Abstract
Background
Enhanced recovery after surgery (ERAS) protocols or laparoscopic technique has been applied in various surgical procedures. However, the clinical efficacy of combination of the two methods still remains unclear. Thus, our aim was to assess the role of ERAS protocols in laparoscopic abdominal surgery.
Methods
We performed a systematic literature search in various databases from January 1990 to October 2017. The results were analyzed according to predefined criteria.
Results
In the present meta-analysis, the outcomes of 34 comparative studies (15 randomized controlled studies and 19 non-randomized controlled studies) enrolling 3615 patients (1749 in the ERAS group and 1866 in the control group) were pooled. ERAS group was associated with shorter hospital stay (WMD − 2.37 days; 95% CI − 3.00 to − 1.73; P 0.000) and earlier time to first flatus (WMD − 0.63 days; 95% CI − 0.90 to − 0.36; P 0.000). Meanwhile, lower overall postoperative complication rate (OR 0.62; 95% CI 0.51–0.76; P 0.000) and less hospital cost (WMD 801.52 US dollar; 95% CI − 918.15 to − 684.89; P 0.000) were observed in ERAS group. Similar readmission rate (OR 0.73, 95% CI 0.52–1.03, P 0.070) and perioperative mortality (OR 1.33; 95% CI 0.53–3.34; P 0.549) were found between the two groups.
Conclusions
ERAS protocol for laparoscopic abdominal surgery is safe and effective. ERAS combined with laparoscopic technique is associated with faster postoperative recovery without increasing readmission rate and perioperative mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The concept of enhanced recovery after surgery (ERAS) was first introduced by Kehlet in the 1990s and has been widely used in various surgeries in past decade [1,2,3,4,5]. The application of ERAS protocols is expected to reduce surgical stress and complication rate by the combination of various interventions in the preoperative period. The key components of ERAS guidelines include preoperative education, epidural or regional anesthesia, no routine use of drains or nasogastric tubes, preventing postoperative vomiting, optimal pain control, early enteral nutrition, and ambulation. Extensive researches have shown that the ERAS protocol is superior to the conventional protocol in the field of many types of surgery [6,7,8]. Nowadays, laparoscopic technique has been applied widely because of the superior surgical and short-term outcomes as compared with open surgery. Accumulating researches have shown that patients undergoing major abdominal surgery could benefit from both ERAS programs and laparoscopic technique [9,10,11]. However, some surgeons still ignore the benefits of ERAS when performing laparoscopic surgery. The aim of this meta-analysis was to assess the role of ERAS protocols in laparoscopic abdominal surgery.
Search strategy
Materials and methods
A systematic literature search of PubMed, EMBASE, the Cochrane Library, and Web of Science from January 1990 to October 2017 was undertaken with restriction to English. Search terms “ERAS” (enhanced recovery, fast track) and “laparoscopy” (laparoscopic, laparoscopy-assisted) were used in combination with the Boolean operators AND or OR. The reference lists of articles obtained were also reviewed to find the relevant literature. Two investigators (ZYL and BB) performed the literature search independently.
Study selection
Included criteria for this meta-analysis were as follows: (1) randomized controlled trials (RCTs) or non-randomized controlled trials (nRCTs) that compared ERAS with traditional care in laparoscopic surgery; (2) studies that focus on major abdominal surgery, including gastrectomy, hepatectomy, cystectomy, pancreatectomy, prostatectomy, nephrectomy, and colorectal surgery; (3) studies that reported at least one outcome of interest, including length of hospital stay, postoperative complication, readmission rate, time to first flatus, hospital costs, and perioperative mortality; and (4) the ERAS group should include at least five intervention items of ERAS protocol that distinct from the control group. The excluded criteria were: (1) studies such as reviews, comments, letters, case reports, or cohort studies including fewer than ten patients; (2) study included vascular or emergency operation cases; and (3) studies published in a language other than English.
Data extraction, outcome measures, and bias assessment
Data were extracted independently by two reviewers (ZYL and BB), and discrepancies were adjudicated by a third reviewer (QCZ).Primary outcomes were length of hospital stay, postoperative complication, and readmission rate. Secondary outcomes were time to first flatus, hospital costs, and perioperative mortality. Hospital costs were converted into US dollar according to the exchange rate of October 2017.
