggested that the incidence of DGE is lower with the antecolic route than with the retrocolic route. A recent prospective randomized study by Tani et al. [18]. yielded an incidence of 50% for the retrocolic route, but 5% for the antecolic route. In the current prospective randomized controlled trial, the incidence of DGE was 22% with the vertical retrocolic route and 6% with the antecolic route, but the difference was not statistically significant. Although the purpose of this study was to show the superiority of the vertical retrocolic route, an interim analysis did not show any advantage of the vertical retrocolic route; hence, we decided to terminate the study.
In addition to clinically defined DGE, the 13C-acetate gastric emptying test showed that gastric emptying on POD 30 did not differ between the antecolic route and the vertical retrocolic route. Moreover, the gastric emptying did not recover to the preoperative level by 30 days in approximately 20% of patients, regardless of the reconstruction route. A greater increase in Tmax and T1/2 was observed with the vertical retrocolic route than with the antecolic route. These results suggest that the vertical retrocolic route offers no advantage. An analysis of the individuals showed that gastric emptying variables (Tmax, T1/2) had recovered to the preoperative level in approximately 80% of patients on POD 30, regardless of the reconstruction route. The day of analysis and type of meal selected (POD 30, liquid meal) should be reconsidered in another study.
In conclusion, the incidence of DGE and gastric emptying variables (Tmax, T1/2) after PpPD were similar between patients in whom reconstruction was performed by the antecolic route and those in whom it was performed by the vertical retrocolic route. On POD 30, gastric emptying was impaired in both groups compared to the preoperative level, but an analysis of individuals showed that it had recovered to the preoperative level in most patients, regardless of the reconstruction route.
References
Traverso LW, Longmire WP Jr. Preservation of the pylorus in pancreaticoduodenectomy. Surg Gynecol Obstet. 1978;146:959–62.
Kozuschek W, Reith HB, Waleczek H, Haarmann W, Edelmann M, Sonntag D. A comparison of long term results of the standard Whipple procedure and the pylorus preserving pancreatoduodenectomy. J Am Coll Surg. 1994;178:443–53.
Zerbi A, Balzano G, Patuzzo R, Calori G, Braga M, Carlo VD. Comparison between pylorus-preserving and Whipple pancreatoduodenectomy. Br J Surg. 1995;82:975–9.
Mosca F, Giulianotti PC, Balestracci T, Candio GD, Pietrabissa A, Sbarana F. Long-term survival in pancreatic cancer: pylorus-preserving versus Whipple pancreatoduodenectomy. Surgery. 1997;122:553–66.
Warshaw AL, Torchiana DL. Delayed gastric emptying after pylorus-preserving pancreaticoduodenectomy. Surg Gynecol Obstet. 1985;160:1–4.
Jiminez RE, Fernandez-del Castillo C, Rattner DW, Chang YC, Warshaw AL. Outcome of pancreaticoduodenectomy with pylorus preservation or with antrectomy in the treatment of chronic pancreatitis. Ann Surg. 2000;231:293–300.
Itani KM, Coleman RE, Meyers WC, Akwari OE. Pylorus-preserving pancreaticoduodenectomy. A clinical and physiological appraisal. Ann Surg. 1986;204:655–64.
Yeo CJ, Cameron JL, Sohn TA, Lillemoe KD, Pitt HA, Talamini MA. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg. 1997;226:248–57.
Fabre JM, Burgel JS, Navarro F, Boccarat G, Lemoine C, Domergue J. Delayed gastric emptying after pancreaticoduodenectomy and pancreaticogastrostomy. Eur J Surg. 1999;165:560–65.
Patel AG, Toyama MT, Kusske AM. Pylorus-preserving resection for pancreatic cancer. Is it any better? Arch Surg. 1995;130:838–43.
Braasch JW, Deziel DJ, Rossi RL, Watkins E Jr, Winter PF. Pyloric and gastric preserving pancreatic resection: experience with 87 patients. Ann Surg. 1986;204:411–8.
