Abstract
Anthropogenic and climatic changes are continuously altering the freshwater plankton, necessitating an evaluation of the complex structure of plankton communities to understand and mitigate these impacts. In this context, the present study focuses on evaluating the structure of plankton communities, specifically Phytoplankton Functional Groups (FGs) for assessing the environmental sensitivity of wetlands under changing scenario. These FGs are defined by shared adaptive features rather than taxonomic traits. Over the period from 2016 to 2018, two ecologically distinct wetlands were examined, analysing their phytoplankton FGs and their relationship with water quality parameters. Ecohydrological data revealed significant seasonal variations (p ≤ 0.05) in key parameters such as water depth, temperature, pH, electrical conductivity, dissolved oxygen, total alkalinity, total hardness, NO3-N, and PO4-P. Notably, there were no significant differences observed among the sampling stations within each wetland. A total of 125 phytoplankton genera/species were classified into 23 FGs in the open wetland and 22 FGs in the closed wetland. Spatial and seasonal analyses of dominant FGs suggested both wetlands were experiencing pollution pressures. This study highlights the powerful role of phytoplankton functional groups (FGs) as bioindicators of wetland health, uncovering pollution pressures. In open wetlands, 15 phytoplankton FGs with 36 key taxa (Indicator Value ≥ 40%) emerged as critical indicators, while in closed wetlands, only 10 FGs with 17 taxa were identified. To assess eutrophication, the occurrence of these indicator species was evaluated using BVSTEP function analysis. The study recommends pollution reduction in catchment areas and restoration of riverine connectivity to enhance FG diversity. Phytoplankton FG methodologies are deemed effective for assessing the environmental sensitivity of wetlands significantly impacted by human activities. This research offers a scientific foundation for the evaluation and restoration of wetland ecosystems.
Highlights
-
Phytoplankton functional groups (FGs) serve as precise bioindicators of wetland environmental sensitivity and pollution pressures.
-
Seasonal and spatial assessments of water quality reveal insights into wetland ecological health.
-
The response of phytoplankton FGs to environmental factors effectively indicates water quality.
-
Key plankton species were identified as indicators of wetland ecological sensitivity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Inland freshwater ecosystems and their finite resources are increasingly affected by climatic and anthropogenic factors. Ecology plays a crucial role in understanding the distribution and interactions of organisms within their environment. Rapid environmental changes, including reduced water depth, extreme events, pollution, eutrophication, and ecological degradation (Sarkar et al. 2020; Zhang et al. 2019; Zhu et al. 2021; Kumari et al. 2023), are growing concerns. Wetlands, as primary renewable water resources, provide diverse services that enhance livelihoods (Sarkar et al. 2020). Shallow lakes, dynamic aquatic ecosystems, are significantly impacted by rising temperatures and nutrient fluctuations, leading to rapid phytoplankton succession (Allende et al. 2019; Poniewozik & Lenard 2022). Phytoplankton abundance and diversity are highly responsive indicators of ecological changes in aquatic environments (Gogoi et al., 2021; Mohanty et al. 2021). While traditional research has focused on phytoplankton assemblages and diversity for monitoring various aquatic ecosystems (Sarkar et al. 2020; Gogoi et al., 2021; Mohanty et al. 2021), these methods do not fully capture ecosystem functions. Understanding the functional roles and structural adaptations of phytoplankton is crucial for comprehending their ecological interactions (Reynolds et al. 2002). Taxonomic classification provides morphological and genetic information but falls short in understanding ecological functional similarities.
The categorization of phytoplankton based on functional roles and morphology has been explored by ecologists in freshwater (Salmaso et al. 2015; Allende et al. 2019; Wang et al. 2021; Abonyi et al. 2021) and marine ecosystems (Xu et al. 2016). For instance, within Bacillariophyta, various cell forms and life strategies from unicellular to colonial and filamentous—are documented. Ecologists have sought to address this complexity by grouping organisms based on functional characteristics such as size, form, nutrient requirements, life strategies, distribution patterns, and common structural and functional features.
The FG classification of phytoplankton has benn primarily fostered to understand how environmental factors shape the functional composition of phytoplankton (Reynolds et al. 2002). This FG approach allows for the distinction between “natural constraints” and “human impacts” on phytoplankton, facilitating the evaluation of a river’s ecological status (Yan et al. 2023). The concept gained traction as ecological understanding of phytoplankton species’ functional qualities improved. Today, the approach proves effective in understanding seasonal changes in phytoplankton communities and assessing the ecological status of aquatic ecosystems based on physiological, morphological, and environmental distinctiveness (Dochin, 2019; Latinopoulos et al. 2020). Initially, identified 14 phytoplankton functional groups, later expanded to 31 (Reynolds et al., 2002), and further to over 40 groups (Padisak et al. 2009). Methods for classifying functional groups based on shared ecological features and functional connectivity have since been refined (Reynolds et al. 2002; Padisak et al. 2009; Salmaso et al. 2015; Wang et al. 2021). The concept of phytoplankton functional groups (FGs) has been widely applied to assess the ecological status of diverse aquatic habitats (Borics et al., 2014; Toporowska et al. 2018; Varol, 2019; Allende et al. 2019; Latinopoulos et al. 2020; Wang et al. 2021).
This study focused on two shallow oxbow lakes in eastern India, each with distinct morphological features and significant human impact. These lakes serve multiple purposes beyond fisheries enhancement, making them ideal for examining phytoplankton and identifying key phytoplankton functional groups (FGs). Anthropogenic and climatic changes are driving significant alterations in freshwater plankton diversity, specifically within phytoplankton Functional Groups (FGs). These changes are reflected in the spatial and seasonal variations in phytoplankton community structure, which are closely linked to water quality parameters. By analyzing phytoplankton FGs as bioindicators, it is possible to detect pollution pressures and assess the ecological health of wetland ecosystems. This study hypothesizes that the diversity and composition of phytoplankton FGs are sensitive indicators of environmental changes in wetlands, and that effective management strategies can mitigate these impacts by enhancing FG diversity through pollution reduction and restoration efforts. By gathering ecological data on these lakes, this study contributes to the understanding of their environmental sensitivity and provides essential insights for informing government policies on aquatic ecosystem health management. The findings establish a foundation for further ecological assessments and efforts to improve and restore these unique shallow lakes.
