Abstract
Osteoarthritis (OA) is one of the leading causes of disability worldwide. As our understanding of OA progressively has moved from a purely mechanical “wear and tear” concept toward a complex multi-tissue condition in which inflammation plays a central role, the possible role of crystal-induced inflammation in OA incidence and progression may be relevant. In addition to gout, which affects 4% of the US population, basic calcium phosphate and calcium pyrophosphate deposition both may induce joint inflammation and may play a role in pain in OA. This narrative review article discusses the possible mechanisms underlying the associations between crystal-induced arthropathies and OA, and the important implications of these for clinical practice and future research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is one of the leading causes of disability worldwide, affecting 10–15% of adults, with a lifetime risk as high as 50% [1]. Currently, OA affects 30 million individuals in the U.S.A. [2] and 302 million worldwide [3]. Despite the advances in understanding the roles of biomechanical stress and cellular responses in OA pathophysiology [4], there are no disease modifying OA drugs to prevent or halt the progression of the disease.
Although OA has not been considered an inflammatory joint disorder, inflammation is an important feature in disease incidence and progression that is commonly assessed using either ultrasound or magnetic resonance imaging (MRI) [5,6,7]. Synovitis in OA is associated with inflammatory cytokines [8], structural OA severity [9], and fluctuation of pain in the same direction [7]. The etiopathogenesis of synovitis in knee OA is controversial. One hypothesis is that synovitis is a secondary reaction to cartilage fragments and other debris that promote synovial production of inflammatory mediators [10]. As our understanding of OA moves away from a purely mechanical “wear and tear” condition towards a complex multifaceted disease of the whole joint in which inflammation plays a central role, the question of a possible relationship between OA and calcium- and monosodium urate (MSU) crystal-induced inflammation in the context of OA has gained increasing attention [11, 12]. Both gout and OA are associated with calcium pyrophosphate (CPP) crystal deposition.
From a clinical perspective, there are several indicators suggesting an association between OA and crystal-induced arthropathies. For instance, OA and radiographically detected intra-articular mineralization (i.e., chondrocalcinosis, are often seen concurrently). In addition, gout and OA have many overlapping joint distributions, including the first metatarsophalangeal (MTP), knee, midfoot, and interphalangeal joints [11]. This clinically observed co-occurrence may be not merely coincidental.
In this review, we will discuss in narrative fashion the current knowledge of crystal-induced arthropathies and their role in structural OA incidence and progression, including the role of imaging in helping to understand such associations.
Calcium crystal arthropathy and OA
Calcium crystal arthropathies can be secondary to CPP and/or basic calcium phosphate (BCP). CPP crystals are rod- or rhomboid-shaped, 1–22 μm in size, and birefringent in polarized light [13]. BCP crystals, however, are much smaller (20–100 nm) and cannot be readily visualized on light or polarized microscopy [14]. There are three types of BCP crystals: partially carbonate-substituted hydroxyapatite, octacalcium phosphate, and tricalcium phosphate [15]. Of note, BCP is often termed “hydroxyapatite” in the radiological literature, although the “BCP” terminology is more accurate, since hydroxyapatite is only one subtype of BCP, and any of the BCP elements can be associated with calcific periarthritis. BCP crystals are similar to the mineral calcium phosphate that is a normal component of bone, but in pathologic situations, BCP crystals can produce a strong inflammatory response with large amounts of destructive cytokines and prostaglandins [16], such as have been associated with the highly destructive Milwaukee shoulder syndrome [17].
A major hurdle to understanding the contribution of CPP and BCP crystals to OA incidence and progression is that they have an identical presentation on imaging, which is chondrocalcinosis. When chondrocalcinosis is present on radiographs, computed tomography (CT), or ultrasound imaging, it may reflect either CPP or BCP crystal deposition. Outside of relatively rare metabolic diseases, such as hereditary hemochromatosis, hyperparathyroidism, and hypomagnesemia [18], the mechanism of occurrence of chondrocalcinosis is not well understood. It is known that the prevalence of chondrocalcinosis increases with a history of prior joint damage, such as prior meniscectomy [19]. The association between chondrocalcinosis and OA is also well established and supported by evidence from large epidemiological cohorts [20, 21] (Fig. 1), but the causal relationship between the two remains largely unclear. The Boston University Calcium Knee Score (BUCKS) is a new CT-based scoring system for the evaluation of knee chondrocalcinosis [22]. The BUCKS has been used in a large epidemiological cohort, to describe the prevalence of chondrocalcinosis and study its association with knee pain and other features of OA [23]. The use of the BUCKS system will help studying correlations between intra-articular mineralization deposition and MRI-based semi-quantitative features of knee OA, which in turn will lay the groundwork for a better understanding of knee OA pathophysiology and more specifically the role of crystal deposition in knee OA. Additional research is needed on the relationship between chondrocalcinosis and OA. Both CPP and BCP are commonly detected in cartilage and synovial fluid of the joints affected by OA [24]. Both crystals are also associated with the severity of OA and may become easier to detect as OA progresses [25, 26].
