Abstract
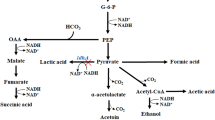
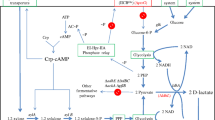
Acetoin, an important and high-value added bio-based platform chemical, has been widely applied in fields of foods, cosmetics, chemical synthesis, and agriculture. Lactate is a significant intermediate short-chain carboxylate in the anaerobic breakdown of carbohydrates that comprise ~ 18% and ~ 70% in municipal wastewaters and some food processing wastewaters, respectively. In this work, a series of engineered Escherichia coli strains were constructed for efficient production of acetoin from cheaper and abundant lactate through heterogenous co-expression of fusion protein (α-acetolactate synthetase and α-acetolactate decarboxylase), lactate dehydrogenase and NADH oxidase, and blocking acetate synthesis pathways. After optimization of whole-cell bioconversion conditions, the engineered strain BL-11 produced 251.97 mM (22.20 g/L) acetoin with a yield of 0.434 mol/mol in shake flasks. Moreover, a titer of 648.97mM (57.18 g/L) acetoin was obtained in 30 h with a yield of 0.484 mol/mol lactic acid in a 1-L bioreactor. To the best of our knowledge, this is the first report on the production of acetoin from renewable lactate through whole-cell bioconversion with both high titer and yield, which demonstrates the economy and efficiency of acetoin production from lactate.
Key Points • The lactate dehydrogenases from different organisms were expressed, purified, and assayed. • It is the first time that acetoin was produced from lactate by whole-cell biocatalysis. • The highest titer of 57.18 g/L acetoin was obtained with high theoretical yield in a 1-L bioreactor. |
Graphical abstract










Similar content being viewed by others
References
Ahmad A, Banat F, Taher H (2022) Enhanced lactic acid production from food waste in dark fermentation with indigenous microbiota. Biomass Convers Biorefin 12(8):3425–3434. https://doi.org/10.1007/s13399-020-00801-2
Arslan D, Steinbusch KJJ, Diels L, De Wever H, Hamelers HVM, Buisman CJN (2013) Selective carboxylate production by controlling hydrogen, carbon dioxide and substrate concentrations in mixed culture fermentation. Bioresour Technol 136:452–460. https://doi.org/10.1016/j.biortech.2013.03.063
Aydın G, Çelebi SS, Özyörük H, Yıldız A (2002) Amperometric enzyme electrode for L-(+)-lactate determination using immobilized L-(+)-lactate oxidase in poly(vinylferrocenium) film. Sens Actuators B 87(1):8–12. https://doi.org/10.1016/S0925-4005(02)00167-3
Bae SJ, Kim S, Hahn JS (2016) Efficient production of acetoin in Saccharomyces cerevisiae by disruption of 2,3-butanediol dehydrogenase and expression of NADH oxidase. Sci Rep 6:27667. https://doi.org/10.1038/srep27667
Bae S-J, Kim S, Park HJ, Kim J, Jin H, Kim B-g, Hahn J-S (2021) High-yield production of (R)-acetoin in Saccharomyces cerevisiae by deleting genes for NAD(P)H-dependent ketone reductases producing meso-2,3-butanediol and 2,3-dimethylglycerate. Metab Eng 66:68–78. https://doi.org/10.1016/j.ymben.2021.04.001
Bao Z-Z, Xu J-y, Jiang H-y, Huang Z-y (2009) Optimization beer yeast strain JX-07 production GSH through increase oxygen stress and yeast cell membrane permeability. Pharm Biotechnol 16(1):60–63 https://www.webofscience.com/wos/alldb/full-record/CSCD:3541368