Risk of bias of RCTs was assessed independently using the Cochrane risk of bias method [12]. Other studies were methodologically assessed by the same reviewers using the Newcastle–Ottawa scale (NOS), which has been widely used for the assessment of the quality of non-randomized studies in meta-analyses [13].
Statistical analysis
The data of primary and secondary outcomes were calculated using a random effects model and reported with 95% confidence interval (CI). Odds ratio (OR) was used to calculated categorical outcomes (postoperative complication, readmission rate, and perioperative mortality). Weighted mean difference (WMD) was used to calculate continuous outcomes (length of hospital stay, time to first flatus, and hospital costs). I2 statistics were used to quantify the heterogeneity among studies. Heterogeneity was graded as low (I2 < 25%), moderate (I2 = 25% to 75%), or high (I2 > 75%). Potential publication bias was tested by Begg test with visual inspection of the funnel plot (Supplemental Figure 1).
We performed sensitivity analysis to assess the stability of primary outcomes. The results were regarded as statistically significant at two-sided P < 0.05. All statistical analyses for meta-analyses were performed using Stata, version 14.1 (Stata Corp, College Station, TX). Risk of bias was assessed using the dedicated Cochrane tool of Review Manager software (RevMan version 5.3; Cochrane Collaboration).
Results
Study selection
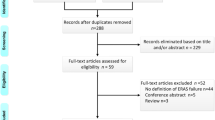
We conduct this systematic review and meta-analysis in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. Finally, 34 studies [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47] were eligibly included in the pooled analysis, which contained 15 randomized controlled studies [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28] and 19 non-randomized controlled studies [29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47]. The detailed search steps are illustrated in Fig. 1.
Study characteristics and quality assessment
Table 1 presents the characteristics of the final included studies. These studies were published from 2005 to 2017. A total of 3615 patients were included in 34 studies with 1749 in ERAS group and 1866 in control group. The 15 randomized controlled studies included 1247 patients: 606 in the ERAS group and 641 in the control group. The non-randomized controlled studies included 2368 patients: 1143 in the ERAS group and 1225 in the control group. The numbers of intervention items were ranged from 5 to 16. Risk of bias for randomized controlled studies is shown in Fig. 2. All these studies presented low to moderate risks. The quality assessment of non-randomized studies the NOS is presented in Table 2. Most of them had a score of 6–8, and only 2 studies had a score of 9.
Primary outcomes measures
Length of hospital stay
Twenty-one studies with appropriate data reported the length of hospital stay. This meta-analysis indicated that ERAS group was associated with a shorter hospital stay as compared with the control group (WMD − 2.37 days; 95% CI − 3.25 to − 1.73; P 0.000). This result showed a significant heterogeneity among the studies (I2 = 94.2, P = 0.000) (Fig. 3a). There were 12 randomized trials reported postoperative hospital stay. Our results illustrate that the ERAS group is associated with a shorter hospital stay based on analysis of RCTs (WMD − 1.89 days; 95% CI − 2.46 to − 1.32; P 0.000). The result also showed a significant heterogeneity among these studies (I2 = 88.1, P = 0.000) (Fig. 3b).
Postoperative complication rate
Thirty-one studies reported the postoperative complication. A lower postoperative complication rate was observed in the ERAS group (OR 0.61, 95% CI 0.49–0.75, P 0.000). This result showed a moderate heterogeneity (I2 26.6, P 0.089) (Fig. 3c). Analysis of the RCTs also revealed a lower postoperative complication rate favoring ERAS group (OR 0.57, 95% CI 0.37–0.86, P 0.000). This result also showed a moderate heterogeneity (I2 41.6, P 0.008) (Fig. 3d).
Readmission rate
Twenty-two studies reported the readmission rate, whereas nine of them had no readmission. The pooled data based on 13 studies identified no significant difference in readmission rate between the ERAS group and control group (OR 0.73, 95% CI 0.52–1.03, P 0.070) (Fig. 3e). Analysis of the RCTs also indicates that the readmission rate was similar between the two groups (OR 0.88; 95% CI 0.48–1.62, P 0.688) (Fig. 3f).
Subgroup analysis of primary outcomes measures
For the subgroup analysis based on study types (RCTs or nRCTs), number of intervention items (intervention items ≥ 10 or intervention items < 10), and surgery types (laparoscopic non-colorectal surgery or laparoscopic colorectal surgery), the ERAS group still had the advantage of shorter hospital stay and lower postoperative complication rate. The subgroup analysis also revealed that ERAS and control groups had similar readmission rate. Table 3 shows the results of subgroup analysis.