Example of co-occurrence of chondrocalcinosis and knee OA. Coronal CT reformation shows definite medial and lateral tibiofemoral OA. There are large osteophytes at the femoral joint margins (arrowheads). In addition, there is severe meniscal calcification at the lateral meniscus (large arrow). There is a large osteophyte at the medial tibia (small arrow) and signs of severe sclerosis at the subchondral medial tibia (asterisk)
The studies that examined what types and proportion of calcium crystals are involved in chondrocalcinosis are rather limited, and to further complicate the matter, some published research has been based on the assumption that chondrocalcinosis is related to CPP deposition without consideration for the role of BCP [27]. Partly because of the difficulty in identifying BCP, these crystals are often ignored [28]; as a result, the clinical research on this matter is limited, and the clinical significance of BCP crystals is poorly understood.
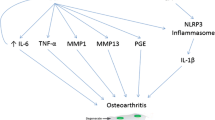
A cadaveric study that included 106 knee specimens from cadaveric donors showed that every knee contained some measurable cartilage mineralization, and that cartilage mineralization was more strongly associated with age rather than the grade of OA. These findings seem to suggest that intra-articular mineralization is an “innocent bystander” rather than the cause of damage and senescence [29]. However, several other studies have emphasized the oxidative stress induced by intra-articular mineralization through the release of inflammatory cytokines and other inflammatory mediators [30]. For instance, CPP crystals were shown to activate nucleotide-binding domain-like receptor protein-3 (NLRP3) inflammasome (in human and mice cells), which cleaves and expresses interleukin-1β (IL-1β), in a similar way to gout [14, 31]. BCP on the other hand can operate via dependent and independent NLRP3 pathways [32]. For instance, BCP increases the production of prostaglandin E2 production [33], matrix metalloproteinases-13 from human fibroblasts [34], and the release of tumor necrosis factor-α [35].
A recent study showed that BCP, but not CPP, is associated with hypertrophy of chondrocytes [27], a feature that is thought to be associated with the initiation and progression of OA [36]. CPP on the other hand was found to be associated with cellular senescence [27]. In other ex vivo studies, BCP crystals were shown to increase chondrocyte apoptosis [37] and cartilage degradation [32]. In an animal study, intra-articular injection of CPP resulted in accelerated OA [38]. Finally, a large community-based case–control study in the UK showed CPP deposits were associated with osteophyte formation [39].
Of note, therapeutic strategies targeting various inflammatory pathways, including IL-1β blockade, have been trialed in OA with varying success [40, 41]. The role of calcium-containing crystals in the pathogenesis of OA remains to be determined. Although developments in clinical and translational research suggest an important role, particularly for BCP crystals, the importance of calcium crystals in overall OA initiation and propagation requires further investigation.
Gout and osteoarthritis
Gout is an increasingly prevalent disorder, with a reported U.S. prevalence of nearly 4% in 2008 [42]. Hyperuricemia, which had a U.S. prevalence of 21.4% in 2008 [42], is defined as a serum urate level of > 6.8 mg/dL. This concentration is the threshold above which the risk of MSU crystallization increases [43]. The progression from asymptomatic hyperuricemia to symptomatic gout implies a state of increased MSU crystal deposition in the tissues around the joint, including the formation of tophi, complex biologically—active structures that reflect chronic inflammatory granulomatous and fibrotic tissue responses to MSU crystals [11, 44]. Gouty arthropathy is clinically characterized by episodes of intense inflammatory arthritis and sometimes a chronic inflammatory arthritis. Gouty flares are usually mono-articular or less commonly polyarticular, whereas painful flares in OA are usually less intense, more variable in its manifestations and less inflammatory (e.g., effusions may be cool to touch). Pain in OA has been associated with the presence of synovitis and bone marrow lesions on MRI [45, 46].
