Abstract
Objective
To determine T2* normal reference values for anterior talofibular ligament (ATFL) and to investigate the feasibility of the quantitative ATFL quality evaluation in chronic lateral ankle instability (CLAI) using T2* values.
Materials and methods
This study enrolled 15 patients with CLAI and 30 healthy volunteers. The entire ATFL T2* values from the MRI T2* mapping were measured. The prediction equation (variables: age, height, and weight) in a multiple linear regression model was used to calculate the T2* normal reference value in the healthy group. T2* ratio was defined as the ratio of the actual T2* value of the patient’s ATFL to the normal reference value for each patient. A Telos device was used to measure the talar tilt angle (TTA) from the stress radiograph.
Results
T2* values of ATFL in the healthy and CLAI groups were 10.82 ± 1.84 ms and 14.36 ± 4.30 ms, respectively, which are significantly higher in the CLAI group (P < 0.05). The prediction equation of the normal reference T2* value was [14.9 + 0.14 × age (years) − 4.7 × height (m) − 0.03 × weight (kg)] (R2 = 0.65, P < 0.0001). A significant positive correlation was found between the T2* ratio and TTA (r = 0.66, P = 0.007).
Conclusion
MRI T2* values in patients with CLAI were higher than those in healthy participants, and the T2* ratio correlated with TTA, suggesting that T2* values are promising for quantitative assessment of ATFL quality preoperatively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
An ankle sprain is one of the most frequent injuries during daily living and sports activities [1, 2], most commonly associated with anterior talofibular ligament (ATFL) injury. A common problem after ATFL injury includes chronic lateral ankle instability (CLAI), and surgery is indicated if conservative treatment fails. ATFL repair is the first-line CLAI surgical treatment when the residual ligament remains. Arthroscopic lateral ankle repair has been widely used to achieve good clinical results, such as providing stabilization, reducing postoperative pain, and facilitating early recovery [3]. Michels et al. reported that the quality of the residual ligament influences treatment outcome [4]. Moreover, the previous study reported that a repair procedure against poor tissue quality in the ATFL remnant eventually required additional allograft reconstruction [5]. However, few studies have investigated a method of a preoperative quantitative ATFL quality assessment.
Magnetic resonance imaging (MRI) can provide a detailed morphological assessment of ligaments [6,7,8,9] and has recently become available for quantitative assessment [10, 11]. T2* values reflect the water content and collagen fiber content and orientation in the tissue [12], which can quantitatively assess the ligament quality with shorter T2* relaxation times. The normal T2* value of the anterior cruciate ligament (ACL) is 11 to 19 ms [13, 14], and the higher T2* value indicates the lower fascicle-specific ACL surface in histomorphometry measurement [15] and increases anterior knee laxity [14].
Few studies have examined the T2* value of the ankle ligament. Surgeons appropriately may select the surgical procedure, such as ATFL repair or ATFL reconstruction if the ATFL quality can be assessed preoperatively using the T2* value, resulting in a better clinical outcome. Therefore, quantitative assessment is clinically important. This study aimed to determine T2* normal reference values for ATFL and investigate the feasibility of quantitative ATFL quality evaluation in CLAI using T2* values. We hypothesized that it would be possible to conduct a quantitative assessment of ATFL quality in CLAI using MRI T2* value. Furthermore, we also hypothesized that the MRI T2* values would be correlated with the anterior drawer distance as measured in patients.
Materials and methods
Patient cohort
The institutional review board of our hospital approved this study. This study enrolled 17 consecutive patients with CLAI and 30 healthy volunteers (16 males and 14 females; mean age, 39.8 ± 10.4 years [standard deviation]). The CLAI group consisted of patients who underwent surgery from November 2020 to November 2022, 3 months after ankle sprains, and those without ankle instability improvement after conservative treatment. The exclusion criteria include ankle osteoarthritis, rheumatoid arthritis, and incomplete MRI data. The final number of patients was 15 [6 males and 9 females; mean age, 34.7 ± 15.3 years (standard deviation)] based on these exclusion criteria. The healthy group consisted of participants with no history of sprain and no ankle pain during the MRI scan.
Imaging
All patients and healthy volunteers underwent 3.0-T MRI (Ingenia, Philips Healthcare, Netherlands) with a dStream FootAnkle 8ch coil consisting of structural three-dimensional (3D) T2-weighted images (repetition time/echo time = 1400/ 62 ms, voxel size = 0.5 × 0.5 × 0.6 mm3) along with T2* map images in a neutral ankle position. The high-resolution T2* map image (multi-echo–Fast Field echo sequence) parameters were as follows: repetition time = 32 ms; echo time = 2.3, 4.6, 6.9, 9.2, 11.5, 13.8, and 16.1 ms; flip angle = 13°; and voxel size = 0.6 × 0.6 × 0.6 mm3; compressed sensing acceleration factor = 4.6. The acquisition time was 5 min 4 s.