Materials and Methods
Study Area and Sampling Sites
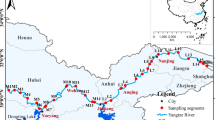
The wetlands 25059'54.02“N 88038'27“E (Khalsi) and 23005'14.14“N 88042'56.22“E (Akaipur) located in West Bengal, are two distinct wetlands and productive in terms of fisheries (Fig. 1). The water spread area of Khalsi ranges from 36 to 65 hectares, while Akailpur varies between 20 and 28 hectares. Our study spanned from March 2016 to February 2018, with seasonal sampling across three stations in each wetland: pre-monsoon, monsoon, post-monsoon, and winter. These ecosystems are nestled among farmlands and residential areas. They support capture-based and culture-based fisheries, sustaining approximately 300 families in Akaipur and 500 in Khalsi (Meena et al., 2019). Both wetlands lie within the lower Ganga basin and are affected by agricultural runoff, domestic waste, jute retting, silt from monsoon floods, and other human activities. Akaipur is a closed wetland with no river connection, while Khalsi reconnects to its parent river during the monsoon. Their abundant macrophytes provide refuge and breeding grounds for various indigenous fish species. The determination of sampling sites in both lakes was done in such a way that the sample has a possible resemblance to the present ecological status of the lakes. Khalsi (seasonally open wetland) Sector I is the middle zone (free from macrophytes), Sector II is open with a river channel and infested with Eichhornia hyacinth, and Sector III is dominated by Nymphaea. In the closed wetlands wetland (Akaipur), station 1 surroundings are dominated by human habitation Station II clear zone free from macrophytes, and Station 3 is dominated by macrophytes and surrounding agricultural crop fields.
Water Sampling
Water samples were collected from each sampling site to assess various water quality parameters. Water temperature (Temp.), pH, and electrical conductivity (EC) were measured in the field using a multi-parameter probe (Eutech PCS Test 35). Transparency (SD) was determined using a Secchi disc. Dissolved oxygen levels were measured on-site using the modified Winkler titrimetric method (Strickland and Parsons, 1972). Additionally, total alkalinity (TA), total hardness (TH), and nutrient parameters such as nitrate-nitrogen (NO3-N) and phosphate-phosphorus (PO4-P) were analyzed following standard methodologies outlined by the American Public Health Association (APHA, 2012). Chlorophyll a (Chl a) concentration was determined using the standard spectrophotometric method with a HACH Spectrophotometer (DR 2800, Germany) in accordance with APHA (2012) guidelines. In addition, the WQI was calculated based on the estimated water quality parameters (water temperature, pH, electrical conductivity, dissolved oxygen, total alkalinity, total hardness, NO3-N and PO4-P) to understand the grade of water suitability for human use and aquatic biodiversity and propagation. Here, we followed the Weighted Arithmetic Water Quality Index (WAWQI) (Brown et al., 1972) for WQI calculation. The degree of purity or potability of the water variables was specified by the Bureau of Indian Standards (BIS, 2012). The WAWQI rating is 0–25: Excellent; 26–50: Good; 51–75: Poor; 76–100: Very poor and >100: unfit for consumption.
Phytoplankton Sampling
Subsurface phytoplankton samples (0.5 meters below the surface) were collected seasonally across four distinct period: pre-monsoon (March–June), monsoon (July-September), post-monsoon (October-November), and winter (December–February) from April 2016 to March 2018. For qualitative and quantitative study of phytoplankton, water samples were collected by using a hand-held plankton net of mesh size 20 μm mesh size bolting silk cloth. 50 L volume of sub-surface water was filtered from each sampling location and the concentrated sample was transferred to a 50 ml plastic container and stabilized in situ with Lugol’s iodine solution (1.5% v/v). fixed into 4% buffered formalin. The algal cell count was enumerated using the Sedgwick–Rafter counting cell method by using a trinocular light microscope ‘Axioster plus − Carl Zeiss’(Zeiss 37081). Taxa were identified and classified following standard literature (Prescott, 1962; Ward and Whipple, 1992; Cox, 1996; Belinger and Sigee, 2010). The updated algal taxonomic nomenclature was reaffirmed by following the Algae Base (Guiry and Guiry 2020). Phytoplankton biomass was assessed following the geometric approximation of each taxon (Hillebrand et al. 1999) and converted into biomass (1 mm3 ≈ 1 mg fresh weight). Phytoplankton biomass was assessed using geometric approximations for each taxon (Hillebrand et al. 1999) and converted into biomass (1 mm3 ≈ 1 mg fresh weight). Phytoplankton taxa were classified into functional groups (FGs) based on criteria established by Reynolds et al. (2002), Padisak et al. (2009), and Borics et al. (2020).
Statistical Analysis
The summary statistics (mean and standard deviation) of the abiotic and biotic data were performed in MS Excel 2010. The hydro chemical parameters in different seasons for both wetlands were subjected to one-way analysis of variance (ANOVA) and post hoc test (Duncan’s) using SPSS v.21. To comprehend the major water variables in the studied wetlands, principal component analysis (PCA) was performed. In addition, we performed permutational MANOVA (PERMANOVA) to test the differences (p ≤ 0.05) between the phytoplankton FGs (biomass value) with respect to seasons and stations for both wetlands. To examine the similarity of community structure between FGs, non-metric multidimensional scaling (with the distance-based trajectory) was carried-out using ‘Bray-Curtis’ similarity measures. Pre-treatment of all biological data was done by square root transformation before analysis. Canonical correspondence analysis (CCA) between biotic (FGs) and the abiotic variables (10 water variables) was performed to examine their relationship for open and closed wetlands. In addition, distance- based linear model (DistLM) was performed using the distance−based redundancy analysis constrained ordination (dbRDA). Then we obtained the model using the AICc selection criteria and the stepwise selection procedure. The indicator species analysis was carried-out to identify the potential indicator species of phytoplankton in the seasonally open and closed wetland environment in each season. An indicator value (IndVal) of ≥25% can be considered the threshold to identify the indicator taxa in this method, meaning that a taxon is present in ≥50% of the samples in a group and that its relative abundance in each group is ≥50% (Hunt and Hosie, 2006). All multivariate analyses were performed using statistical tools such as PRIMER version 6.1.6 and PAST version 4.08.