The similar distribution of affected joints in OA and gout and, in particular, the striking predilection of gout for the first MTP joint [47] has long been considered a clue of a relationship between the two conditions (Fig. 2). In addition, the common occurrence of acute gouty attacks in nodal OA has long been recognized [48]. Moreover, the two entities share similar risk factors, including age, obesity, and prior joint injury [49, 50]. A cadaveric study including 7855 adult human tali found a strong correlation between MSU deposition and sites of cartilage lesions in zones of biomechanical stresses (typical of OA) [51]. In addition, OA has been associated with interleukin (IL)-1β response genes [52]. This is similar to gouty arthropathy since IL-1β is also an important mediator in the inflammatory response of gout [53] and is released in response to the presence of MSU crystals [54].
Example of gout in a 65-year-old man. A Axial CT of the foot shows an erosion of the medial aspect of the first metatarsal head (arrowhead). B Three-dimensional volume rendering technique (VRT) image post gout processing shows green-color coded deposits in the hallux and along the Achilles tendon (dashed arrows). The first MTP joint is also a common location for osteoarthritis
There are also non-overlapping features between gout and OA, however, including differences in risk factors and joint distributions. For instance, men are more likely to develop gout whereas women are more likely to develop OA [11]. Renal insufficiency is a risk factor for gout but not for OA [11]. The genetic associations of both diseases are also different, with OA genetics relating to BMI and cartilage biology [55], whereas gout genetics largely relate to renal urate excretion [56]. In terms of distribution, the hip joint is commonly affected in OA but not in gout [11].
A prior in vitro study demonstrated a deleterious effect of microcrystalline urate leading to the generation of chondrocyte death, which in turn leads to the generation of additional urate in a feed-forward amplification loop [57]. These responses only occurred at high concentrations of MSU, consistent with a higher likelihood MSU crystal deposition. In contrast, at lower (physiological) concentration, soluble uric acid may have an antioxidant chondroprotective and anti-inflammatory effect, thereby acting as a protective against OA [58]. In a recent animal study, the repeated injection of MSU into the knee joint was found to cause distinctive pathological changes reflecting OA as investigated by translational imaging [59].
In a large community-based study of 4249 participants, a highly significant association between the site of acute attacks of gout and the presence of OA was demonstrated, including in the first MTP, the midfoot joint, knee, and DIP joints [60].
High levels of uric acid have been associated with generalized OA among those undergoing hip replacement, but not among those undergoing knee replacement [61]. Although another case–control study found generalized OA to be no more common among people with gout than those without gout after matching for age and gender and adjusting for BMI and diuretic use [62], chronic knee and hallux pain, and hallux valgus were more frequent among those with gout. This suggests that the association between OA and gout lies at the individual-affected joint level. In addition, a study using patient databases from Taiwan and the UK showed an association between gout and both hip and knee replacement [63]; the risk of joint replacement was not reduced with urate-lowering therapy, however.
In a large case–control study of 39,111 patients with incident gout [64], a review of medical records over a 10-year period prior to and after the index date showed that the risk of incident OA was significantly higher in subjects with gout than in controls (hazard ratio 1.45 (95% C.I., 1.35–1.54)). However, the subjects with gout were also more likely to have had a preceding diagnosis of OA (OR 1.27) versus controls.
From an imaging perspective, a cross-sectional study of 92 patients with tophaceous gout of the feet reported that joints with MSU crystal deposition detected on dual-energy CT (DECT) were up to 10 times more likely to display features such as osteophytes, subchondral sclerosis, and joint space narrowing, which are typical of OA but not of gout [65]. DECT is commonly used in clinical practice to differentiate between gout and CPP arthropathy (Fig. 3) though a recent study showed that ultrasound has higher sensitivity than DECT for the detection of MSU and CPP crystals [66].