Imaging analysis
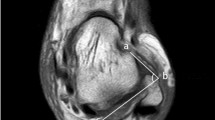
The entire ATFL of each ankle was manually segmented from the T2* map images using Ziostation2 (ZIOSOFT Inc., Tokyo, Japan) with reference to the 3D T2-weighted images by an MRI researcher with adequate ankle anatomy knowledge (Fig. 1). In this case, three planes were used for the identification and segmentation of the ATFL.
The T2* value of each ATFL was determined as the median value from these segmented pixels. Additionally, another MRI researcher performed image analysis to assess measurement interobserver variability. The prediction equation (variables: age, height, weight) in a multiple linear regression model calculated the T2* normal reference value in the healthy group. T2* ratio was defined as the ratio of the actual T2* value of the patient’s ATFL to the normal reference value of ATFL obtained by substituting the patient’s age, height, and weight into the prediction equation. Additionally, the MRI findings on T2-weighted image were evaluated for the ATFL morphological characteristics classified into the following categories: normal, wavy, thin, and thickened ATFL [16].
A Telos device was used to measure the talar tilt angle (TTA) and anterior drawer distance (ADD) from the stress radiograph in the CLAI group. Anterior translation (AT) was measured by a manual test using a capacitance-type strain sensor [17] in the healthy group and the CLAI group. All patients with CLAI underwent arthroscopic ATFL repair after an MRI examination [18]. The ATFL tension (taut, mild laxity, and laxity) and quality (excellent, moderate, and poor) were evaluated from arthroscopic findings by two foot and ankle orthopedic surgeons [19].
Statistical analysis
All statistical analyses were performed using JMP 17.0 software (SAS Institute, Cary, NC, USA). The chi-squared test and Mann–Whitney U test for categorical and continuous variables, respectively, were used to evaluate the differences between the two groups. Accuracy was tested by analyzing the interinvestigator intraclass correlation coefficient (ICC) of the T2* values, and the measured values were rated as follows: <0.50 (considered poor), 0.50 to 0.75 (moderate), 0.75 to 0.90 (good), and >0.90 (excellent). First-order multiple linear regression analyses, with the T2* value as a dependent variable and age, height, and weight as independent variables, were used to determine the best-fit parameters and evaluate the relationship between the T2* value and age, height, or weight. The R2 values were reported as indicators of the relationship strength and goodness of fit, and the measured values were rated as follows: 0 to 0.25 (weak), 0.26 to 0.50 (moderate), and 0.51 to 1.00 (substantial) [20]. Spearman correlation coefficients were used to investigate the correlation of the T2* value or T2* ratio and the TTA, ADD, or AT. The Kruskal–Wallis test was used to test the statistical significance of T2* values between arthroscopic findings. P values less than 5% were considered statistically significant.
A prior power analysis (α error: 0.05, 1-β error: 0.85, number of predictors: 3, sample size: 29) was performed using the G*power program (version 3.1.9.6) to calculate the sample size for the healthy group. Additionally, post hoc analysis for correlation T2* ratio and TTA revealed that a sample size of 15 would achieve 88% power. A power level of more than 80% was considered sufficient. Thus, we determined that our study included a sufficient sample size.
Results
There were no signal changes of the ATFL on T2-weighted images in all healthy volunteers. T2* values of ATFL in the healthy and CLAI groups were 10.82 ± 1.84 ms (range, 7.54–14.09 ms) and 14.05 ± 4.60 ms, respectively, and were significantly higher in the CLAI group (P = 0.0284) (Table 1). The ICC index for the interobserver reliability of the T2* value between the two investigators was 0.94 (95% confidence interval: 0.83–0.99), indicating excellent interobserver reliability. Multiple regression analysis revealed that the T2* value was predicted by age, height, and weight (R2 = 0.65, P < 0.0001) (Fig. 2). The prediction equation of the T2* normal reference value is shown as follows: T2* value (ms) = 14.9 + 0.14 × age (years) − 4.7 × height (m) − 0.03 × weight (kg). The variance inflation factor (VIF) of each variable was 1.3 for age, 1.9 for height, and 2.2 for weight, indicating no problematic level of collinearity as the VIF was less than 10 [21]. Age showed significantly high positive [22] correlations with T2* value (r = 0.72, P = 0.001). Also, R2 was 0.51 in the parsimonious model where age was the only predictor variable (P < 0.0001).