Result
Water Quality Parameters
The water quality parameters of open and closed wetlands are presented in Table 1, revealing significant variations between them, as indicated by PERMANOVA analysis (F = 5.673; p = 0.01). Maximum water depth was observed during the monsoon season in open wetlands and during winter in closed ones, with significant seasonal differences (p < 0.05) in both. Transparency was lower in closed wetlands but not significantly (p > 0.05), while temperature variations were significant across seasons, especially in closed wetlands. Water remained alkaline in both wetlands with pH values between 7.7 and 8.7, and electrical conductivity (EC) was significantly higher during pre-monsoon in open wetlands. Nutrient concentrations varied significantly between seasons, with nitrate-nitrogen (NO₃–N) highest during the monsoon in open wetlands and during pre-monsoon in closed ones, and phosphate-phosphorus (PO4-P) levels highest in winter for open wetlands and monsoon for closed ones. Principal Component Analysis (PCA) revealed that axes 1 and 2 explained 61.8% of the seasonal variation in open wetlands and 58.0% in closed wetlands, with pH, temperature, EC, TA, NO₃–N, and water depth being key variables (Fig. 2a, b). Based on the calculated value of Water Quality Index (WQI), the water quality was found to be ‘good’ and ranged from 24.92–31.65 and 29.77–35.54 for Khalsi and Akaipur wetlands, respectively. While comparing both wetlands, the index value was comparatively higher in the closed wetland, indicating that the closed wetland had slightly low water quality than the open one.
Phytoplankton FGs Biomass Composition
In the present study, a total number of 125 phytoplankton genera/species was classified into 23 phytoplankton FGs – TB, P, G, X1, T, S3, J, X2, N, H2, H1, M, S2, K, Lo, MP, TD, S1, W1, W2, C, D, F of which 22 FGs from the closed wetland. W2 FG is absent from the closed wetland (Supplementary file: Table S2). Single species Trachelomonas sp. of functional group W2 was dominant during the post-monsoon season in open wetland. Phytoplankton FGs biomass (mgL−1) of seasonally open wetland was calculated highest (90.92 ± 19.56) in the post-monsoon season and lowest (26.38 ± 8.63) in the monsoon season. The average biomass of FGs was higher in open wetlands than the closed ones. In the closed wetland, phytoplankton FGs biomass (mgL−1) was slightly higher (29.52 ± 16.23) in winter season than post-monsoon season (29.21 ± 14.86) and lowest (15.11 ± 6.78) in pre-monsoon season (Fig. 3). We have also studied the assemblage pattern of FGs in the different sampling sites in both closed and open wetlands to know the intra variations of wetlands. The finding is very interesting, among the 22 FGs recorded from closed wetland dominance of Functional group M (more than 40% contribution) followed by K (more than 20% contribution) in all three sectors. Whereas in seasonally open wetland, more homogeneous distribution of FGs, but dominance of M (more than 20%) contribution in Sector III and I but in the clear zone (Sector II) dominance of J followed by P (Fig. 4). PERMANOVA analysis exhibited a significant variation of phytoplankton composition between seasons (Open: F = 2.116; p = 0.016; Closed: F = 2.1331; p = 0.022) and insignificant between stations in both the wetlands.
FGs-wise seasonal distribution pattern was assessed for both open and closed wetlands. In open wetland, 23 FGs have been recorded throughout the study. Their contribution to each season has been given in Fig. 5. TD was the most dominant FG during Pre-monsoon followed by X2 and H2. Twenty-one FGs have been recorded from post-monsoon except S1 and X1. The lowest contribution (<2%) was made by F during the post-monsoon season. Biomass of FGs – T and F (more than 70%) was contributed during the winter season followed by S1, C, and N. Except for these two FGs–TD and S3 rest were recorded during the winter season. FG–TD was found to be most dominant throughout the study period in open wetland.
In closed wetlands, the dominance of FGs–S1, S2, S3, H2, X1, MP in pre-monsoon season. The contribution of FGs–G, N, H1, and Lo were almost negligible in pre-monsoon. Single FG S1 was found to be most dominant in the pre-monsoon season. The FGs such as N and TD were found to be the most prevailing Group (contributing more than 95%) followed by T and D in the post-monsoon season. H2 is the only FG that has not been recorded during the post-monsoon season. FGs–Lo, X2, and G were found to be dominating during the monsoon season while F was in the winter season. The FGs–G, X1, T, X2, N, TD, S1 and D were least or not contributing to the biomass of winter seasons (Fig. 5).
Non-metric multidimensional scaling based on Bray-Curtis similarity matrix for the FGs in open wetland showed overall 54% similarity of the phytoplankton FG (biomass) and their resemblance increased steadily from monsoon to winter and pre-monsoon (Fig. 6a). Likewise, the overall similarity of phytoplankton biomass in the closed wetland was slightly lower at 43% (Fig. 6b). In addition, the FGs biomass in the studied wetlands was multimodal as samples in both the wetlands were widely separated in the same season. The cumulative dominance curve (k − dominance curve) extracted for the seasonally open wetland reflected that the biomass of phytoplankton functional groups was noticeably higher during the monsoon than in other seasons (Fig. 7a). However, in the closed wetland, the plotted curves indicate similar pattern of dominance (biomass) with their overlapping nature (Fig. 7b). In addition, the nature of phytoplankton biomass between post-monsoon and winter period is closely resembles than the seasons between pre-monsoon and monsoon. In winter, the biomass of the phytoplankton functional groups in the closed wetland increased substantially.