Gout, CPPD, and OA may be present concomitantly in a painful knee. A Color-coded coronal dual-energy CT reformation shows definitive mono-sodium urate crystal deposition in the lateral joint space (arrow). In the medial joint space, meniscal calcification is observed consistent with CPPD (arrowhead). B Plain CT reformation shows a definite osteophyte at the lateral joint margin (small arrow) and large notch osteophytes (large arrows)
Advanced MRI sequences that included T2 mapping and T1rho, which are biomarkers for early-stage OA [67, 68], showed abnormal cartilage and meniscal changes in patients with hyperuricemia [69, 70].
Summary
The links between OA, uric acid, CPP, and BCP are complex and require further investigation. Both gout and OA are associated with CPP crystal deposition. Gout and OA appear to be risk factors for each other, with some overlap in terms of the joint distribution. The associations among OA, CPP, and BCP are complex and less understood, partly because of the challenges for the detection of BCP. As for CPP deposition, the lack of longitudinal studies limits our understanding of the causality direction between CPP and OA.
References
Murphy L, Schwartz TA, Helmick CG, Renner JB, Tudor G, Koch G, et al. Lifetime risk of symptomatic knee osteoarthritis. Arthritis Rheum. 2008;59:1207–13.
Ma VY, Chan L, Carruthers KJ. Incidence, prevalence, costs, and impact on disability of common conditions requiring rehabilitation in the United States: stroke, spinal cord injury, traumatic brain injury, multiple sclerosis, osteoarthritis, rheumatoid arthritis, limb loss, and back pain. Arch Phys Med Rehabil. 2014;95:986-995.e1.
GBD 2015 Disease and injury incidence and prevalence collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Lond Engl. 2016;388:1545–602.
Abramson SB, Attur M, Yazici Y. Prospects for disease modification in osteoarthritis. Nat Clin Pract Rheumatol. 2006;2:304–12.
Crema MD, Felson DT, Roemer FW, Niu J, Marra MD, Zhang Y, et al. Peripatellar synovitis: comparison between non-contrast-enhanced and contrast-enhanced MRI and association with pain. MOST Study Osteoarthr Cartil. 2013;21:413–8.
Roemer FW, Guermazi A, Felson DT, Niu J, Nevitt MC, Crema MD, et al. Presence of MRI-detected joint effusion and synovitis increases the risk of cartilage loss in knees without osteoarthritis at 30-month follow-up: the MOST study. Ann Rheum Dis. 2011;70:1804–9.
Zhang Y, Nevitt M, Niu J, Lewis C, Torner J, Guermazi A, et al. Fluctuation of knee pain and changes in bone marrow lesions, effusions, and synovitis on magnetic resonance imaging. Arthritis Rheum. 2011;63:691–9.
Attur M, Krasnokutsky S, Statnikov A, Samuels J, Li Z, Friese O, et al. Low-grade inflammation in symptomatic knee osteoarthritis: prognostic value of inflammatory plasma lipids and peripheral blood leukocyte biomarkers. Arthritis Rheumatol Hoboken NJ. 2015;67:2905–15.
Krasnokutsky S, Belitskaya-Lévy I, Bencardino J, Samuels J, Attur M, Regatte R, et al. Quantitative magnetic resonance imaging evidence of synovial proliferation is associated with radiographic severity of knee osteoarthritis. Arthritis Rheum. 2011;63:2983–91.
Berenbaum F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr Cartil. 2013;21:16–21.
Neogi T, Krasnokutsky S, Pillinger MH. Urate and osteoarthritis: evidence for a reciprocal relationship. Joint Bone Spine. 2019;86:576–82.
Conway R, McCarthy GM. Calcium-containing crystals and osteoarthritis: an unhealthy alliance. Curr Rheumatol Rep. 2018;20:13.
Zell M, Aung T, Kaldas M, Rosenthal AK, Bai B, Liu T, et al. Calcium pyrophosphate crystal size and characteristics. Osteoarthr Cartil Open. 2021;3:100133.
McCarthy GM, Dunne A. Calcium crystal deposition diseases - beyond gout. Nat Rev Rheumatol. 2018;14:592–602.
Rosenthal AK, Ryan LM. Nonpharmacologic and pharmacologic management of CPP crystal arthritis and BCP arthropathy and periarticular syndromes. Rheum Dis Clin North Am. 2014;40:343–56.
Rosenthal AK. Basic calcium phosphate crystal-associated musculoskeletal syndromes: an update. Curr Opin Rheumatol. 2018;30:168–72.
