Abstract
Background
Cerebral sinovenous thrombosis (CSVT) has been proposed in legal settings to be an atraumatic mimic of abusive head trauma (AHT).
Objective
The objective of this study was to determine the prevalence of CSVT and subdural hemorrhage (SDH) in a large AHT population.
Materials and methods
This retrospective cohort study measured the prevalence of CSVT and SDH on magnetic resonance venograms in 243 patients diagnosed with AHT at a single center. We also reported additional intra- and extracranial injuries, head injury severity and length of hospital stay.
Results
Among 243 patients diagnosed with AHT, 7% (16/243) had CSVT. SDH was present in 94% (15/16) of the CSVT cases. Cytotoxic edema and subarachnoid hemorrhage were in 88% (14/16) and 69% (11/16) of the CSVT cases, respectively. Extracranial signs of abuse were also in 100% (16/16) of the patients with CSVT. Critical to maximal head injury severity (abbreviated injury scale >=5) was in 75% (12/16) of the CSVT population vs. 33% (82/243) in the total AHT population. Length of hospital and pediatric intensive care unit stay was greater in those with CSVT (10 vs. 21.9 and 3.5 vs. 7.3 days).
Conclusion
These findings suggest that CSVT is uncommon in AHT and is associated with additional traumatic injuries and greater injury severity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Child maltreatment is a global health emergency [1]. In 2018, in the United States (U.S.) alone, there were 3.5 million investigations of suspected child abuse and neglect, and 678,000 victims of maltreatment [2]. Among this population, abusive head trauma (AHT) is common [3] and is one of the leading causes of mortality [4]. Common imaging findings associated with AHT include skull fractures, subdural hematomas, cytotoxic brain injury and injured bridging veins [5]. These findings are often the result of direct impact or rotational acceleration/deceleration forces that result in the disruption of the cortical bridging veins that traverse the subdural space.
Some medical conditions can produce findings similar to those of abuse and result in the misdiagnosis of child abuse. For example, fractures seen in patients with osteogenesis imperfecta, a disease of bone formation, can mimic fractures that are often observed in abuse [6]. Conversely, some theories of alternative etiology for the findings we observe in child abuse have been proposed, primarily in medicolegal settings, with little or no supporting evidence [7,8,9]. One theory, based on case reports, suggests that the subdural hematomas associated with AHT are the result of cerebral sinovenous thrombosis (CSVT) and not inflicted trauma [10,11,12,13,14]. These case reports, however, failed to determine the cause-effect relationship of CSVT and subdural hematomas or other signs of trauma [15]. If CSVT mimicked AHT by causing subdural hematomas, then atraumatic CSVT cases (i.e. patients who lack other signs of trauma such as bruising, fractures, abdominal injuries and retinal hemorrhages) should not be associated with the witness or confession of abuse. Nonetheless, this theory has impacted the work-up of AHT, specifically as it relates to the utilization of magnetic resonance (MR) venography to evaluate for CSVT. To that end, some institutions utilize contrast-enhanced MR venography in children with concern for AHT, as it is more sensitive for detecting thrombus than non-contrast MR venography [16,17,18,19]. While intracranial findings such as injured bridging veins are common in AHT, the prevalence of CSVT in AHT is unknown.
The objective of this study is to determine the prevalence of CSVT in a large AHT population and determine the relative prevalence of intra- and extracranial trauma in AHT victims with and without CSVT. Our hypothesis is that CSVT is associated with other abusive injuries and with witnessed or confessed abuse.
Materials and methods
Study design
This is a single-center, retrospective study of patients who had a contrast-enhanced MR venogram during a hospitalization in which they were diagnosed with AHT. The study protocol was approved by the Colorado Multiple Institution Review Board.
Setting, participant identification and data collection
The center is a 434-bed tertiary referral center with an established child protection team (CPT). Patients were identified from a CPT database of all children diagnosed with AHT since 2012. Inclusion criteria for this study were: diagnosis of AHT by the CPT at the Children's Hospital Colorado (Aurora, CO, USA) between Jan. 1, 2012 and Dec. 30, 2020, age <18 years at the time of presentation, and completion of a contrast-enhanced intracranial MR venogram during the acute phase of their initial hospitalization. Data were extracted by clinicians and research assistants from the medical record.
Race and ethnicity were based on self-reported data from the medical record. Each patient was individually verified by a member of the CPT to confirm a diagnosis of AHT from the assessment of the multidisciplinary staffing note. Past medical history was included as documented in the initial hospitalization. Admission of harm was defined as admission from a caregiver to causing the injuries to a medical provider, or if the medical team was informed of an admission of harm per investigation by human services or law enforcement. All patients were evaluated for additional signs of trauma that included abdominal injuries, fractures, retinal hemorrhage and cutaneous injury.