Bao T, Zhang X, Rao Z, Zhao X, Zhang R, Yang T, Xu Z, Yang S (2014) Efficient whole-cell biocatalyst for acetoin production with NAD+ regeneration system through homologous co-expression of 2,3-butanediol dehydrogenase and NADH oxidase in engineered Bacillus subtilis. PloS One 9(7):e102951. https://doi.org/10.1371/journal.pone.0102951
Chen X, Zaro JL, Shen WC (2013) Fusion protein linkers: property, design and functionality. Adv Drug Deliv Rev 65(10):1357–1369. https://doi.org/10.1016/j.addr.2012.09.039
Conrado RJ, Varner JD, DeLisa MP (2008) Engineering the spatial organization of metabolic enzymes: mimicking nature’s synergy. Curr Opin Biotechnol 19(5):492–499. https://doi.org/10.1016/j.copbio.2008.07.006
Crout DHG, Hedgecock CJR (1979) The base-catalysed rearrangement of α-acetolactate (2-hydroxy-2-methyl-3-oxobutanoate) : a novel carboxylate ion migration in a tertiary ketol rearrangement. J Chem Soc, Perkin Trans 1:1982–1989. https://doi.org/10.1039/P19790001982
Cui Z, Mao Y, Zhao Y, Chen C, Tang Y-J, Chen T, Ma H, Wang Z (2018) Concomitant cell-free biosynthesis of optically pure D-(-)-acetoin and xylitol via a novel NAD+ regeneration in two-enzyme cascade. J Chem Technol Biotechnol 93(12):3444–3451. https://doi.org/10.1002/jctb.5702
Cui Z, Zhao Y, Mao Y, Shi T, Lu L, Ma H, Wang Z, Chen T (2019) In vitro biosynthesis of optically pure D-(-)-acetoin from meso-2,3-butanediol using 2,3-butanediol dehydrogenase and NADH oxidase. J Chem Technol Biotechnol 94(8):2547–2554. https://doi.org/10.1002/jctb.6050
Cui Z, Mao Y, Zhao Y, Zheng M, Wang Z, Ma H, Chen T (2020) One-pot efficient biosynthesis of (3R)-acetoin from pyruvate by a two-enzyme cascade. Cat Sci Technol 10(22):7734–7744. https://doi.org/10.1039/D0CY01332C
Cui Z, Wang Z, Zheng M, Chen T (2022) Advances in biological production of acetoin: a comprehensive overview. Crit Rev Biotechnol 42(8):1135–1156. https://doi.org/10.1080/07388551.2021.1995319
Dai J, Wang Z, Xiu ZL (2019) High production of optically pure (3R)-acetoin by a newly isolated marine strain of Bacillus subtilis CGMCC 13141. Bioprocess Biosyst Eng 42(3):475–483. https://doi.org/10.1007/s00449-018-2051-8
Fan L, Wang Y, Tuyishime P, Gao N, Li Q, Zheng P, Sun J, Ma Y (2018) Engineering artificial fusion proteins for enhanced methanol bioconversion. Chembiochem 19(23):2465–2471. https://doi.org/10.1002/cbic.201800424
Gao J, Xu Y, Li F, Ding G (2013) Production of S-acetoin from diacetyl by Escherichia coli transformant cells that express the diacetyl reductase gene of Paenibacillus polymyxa ZJ-9. Lett Appl Microbiol 57(4):274–281
Gao C, Li Z, Zhang L, Wang C, Li K, Ma C, Xu P (2015) An artificial enzymatic reaction cascade for a cell-free bio-system based on glycerol. Green Chem 17(2):804–807. https://doi.org/10.1039/c4gc01685h
Guo Z, Zhao X, He Y, Yang T, Gao H, Li G, Chen F, Sun M, Lee J, Zhang L (2017) Efficient (3R)-acetoin production from meso-2, 3-butanediol using a new whole-cell biocatalyst with co-expression of meso-2, 3-butanediol dehydrogenase, NADH oxidase and Vitreoscilla hemoglobin. J Microbiol Biotechnol 27(1):92–100
Guo S, Tan X, Wang Y, Li K, Lü C, Ma C, Gao C (2021) Enhanced in vitro cascade catalysis of glycerol into pyruvate and acetoin by integration with dihydroxy acid dehydratase from Paralcaligenes ureilyticus. Catalysts 11(11):1282