The mean AT of the healthy subjects was 3.8 ± 1.4 mm. The mean TTA, ADD, and AT were 13.1° ± 4.6°, 7.7 ± 1.9 mm, and 8.9 ± 3.0 mm, respectively, in the CLAI group. A significant positive correlation was found between the T2* ratio and TTA (r = 0.66, P = 0.007) and between the T2* value and ADD (r = 0.57, P = 0.033) (Table 2) (Fig. 3). The T2* value for each morphologic finding were 9.61 ms for normal thick without wavy contour in 1 (6.7%), 13.80 ± 3.95 ms for normal thick with wavy contour in 2 (13.3%), 13.58 ± 4.21 ms for thickened ATFL in 6 (40%), and 18.18 ± 8.87 ms for thin ATFL in 2 (13.3%) (Figs. 4, 5, and 6). There was no relationship between morphologic characteristics and T2* values. Arthroscopic findings included ATFL tension (taut: 0, mild laxity: 10 cases, laxity: 5 cases) and quality (excellent: 3 cases, moderate: 8 cases, poor: 4 cases). A trend toward higher T2* values as the intraoperative findings progressed from mild laxity to laxity, from excellent to poor, or from moderate to poor. No significant differences were found between T2* values and intraoperative arthroscopic findings (Table 3).
Discussion
To our best knowledge, this is the first report related to the quantitative evaluation of ATFL using MRI T2* values. The most important finding in this study was that the T2* ratio revealed a significant positive correlation with the TTA of the stress radiograph in patients with CLAI.
ACL, posterior cruciate ligament, and patella tendon differ in collagen content and collagen fiber orientation [23, 24], as reflected by T2* values [25]. Previous prospective studies have shown longitudinal changes in graft maturation after ACL reconstruction using T2* values [26, 27]. Additionally, direct measurement of T2* values of the ACL helped determine the elastic modulus [15] and the difference in anterior knee laxity in clinical practice [14].
This study revealed that the T2* value of ATFL has a significant positive correlation with age in healthy groups. A high percentage of age-related ACL histopathologic abnormalities had collagen fiber disorientation [28]. Furthermore, higher T2* values reflect collagen fiber disorientation [29]. Therefore, the increase in T2* values with age may indicate degenerative changes in ankle ligaments. This study derived a substantial equation using age as a variable to calculate a normal reference value.
CLAI is a condition that is primarily characterized by insufficient ATFL. Instability assessment using stress radiographs has been performed. Hashimoto et al. reported that the condition of the injured ligaments in patients with a TTA of more than 15° had almost avulsed and degenerated or completely absent injured ligaments [30]. However, stress radiographs have not directly assessed ATFL insufficiency. MRI T2* map images allowed a direct ATFL assessment. A significant correlation between TTA and the T2* ratios was found in the CLAI group. Therefore, the T2* value may provide a more robust ATFL insufficiency assessment. Additionally, a higher T2* value indicates a combination of less densely packed collagen fibrils, periligamentous scar tissue [31], and a histological lower score [32]. No statistically significant difference was found in the arthroscopic finding evaluation, but lower ATFL tension and quality revealed higher T2* values. The current findings reported revealed ATFL quality can be objectively and quantitatively assessed preoperatively. Because there was a wide range of T2* value (7.54–14.09 ms) in the healthy group, the ATFL assessment of the CLAI patients using the pure T2* seemed to be difficult. Therefore, the use of T2* ratio should be considered for the assessment of ATFL quality in clinical practice.
Recently, ATFL arthroscopic is often performed, which is less invasive than open repair or reconstruction, and good clinical outcomes have been reported. Arthroscopic repair procedure also has the advantage of a shorter rehabilitation period [33]; therefore, surgical technique selection for arthroscopic repair, open repair, or reconstruction is important. Future work will need to investigate the relationship between time-dependent changes in T2* and the postoperative outcome with more participants to determine the cut-off value for surgical procedures.