Relations of the Phytoplankton FGs to Environmental Factors
Canonical Correspondence analysis was performed between water variables (depth, transparency, SD, pH, EC, DO, TA, TH, NO3-N, PO4-P) and phytoplankton functional groups (biomass value) to comprehend the interactions between the factors. The eigen value and percent variation were calculated, and the scaler explained 0.17616 and 40.04% correlations in axis 1 and 0.10613 and 24.73% correlations in axis 2 in the data set. The variables TA, Temp., depth, SD and nutrients (NO3-N, PO4-P) had close affinity with all the functional groups in the open wetland. Water variables such as TA, TH, pH and depth were positively associated in Axis 1 and SD, Temp., pH, NO3-N, PO4-P in axis 2. Axis 1 scores 10 functional groups (G, X2, N, H2, H1, K, Lo, TD, S1 and F) and is more closely related to TA, TH, EC, pH and NO3-N. Similarly, axis 2 scores six groups (S3, W2, X2, Lo, TD and W1) and showed strong affinity for depth, SD, Temp. and PO4-P. Functional groups (TB and X1) showed a negative correlation in both axes. Overall, water depth and transparency showed a strong negative affinity with all functional groups in the open wetland (Fig. 8). In the close wetland, CCA revealed 35.13% and 20.76% variation of the data set with the eigen value 0.1488 and 0.0879 in axis 1 and 2, respectively. Axis 2 achieves higher number of functional groups (TB, C, D, MP, T, H2, S1 and H) and shows a correlation with Temp., depth, TH, EC and SD. The functional groups – W, J, TB, C, D and T exhibited positive correlation in both the axis unlike F and K. Water variables Temp. and total alkalinity acted as effective factors for functional groups TB, J, T, D and C. Similarly, TD, X1, and S3 showed a strong positive correlation with depth. It was also shown that nutrient parameters (NO3-N, PO4-P) had an overall negative influence on all functional groups in the closed wetland (Fig. 8). The NMDS trajectory showed at the similarity of samples biomass increased from winter to pre-monsoon for both the wetlands (Fig. 8). In addition, to quantify the explained variables, distance based linear model (DistLM) was performed to obtain a significant correlation between the phytoplankton functional groups and each of the environmental variables. The linear model explained 72.11% (axes 1 and 2) with the predictor variables TA (F = 4.4111, p = 0.001), Temp. (F = 3.2371, p = 0.008), PO4-P (F = 3.4139; p = 0.009) and NO3-N (F = 2.8967, p = 0.043) with the AICc ( = 170.34) and R2 = 0.47 in the open wetland. Similarly, in the closed wetland also explained 67.42% variation with 6 environmental variables (step-wise selection) viz., depth, SD, Temp. EC, TH and PO4-P could explain the discrepancies in phytoplankton biomass in the studied stations. The fitted model explained AICc value of 138.78 with R2 ( = 0.80675).
Seasonal Occurrence of Indicator FGS
The seasonal occurrence of indicator species among different FGS (IndVal) for wetland sensitivity was analyzed and species contributing more than 30% were considered as Indicator species for both the ecosystem (open and closed wetland). The best set of 70 phytoplankton species found to be near a perfect match in the seasonally open wetland. Likewise, a set of 38 species of phytoplankton were found in the closed wetland, which could be potential indicator species. Based on the IndVal analysis (>30% threshold value), a total of 92 phytoplankton species were recognized as key indicators across seasons, considering both wetlands (Table 2).
In the open wetland, a total of 9 (J, H1, TB, H2, TD, X2, K, T, D) phytoplankton FGs were represented as indicators during the pre-monsoon period followed by 8 in post-monsoon. The maximum number of phytoplankton species (36 taxa) was marked as indicator species (IndVal ≥ 40%) for open werland. However, the lowest FGs were found in winter with 2 Phytoplankton FGs (F and TB). In the closed wetland, the FGs indicator was 10 Phytoplankton FGs, comparatively lower than in the open wetland 15 FGs with IndVal (≥40%). In contrast to open wetland, maximum FGs (9 FGs–J, S2, TB, TD, X2, G, W1, T, D) during the post-monsoon period. The indicator species that reached IndVal (≥ 40%) were Aphanothece sp., Caloneis sp., Centritractus sp., Fragilaria sp., Mougeotia sp. Phacus sp., Pinnularia sp., Scenedesmus longus, and Synedra sp. While in the monsoon season, only 2 FGs characterized as IndVal (≥ 40%) represented by two taxa Coelastrum reticulatum (FG-J) and Gomphospheria sp. (FG-K). The indicator species were somewhat lower in both the wetlands during winter season. The IndVal (>40%) was characterized by 2 taxa i. e., Rhopalodia sp. (TB) and Westella sp. (F) in the open wetland and only one species occurs in the closed wetland under FGs (F) during winter. Quantitatively several phytoplankton species characterized by IndVal (≥ 30%) as indicator species Caloneis sp., Coelastrum sp., Coscinodiscus sp., Dictyosphaerium sp., Euastrum sp., Euglena elongate, Penium sp., Rhodomonas sp., Synedra sp., Synedra ulna, Tetrastrum sp., Ulothrix sp. are common in both wetlands. The FGs for common indicators are depicted in Table 2.
Discussion
The human activities such as agricultural runoff, urbanization, industrial discharges, and aquaculture practices significantly affect the water quality and planktonic algae in the wetlands of West Bengal’s wetlands. The wetlands in West Bengal often bordered by agricultural lands, where the use of fertilizers cuses nutrient-rich runoff (particularly nitrogen and phosphorus) to enter these wetlands. These activities contribute to nutrient loading, pollution, and habitat alteration, leading to issues like eutrophication, toxic algal blooms, and changes in species composition (Bera et al. 2021). The study revealed that nutrient parameters such as NO₃-N were highest in both wetlands during the monsoon season. However, the concentration was greater in open wetlands compared to closed ones. In contrast, PO₄-P levels peaked in open wetlands during the post-monsoon period but were subsequently lower than in closed wetlands. This difference is attributed to the increased decomposition process in open wetlands during the post-monsoon season, which is due to reduction in water outflow. Meanwhile, continuous decay of organic matter in closed wetlands resulted in higher PO₄-P concentrations throughout the seasons (Bao et al. 2023).
