Abstract
Background
The ratio of C-reactive protein to albumin (CRP/Alb) is a biochemical marker of systemic inflammatory response and has been associated with poor survival in cancer. This study retrospectively investigated the relationship between the CRP/Alb ratio and prognosis in gastric cancer patients.
Methods
This study enrolled 453 patients with a histopathological diagnosis of gastric adenocarcinoma who underwent curative surgery.
Results
A statistically significant weak correlation was observed between CRP/Alb ratio and neutrophil/lymphocyte ratio (NLR) (r = 0.19; P < 0.0001). There were statistically significant correlations between high CRP/Alb ratio and age (P = 0.0004), tumor size (P = 0.02), depth of invasion (P = 0.012), and lymph node metastasis (P = 0.022). A high NLR was significantly correlated with age (P = 0.0027), tumor size (P = 0.0006), depth of invasion (P < 0.0001), lymphatic involvement (P = 0.0031), venous involvement (P = 0.0022), and stage of disease (P = 0.0024). Based on results by receiver operating characteristic analysis, patients were divided as follows: CRP/Alb ratio ≥ 0.0232 (CARHigh), CRP/Alb ratio < 0.0232 (CARLow), NLR ≥ 2.43 (NLRHigh), and NLR < 2.43 (NLRLow). Five-year survival rates of patients with both CARHigh and NLRHigh, either CARHigh or NLRHigh, and both CARLow and NLRLow were 59.6, 75.8, and 87.5%, respectively, with statistically significant differences (P < 0.0001). Multivariate analysis revealed that the combination of CRP/Alb ratio and NLR was an independent prognostic indicator.
Conclusions
The combination of CRP/Alb ratio and NLR may be useful in predicting prognosis in gastric cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It was estimated that there were approximately 989,600 new cases of gastric cancer and 738,000 deaths worldwide in 2008, accounting for 8 and 10% of total cancer cases and cancer deaths, respectively [1]. As such, determining postoperative prognosis factors in gastric cancer patients has clinical importance. To this end, serum markers are easy to measure and are useful for diagnosis, predicting survival rates, and monitoring recurrence following surgery [2, 3].
The close correlation between inflammation and cancers was first discovered by Virchow in 1863 [4]. The systemic inflammatory response plays an important role in cancer development and progression. Pretreatment serum-based inflammatory indicators such as the neutrophil–lymphocyte ratio (NLR), the modified Glasgow prognostic score (mGPS), platelet–lymphocyte ratio (PLR), and C-reactive protein (CRP) can be derived from routine tests and have been associated with prognosis in various types of cancer [5, 6]. Recently, the ratio of C-reactive protein to albumin (CRP/Alb ratio) has been identified as another serum-based inflammatory indicator. An elevated CRP/Alb ratio was linled to poor prognosis in septic patients [7, 8]. In addition, several studies have reported that the CRP/Alb ratio is significantly related to prognosis in various types of cancers [9, 10]. However, whether the CRP/Alb ratio is associated with prognosis in gastric cancer patients has not yet been elucidated.
The CRP/Alb ratio is believed to reflect nutritional status as well as inflammation status of patients because serum albumin level is included in this marker. Since it has been well known that immune status is also closely correlated with prognosis in various types of cancers, we speculate that combination analysis of the CRP/Alb ratio and some markers that reflect patient’s immune status can predict the prognosis of patients more accurately than single usage of the CRP/Alb ratio. To this end, we used NLR because NLR includes lymphocyte count and is believed to reflect patient’s immune status as well as inflammation status. In this regard, Choi et al. [11 ] recently demonstrated that NLR was associated with the density of CD4 + T cells within the tumor microenvironment, indicating the close correlation between NLR and patient’s immune status. Therefore, the aim of this study was to validate the prognostic significance of combination of the CRP/Alb ratio and NLR in gastric cancer patients.