Secondary outcomes
Nine studies with the appropriate data reported the time to first flatus. The pooled result indicated that ERAS group is associated with earlier time to first flatus than control group (WMD − 0.63 days; 95% CI − 0.90 to − 0.36; P 0.000). The statistical outcome showed a significant heterogeneity among these studies (I2 89.8, P 0.000) (Fig. 4a). Hospital cost with appropriate data was reported in seven of the included studies. Our result indicated that the ERAS group had less hospital cost (WMD − 801.52 US dollar; 95% CI − 918.15 to − 684.89; P 0.000) (Fig. 4b). Twenty-one studies reported perioperative mortality outcome, whereas 13 of them had no perioperative mortality. No significant difference was observed in perioperative mortality between the two groups (OR 1.33; 95% CI 0.53–3.34; P 0.549). This result showed no significant heterogeneity (I2 = 0, P = 0.983) (Fig. 4c).
Publication bias and sensitivity analysis
Begg test with funnel plot for primary and secondary outcomes revealed no significant publication bias except for time to first flatus (Supplemental Figure 1). Sensitivity analysis was conducted by subgroup analysis on primary outcomes. Table 3 shows the result of subgroup analysis of primary outcomes. The result indicated that the primary outcomes are stable.
Discussion
Over the past decades, laparoscopic techniques and ERAS program have been applied in various kinds of surgery. However, the clinical efficacy of combination of the two methods still remains unclear. This systematic review and meta-analysis include comparative studies of the safety and efficacy of ERAS protocol and conventional care for patients underwent laparoscopic abdominal surgery from 2005 to 2017. To our knowledge, this is the first meta-analysis to give an overview of the clinical efficacy of ERAS protocol combined with laparoscopic technique for major abdominal surgery.
Hospital stay is a key index to assess the outcome of postoperative recovery. We hypothesize previously that optimal surgical outcomes may have already been achieved with minimally invasive techniques, leaving limited room for improvement via ERAS protocols. With the results of our meta-analysis, we observed that ERAS group had shorter hospital stay. This reduction was 2.37 days (95% CI − 3.00 to − 1.73) in the analysis of all included studies. This result not just statistically significant, but also have clinical significance.
In this study, almost half of the studies with appropriate data reported the outcomes of length of hospital stay are about the colorectal surgery. Therefore, we perform the subgroup analysis to test the stability of the result by excluding the studies related to colorectal surgery. The result was also consistent when only non-colorectal surgeries were evaluated, indicating that the outcome is stable. In the included studies, the intervention items are ranged from 5 to 16. We infer that the benefit of patients may associated with the number of intervention items be applied. Interestingly, the subgroup analysis revealed that the shorter hospital stay could also be observed in ERAS group when less intervention items (intervention items < 10) were taken for consideration. However, considerable heterogeneity still existed when subgroup analysis was conducted. This may attributed to the different surgical types and discharge criteria among the included studies.
Postoperative complication is always regarded as one of the major concerns in clinical practice. Extensive researches have demonstrated that patients who underwent laparoscopic surgery are always associated with lower complication rate when compared with open surgery. Our result indicated that ERAS group was associated with lower overall postoperative complication rate. Furthermore, the subgroup analysis based on study types (RCTs or nRCTs), intervention items (intervention items ≥ 10 or intervention items < 10), and surgery types (laparoscopic non-colorectal surgery or laparoscopic colorectal surgery) still proved that ERAS was beneficial to lower postoperative complications after laparoscopic surgery. However, postoperative complications were not recorded in majority of included studies according to the Clavien–Dindo grade. Moreover, postoperative complications reported in the included studies were varied due to different types of surgery and uniform standards. So, it was not feasible for us to perform a stratified analysis according to the severity of complications.
Whether the ERAS protocol is accompanied by higher potentially readmission rate is a big concern in clinical practice, since readmission has negative effect on quality of life [48,49,50]. Twenty-two included studies reported the outcomes of the readmission rate, whereas nine of them had no readmission. Our results revealed no significant difference between ERAS and control groups in readmission rate and perioperative mortality, indicating that the combination of ERAS and laparoscopic had the superiority of shorter hospital stay without increasing readmission rates and perioperative mortality. Moreover, we also noticed that these results are consistent among all included studies.