Electrical conductivity in wetlands tends to increase during both pre-monsoon and post-monsoon periods due to higher water saturation. Following the monsoon, the geochemical characteristics of surface water in these wetlands change, resulting in higher electrical conductivity, indicating shifts in ion concentrations and overall water quality (Baruah and Baruah 2024). Additionally, pH in wetlands often rise during the winter season, further affecting electrical conductivity (Gerla 2013). Interestingly, dissolved oxygen levels are lower in open wetlands compared to closed ones, likely due to higher DO consumption for decomposition of debris and organic matter, which is replenished during seasonal restoration of connectivity with the main river channel. In contrast, closed wetlands, due to their isolated nature, receive less debris. However, the lowest DO levels in closed wetlands are observed during the pre-monsoon period when water saturation is high and organic decomposition is intensified. WQI was comparatively higher in closed wetland than in open one, fall within the range of scale ‘good’ (BIS, 2012) as evident in our study.
Understanding the spatial distribution of phytoplankton functional groups is a key focus in ecology, and its importance is heightened by the current challenges of habitat alteration, fragmentation, wetland shrinkage, and ongoing biodiversity loss (Pinel-Alloul et al. 2013; Tinnevelt et al. 2022). The present study broadly contributes to understanding the diversity of phytoplankton functional groups and their ecohydrological interactions in two ecologically district wetlands. A large number of species based on similar taxonomic traits but having different ecological characteristics, their linkage and ecological functions are different in both the wetlands. Phytoplankton species were grouped based on their similarity in ecological adaptation and classified into 23 FGs for open and 22 FGs for closed wetlands for assessing ecological status. High fluctuation in water level and variation in water spread area are natural phenomena of the wetlands, seasonally open with parent River channel (Md. Aftabuddin et al. 2017). Flood riparian connection in wetlands is a prime factor for infusing lots of nutrients into the seasonally open wetland (Gogoi et al., 2021). Ecohydrological changes become drivers for changes in phytoplankton FGs diversity and increasing their biomass in post-monsoon (Townsend and Doughlas 2017). The average annual biomass of phytoplankton functional groups was higher in seasonally open than closed wetland. The spatial pattern of phytoplankton functional groups did not depict significant differences between the wetlands. However, ecohydrological spatial variation between wetlands is significant. In contrast, to the river ecosystem, Yang et al. (2017) water quality parameters become more favorable in late monsoon than the onset of monsoon in wetland ecosystem. This was due to the residential period of influx water from the catchment is higher in wetland than river ecosystem. FG biomass in monsoon was lowest in seasonally open wetland but comparatively higher in closed wetlands. This could be related to the connectivity of the main river channel and the mixing of nutrients leads to higher plankton diversity and biomass in open than closed wetlands (Gogoi et al., 2021). FG biomass was recorded highest during the post-monsoon season in open wetlands due to the increased growth of a few phytoplankton functional groups in the presence of favorable temperatures and availability of organic nutrients (Padisak et al. 2009; Reinl et al. 2022).
In terms of FG composition, TB was the most diverse group composition of 22 species. Generally, TB prefers clear lotic environment, low nutrient and considerably good, oxygenated water, representative species mostly diatoms include Mastogloia sp., Cymbella sp., Navicula radiosa, Navicula rhynchocephala, Navicula sp., Gomphonema gracile, Gomphonema costrictum, Amphipleura sp., Nitzschia sp. etc. Wang et al. (2021) has studied in estuarine river delta, reported TB dominates during the dry seasons. In the present finding, similarly, TB dominates during premonsoon season in open wetland, in contrast to closed wetland during post-monsoon season. The recent application of the Reynolds FG system to the middle Danube River has demonstrated its utility in identifying long-term compositional changes and potential regime shifts, but future advancements depend on developing quantitative parameters to model and quantify the effects of global warming and human impacts on river phytoplankton and ecosystem functioning (Abonyi et al. 2021). However, TB dominates during pre-monsoon in open wetland and post-monsoon in closed wetland but lower contribution in biomass. Occurrences of several FGs such as X1, H1, C, F, and X2 are reasonably higher but have less contribution in biomass. Gogoi et al., (2021) reported a lower contribution of Bacillariphyceae and Chlorophyceae during the monsoon season. The FGs such as G, X2, and Lo species Chlamydomonas sp., Volvox sp., Eudorina sp., Pandorina sp., Merismopedia sp. were contributing higher in closed wetland compared to open wetland. These species are an indicator of the stagnant organic pollution-rich ecological status of water (Cardoso et al. 2017; Reinl et al. 2022). The FG term W2, dominance of single species Trachelomonas spp. during the post-monsoon season in open wetland. This is one of the species that flourish in certain environmental conditions such as pH closer to neutral and organically rich conditions (Grabowksa and Wołowski 2013). In open wetland, pH was most suitable for their growth, and it was no longer supported by changes in environmental factors, hence was absent in closed wetland. The dominance in biomass of Functional group TD in the pre-monsoon season was influenced by the rise in temperature and reduction in depth in the open wetland and S1 in the closed wetland due to higher organic nutrients (Wanganeo and Wanganeo 1991). The biomass of functional groups TD, N, T, and D associated with Chlorophyceae and Bacillariophyceae was comparatively higher during the post-monsoon season in closed wetlands due to benign environmental water quality parameters and bioavailability of silica content in water (Sharma 2015; Gogoi et al., 2021). Biomass of functional groups of W1, W2, X1, J, S2, S3, and M contributes to individual peaks in post-monsoon season in open wetland due to higher individual biomass and abundance of species associated with the functional groups. In contrast to the finding of dominance of Euglenophyceae such as Trachelomonas sp. and Euglena sp. in closed wetlands, their abundance increased due to the presence of labile organic load in combination with seasonal alteration especially physical water quality parameters (Li et al. 2018). Non-motile green algae associated with F is dominant in both closed and open wetlands during the winter season due to more stable water and moderate nutrient concentration (Padisak et al. 2009). This implies that the diversity of functional groups is a potential indicator of spatial and temporal changes in aquatic ecosystems.