Yeo CJ, Barry K, Sauter PK, Sostre S, Lillemoe KD, Pitt HA, et al. Erythromycin accelerates gastric emptying after pancreaticoduodenectomy. Ann Surg. 1993;218:229–37.
Yamaguchi K, Tanaka M, Chijiiwa K, Nagakawa T, Imamura M, Takada T. Early and late complications of pylorus-preserving pancreatoduodenectomy in Japan 1998. J Hepatobiliary-Pancreat Surg. 1999;6(3):303–11.
Lin P, Lin YJ. Prospective randomized comparison between pylorus-preserving and standard pancreaticoduodenectomy. Br J Surg. 1999;86:603–7.
van Berge Henegouwen MI, van Gulik TM, de Wit LT, Allema JH, Rauws EA, Obertop H, et al. Delayed gastric emptying after standard pancreaticoduodenectomy versus pylorus-preserving pancreaticoduodenectomy: an analysis of 200 consecutive patients. J Am Coll Surg. 1997;185:373–9.
Neoptolemos JP, Russell RC, Bramhall S, Theis B. Low mortality following resection for pancreatic and periampullary tumours in 1026 patients: UK surgery of specialist pancreatic units. Br J Surg. 1997;84:1370–6.
Horstmann O, Markus PM, Ghadimi MB, Becker H. Pylorus preservation has no impact on delayed gastric emptying after pancreatic head resection. Pancreas. 2004;28:69–74.
Tani M, Terasawa H, Kawai M, Ina S, Hirono S, Uchiyama K, et al. Improvement of delayed gastric emptying in pylorus-preserving pancreaticoduodenectomy Results of a prospective, randomized, controlled trial. Ann Surg. 2006;243:316–20.
Chijiiwa K, Ohuchida J, Hiyoshi M, Nagano M, Kai M, Kondo K. Vertical retrocolic duodenojejunostomy decreases delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. Hepato Gastroenterol. 2007;54:1874–7.
Braden B, Adams S, Duan LP, Orth KH, Maul FD, Lembcke B, et al. The [13C] acetate breath test accurately reflects gastric emptying of liquids in both liquid and semisolid test meals. Gastroenterology. 1995;108:1048–55.
Hocking MP, Harrison WD, Sninsky CA. Gastric dysrhythmias following pylorus-preserving pancreaticoduodenectomy. Possible mechanism for early delayed gastric emptying. Dig Dis Sci. 1990;35:1226–30.
Riediger H, Makowiec F, Schareck WD, Hopt UT, Adam U. Delayed gastric emptying after pylorus-preserving pancreatoduodenectomy is strongly related to other postoperative complications. J Gastrointest Surg. 2003;7:758–65.
Park YC, Kim SW, Jang JY, Ahn YJ, Park YH. Factors influencing delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. J Am Coll Surg. 2003;196:859–65.
Hartel M, Wente MN, Hinz U, Kleef J, Wagner M, Muller MW, et al. Effect of antecolic reconstruction on delayed gastric emptying after the pylorus-preserving Whipple procedure. Arch Surg. 2005;140:1094–9.
Sugiyama M, Abe N, Ueki H, Masaki T, Mori T, Atomi Y. A new reconstruction method for preventing delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. Am J Surg. 2004;187:743–6.
Acknowledgments
A part of this study was supported by grant-in-aid (No. 17591417) from Japanese Ministry of Education, Culture, Sports, Science and Technology. Dr. Imamura is an equally contributed first author.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Chijiiwa, K., Imamura, N., Ohuchida, J. et al. Prospective randomized controlled study of gastric emptying assessed by 13C-acetate breath test after pylorus-preserving pancreaticoduodenectomy: comparison between antecolic and vertical retrocolic duodenojejunostomy. J Hepatobiliary Pancreat Surg 16, 49–55 (2009). https://doi.org/10.1007/s00534-008-0004-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00534-008-0004-3