Image and data analysis
A review of the imaging reports for all patients who met inclusion criteria was performed. The primary outcome measure was prevalence of CSVT in AHT, whereby a 95% normal proportion confidence interval (CI) was calculated. All positive or indeterminate cases of CSVT were examined by a board-certified pediatric neuroradiologist (D.M.M., 10 years of experience) to confirm the presence or absence of CSVT. While the assessors of outcomes were not formally blinded to the presence or absence of CSVT, outcomes were recorded before the planning of this retrospective study, and were recorded by different data operators than those who recorded the presence of CSVT, which minimizes the chance for confirmation bias. Any cases that remained indeterminate were adjudicated by a second pediatric neuroradiologist (N.V.S., 11 years of experience). The location and length of the CSVT was recorded, as well as whether the clot appeared to originate within an injured bridging vein and extend into the sinus (i.e. the bulk of the clot was within the bridging vein) versus originate within the sinus (i.e. the bulk of the clot was within the venous sinus). All cases were also examined for subdural hematomas, cytotoxic edema, parenchymal laceration, contusion, shear injury and subarachnoid hemorrhage.
Results
In all, 243 patients were identified with AHT who had contrast-enhanced MR venograms. Characteristics of the 243 identified participants are shown in Table 1. Participants were mostly white, with a slight male predominance (65%). CSVT was present in 7% (16/243; 95% CI, 4–11%) and indeterminate in 2% (5/243; 95% CI, 1–5%) of cases within the AHT population. The most common location for CSVT was within the superior sagittal sinus (Fig. 1, Table 2). Thrombi ranged from 4 to 206 mm in length with an average length of 53 mm (Table 2). Among confirmed cases of CSVT, 7 demonstrated evidence of the clot being centered upon an injured bridging vein and propagating back into the dural venous sinus (Fig. 2).
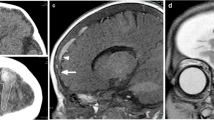
A 3-month-old boy with abusive head trauma. a An axial diffusion-weighted magnetic resonance (MR) image shows diffuse cytotoxic edema throughout the supratentorial brain (arrowheads), largely sparing the deep gray nuclei. Axial T2-weighted (b) and (c) gradient recalled echo MR images reveal subdural hematomas about the bilateral convexities (arrows). d A sagittal contrast-enhanced, fat-suppressed, T1-weighted MR venogram image demonstrates a filling defect within the superior sagittal sinus (arrowheads)
An 8-month-old boy with abusive head trauma. a An axial diffusion-weighted magnetic resonance (MR) image shows faint cytotoxic edema involving the supratentorial cortices, most evident posteriorly (arrowheads). b An axial T2-weighted MR image demonstrates subdural hematomas about the bilateral convexities (arrows), left larger than right. c An axial T2-weighted MR image, at the cranial vertex, illustrates abnormal signal intrinsic to a left-side bridging vein that extends into the superior sagittal sinus (arrows). d A coronal contrast-enhanced, fat-suppressed, T1-weighted MR venogram image confirms thrombus within a left-side bridging vein extending into the superior sagittal sinus (arrowheads)
Subdural hematomas were present in 94% (228/243) of the total AHT population, and 94% (15/16) of the confirmed CSVT cases. Subdural hematoma was absent in one case of CSVT. There were, however, additional injuries including extracranial fractures and liver injury.
Among confirmed CSVT patients, 75% (12/16) were determined to have critical or greater (abbreviated injury scale [AIS]> or =5) severity of head injury, compared with 33% (82/243) in the total AHT population. Cytotoxic edema and subdural hematomas were reported in 88% (14/16) and 69% (11/16) of CSVT cases, respectively (Table 3). Brain contusions were present in 38% (6/16) of CSVT cases. Extracranial injuries associated with abuse were present in 100% (16/16) of confirmed CSVT cases. Specifically, 75% (12/16) with cutaneous injury, 56% (9/16) with retinal hemorrhage, 56% (9/16) with one or more fractures, and 19% (3/16) with abdominal visceral injury.
CSVT was treated with anticoagulation in 25% (4/16) of patients. The mean length of hospital and pediatric intensive care unit (PICU) stays increased from 10 to 21.9 and 3.5 to 7.3 days, respectively, for confirmed CSVT cases.
Discussion
To the of best our knowledge, the prevalence of CSVT in AHT is not known. We found a low prevalence of CSVT and high prevalence of subdural hematoma in a large AHT population. These findings contradict the theory that CSVT is the cause of subdural hematomas [10,11,12,13,14]. To that end, if any subdural hematomas in our study resulted from CSVT, then these cases would likely be absent of other signs of abuse including subarachnoid hemorrhage, cytotoxic injury, brain contusions, parenchymal laceration, shear injury, bruising, fractures, abdominal injury and retinal hemorrhage; which was not the case. Our results do not support the theory that CSVT is often a nontraumatic source of subdural hematomas that mimic AHT. All cases had findings that raise significant concern for abuse, irrespective of the presence of subdural hematoma or CSVT. Furthermore, a single case of CSVT lacked subdural hematoma. In our cohort, CSVT was associated with additional traumatic injuries and greater injury severity; increased AIS and PICU length of stay. This would suggest that CSVT is the result of critical illness and brain injury in an AHT child, not the cause of it. Compared to historical cohorts of children with CSVT that resulted from dehydration or metabolic illness, children in our cohort who had CSVT and were diagnosed with AHT had significant signs of abuse. Future studies with larger sample sizes should address this hypothesis, and look at clinical indicators (e.g., impact injury, dehydration, degree of swelling, location/size of ADH) that may predispose AHT patients to CVST. Given the low, but not insignificant, prevalence of CSVT demonstrated in this patient population, we support the use of MR venography in AHT to diagnose CSVT. Furthermore, in light of the unknown risks of gadolinium retention, non-contrast MR venography should suffice.