Phytoplankton species appeared as indicator species that vary seasonally in two different ecohydrological scenarios. There were no common taxa that appeared as indicator species during pre-monsoon with IndVal (≥30%). Therefore, it indicated that taxa present during this period are influenced by light intensity with higher temperature and bottom visible conditions in open wetlands as transparency equal to water depth. The abundance of taxa such as Ankistodesmus spiralis, Calothrix sp., Coelosphaerium sp., Coscinodiscus sp., Gleotrichia natans, Microchaete sp., Netrium digitus, Rivularia sp., Spirogyra vandalurensis, Surirella sp. Zygema sp., mostly benthic favored by shallow depth, good light, stagnant water, and moderately organic nutrient-rich conditions followed by higher temperature (Palmer 1980; Sin and Lee 2020). In the closed wetland, only filamentous algae appeared as the dominant indicator species Ulothrix sp. influenced by higher temperature and favorable nutrients in water. Chen et al. (2011) assessed the gut content of Daphnia sp. from February to May (summer) and found most dominant one was Ulothrix sp. However, the author has not confirmed the dominance of Ulothrix sp. in the gut content of Daphnia either due to its abundance in water or selectively fed upon. Riverine connectivity and their change in hydrological regimes during monsoon season are one of the driving factors (Marshall et al. 2005) for influencing the abundance of these species (Mastogloia sp., Microspora sp., Rhodomonas sp., Schroederia sp., Volvox sp., Tetraedron regulare, Synedra ulna) in open wetland and species (Coelastrum reticulatum, Gomphospheria sp.,) dominance IndVal (≥50%) in closed wetland. Thus, it revealed that taxa respond differently in varying monsoon water influx regimes. Belinger and Sigee (2010) found that Volvox sp., Synedra ulna, Microspora sp., and Rhodomonas sp., were more abundant during the wet season, which was consistent with our results. The result suggested that the highest number of indicator species (IndVal ≥ 30%) was recorded during post-monsoon in open wetland. This change was possible due to riverine connectivity becoming more stagnant and favoring nutrient utilization at suitable temperatures leading to an increase in the abundance of certain groups of algae. In shallow lakes, an abundance of nutrients is primarily phosphate as one of the limiting factors controlling the phytoplankton growth especially Chlorophyceae (Prusty et al. 2010). Increasing the algal biomass and abundance during the post-monsoon seasons in freshwater wetland ecosystems has suggested that environmental parameters and dissolving nutrients are driving factors of those changes (Sin and Lee 2020; Ray et al. 2023). The phytoplankton indicator species response in terms of IndVal was low in both wetlands during the winter season due to a reduction in nutrient concentration and grazing by zooplankton (Kumari et al. 2023). The present finding suggests that algal taxonomic structure shifts are likely due to monsoon flux in combination with nutrient and water temperature. Therefore, increasing temperature in freshwater wetland ecosystems may affect the eco-trophic structure and dominant harmful algal bloom.
Conclusion
In conclusion, our study highlights the crucial role of wetland connectivity with parent rivers in preserving ecological health and boosting plankton productivity. Open wetlands demonstrate greater variability in water quality parameters compared to closed ones, underscoring the importance of water mixing through link channels for sustaining plankton ecology. Re-excavation or dredging of blocked channels and reconnection to natural water sources can reestablish the water flow in the closed wetland. Controlled water management practices, like regulating sluice gates, can further maintain hydrological balance and improve water quality. The plankton community and biomass are essential for supporting sustainable fish production through ecosystem-based fisheries management, with phytoplankton functional groups (FGs) playing key roles in nutrient cycling, oxygen production, and maintaining ecological balance. Monitoring these groups is vital for safeguarding aquatic ecosystems. Canonical Correspondence Analysis (CCA) revealed significant relationships between water variables and phytoplankton FGs, highlighting their ecological interactions in both wetland types. Our study confirmed the value of identifying indicative phytoplankton species and FGs for assessing seasonal and spatial diversity. Using FGs as bioindicators can serve as early warnings of environmental changes, and the FG methodology effectively characterizes the spatiotemporal dynamics of aquatic environments. Continuous efforts to promote FG diversity and restore link channels are necessary to enhance the resilience and ecological health of aquatic ecosystems.
References
Abonyi A, Descy JP, Borics G et al. (2021) From historical backgrounds towards the functional classification of river phytoplankton sensu Colin S. Reynolds: what future merits the approach may hold? Hydrobiologia 848:131–142. https://doi.org/10.1007/s10750-020-04300-3
Allende L, Fontanarrosa MS, Murno A, Sinistro R (2019) Phytoplankton functional group classifications as a tool for biomonitoring shallow lakes: a case study. Knowl Manag Aquat Ecosyst 420(5):14. https://doi.org/10.1051/kmae/2018044
APHA (2012) Standard methods for the examination of water and wastewater. Rice, E. W. Baird, R. B., Eaton, A. D. & Clesceri, L. S. (Eds.). American Public Health Association (APHA), American Water Works Association (AWWA) and Water Environment Federation (WEF) (22nd ed.). Washington, D.C., USA.
Bao S, Jin L, Wu Q, Xie S, Xu J, Peng S, Liu C, Pan Y (2023) Effects of interspecific interactions on aquatic macrophyte litter decomposition and its influencing factors. ACS EST Water 3(12):3755–3766
Baruah D, Baruah PP (2024) Evaluation of water quality through water quality index at sorbhog beel, a critical ecotone along Indo-Bhutan boundary. Water Air Soil Pollut 235:525. https://doi.org/10.1007/s11270-024-07335-7
Belinger GE, Sigee CD (2010) Freshwater algae: Identification and use as bioindicators. Blackwell publishing. 271.
Bera B, Bhattacharjee S, Shit PK, Sengupta N, Saha S (2021) Anthropogenic stress on a Ramsar site, India: Study towards rapid transformation of the health of aquatic environment. Environ Chall 4:100158. https://doi.org/10.1016/j.envc.2021.100158.
Borics G, Abonyi A, Salmaso N, Ptacnik NR (2020) Freshwater phytoplankton diversity: Models, drivers, and implications for ecosystem properties. Hydrobiologia. https://doi.org/10.1007/s10750-020-04332-9.