This study has several limitations. First, the sample size in the CLAI group was small. Further investigations should be performed in a larger sample size when more patient data are available. Second, ATFL tissue samples were impossible to evaluate because all surgical procedures included a repair; therefore, the actual ATFL quality, such as collagen content and orientation is unknown. A histological evaluation may be possible in the case of ATFL reconstruction, but it also suffers from subject bias. Third, an arthroscopic evaluation of the entire ATFL is difficult to perform due to the limited field of view of arthroscopy, while MRI allows T2* value measurement for total ATFL. Forth, the T2* values may also differ for different vendors or different magnetic field strength, and T2* values should probably be locally verified for each center. Finally, the relationship between T2* value and clinical results is unclear. The clinical score and preoperative outcome should be investigated. However, to our best knowledge, the present study first demonstrated measuring the T2* value of ATFL. Furthermore, the T2* ratio, defined as the ratio of normal calculation from the prediction equation, was correlated with TTA.
In conclusion, MRI T2* values in patients with CLAI were higher than those in healthy participants, and the T2* ratio correlated with TTA, suggesting that T2* values are promising for quantitative ATFL quality assessment preoperatively.
Data Availability
The data that support the findings of this study are available from the corresponding author, AT, upon reasonable request.
References
Doherty C, Delahunt E, Caulfield B, Hertel J, Ryan J, Bleakley C. The incidence and prevalence of ankle sprain injury: a systematic review and meta-analysis of prospective epidemiological studies. Sports Med. 2014;44(1):123–40.
Waterman BR, Owens BD, Davey S, Zacchilli MA, Belmont PJ Jr. The epidemiology of ankle sprains in the United States. J Bone Joint Surg Am. 2010;92(13):2279–84.
Matsui K, Takao M, Miyamoto W, Matsushita T. Early recovery after arthroscopic repair compared to open repair of the anterior talofibular ligament for lateral instability of the ankle. Arch Orthop Trauma Surg. 2016;136(1):93–100.
Michels F, Pereira H, Calder J, et al. Searching for consensus in the approach to patients with chronic lateral ankle instability: ask the expert. Knee Surg Sports Traumatol Arthrosc. 2018;26(7):2095–102.
Dierckman BD, Ferkel RD. Anatomic reconstruction with a semitendinosus allograft for chronic lateral ankle instability. Am J Sports Med. 2015;43(8):1941–50.
Akatsuka Y, Teramoto A, Takashima H, Watanabe K, Yamashita T. Morphologicalevaluation of the calcaneofibular ligament in different ankle positions using a three-dimensional MRI sequence. Surg Radiol Anat. 2019;41(3):307–11.
Teramoto A, Akatsuka Y, Takashima H, et al. 3D MRI evaluation of morphological characteristics of lateral ankle ligaments in injured patients and uninjured controls. J Orthop Sci. 2020;25(1):183–7.
Yi J, Lee YH, Hahn S, Albakheet SS, Song H-T, Suh J-S. Fast isotropic volumetric magnetic resonance imaging of the ankle: acceleration of the three-dimensional fast spin echo sequence using compressed sensing combined with parallel imaging. Eur J Radiol. 2019;112:52–8.
Hong CC, Lee JC, Tsuchida A, et al. Individual fascicles of the ankle lateral ligaments and the lateral fibulotalocalcaneal ligament complex can be identified on 3D volumetric MRI. Knee Surg Sports Traumatol Arthrosc. 2023;31(6):2192–8.
Haraguchi N, Ota K, Nishida N, Ozeki T, Yoshida T, Tsutaya A. T1rho mapping of articular cartilage grafts after autologous osteochondral transplantation for osteochondral lesions of the talus: a longitudinal evaluation. J Magn Reson Imaging. 2018;48(2):398–403.
Takashima H, Yoshimoto M, Ogon I, et al. Lumbar disc degeneration assessment using T2* relaxation time with ultra-short TE. Magn Reson Imaging. 2020;73:11–4.
Matzat SJ, van Tiel J, Gold GE, Oei EH. Quantitative MRI techniques of cartilage composition. Quant Imaging Med Surg. 2013;3(3):162–74.
Anz AW, Edison J, Denney TS, et al. 3-T MRI mapping is a valid in vivo method of quantitatively evaluating the anterior cruciate ligament: rater reliability and comparison across age. Skelet Radiol. 2020;49(3):443–52.
Wang HM, Shultz SJ, Ross SE, Henson RA, Perrin DH, Schmitz RJ. Relationship of anterior cruciate ligament volume and T2* relaxation time to anterior knee laxity. Orthop J Sports Med. 2021;9(2):2325967120979986.
Jerban S, Hananouchi T, Ma Y, et al. Correlation between the elastic modulus of anterior cruciate ligament (ACL) and quantitative ultrashort echo time (UTE) magnetic resonance imaging. J Orthop Res. 2022;40(10):2330–9.