He Y, Chen F, Sun M, Gao H, Guo Z, Lin H, Chen J, Jin W, Yang Y, Zhang L, Yuan J (2018) Efficient (3S)-acetoin and (2S, 3S)-2, 3-butanediol production from meso-2, 3-butanediol using whole-cell biocatalysis. Molecules 23(3):691. https://doi.org/10.3390/molecules23030691
Hohagen H, Schwarz D, Schenk G, Guddat LW, Schieder D, Carsten J, Sieber V (2017) Deacidification of grass silage press juice by continuous production of acetoin from its lactate via an immobilized enzymatic reaction cascade. Bioresour Technol 245:1084–1092. https://doi.org/10.1016/j.biortech.2017.08.203
Hull SR, Yang BY, Venzke D, Kulhavy K, Montgomery R (1996) Composition of corn steep water during steeping. J Agric Food Chem 44(7):1857–1863. https://doi.org/10.1021/jf950353v
Jang JW, Jung HM, Im DK, Jung MY, Oh MK (2017) Pathway engineering of Enterobacter aerogenes to improve acetoin production by reducing by-products formation. Enzyme Microb Technol 106:114–118. https://doi.org/10.1016/j.enzmictec.2017.07.009
Ji X, Huang H, Ouyang P (2011) Microbial 2, 3-butanediol production: a state-of-the-art review. Biotechnol Adv 29(3):351–364. https://doi.org/10.1016/j.biotechadv.2011.01.007
Jia X, Kelly RM, Han Y (2018) Simultaneous biosynthesis of (R)-acetoin and ethylene glycol from D-xylose through in vitro metabolic engineering. Metab Eng Commun 7:e00074. https://doi.org/10.1016/j.mec.2018.e00074
Jiang T, Gao C, Ma C, Xu P (2014a) Microbial lactate utilization: enzymes, pathogenesis, and regulation. Trends Microbiol 22(10):589–599. https://doi.org/10.1016/j.tim.2014.05.008
Jiang T, Xu Y, Sun X, Zheng Z, Ouyang J (2014b) Kinetic characterization of recombinant Bacillus coagulans FDP-activated L-lactate dehydrogenase expressed in Escherichia coli and its substrate specificity. Protein Expr Purif 95:219–225. https://doi.org/10.1016/j.pep.2013.12.014
Khang Y-H, Kim I-W, Hah Y-R, Hwangbo J-H, Kang K-K (2003) Fusion protein of Vitreoscilla hemoglobin with D-amino acid oxidase enhances activity and stability of biocatalyst in the bioconversion process of cephalosporin C. Biotechnol Bioeng 82(4):480–488. https://doi.org/10.1002/bit.10592
Kleerebezem R, van Loosdrecht MCM (2007) Mixed culture biotechnology for bioenergy production. Curr Opin Biotechnol 18(3):207–212. https://doi.org/10.1016/j.copbio.2007.05.001
Kromus S, Wachter B, Koschuh W, Mandl M, Narodoslawsky M (2004) The green biorefinery Austria - developement of an integrated system for green mass utilization. Chem Biochem Eng Q 18(1):7–12
Li Z, Zhang M, Jiang T, Sheng B, Ma C, Xu P, Gao C (2017) Enzymatic cascades for efficient biotransformation of racemic lactate derived from corn steep water. ACS Sustain Chem Eng 5(4):3456–3464. https://doi.org/10.1021/acssuschemeng.7b00136
Li JX, Huang YY, Chen XR, Du QS, Meng JZ, Xie NZ, Huang RB (2018) Enhanced production of optical (S)-acetoin by a recombinant Escherichia coli whole-cell biocatalyst with NADH regeneration. RSC Adv 8(53):30512–30519. https://doi.org/10.1039/c8ra06260a
Lin Z, Xu Z, Li Y, Wang Z, Chen T, Zhao X (2014) Metabolic engineering of Escherichia coli for the production of riboflavin. Microb Cell Fact 13:104 doi: https://doi.org/10.1186/s12934-014-0104-5
Liu Z, Qin J, Gao C, Hua D, Ma C, Li L, Wang Y, Xu P (2011) Production of (2S,3S)-2,3-butanediol and (3S)-acetoin from glucose using resting cells of Klebsiella pneumonia and Bacillus subtilis. Bioresour Technol 102(22):10741–10744