McCarty DJ, Halverson PB, Carrera GF, Brewer BJ, Kozin F. “Milwaukee shoulder”–association of microspheroids containing hydroxyapatite crystals, active collagenase, and neutral protease with rotator cuff defects. I Clinical aspects. Arthritis Rheum. 1981;24:464–73.
Jones AC, Chuck AJ, Arie EA, Green DJ, Doherty M. Diseases associated with calcium pyrophosphate deposition disease. Semin Arthritis Rheum. 1992;22:188–202.
Doherty M, Watt I, Dieppe PA. Localised chondrocalcinosis in post-meniscectomy knees. Lancet Lond Engl. 1982;1:1207–10.
Ramonda R, Musacchio E, Perissinotto E, Sartori L, Punzi L, Corti MC, et al. Prevalence of chondrocalcinosis in Italian subjects from northeastern Italy. The Pro.V.A. (PROgetto Veneto Anziani) study. Clin Exp Rheumatol. 2009;27:981–4.
Felson DT, Anderson JJ, Naimark A, Kannel W, Meenan RF. The prevalence of chondrocalcinosis in the elderly and its association with knee osteoarthritis: the Framingham Study. J Rheumatol. 1989;16:1241–5.
Guermazi A, Jarraya M, Lynch JA, Felson DT, Clancy M, Nevitt M, et al. Reliability of a new scoring system for intraarticular mineralization of the knee: Boston University Calcium Knee Score (BUCKS). Osteoarthr Cartil. 2020;28:802–10.
Liew J, Guermazi A, Jarraya M, Wang N, Felson D, Lewis CE, et al. The Association of Radiographic Chondrocalcinosis with Localized Structural Outcomes in Knee OA: the multicenter osteoarthritis study [abstract]. Arthritis Rheumatol [Internet]. 2022;74. Available from: https://acrabstracts.org/abstract/the-association-of-radiographic-chondrocalcinosis-with-localized-structural-outcomes-in-knee-oa-the-multicenter-osteoarthritis-study/. Accessed September 17, 2022.
Derfus BA, Kurian JB, Butler JJ, Daft LJ, Carrera GF, Ryan LM, et al. The high prevalence of pathologic calcium crystals in pre-operative knees. J Rheumatol. 2002;29:570–4.
Nalbant S, Martinez JAM, Kitumnuaypong T, Clayburne G, Sieck M, Schumacher HR. Synovial fluid features and their relations to osteoarthritis severity: new findings from sequential studies. Osteoarthr Cartil. 2003;11:50–4.
Fuerst M, Bertrand J, Lammers L, Dreier R, Echtermeyer F, Nitschke Y, et al. Calcification of articular cartilage in human osteoarthritis. Arthritis Rheum. 2009;60:2694–703.
Meyer F, Dittmann A, Kornak U, Herbster M, Pap T, Lohmann CH, et al. Chondrocytes from osteoarthritic and chondrocalcinosis cartilage represent different phenotypes. Front Cell Dev Biol. 2021;9:622287.
Dieppe P, Swan A. Identification of crystals in synovial fluid. Ann Rheum Dis. 1999;58:261–3.
Mitsuyama H, Healey RM, Terkeltaub RA, Coutts RD, Amiel D. Calcification of human articular knee cartilage is primarily an effect of aging rather than osteoarthritis. Osteoarthr Cartil. 2007;15:559–65.
Ea H-K, Nguyen C, Bazin D, Bianchi A, Guicheux J, Reboul P, et al. Articular cartilage calcification in osteoarthritis: insights into crystal-induced stress. Arthritis Rheum. 2011;63:10–8.
Campillo-Gimenez L, Renaudin F, Jalabert M, Gras P, Gosset M, Rey C, et al. Inflammatory potential of four different phases of calcium pyrophosphate relies on NF-κB activation and MAPK pathways. Front Immunol. 2018;9:2248.
Ea H-K, Chobaz V, Nguyen C, Nasi S, van Lent P, Daudon M, et al. Pathogenic role of basic calcium phosphate crystals in destructive arthropathies. PLoS One. 2013;8:e57352.
Molloy ES, Morgan MP, Doherty GA, McDonnell B, O’Byrne J, Fitzgerald DJ, et al. Microsomal prostaglandin E2 synthase 1 expression in basic calcium phosphate crystal-stimulated fibroblasts: role of prostaglandin E2 and the EP4 receptor. Osteoarthr Cartil. 2009;17:686–92.