Materials and methods
This study was based on a retrospective analysis of 453 gastric adenocarcinoma patients who underwent curative gastrectomy (R0 resection) at our institution between January 2005 and December 2013. The clinicopathologic findings were determined according to the Japanese Classification of Gastric Carcinoma [12]. All patients underwent either distal partial, proximal partial, or total gastrectomy with regional dissection of the lymph nodes. Five patients were treated with neoadjuvant chemotherapy, 64 patients were treated with adjuvant chemotherapy, and 10 patients were treated with perioperative chemotherapy. No patients were treated by radiation therapy. Patients were periodically checked for early recurrence by diagnostic imaging including chest X-ray, double-contrast barium meal study, upper gastrointestinal fiberscopy, ultrasonography, and computed tomography. Causes of death and patterns of recurrence were determined by reviewing medical records including laboratory data, ultrasonography, computed tomography, scintigrams, peritoneal punctures, and laparotomies, or by direct inquiry with family members. In some cases, postmortems were undertaken to determine the cause of death. The detailed cause of death for every patient in the current study was known. At the time of analysis, the median follow-up of the 343 surviving patients was 61.9 months. Of the 110 deaths, 53 were related to recurrence of gastric cancer, 57 to an unrelated malignancy and an unrelated disease or accident. The patients who died of other disease (74.5 ± 8.9) were significantly older than either the patients who were alive (66.2 ± 12.0: P < 0.0001) or those who died of recurrence of gastric cancer (69.8 ± 10.9; P = 0.013).
From the patient’s records, we collected preoperative blood test data including serum Alb (g/dL), CRP (mg/dL), and total peripheral blood lymphocyte and neutrophil counts (/mm3). The CRP/Alb ratio was defined by dividing the CRP level by the Alb level. The NLR was defined by dividing the peripheral neutrophil count by the peripheral lymphocyte count. We obtained institutional review board approval for this study, and the informed consent requirement was waived.
Statistical analysis
The Mann–Whitney U test was used to evaluate the differences between the two groups. The Youden index was calculated using the receiver operating characteristic (ROC) analysis to determine an optimal cutoff value for CRP/Alb ratio and NLR for survival analysis. Survival curves were calculated according to the Kaplan–Meier method. Differences between survival curves were examined with the log rank test. We used multivariate analysis of factors considered prognostic of overall survival (OS), with Cox’s proportional hazards model and a stepwise procedure. P < 0.05 was considered significant. GraphPad Prism (GraphPad Software, Inc., La Jolla, CA, USA) and Stat View (Abacus Concepts, Inc., Berkeley, CA, USA) software were used for the statistical analyses.
Results
The mean CRP/Alb ratio and NLR were 0.0856 (range 0.002–4.23) and 2.53 (range 0.52–13.0), respectively. There was a statistically significant weak correlation between the CRP/Alb ratio and the NLR (r = 0.19; P < 0.0001) (Fig. 1). Table 1 shows the correlations among the CRP/Alb ratio, the NLR, and clinicopathological variables in gastric cancer patients. There were statistically significant correlations between a high CRP/Alb ratio and age (P = 0.0004), tumor size (P = 0.02), depth of invasion (P = 0.012), and lymph node metastasis (P = 0.022). On the other hand, a high NLR was significantly correlated with age (P = 0.0027), tumor size (P = 0.0006), depth of invasion (P < 0.0001), lymphatic involvement (P = 0.0031), venous involvement (P = 0.0022), and stage of disease (P = 0.0024).
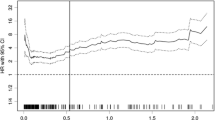
ROC curves were constructed for the survival status, and the area under the curve (AUC) values were compared to assess the discrimination ability of each inflammation-based prognostic score, including CRP/Alb ratio, NLR, and modified glasgow prognostic score (mGPS) (Fig. 2). Among the three prognostic scores, the CRP/Alb ratio had the highest AUC values, followed by the NLR and mGPS. We next investigated the prognostic significance of the CRP/Alb ratio and the NLR in patients with gastric cancer. ROC analysis showed the optimal cutoff value of CRP/Alb ratio and NLR to be 0.0232 (AUC 0.57–96, P = 0.0024) and 2.43 (AUC 0.585, P = 0.0071), respectively. Based on these results, patients were divided as follows: CRP/Alb ratio ≥ 0.0232 (CARHigh, n = 170), CRP/Alb ratio < 0.0232 (CARLow, n = 283), NLR ≥ 2.43 (NLRHigh, n = 197), and NLR < 2.43 (NLRLow, n = 256). The 5-year survival rates were 81.7 and 90.3% in patients with CARHigh and CARLow, respectively, and the difference was statistically significant (P = 0.026, Fig. 3a). Furthermore, 5-year survival rates were significantly related to NLR (NLRLow: 91.1%; NLRHigh: 82.1%; P = 0.041; Fig. 3b).