The major limitation of this study is that it represents a single-center cohort (n=243) whereby the population is comprised of a single geographical region with limited racial, ethnic and gender diversity. These findings need to be replicated across several diverse populations in order to generalize the evidence and potentially alter routine imaging protocols related to AHT.
Conclusion
Cerebral sinovenous thrombosis was present in the minority of cases within a large abusive head trauma population; conversely, subdural hemorrhage was present in the majority of cases. All confirmed CSVT cases included additional injuries associated with abuse. CSVT was associated with greater head injury severity and a longer stay in the hospital and intensive care unit.
References
Violence Against Children. Health Topics. World Health Organization. https://www.who.int/health-topics/violence-against-children#tab=tab_1. Accessed 15 June 2021
(2018) Child maltreatment. In: Administration for children and families AoC, youth and families, Children’s Bureau (ed). U.S. Department of Health & Human Services. https://www.acf.hhs.gov/sites/default/files/documents/cb/cm2018.pdf. Accessed 15 June 2021
Wright JN (2017) CNS injuries in abusive head trauma. AJR Am J Roentgenol 208:991–1001
Klevens J, Leeb RT (2010) Child maltreatment fatalities in children under 5: findings from the National Violence Death Reporting System. Child Abuse Negl 34:262–266
Vilanilam GK, Jayappa S, Desai S et al (2021) Venous injury in pediatric abusive head trauma: a pictorial review. Pediatr Radiol 51:918–926
Singh Kocher M, Dichtel L (2011) Osteogenesis imperfecta misdiagnosed as child abuse. J Pediatr Orthop B 20:440–443
Choudhary AK, Servaes S, Slovis TL et al (2018) Consensus statement on abusive head trauma in infants and young children. Pediatr Radiol 48:1048–1065
Edwards GA (2015) Mimics of child abuse: can choking explain abusive head trauma? J Forensic Legal Med 35:33–37
Leventhal JM, Edwards GA (2017) Flawed theories to explain child physical abuse: what are the medical-legal consequences? JAMA 318:1317–1318
Bailey OT, Hass GM (1937) Dural sinus thrombosis in early life: recovery from acute thrombosis of the superior longitudinal sinus and its relation to certain acquired cerebral lesions in childhood. Brain 60:293–314
Bucy PC, Lesemann FJ (1942) Idiopathic recurrent thrombophlebitis: with cerebral venous thrombosis and an acute subdural hematoma. JAMA 119:402–405
Matsuda M, Matsuda I, Sato M, Handa J (1982) Superior sagittal sinus thrombosis followed by subdural hematoma. Surg Neurol 18:206–211
Singh S, Kumar S, Joseph M et al (2005) Cerebral venous sinus thrombosis presenting as subdural haematoma. Australas Radiol 49:101–103
Takamura Y, Morimoto S, Uede T et al (1996) Cerebral venous sinus thrombosis associated with systemic multiple hemangiomas manifesting as chronic subdural hematoma--case report. Neurol Med Chir (Tokyo) 36:650–653
McLean LA, Frasier LD, Hedlund GL (2012) Does intracranial venous thrombosis cause subdural hemorrhage in the pediatric population? AJNR Am J Neuroradiol 33:1281–1284
Ayanzen RH, Bird CR, Keller PJ et al (2000) Cerebral MR venography: normal anatomy and potential diagnostic pitfalls. AJNR Am J Neuroradiol 21:74–78
Lettau M, Sartor K, Heiland S, Hahnel S (2009) 3T high-spatial-resolution contrast-enhanced MR angiography of the intracranial venous system with parallel imaging. AJNR Am J Neuroradiol 30:185–187
Liang L, Korogi Y, Sugahara T et al (2001) Evaluation of the intracranial dural sinuses with a 3D contrast-enhanced MP-RAGE sequence: prospective comparison with 2D-TOF MR venography and digital subtraction angiography. AJNR Am J Neuroradiol 22:481–492
Widjaja E, Shroff M, Blaser S et al (2006) 2D time-of-flight MR venography in neonates: anatomy and pitfalls. AJNR Am J Neuroradiol 27:1913–1918
Acknowledgments
National Institutes of Health/National Center for Advancing Translational Sciences Colorado Clinical & Translational Science Award Grant Number UL1 TR002535, End Child Abuse and Neglect (EndCAN)-Helfer Society Grant Program for Child Maltreatment Research June 2020–June 2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Burtard, C., Panks, J., Silverman, L.B. et al. Prevalence of cerebral sinovenous thrombosis in abusive head trauma. Pediatr Radiol 53, 78–85 (2023). https://doi.org/10.1007/s00247-022-05462-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-022-05462-z