With an aging population, it can be predicted that the number of elderly patients with disease that requires the surgery will increase. To date, the role of ERAS protocol in elderly patients still remains controversial. Zeng et al. [47] and Wang et al. [21] reported that the application of ERAS is safe and effective for elderly patients underwent laparoscopic colorectal surgery. However, a study conducted by Bu et al. [51] found that the elderly gastric cancer patients (range from 75 to 89 years old) did not benefit from ERAS protocol. What is more, we also needed to be alert that the ERAS group had a higher readmission rate as compared with the control group (19 vs. 5% P 0.013). No routine abdominal drainage is one of intervention items, and the superiority and feasibility of this have been confirmed in colorectal surgery [52, 53]. However, whether the safe and effective measures confirmed in colorectal surgery are suitable for other abdominal surgery should be evaluated in further studies.
In the present meta-analysis, only three studies mainly focus on the clinical outcome of ERAS protocol in elderly patients. In addition, large cohort studies have yet to determine the outcomes of ERAS protocol in elderly population. Studies that have reported that elderly patients who accepted ERAS protocol have a varied definition of elderly (range from 60 to 75 year old) which may limit the general application of ERAS protocol. More high-quality researches are needed to evaluate the safety and feasibility of ERAS protocol for elderly patients. Additionally, the ERAS guidelines should also consider the unique physiological and anatomical characteristics of elderly population.
Recovery of gastrointestinal function is important in postoperative recovery. We observed that the ERAS group had an earlier time to first flatus of 0.6 day, indicating that the ERAS group is superior to the control group in the recovery of gastrointestinal function. Compared with previous studies focus on open surgery, 0.6 day may seem like a relatively low reduction. However, it also means that ERAS protocol could further promote the recovery of patients based on the beneficial effect of minimally invasive techniques.
Economic burden on patients is an important issue needed to be considered in clinical practice. Our results indicate that the ERAS group had a reduction of about eight hundred dollars in hospital costs. This reduction was mainly accompanied with shorter hospital stay, earlier oral feeding, and lower complication rate. Inadequate compliance is a problem in the implementation and maintaining of ERAS protocol. Previous studies showed that the increased compliance to ERAS protocol was associated with better postoperative outcomes [54,55,56]. In this meta-analysis, only six studies [17, 18, 25, 42, 57, 58] mentioned the compliance of ERAS protocol. Lee et al. [18] reported that the compliance rates for major items in ERAS group were more than 80%. Vignali et al. [42] observed that the overall compliance rate for the ERAS group was 85.7%. Regarding different ERAS items, we found that the compliance rates are varied widely and the number of items applied in current meta-analysis is ranged from 5 to 16. Meanwhile, the control group may also be implemented several ERAS items. These might create potential bias, which may decrease the benefit of ERAS group when compared with the control group.
Quality of life (QOL) after surgery is often used to assess the subjective of illness and its treatment which should also be considered [59, 60]. Compared with open surgery, studies have demonstrated that the application of laparoscopic technique is always associated with better QOL [61, 62]. In all include studies, six studies [14, 16,17,18,19, 25] assessed the patient’s QOL. These results indicated that the patients in the ERAS group experience no negative satisfaction and even better QOL score.
Some limitations of our present study need to be noted. First, there are no ERAS guidelines for several abdominal surgeries, so the number and details of ERAS items are varied in these studies. Second, the compliance and ERAS items applied are varied among included studies. These might create potential bias which may therefore reduce the benefits in the ERAS group. Third, only three studies focus on the application of ERAS protocol for elderly patients which may therefore limited the generalization of our results. Finally, high heterogeneity was observed in some outcomes except for postoperative complication rate, readmission rate, and perioperative mortality.
In conclusion, the results showed that ERAS protocol for laparoscopic abdominal surgery is safe and effective. ERAS combined with laparoscopic technique is associated with faster postoperative recovery without increasing readmission rate and perioperative mortality.