a Survival curves according to the CRP/Alb ratio. The 5-year survival rate was significantly worse in patients with CARHigh than in those with CARLow (81.7 vs. 90.3%, P = 0.025). b Survival curves according to the NLR. The 5-year survival rate was significantly worse in patients with NLRHigh than in those with NLRLow (82.1 vs. 91.1%, P = 0.041)
Among the 453 patients included in the current study, 95 were both CARHigh and NLRHigh (5-year survival: 59.6%); 179 were either CARHigh or NLRHigh (5-year survival: 75.8%); and 179 were both CARLow and NLRLow (5-year survival: 87.5%). The patients with both CARLow and NLRLow, those with either CARHigh or NLRHigh, those with both CARHigh and NLRHigh were assigned 0, 1, and 2, respectively. ROC analysis indicated that AUC of the combination of CRP/Alb and NLR for the survival status was 0.654, which was much higher than that of the individual use of either the CRP/Alb ratio or NLR (Fig. 4). The prognosis of the CARHigh/NLRHigh group was significantly worse than for those who were either CARHigh or NLRHigh, and the CARLow/NLRLow group (Fig. 5). Finally, multivariate analysis indicated that the combination of CRP/Alb ratio and NLR was an independent prognostic indicator (Table 2).
a Overall survival curves according to the combination of CRP/Alb ratio and NLR. The 5-year survival rates were 59.6, 75.8, and 87.5% in patients with both CARHigh and NLRHigh, either CARHigh or NLRHigh, and both CARLow and NLRLow, respectively. The difference was statistically significant (P < 0.0001). b Disease-specific survival curves according to the combination of CRP/Alb ratio and NLR. The 5-year survival rates were 76.0, 89.6, and 91.4% in patients with both CARHigh and NLRHigh, either CARHigh or NLRHigh, and both CARLow and NLRLow, respectively. The difference was statistically significant (P = 0.0013)
Discussion
Recent reports suggest that the outcomes for cancer patients are determined by tumor-related factors as well as by patient-related factors. Several studies have demonstrated that depth of invasion and lymph node metastasis are the most important tumor-related prognostic factors in gastric cancer [13, 14]. Inflammation, malnutrition, and immune status are patient-related factors related to prognosis in gastric cancer patients.
In the current study, we demonstrated that the CRP/Alb ratio, thought to be an inflammatory marker, was significantly related to prognosis in gastric cancer patients. CRP is an acute-phase reactant produced by hepatocytes and regulated by inflammatory cytokines, particularly IL-6 [15]. CRP is one of the most frequently used serum markers to evaluate inflammation status in patients and has been identified as a prognostic indicator in gastric cancer patients [16]. Because a high CRP/Alb ratio reflects high serum level of CRP, it follows that the CRP/Alb ratio would also be a prognostic indicator in gastric cancer patients. A high CRP/Alb ratio also reflects decreased serum level of albumin. Serum albumin is produced in the liver and the most abundant blood plasma protein. Serum albumin is the traditional standard factor used to assess a patient’s nutritional status. Gastric cancer often causes lumen obstruction and bleeding, which leads to malnutrition and increases a patient’s risk of postoperative complications. Therefore, it is important to improve a patient’s preoperative nutritional status before surgery to avoid postoperative complications. There are several assessment tools used in nutritional evaluation including the subjective global assessment (SGA) [17], the mini nutritional assessment (MNA) [18], and the Nutritional Risk Screening 2002 (NRS 2002) [19]. Onodera’s prognostic nutritional index (PNI) is also used to evaluate patient’s nutritional assessment. It is a simple index calculated by serum albumin and total lymphocyte count and has been proposed as a prognostic indicator in patients with gastrointestinal malignancies [20]. Recent report indicated that PNI could predict the prognosis of gastric cancer patients [21]. CRP/Alb ratio is an indicator that reflects both inflammation and nutritional status that are closely related to prognosis of gastric cancer patients.
The mGPS is also a serum-based inflammatory indicator that is assessed by both serum albumin level and CRP. Previous studies have suggested that the mGPS represents potential prognostic indicators in various types of cancers including gastric cancer [22,23,24]. Of note, although both mGPS and CRP/Alb used the same variables, the CRP/Alb ratio predicts the patients’ outcomes more precisely than the mGPS due to the presence of continuous variables. In addition, Kinoshita has speculated that the mGPS may overestimate or underestimate the inflammatory status because it scores the serum CRP and Alb value separately [9]. Conversely, the CRP/Alb ratio integrates the CRP and Alb values more reasonably, thus decreasing the potential overestimate or underestimate. In fact, it has been demonstrated that the CRP/Alb ratio was a more useful prognostic indicator in pancreas cancer [25] and hepatocellular carcinoma [9] than the mGPS. Furthermore, our study also demonstrated that the CRP/Alb ratio was more useful than the mGPS at predicting the prognosis of gastric cancer.