McCarthy GM, Westfall PR, Masuda I, Christopherson PA, Cheung HS, Mitchell PG. Basic calcium phosphate crystals activate human osteoarthritic synovial fibroblasts and induce matrix metalloproteinase-13 (collagenase-3) in adult porcine articular chondrocytes. Ann Rheum Dis. 2001;60:399–406.
Grandjean-Laquerriere A, Tabary O, Jacquot J, Richard D, Frayssinet P, Guenounou M, et al. Involvement of toll-like receptor 4 in the inflammatory reaction induced by hydroxyapatite particles. Biomater. 2007;28:400–4.
van der Kraan PM, van den Berg WB. Chondrocyte hypertrophy and osteoarthritis: role in initiation and progression of cartilage degeneration? Osteoarthr Cartil. 2012;20:223–32.
Ea HK, Monceau V, Camors E, Cohen-Solal M, Charlemagne D, Lioté F. Annexin 5 overexpression increased articular chondrocyte apoptosis induced by basic calcium phosphate crystals. Ann Rheum Dis. 2008;67:1617–25.
Fam AG, Morava-Protzner I, Purcell C, Young BD, Bunting PS, Lewis AJ. Acceleration of experimental lapine osteoarthritis by calcium pyrophosphate microcrystalline synovitis. Arthritis Rheum. 1995;38:201–10.
Neame RL. UK community prevalence of knee chondrocalcinosis: evidence that correlation with osteoarthritis is through a shared association with osteophyte. Ann Rheum Dis. 2003;62:513–8.
Auw Yang KG, Raijmakers NJH, van Arkel ERA, Caron JJ, Rijk PC, Willems WJ, et al. Autologous interleukin-1 receptor antagonist improves function and symptoms in osteoarthritis when compared to placebo in a prospective randomized controlled trial. Osteoarthr Cartil. 2008;16:498–505.
Schieker M, Conaghan PG, Mindeholm L, Praestgaard J, Solomon DH, Scotti C, et al. Effects of interleukin-1β inhibition on incident hip and knee replacement : exploratory analyses from a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2020;173:509–15.
Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum. 2011;63:3136–41.
Martillo MA, Nazzal L, Crittenden DB. The crystallization of monosodium urate. Curr Rheumatol Rep. 2014;16:400.
Chhana A, Dalbeth N. The gouty tophus: a review. Curr Rheumatol Rep. 2015;17:19.
Mathiessen A, Conaghan PG. Synovitis in osteoarthritis: current understanding with therapeutic implications. Arthritis Res Ther. 2017;19:18.
Felson DT. Developments in the clinical understanding of osteoarthritis. Arthritis Res Ther. 2009;11:203.
Simkin PA. The pathogenesis of podagra. Ann Intern Med. 1977;86:230–3.
Fam AG, Stein J, Rubenstein J. Gouty arthritis in nodal osteoarthritis. J Rheumatol. 1996;23:684–9.
Choi HK, Atkinson K, Karlson EW, Curhan G. Obesity, weight change, hypertension, diuretic use, and risk of gout in men: the health professionals follow-up study. Arch Intern Med. 2005;165:742–8.
Oliveria SA, Felson DT, Cirillo PA, Reed JI, Walker AM. Body weight, body mass index, and incident symptomatic osteoarthritis of the hand, hip, and knee. Epidemiol Camb Mass. 1999;10:161–6.
Muehleman C, Li J, Aigner T, Rappoport L, Mattson E, Hirschmugl C, et al. Association between crystals and cartilage degeneration in the ankle. J Rheumatol. 2008;35:1108–17.
Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier J-P, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7:33–42.
Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–41.
Giamarellos-Bourboulis EJ, Mouktaroudi M, Bodar E, van der Ven J, Kullberg B-J, Netea MG, et al. Crystals of monosodium urate monohydrate enhance lipopolysaccharide-induced release of interleukin 1 beta by mononuclear cells through a caspase 1-mediated process. Ann Rheum Dis. 2009;68:273–8.
Zengini E, Hatzikotoulas K, Tachmazidou I, Steinberg J, Hartwig FP, Southam L, et al. Genome-wide analyses using UK Biobank data provide insights into the genetic architecture of osteoarthritis. Nat Genet. 2018;50:549–58.