Borics G, Görgényi J, Grigorszky I, László-Nagy Z, Tóthmérész B, Krasznai E, Várbíró G (2014) The role of phytoplankton diversity metrics in shallow lake and river quality assessment. Ecol Ind 45:28–36
Bureau of Indian Standards (BIS) (2012) Indian standard drinking water specification (second revision) BIS 10500:2012, New Delhi
Cardoso S, Nabout J, Farjalla V, Lopes PM, Bozelli R, Husza V, Roland F (2017) Environmental factors driving phytoplankton taxonomic and functional diversity in Amazonian floodplain lakes. Hydrobiologia 802:115–130. https://doi.org/10.1007/s10750-017-3244-x
Chen F, Gulati RD, Li J, Liu Z (2011) A comparison of the size distribution of the filamentous green alga Ulothrix in Daphnia guts and lake water from Lake Taihu, China. J Plank Res 33(8):1274–1283. https://doi.org/10.1093/plankt/fbr029
Cox EJ (1996) Identification of freshwater diatoms from live material (158). Chapman and Hall Publisher.
Dochin K (2019) Functional and morphological groups in the phytoplankton of large reservoirs used for aquaculture in Bulgaria. Bulgarian J Agricul Sci 25(1):166–175
Gerla PJ (2013) Can pH and electrical conductivity monitoring reveal spatial and temporal patterns in wetland geochemical processes? Hydrol Earth Syst Sci Discuss 10:699–728. https://doi.org/10.5194/hessd-10-699-2013
Gogoi P, Kumari S, Sarkar UK, Lianthuamluaia L, Puthiyottil M, Bhattacharjya BK, Das BK (2021) Dynamics of phytoplankton community in seasonally open and closed wetlands in the Teesta–Torsa basin, India, and management implications for sustainable utilization. Environ Monit Assess 193:810. https://doi.org/10.1007/s10661-021-09587-w
Grabowksa M, Wołowski K (2013) “Development of Trachelomonas species (Euglenophyta) during blooming of Planktothrix agardhii (Cyanoprokaryota)”. Int J Limnol 50:49–57. https://doi.org/10.1051/limn/2013070
Guiry MD, Guiry GM (2020) AlgaeBase. World-wide electronic publication. National University of Ireland, Galway, http://www.algaebase.org Accessed 1 May 2021
Hillebrand H, Dürselen CD, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35(2):403–424
Hunt BPV, Hosie GW (2006) The seasonal succession of zooplankton in the Southern Ocean south of Australia, part II: the sub-Antarctic to polar frontal zones. Deep-Sea Res I 53:1203–1223
Kumari S, Lianthuamluaia L, Sarkar UK et al. (2023) Environmental Characterization of Two Ecologically Distinct Gangetic Oxbow Lakes using Zooplankton Taxonomic Indices Through Comparative Approach for Wetland Monitoring. Wetlands 43:14. https://doi.org/10.1007/s13157-023-01658-7
Latinopoulos D, Ntislidou C, Kagalou I (2020) Relationships of environmental conditions and phytoplankton functional groups in a new re-constructed shallow lentic system in drought conditions. Ecohydrol Hydrobiol 20(3):369–381
Li T, Radley MH, Daniel AB, Liu F, Sun Q, Patrick LK (2018) Long-term projections of temperature-related mortality risks for ischemic stroke, hemorrhagic stroke, and acute ischemic heart disease under changing climate in Beijing, China. Environ Int 112:1–9. https://doi.org/10.1016/j.envint.2017.12.006
Marshall HG, Burchardt L, Lacouture R (2005) A review of phytoplankton composition within the Chesapeake Bay and its tidal estuaries. J Plank Res 27(11):1083–1102. https://doi.org/10.1093/plankt/fbi079
Md. Aftabuddin, Hassan MA, Das AK, Jha BC, Sharma AP (2017) Effect of river connectivity on hydrochemistry, sediment enzyme activity and biotic communities of wetlands. Aquat Ecosys Health Man 20(1-2):140–150. https://doi.org/10.1080/14634988.2017.1301095
Meena DK, Lianthuamluia L, Mishal P, Swain HS, Naskar BK, Saha S, Sandhya KM, Kumari S, Tayung T, Sarkar UK, Das BK (2019) Assemblage patterns and community structure of macrozoobenthos and temporal dynamics of eco-physiological indices of two lakes, in lower Gangetic plains under varying ecological regimes: A tool for wetland management. Ecol Engin 130:1–20
Mohanty TR, Tiwari NK, Kumari S et al. (2021) Variation of Aulacoseira granulata as an eco-pollution indicator in subtropical large river Ganga in India: a multivariate analytical approach. Environ Sci Pollut Res 29:37498–37512. https://doi.org/10.1007/s11356-021-18096-9
Padisak J, Crossetti LO, Naselli-Flores L (2009) Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621:1–19
Palmer C (1980) Algae and water pollution. The identification, significance, and control of algae in water supplies and in polluted water. Castle House Publications Ltd.
Pinel-Alloul B, André A, Legendre P, Cardille JA, Patalas K, Salki A (2013) Large-scale geographic patterns of diversity and community structure of pelagic crustacean zooplankton in Canadian lakes. Glob Ecol Biogeogr 22:784–795. https://doi.org/10.1111/geb.12041
Poniewozik M, Lenard T (2022) Phytoplankton Composition and Ecological Status of Lakes with Cyanobacteria Dominance. Int J Environ Res Public Health 19(7):3832. https://doi.org/10.3390/ijerph19073832.
Prescott GW (1962) Algae of the Western Great Lakes area with an illustrated key to the genera of desmid and freshwater diatoms (Revised, p. 1000). C. Brown Company Publishers, Dubuque, Lowa
Prusty AKB, Chandra R, Azeez PA (2010) Macronutrients along the sediment profile in a subtropical monsoonal wetland in India. Wetl Ecol Manag 18:91–105. https://doi.org/10.1007/s11273-009-9151-9
Ray A, Mondal D, Chakraborty N, Ganguly S (2023) Phytoplankton diversity in wastewater impacted Indian Ramsar site: a study from East Kolkata Wetland. Environ Monit Assess 195(8):938. https://doi.org/10.1007/s10661-023-11541-x.