The NLR is another serum-based inflammatory indicator shown in several studies to be a useful prognostic indicator in various types of cancers [26,27,28]. A high NLR reflects a decreased lymphocyte count, as well as an increased neutrophil count. Neutrophil and lymphocyte are related to inflammation and immune activity, respectively. Although both the CRP/Alb ratio and NLR are considered to be serum-based inflammatory indicators, our results indicated that the correlation between the CRP/Alb ratio and NLR was very weak. Therefore, we hypothesized that the combination of CRP/Alb ratio and NLR might be more useful in predicting the prognosis of gastric cancer patients than the individual use of either the CRP/Alb ratio or NLR. In fact, AUC of the combination of CRP/Alb ratio and NLR was much higher than that of the individual use of either the CRP/Alb ratio or NLR, indicating that the combination of CRP/Alb ratio and NLR might be more useful to predict the prognosis of gastric cancer patients than the individual use of either the CRP/Alb ratio or NLR. Furthermore, our results clearly demonstrated that the combination of CRP/Alb ratio and NLR was an independent prognostic indicator. The combination of CRP/Alb ratio and NLR makes it possible to evaluate inflammation, immune status, and nutritional status, which are all thought to be patient-related prognostic factors.
There are a few limitations in the present study. First, because our study was retrospective there is some bias. In particular, the majority of the patients included in this study are early gastric cancer. Therefore, the usefulness of the combination of the CRP/Alb ratio and NLR as prognostic indicator in advanced gastric cancer patients should be confirmed in a large-scale study. Second, we used 0.0232 as a cutoff value for the CRP/Alb ratio. In other studies, Kinoshita et al. [9] used 0.037 and Haruki et al. [29] used 0.03, suggesting the optimal cutoff value of the CRP/Alb ratio might be dependent on the type of cancer. It will be important to determine an optimal cutoff value of the CRP/Alb ratio for routine clinical usage by analysis of a large-scale study. Third, the number of patients included in the current study was small; therefore, a large-scale, prospective, randomized, controlled trial is needed to confirm the results. Fourth, it is known that inflammation, immune status, and nutritional status are influenced by aging. In the current study, AUC of the combination of the CRP/Alb ratio in patients aged 75 and more (AUC 0.620; P = 0.025) was much lower than that in those aged under 75 years (AUC 0.688; P < 0.0001). Therefore, it is unclear that the combination of the CRP/Alb ratio and NLR is useful to accurately predict the prognosis of elderly gastric cancer patients. Therefore, the usefulness of the combination of the CRP/Alb ratio and NLR as prognostic indicator in elderly patients should be confirmed in a large-scale study.
In conclusion, the combination of the CRP/Alb ratio and NLR appears to be useful in predicting prognosis in gastric cancer patients. Because serum markers are a quick, easy, and noninvasive assay, measuring the CRP/Alb ratio and NLR may be a useful biological marker in routine clinical settings.