Major TJ, Dalbeth N, Stahl EA, Merriman TR. An update on the genetics of hyperuricaemia and gout. Nat Rev Rheumatol. 2018;14:341–53.
Shi Y, Evans JE, Rock KL. Molecular identification of a danger signal that alerts the immune system to dying cells. Nature. 2003;425:516–21.
Lai J-H, Luo S-F, Hung L-F, Huang C-Y, Lien S-B, Lin L-C, et al. Physiological concentrations of soluble uric acid are chondroprotective and anti-inflammatory. Sci Rep. 2017;7:2359.
Accart N, Dawson J, Obrecht M, Lambert C, Flueckiger M, Kreider J, et al. Degenerative joint disease induced by repeated intra-articular injections of monosodium urate crystals in rats as investigated by translational imaging. Sci Rep. 2022;12:157.
Roddy E, Zhang W, Doherty M. Are joints affected by gout also affected by osteoarthritis? Ann Rheum Dis. 2007;66:1374–7.
Sun Y, Brenner H, Sauerland S, Günther KP, Puhl W, Stürmer T. Serum uric acid and patterns of radiographic osteoarthritis–the Ulm Osteoarthritis Study. Scand J Rheumatol. 2000;29:380–6.
Roddy E, Zhang W, Doherty M. Gout and nodal osteoarthritis: a case-control study. Rheumatol. 2008;47:732–3.
Kuo C-F, Chou I-J, See L-C, Chen J-S, Yu K-H, Luo S-F, et al. Urate-lowering treatment and risk of total joint replacement in patients with gout. Rheumatol Oxf Engl. 2018;57:2129–39.
Kuo C-F, Grainge MJ, Mallen C, Zhang W, Doherty M. Comorbidities in patients with gout prior to and following diagnosis: case-control study. Ann Rheum Dis. 2016;75:210–7.
Dalbeth N, Aati O, Kalluru R, Gamble GD, Horne A, Doyle AJ, et al. Relationship between structural joint damage and urate deposition in gout: a plain radiography and dual-energy CT study. Ann Rheum Dis. 2015;74:1030–6.
Kravchenko D, Karakostas P, Kuetting D, Meyer C, Brossart P, Behning C, et al. The role of dual energy computed tomography in the differentiation of acute gout flares and acute calcium pyrophosphate crystal arthritis. Clin Rheumatol. 2022;41:223–33.
Baum T, Joseph GB, Karampinos DC, Jungmann PM, Link TM, Bauer JS. Cartilage and meniscal T2 relaxation time as non-invasive biomarker for knee osteoarthritis and cartilage repair procedures. Osteoarthr Cartil. 2013;21:1474–84.
Le J, Peng Q, Sperling K. Biochemical magnetic resonance imaging of knee articular cartilage: T1rho and T2 mapping as cartilage degeneration biomarkers. Ann N Y Acad Sci. 2016;1383:34–42.
Zhu J, Hu N, Hou J, Liang X, Wang Y, Zhang H, et al. T1rho mapping of cartilage and menisci in patients with hyperuricaemia at 3 T: a preliminary study. Clin Radiol. 2021;76:710.e1-710.e8.
Hu N, Zhu J, Liang X, Wang Y, Guan J, Wen W, et al. T2 MRI at 3T of cartilage and menisci in patients with hyperuricemia: initial findings. Skeletal Radiol. 2022;51:607–18.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AG: received consultancy fees from Pfizer, Novartis, MerckSerono, TissueGene, AstraZeneca, and Regeneron. He is a shareholder of Boston Imaging Core Lab., LLC. FWR: Consultant to Calibr and Grünenthal. He is a shareholder of Boston Imaging Core Lab., LLC. CKK has received consultancy fees from Thuasne, Regeneron, Novartis, Kolon Tissue Gene, Taiwan Liposome, Amzell AZ, LG Chem, Express Scripts, and has received grants from Lilly, Pfizer GSK, Cumberland.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jarraya, M., Roemer, F., Kwoh, C.K. et al. Crystal arthropathies and osteoarthritis—where is the link?. Skeletal Radiol 52, 2037–2043 (2023). https://doi.org/10.1007/s00256-022-04246-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-022-04246-8