References
Bardram LF-JP, Jensen P, Crawford ME, Kehlet H (1995) Recovery after laparoscopic colonic surgery with epidural analgesia, and early oral nutrition and mobilisation. Lancet 345:763–764
Kehlet HWD (2002) Multimodal strategies to improve surgical outcome. Am J Surg 83:630–641
Grant MCYD, Wu CL, Makary MA, Wick EC (2017) Impact of enhanced recovery after surgery and fast track surgery pathways on healthcare-associated infections: results from a systematic review and meta-analysis. Ann Surg 265:68–79
Zhao Y, Qin H, Wu Y et al (2017) Enhanced recovery after surgery program reduces length of hospital stay and complications in liver resection: a PRISMA-compliant systematic review and meta-analysis of randomized controlled trials. Medicine 96:e7628
Deng XCX, Huo Z, Shi Y, Jin Z, Feng H, Wang Y, Wen C, Qian H, Zhao R, Qiu W, Shen B, Peng C (2017) Modified protocol for enhanced recovery after surgery is beneficial for Chinese cancer patients undergoing pancreaticoduodenectomy. Oncotarget 8:47841–47848
Tanaka R, Lee SW, Kawai M et al (2017) Protocol for enhanced recovery after surgery improves short-term outcomes for patients with gastric cancer: a randomized clinical trial. Gastric Cancer 20:861–871
Visioni A, Shah R, Gabriel E et al (2017) Enhanced recovery after surgery for noncolorectal surgery? A systematic review and meta-analysis of major abdominal surgery. Ann Surg 267:57–65
Wang C, Zheng G, Zhang W et al (2017) Enhanced recovery after surgery programs for liver resection: a meta-analysis. J Gastrointest Surg 21:472–486
Jiang L, Yang KH, Guan QL et al (2013) Laparoscopy-assisted gastrectomy versus open gastrectomy for resectable gastric cancer: an update meta-analysis based on randomized controlled trials. Surg Endosc 27:2466–2480
Feng FJG, Li JP, Li XH, Shi H, Zhao ZW, Wu GS, Liu XN, Zhao QC (2013) Fast-track surgery could improve postoperative recovery in radical total gastrectomy patients. World J Gastroenterol 19:3642–3648
Geltzeiler CB, Rotramel A, Wilson C et al (2014) Prospective study of colorectal enhanced recovery after surgery in a community hospital. JAMA Surg 149:955–961
Higgins JP, Altman DG, Gotzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Stang A (2010) Critical evaluation of the Newcastle–Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605
Recart ADD, White PF, Thomas T, Johnson DB, Cadeddu JA, Recart A, Duchene D, White PF et al (2005) Efficacy and safety of fast-track recovery strategy for patients undergoing laparoscopic nephrectomy. J Endourol 19:1165–1169
Gralla O, Haas F, Knoll N et al (2007) Fast-track surgery in laparoscopic radical prostatectomy: basic principles. World J Urol 25:185–191
Magheli A, Knoll N, Lein M et al (2011) Impact of fast-track postoperative care on intestinal function, pain, and length of hospital stay after laparoscopic radical prostatectomy. J Endourol 25:1143–1147
Vlug MS, Wind J, Hollmann MW et al (2011) Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study). Ann Surg 254:868–875
Lee TG, Kang SB, Kim DW et al (2011) Comparison of early mobilization and diet rehabilitation program with conventional care after laparoscopic colon surgery: a prospective randomized controlled trial. Dis Colon Rectum 54:21–28
Kim JW, Kim WS, Cheong J-H et al (2012) Safety and efficacy of fast-track surgery in laparoscopic distal gastrectomy for gastric cancer: a randomized clinical trial. World J Surg 36:2879–2887. https://doi.org/10.1007/s00268-012-1741-7
Chen HJ, Xin JL, Cai L et al (2012) Preliminary experience of fast-track surgery combined with laparoscopy-assisted radical distal gastrectomy for gastric cancer. J Gastrointest Surg 16:1830–1839
Wang Q, Suo J, Jiang J et al (2012) Effectiveness of fast-track rehabilitation vs conventional care in laparoscopic colorectal resection for elderly patients: a randomized trial. Colorectal Dis 14:1009–1013
Wang G, Jiang Z, Zhao K et al (2012) Immunologic response after laparoscopic colon cancer operation within an enhanced recovery program. J Gastrointest Surg 16:1379–1388
Lemanu DP, Singh PP, Berridge K et al (2013) Randomized clinical trial of enhanced recovery versus standard care after laparoscopic sleeve gastrectomy. Br J Surg 100:482–489