Liu J-M, Chen L, Dorau R, Lillevang SK, Jensen PR, Solem C (2020) From waste to taste—efficient production of the butter aroma compound acetoin from low-value dairy side streams using a natural (nonengineered) Lactococcus lactis dairy isolate. J Agric Food Chem 68(21):5891–5899. https://doi.org/10.1021/acs.jafc.0c00882
Lu L, Mao Y, Kou M, Cui Z, Jin B, Chang Z, Wang Z, Ma H, Chen T (2020) Engineering central pathways for industrial-level (3R)-acetoin biosynthesis in Corynebacterium glutamicum. Microb Cell Fact 19(1):102. https://doi.org/10.1186/s12934-020-01363-8
Ma CQ, Xu P, Dou YM, Qu YB (2003) Highly efficient conversion of lactate to pyruvate using whole cells of Acinetobacter sp. Biotechnol Prog 19(6):1672–1676. https://doi.org/10.1021/bp0341242
Ma CQ, Xu P, Qiu JH, Zhang ZJ, Wang KW, Wang M, Zhang YN (2004) An enzymatic route to produce pyruvate from lactate. Appl Microbiol Biotechnol 66(1):34–39. https://doi.org/10.1007/s00253-004-1646-y
Mao YF, Fu J, Tao R, Huang C, Wang ZW, Tang YJ, Chen T, Zhao XM (2017) Systematic metabolic engineering of Corynebacterium glutamicum for the industrial-level production of optically pure D-(-)-acetoin. Green Chem 19(23):5691–5702. https://doi.org/10.1039/c7gc02753b
Meng W, Ma C, Xu P, Gao C (2022) Biotechnological production of chiral acetoin. Trends Biotechnol 40(8):958–973. https://doi.org/10.1016/j.tibtech.2022.01.008
Oshiro M, Hanada K, Tashiro Y, Sonomoto K (2010) Efficient conversion of lactic acid to butanol with pH-stat continuous lactic acid and glucose feeding method by Clostridium saccharoperbutylacetonicum. Appl Microbiol Biotechnol 87(3):1177–1185. https://doi.org/10.1007/s00253-010-2673-5
Peng K, Guo D, Lou Q, Lu X, Cheng J, Qiao J, Lu L, Cai T, Liu Y, Jiang H (2020) Synthesis of ligustrazine from acetaldehyde by a combined biological–chemical approach. ACS Synth Biol 9(11):2902–2908. https://doi.org/10.1021/acssynbio.0c00113
Raunkjær K, Hvitved-Jacobsen T, Nielsen PH (1994) Measurement of pools of protein, carbohydrate and lipid in domestic wastewater. Water Res 28(2):251–262. https://doi.org/10.1016/0043-1354(94)90261-5
Sharkey MA, Maher MA, Guyonvarch A, Engel PC (2011) Kinetic characterisation of recombinant Corynebacterium glutamicum NAD+-dependent LDH over-expressed in E. coli and its rescue of an lldD- phenotype in C. glutamicum: the issue of reversibility re-examined. Arch Microbiol 193(10):731–740. https://doi.org/10.1007/s00203-011-0711-z
Tao Y, Hu X, Zhu X, Jin H, Xu Z, Tang Q, Li X (2016) Production of butyrate from lactate by a newly isolated Clostridium sp. BPY5. Appl Biochem Biotechnol 179(3):361–374. https://doi.org/10.1007/s12010-016-1999-6
Tefara SF, Begna Jiru E, G/Meskel Bairu A (2022) Optimization of fermentation condition for production of lactic acid from khat (“Catha edulis”) waste by using immobilized Lactobacillus plantarum. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-022-02797-3
Wang Y, Tashiro Y, Sonomoto K (2015) Fermentative production of lactic acid from renewable materials: recent achievements, prospects, and limits. J Biosci Bioeng 119(1):10–18. https://doi.org/10.1016/j.jbiosc.2014.06.003
Wang S, Hou Y, Chen X, Liu L (2019) Kick-starting evolution efficiency with an autonomous evolution mutation system. Metab Eng 54:127–136. https://doi.org/10.1016/j.ymben.2019.03.010
Wu C-W, Whang L-M, Cheng H-H, Chan K-C (2012) Fermentative biohydrogen production from lactate and acetate. Bioresour Technol 113:30–36. https://doi.org/10.1016/j.biortech.2011.12.130