Morvan A, Klouche S, Thes A, Hardy P, Bauer T. Reliability and validity of preoperative MRI for surgical decision making in chronic lateral ankle instability. Eur J Orthop Surg Traumatol. 2018;28(4):713–9.
Teramoto A, Iba K, Murahashi Y, et al. Quantitative evaluation of ankle instability using a capacitance-type strain sensor. Foot Ankle Int. 2021;42(8):1074–80.
Teramoto A, Murahashi Y, Takahashi K, Watanabe K, Yamashita T. Effect of accelerated rehabilitation on early return to sport after arthroscopic ankle lateral ligament repair. Orthop J Sports Med. 2022;10(9):23259671221121676.
Ahn J, Choi JG, Jeong BO. The signal intensity of preoperative magnetic resonance imaging has predictive value for determining the arthroscopic reparability of the anterior talofibular ligament. Knee Surg Sports Traumatol Arthrosc. 2021;29(5):1535–43.
Adinyira E, Agyekum K, Danku JC, Addison P, Danku JC. Influence of subcontractor risk management on quality performance of building construction projects in Ghana. J Constr Dev Ctries. 2020;25(2):175–97.
James G, Witten D, Hastie T, Tibshirani R (Eds.). An introduction to statistical learning: with applications in R, 1st ed. 2013, Corr. 7th printing 2017 edition. Springer, New York; 2017.
Mukaka MM. Statistics corner: a guide to appropriate use of correlation coefficient in medical research. Malawi Med J. 2012;24(3):69–71.
Hodgson RJ, O'Connor PJ, Grainger AJ. Tendon and ligament imaging. Br J Radiol. 2012;85(1016):1157–72.
Levy YD, Hasegawa A, Patil S, Koziol JA, Lotz MK, D'Lima DD. Histopathological changes in the human posterior cruciate ligament during aging and osteoarthritis: correlations with anterior cruciate ligament and cartilage changes. Ann Rheum Dis. 2013;72(2):271–7.
Okuda M, Kobayashi S, Toyooka K, et al. Quantitative differentiation of tendon and ligament using magnetic resonance imaging ultrashort echo time T2* mapping of normal knee joint. Acta Radiol. 2022;63(11):1489–96.
Chu CR, Williams AA. Quantitative MRI UTE-T2* and T2* show progressive and continued graft maturation over 2 years in human patients after anterior cruciate ligament reconstruction. Orthop J Sports Med. 2019;7(8):2325967119863056.
Warth RJ, Zandiyeh P, Rao M, et al. Quantitative assessment of in vivo human anterior cruciate ligament autograft remodeling: a 3-dimensional UTE-T2* imaging study. Am J Sports Med. 2020;48(12):2939–47.
Hasegawa A, Otsuki S, Pauli C, et al. Anterior cruciate ligament changes in the human knee joint in aging and osteoarthritis. Arthritis Rheum. 2012;64(3):696–704.
Beveridge JE, Machan JT, Walsh EG, et al. Magnetic resonance measurements of tissue quantity and quality using T(2) * relaxometry predict temporal changes in the biomechanical properties of the healing ACL. J Orthop Res. 2018;36(6):1701–9.
Hashimoto T, Inokuchi S, Kokubo T. Clinical study of chronic lateral ankle instability: injured ligaments compared with stress X-ray examination. J Orthop Sci. 2009;14(6):699–703.
Biercevicz AM, Murray MM, Walsh EG, Miranda DL, Machan JT, Fleming BC. T2 * MR relaxometry and ligament volume are associated with the structural properties of the healing ACL. J Orthop Res. 2014;32(4):492–9.
Biercevicz AM, Proffen BL, Murray MM, Walsh EG, Fleming BC. T2* relaxometry and volume predict semi-quantitative histological scoring of an ACL bridge-enhanced primary repair in a porcine model. J Orthop Res. 2015;33(8):1180–7.
Zhi X, Lv Z, Zhang C, Kong C, Wei S, Xu F. Does arthroscopic repair show superiority over open repair of lateral ankle ligament for chronic lateral ankle instability: a systematic review and meta-analysis. J Orthop Surg Res. 2020;15(1):355.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethical approval for this study was obtained from the institutional review board in Sapporo Medical University Hospital (reference no: 322-250).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akatsuka, Y., Teramoto, A., Murahashi, Y. et al. Quantitative assessment of anterior talofibular ligament quality in chronic lateral ankle instability using magnetic resonance imaging T2* value. Skeletal Radiol 53, 733–739 (2024). https://doi.org/10.1007/s00256-023-04480-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-023-04480-8