Feng F, Li XH, Shi H et al (2014) Fast-track surgery combined with laparoscopy could improve postoperative recovery of low-risk rectal cancer patients: a randomized controlled clinical trial. J Dig Dis 15:306–313
He F, Lin X, Xie F et al (2015) The effect of enhanced recovery program for patients undergoing partial laparoscopic hepatectomy of liver cancer. Clin Transl Oncol 17:694–701
Abdikarim I, Cao XY, Li SZ et al (2015) Enhanced recovery after surgery with laparoscopic radical gastrectomy for stomach carcinomas. World J Gastroenterol 21:13339–13344
Liang X, Ying H, Wang H et al (2016) Enhanced recovery program versus traditional care in laparoscopic hepatectomy. Medicine 95:e2835
Liu G, Jian F, Wang X et al (2016) Fast-track surgery protocol in elderly patients undergoing laparoscopic radical gastrectomy for gastric cancer: a randomized controlled trial. Onco Targets Ther 9:3345–3351
Stoot JH, Van Dam RM, Busch OR et al (2009) The effect of a multimodal fast-track programme on outcomes in laparoscopic liver surgery: a multicentre pilot study. Hpb 11:140–144
Tsikitis VL, Holubar SD, Dozois EJ et al (2010) Advantages of fast-track recovery after laparoscopic right hemicolectomy for colon cancer. Surg Endosc 24:1911–1916
Poon JT, Fan JK, Lo OS et al (2011) Enhanced recovery program in laparoscopic colectomy for cancer. Int J Colorectal Dis 26:71–77
Gouvas N, Gogos-Pappas G, Tsimogiannis K et al (2012) Implementation of fast-track protocols in open and laparoscopic sphincter-preserving rectal cancer surgery: a multicenter, comparative, prospective, non-randomized study. Digestive Surgery 29:301–309
Sanchez-Perez B, Aranda-Narvaez JM, Suarez-Munoz MA et al (2012) Fast-track program in laparoscopic liver surgery: theory or fact? World J Gastrointest Surg 4:246–250
Huibers CJA, de Roos MAJ, Ong KH (2011) The effect of the introduction of the ERAS protocol in laparoscopic total mesorectal excision for rectal cancer. Int J Colorectal Dis 27:751–757
Saar M, Ohlmann CH, Siemer S et al (2013) Fast-track rehabilitation after robot-assisted laparoscopic cystectomy accelerates postoperative recovery. BJU Int 112:E99–E106
Guan X, Liu L, Lei X et al (2014) A comparative study of fast-track versus [corrected] conventional surgery in patients undergoing laparoscopic radical cystectomy and ileal conduit diversion: Chinese experience. Sci Rep 4:6820
Sahoo MRGM, Kumar TA (2014) Early rehabilitation after surgery program versus conventional care during perioperative period in patients undergoing laparoscopic assisted total gastrectomy. J Minim Access Surg 10:132–138
Taupyk Y, Cao X, Zhao Y et al (2015) Fast-track laparoscopic surgery: a better option for treating colorectal cancer than conventional laparoscopic surgery. Oncol Lett 10:443–448
Esteban F, Cerdan FJ, Garcia-Alonso M et al (2014) A multicentre comparison of a fast track or conventional postoperative protocol following laparoscopic or open elective surgery for colorectal cancer surgery. Colorectal Dis 16:134–140
Richardson J, Di Fabio F, Clarke H et al (2015) Implementation of enhanced recovery programme for laparoscopic distal pancreatectomy: feasibility, safety and cost analysis. Pancreatology 15:185–190
Alvarez MP, Foley KE, Zebley DM et al (2015) Comprehensive enhanced recovery pathway significantly reduces postoperative length of stay and opioid usage in elective laparoscopic colectomy. Surg Endosc 29:2506–2511
Vignali A, Elmore U, Cossu A et al (2016) Enhanced recovery after surgery (ERAS) pathway vs traditional care in laparoscopic rectal resection: a single-center experience. Tech Coloproctol 20:559–566
Rege A, Leraas H, Vikraman D et al (2016) Could the use of an enhanced recovery protocol in laparoscopic donor nephrectomy be an incentive for live kidney donation? Cureus 8:e889
Fang F, Gao J, Bi X et al (2016) Effect and clinical significance of fast-track surgery combined with laparoscopic radical gastrectomy on the plasma level of vascular endothelial growth factor in gastric antrum cancer. SpringerPlus 5:50