Xiao Z, Lu JR (2014a) Generation of acetoin and its derivatives in foods. J Agric Food Chem 62(28):6487–6497. https://doi.org/10.1021/jf5013902
Xiao Z, Lu JR (2014b) Strategies for enhancing fermentative production of acetoin: a review. Biotechnol Adv 32(2):492–503. https://doi.org/10.1016/j.biotechadv.2014.01.002
Xu P, Qiu J, Gao C, Ma C (2008) Biotechnological routes to pyruvate production. J Biosci Bioeng 105(3):169–175. https://doi.org/10.1263/jbb.105.169
Yadav N, Nain L, Khare SK (2021) One-pot production of lactic acid from rice straw pretreated with ionic liquid. Bioresour Technol 323:124563. https://doi.org/10.1016/j.biortech.2020.124563
Zhang Y, Chen X, Luo J, Qi B, Wan Y (2014) An efficient process for lactic acid production from wheat straw by a newly isolated Bacillus coagulans strain IPE22. Bioresour Technol 158:396–399. https://doi.org/10.1016/j.biortech.2014.02.128
Zhang L, Singh R, Sivakumar D, Guo Z, Li J, Chen F, He Y, Guan X, Kang YC, Lee JK (2018) An artificial synthetic pathway for acetoin, 2,3-butanediol, and 2-butanol production from ethanol using cell free multi-enzyme catalysis. Green Chem 20(1):230–242. https://doi.org/10.1039/c7gc02898a
Zhang X, Han R, Bao T, Zhao X, Li X, Zhu M, Yang T, Xu M, Shao M, Zhao Y, Rao Z (2019) Synthetic engineering of Corynebacterium crenatum to selectively produce acetoin or 2,3-butanediol by one step bioconversion method. Microb Cell Fact 18(1):128. https://doi.org/10.1186/s12934-019-1183-0
Zhao R, Zheng S, Duan C, Liu F, Yang L, Huo G (2013) NAD-dependent lactate dehydrogenase catalyses the first step in respiratory utilization of lactate by Lactococcus lactis. FEBS Open Bio 3:379–386. https://doi.org/10.1016/j.fob.2013.08.005
Zhou X, Zhou X, Zhang H, Cao R, Xu Y (2018) Improving the performance of cell biocatalysis and the productivity of acetoin from 2,3-butanediol using a compressed oxygen supply. Process Biochem 64:46–50. https://doi.org/10.1016/j.procbio.2017.09.027
Zhu CJ, Tao S, Dong L, Wu JL, Yong C, Wang LF, Kai G, Ying HJ, Ouyang PK (2016) Production of liquid hydrocarbon fuels with acetoin and platform molecules derived from lignocellulose. Green Chem 18(7):2165–2174. https://doi.org/10.1039/c5gc02414e
Zhu X, Zhou Y, Wang Y, Wu T, Li X, Li D, Tao Y (2017) Production of high-concentration n-caproic acid from lactate through fermentation using a newly isolated Ruminococcaceae bacterium CPB6. Biotechnol Biofuels 10(1):102. https://doi.org/10.1186/s13068-017-0788-y
Funding
This work was supported by the National Key Research and Development Program of China (2021YFC2100700) and the National Natural Science Foundation of China (grant NSFC-21576191).
Author information
Authors and Affiliations
Contributions
TC and ZC conceived and designed the project. ZC carried out the experiments. ZC and ZW analyzed the data. MZ, MD, and WD provided some support during the experimental phase. ZC and TC wrote the manuscript with contributions from all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cui, Z., Zheng, M., Ding, M. et al. Efficient production of acetoin from lactate by engineered Escherichia coli whole-cell biocatalyst. Appl Microbiol Biotechnol 107, 3911–3924 (2023). https://doi.org/10.1007/s00253-023-12560-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12560-x