References
Jemal A, Bray F, Center MM et al (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Park HJ, Ahn JY, Jung HY et al (2014) Clinical characteristics and outcomes for gastric cancer patients aged 18–30 years. Gastric Cancer 17:649–660
Nam DH, Lee YK, Park JC et al (2013) Prognostic value of early postoperative tumor marker response in gastric cancer. Ann Surg Oncol 20:3905–3911
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420:860–867
Kinoshita A, Onoda H, Imai N et al (2012) Comparison of the prognostic value of inflammation-based prognostic scores in patients with hepatocellular carcinoma. Br J Cancer 107:988–993
Inoue D, Ozaka M, Matsuyama M et al (2015) Prognostic value of neutrophil–lymphocyte ratio and level of C-reactive protein in a large cohort of pancreatic cancer patients: a retrospective study in a single institute in Japan. Jpn J Clin Oncol 45:61–66
Fairclough E, Cairns E, Hamilton J et al (2009) Evaluation of a modified early warning system for acute medical admissions and comparison with C-reactive protein/albumin ratio as a predictor of patient outcome. Clin Med (Lond, Engl) 9:30–33
Ranzani OT, Zampieri FG, Forte DN et al (2013) C-reactive protein/albumin ratio predicts 90-day mortality of septic patients. PLoS ONE 8:e59321
Kinoshita A, Onoda H, Imai N et al (2015) The C-reactive protein/albumin ratio, a novel inflammation-based prognostic score, predicts outcomes in patients with hepatocellular carcinoma. Ann Surg Oncol 22:803–810
Xu XL, Yu HQ, Hu W et al (2015) A novel inflammation-based prognostic score, the C-reactive protein/albumin ratio predicts the prognosis of patients with operable esophageal squamous cell carcinoma. PLoS ONE 10:e0138657
Choi Y, Kim JW, Nam KH et al (2017) Systemic inflammation is associated with the density of immune cells in the tumor microenvironment of gastric cancer. Gastric Cancer 20:602–611
Japanese Gastric Cancer Association (2011) Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 14:101–112
Bozzetti F, Bonfanti G, Morabito A et al (1986) A multifactorial approach for the prognosis of patients with carcinoma of the stomach after curative resection. Surg Gynecol Obstet 162:229–234
Maruyama K (1987) The most important prognostic factors for gastric cancer patients. Scand J Gastroenterol 22:63–68
Morris-Stiff G, Gomez D, Prasad KR (2008) C-reactive protein in liver cancer surgery. Eur J Surg Oncol 34:727–729
Yu Q, Yu XF, Zhang SD et al (2013) Prognostic role of C-reactive protein in gastric cancer: a meta-analysis. Asian Pac J Cancer Prev APJCP 14:5735–5740
Detsky AS, McLaughlin JR, Baker JP et al (1987) What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr 11:8–13
Vellas B, Villars H, Abellan G et al (2006) Overview of the MNA—its history and challenges. J Nutr Health Aging 10:456–463 (Discussion 463–455)
Kondrup J, Rasmussen HH, Hamberg O et al (2003) Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr 22:321–336
Onodera T, Goseki N, Kosaki G (1984) Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients. Nihon Geka Gakkai Zasshi 85:1001–1005
Nozoe T, Iguchi T, Egashira A et al (2011) Significance of modified Glasgow prognostic score as a useful indicator for prognosis of patients with gastric carcinoma. Am J Surg 201:186–191
Imaoka H, Mizuno N, Hara K et al (2016) Evaluation of modified glasgow prognostic score for pancreatic cancer: a retrospective cohort study. Pancreas 45:211–217
Nozoe T, Matono R, Ijichi H et al (2014) Glasgow prognostic score (GPS) can be a useful indicator to determine prognosis of patients with colorectal carcinoma. Int Surg 99:512–517
Hirashima K, Watanabe M, Shigaki H et al (2014) Prognostic significance of the modified Glasgow prognostic score in elderly patients with gastric cancer. J Gastroenterol 49:1040–1046
Liu Z, Jin K, Guo M et al (2017) Prognostic value of the CRP/Alb ratio, a novel inflammation-based score in pancreatic cancer. Ann Surg Oncol 24:561–568
Kim JH, Han DS, Bang HY et al (2015) Preoperative neutrophil-to-lymphocyte ratio is a prognostic factor for overall survival in patients with gastric cancer. Ann Surg Treat Res 89:81–86
Stotz M, Gerger A, Eisner F et al (2013) Increased neutrophil–lymphocyte ratio is a poor prognostic factor in patients with primary operable and inoperable pancreatic cancer. Br J Cancer 109:416–421
Kumar R, Geuna E, Michalarea V et al (2015) The neutrophil–lymphocyte ratio and its utilisation for the management of cancer patients in early clinical trials. Br J Cancer 112:1157–1165
Haruki K, Shiba H, Shirai Y et al (2016) The C-reactive protein to albumin ratio predicts long-term outcomes in patients with pancreatic cancer after pancreatic resection. World J Surg 40:2254–2260. https://doi.org/10.1007/s00268-016-3491-4
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Saito, H., Kono, Y., Murakami, Y. et al. Prognostic Significance of the Preoperative Ratio of C-Reactive Protein to Albumin and Neutrophil–Lymphocyte Ratio in Gastric Cancer Patients. World J Surg 42, 1819–1825 (2018). https://doi.org/10.1007/s00268-017-4400-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4400-1