Pedziwiatr M, Wierdak M, Nowakowski M et al (2016) Cost minimization analysis of laparoscopic surgery for colorectal cancer within the enhanced recovery after surgery (ERAS) protocol: a single-centre, case-matched study. Videosurg Other Miniinvasive Tech 11:14–21
Sugi M, Matsuda T, Yoshida T et al (2017) Introduction of an enhanced recovery after surgery protocol for robot-assisted laparoscopic radical prostatectomy. Urol Int 99:194–200
Zeng WG, Liu MJ, Zhou ZX et al (2017) Enhanced recovery programme following laparoscopic colorectal resection for elderly patients. ANZ J Surg https://doi.org/10.1111/ans.14074. [epub ahead of print]
Tsai TC, Orav EJ, Jha AK (2015) Patient satisfaction and quality of surgical care in US hospitals. Ann Surg 261:2–8
Khorgami Z, Andalib A, Aminian A et al (2016) Predictors of readmission after laparoscopic gastric bypass and sleeve gastrectomy: a comparative analysis of ACS-NSQIP database. Surg Endosc 30:2342–2350
Lyon A, Solomon MJ, Harrison JD (2014) A qualitative study assessing the barriers to implementation of enhanced recovery after surgery. World J Surg 38:1374–1380. https://doi.org/10.1007/s00268-013-2441-7
Bu J, Li N, Huang X et al (2015) Feasibility of fast-track surgery in elderly patients with gastric cancer. J Gastrointest Surg 19:1391–1398
Feroci F, Kroning KC, Lenzi E et al (2011) Laparoscopy within a fast-track program enhances the short-term results after elective surgery for resectable colorectal cancer. Surg Endosc 25:2919–2925
Roulin D, Donadini A, Gander S et al (2013) Cost-effectiveness of the implementation of an enhanced recovery protocol for colorectal surgery. Br J Surg 100:1108–1114
Roulin D, Donadini A, Gander S et al (2015) The Impact of enhanced recovery protocol compliance on elective colorectal cancer resection. Ann Surg 261:1153–1159
Okrainec A, Aarts MA, Conn LG et al (2017) Compliance with urinary catheter removal guidelines leads to improved outcome in enhanced recovery after surgery patients. J Gastrointest Surg 21:1309–1317
Pecorelli N, Hershorn O, Baldini G et al (2017) Impact of adherence to care pathway interventions on recovery following bowel resection within an established enhanced recovery program. Surg Endosc 31:1760–1771
Kennedy RH, Francis EA, Wharton R et al (2014) Multicenter randomized controlled trial of conventional versus laparoscopic surgery for colorectal cancer within an enhanced recovery programme: EnROL. J Clin Oncol 32:1804–1811
van Bree S, Vlug M, Bemelman W et al (2011) Faster recovery of gastrointestinal transit after laparoscopy and fast-track care in patients undergoing colonic surgery. Gastroenterology 141(872–880):e874
Lee SS, Chung HY, Kwon OK et al (2016) Long-term quality of life after distal subtotal and total gastrectomy: symptom- and behavior-oriented consequences. Ann Surg 263:738–744
Bottomley A (2002) The cancer patient and quality of life. Oncologist 7:120–125
Kim YW, Baik YH, Yun YH et al (2008) Improved quality of life outcomes after laparoscopy-assisted distal gastrectomy for early gastric cancer: results of a prospective randomized clinical trial. Ann Surg 248:721–727
Zhang RC, Zhang B, Mou YP et al (2017) Comparison of clinical outcomes and quality of life between laparoscopic and open central pancreatectomy with pancreaticojejunostomy. Surg Endosc 31:4756–4763
Authors’ contributions
Zhengyan Li and Qingchuan Zhao performed the experiment conception and design. Zhengyan Li and Bin Bai performed the research and retrieved the data. Zhengyan Li, Gang Ji, and Yezhou Liu performed the data analysis. Zhengyan Li did the paper writing. All authors read and approved the final manuscript.
Funding
This study was supported by the National Key Basic Research Program of China (No. 2014CBA02002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Zhengyan Li, Qingchuan Zhao, Bin Bai, Gang Ji, and Yezhou Liu have no conflicts of interest or financial ties to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Z., Zhao, Q., Bai, B. et al. Enhanced Recovery After Surgery Programs for Laparoscopic Abdominal Surgery: A Systematic Review and Meta-analysis. World J Surg 42, 3463–3473 (2018). https://doi.org/10.1007/s00268-018-4656-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-018-4656-0