Reinl KL, Harris TD, Elfferich I, Coker A, Zhan Q, De Senerpont Domis LN, Morales-Williams AM, Bhattacharya R, Grossart H, North RL, Sweetman JN (2022) The role of organic nutrients in structuring freshwater phytoplankton communities in a rapidly changing world. Water Res 219:118573. https://doi.org/10.1016/j.watres.2022.118573
Reynolds CS, Huszar V, Kruk C, Naselli-Flores L, Melo S (2002) Towards a functional classification of the freshwater phytoplankton. J Plank Res 24:417–428
Salmaso N, Naselli-Flores L, Judit, Padisak J (2015) Functional classifications and their application in phytoplankton ecology. Freshw Biol 60:603–619
Sarkar UK, Mishal P, Borah S, Karnatak G, Chandra G, Kumari S, Meena DK, Debnath D, Yengkokpam S, Das P, DebRoy P, Yadav AK, Md, Aftabuddin, Gogoi P, Pandit A, Bhattacharjya BK, Tayung T, Lianthuamluaia L, Das BK (2020) Status, Potential, Prospects, and Issues of Floodplain Wetland Fisheries in India: Synthesis and Review for Sustainable Management. Rev Fish Sci Aquac 29(1):1–32. https://doi.org/10.1080/23308249.2020.1779650
Sharma BK (2015) Phytoplankton diversity of Deepor Beel - A Ramsar site in the floodplain of the Brahmaputra River basin, Assam, north-east India. Indian J Fish 62(1):33–40
Sin Y, Lee H (2020) Changes in hydrology, water quality, and algal blooms in a freshwater system impounded with engineered structures in a temperate monsoon river estuary. J Hydrol Reg Stu 32:100744. https://doi.org/10.1016/j.ejrh.2020.100744
Strickland JDH, Parsons, TR (1972) A Practical Hand Book of Seawater Analysis. Fish Res B Canada Bull 157, 2nd edn, p 310
Tinnevelt GH, Lushchikova O, Augustijn D, Lochs M, Geertsma RW, Rijkeboer M, Kools H, Dubelaar G, Veen A, Buydens LMC, Jansen JJ (2022) Water quality monitoring based on chemometric analysis of high-resolution phytoplankton data measured with flow cytometry. Environ Int 170:107587. https://doi.org/10.1016/j.envint.2022.107587
Toporowska M, Ferencz B, Dawidek J (2018) Impact of lake-catchment processes on phytoplankton community structure in temperate shallow lakes. Ecohydrology 11(8). https://doi.org/10.1002/eco.v11.810.1002/eco.2017.
Townsend SA, Douglas MM (2017) Discharge-driven flood and seasonal patterns of phytoplankton biomass and composition of an Australian tropical savannah river. Hydrobiologia 794:203–221
Varol M (2019) Phytoplankton functional groups in a monomictic reservoir: seasonal succession, ecological preferences, and relationships with environmental variables. Environ Sci Pollut Res 26(20):20439–20453
Wang C, Jia H, Wei J, Yang W, Gao Y, Liu Q, Ge D, Wu N (2021) Phytoplankton functional groups as ecological indicators in a subtropical estuarine river delta system. Ecol Indic 126:107651
Wanganeo A, Wanganeo R (1991) Algal population in valley lakes of Kashmir Himalaya. Arch Fur Hydrobiologie (Stuttg) 121:219–233
Ward HB, Whipple GC (1992) Freshwater biology. In: Edmondson WT (Ed.) Second Ed. John Willey & Sons. Inc, New York
Xu H, Yong J, Xu G (2016) Bioassessment of water quality status using a potential bioindicator based on functional groups of planktonic ciliates in marine ecosystem. Mar Poll Bullet 110(1):409–414. https://doi.org/10.1016/j.marpolbul.2016.06.033
Yan G, Yin X, Huang M, Wang X, Huang D, Li D (2023) Dynamics of phytoplankton functional groups in river-connected lakes and the major influencing factors: A case study of Dongting Lake, China. Ecol Indic 149:110177. https://doi.org/10.1016/j.ecolind.2023.110177.
Yang WL, Lai ZN, Zeng YY, Shuai FM, Wang C (2017) Temporal and spatial distribution characteristics of CODMn in surface water of the middle and lower reaches of the Pearl River and water environment assessment. Ecol Environ Sci 26(4):643–648
Zhang YQ, Liu SL, Hou XY, Cheng FY, Shen ZY (2019) Landscape- and climate change-induced hydrological alterations in the typically urbanized Beiyun River basin, Beijing, China. Stoch Environ Res Risk Asses 33:149–68
Zhu L, Chen Y, Wang Y, Wang C, Wei Y (2021) Ecological assessment of water quality in an urban river replenished with reclaimed water: the phytoplankton functional groups approach. Environ Res Commun 3:115006. https://doi.org/10.1088/2515-7620/ac3777
Acknowledgements
The authors thank the Director of ICAR-CIFRI, Bararckpore, for providing the necessary facilities. We are thankful to Y. Ali and Bablu Naskar, who contributed to the sampling for the study. We thank the reviewers for their valuable comments. The authors also thank Research scholar Taniya Koyal for helping prepare map. Authors profusely thanks to unanimous reviewers for their constructive suggestions.
Author contributions
S. K.: Conceptualization, Methodology, Formal analysis, Investigation, Data curation, Writing – original draft, Visualization. P. G. : Methodology, Software, Formal analysis, Writing – review & editing. L. L.: Methodology, Software, Data curation, Writing – review & editing. M.P.: Methodology, Software, Data curation, Writing – review & editing. B. K. D.: Conceptualization, Resources, Data curation. M. A. H.: Conceptualization, Writing – review & editing, A. K. D.: Data curation, Writing – review & editing.
Funding
This research received funding from ICAR-CIFRI, Barrackpotre.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumari, S., Gogoi, P., Lianthuamluaia et al. The Potential Role of Phytoplankton Functional Groups Under Anthropogenic Stressed Wetlands: Characterizing The Environmental Sensitivity. Environmental Management (2024). https://doi.org/10.1007/s00267-024-02036-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00267-024-02036-7