Abstract
Post-pericardiotomy syndrome (PPS) is an inflammatory process involving the pleura, pericardium, or both and occurs after cardiothoracic surgery. Surgical atrial septal defect (ASD) closure is associated with higher incidence of PPS post-operatively as compared to other operations. Reported incidence of PPS varies from 1 to 40%. NSAIDs are often used to treat PPS and in our center, some practitioners have prescribed ibuprofen prophylactically. This study sought to investigate the impact of prophylactic treatment with ibuprofen on the development and severity of PPS following surgical ASD closure, with particular attention to secundum-type ASDs. We retrospectively reviewed clinical and operative data of all surgical ASD repairs in our center from 1/2007 to 7/2017. ASDs were grouped by subtype. PPS was considered positive if the primary cardiologist diagnosed and documented clinical signs of PPS on post-operative outpatient follow-up. Records were reviewed to confirm documented diagnosis of PPS. A total of 245 cases were reviewed with 207 having sufficient data. Median age was 2 years (range 4 months–27 years), female 57%. Overall incidence of PPS was 10%. There was no difference in incidence of PPS in those prescribed ibuprofen as compared to those who were not. This was true for both the entire cohort and the subgroup analysis (P = 1.0). Four patients overall required pericardiocentesis, none of whom received prophylactic ibuprofen. Prophylactic ibuprofen prescription following surgical ASD repair did not reduce the rate of PPS in our cohort.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Post-pericardiotomy syndrome (PPS) is an inflammatory process involving the pleura, pericardium, or both and occurs after cardiothoracic surgery. Atrial Septal Defects (ASDs) are among the most common forms of congenital heart disease (CHD) constituting 8–10% of all forms of CHD in children [1]. Despite the availability of transcatheter closure for some patients, ASDs are commonly surgically addressed during childhood [2]. Surgical ASD closure is associated with a higher incidence of PPS relative to other cardiac operations [3]. Incidence of PPS varies widely, from 1 to 40%, among published series [4]. A pediatric retrospective study of the incidence of PPS following repair of secundum-type ASDs determined the incidence to be 28% (27/97) [4]. A recent single-center French study of patients with acute pericarditis determined PPS to be the etiology in 21% (197/933) of cases [5]. First-line therapy for PPS is typically a non-steroidal anti-inflammatory drug (NSAID) such as ibuprofen.
Starting in 2010, a subset of our practitioners began to prescribe ibuprofen prophylactically in our center after surgical ASD repairs in an attempt to prevent PPS. In this study, we aim to determine whether that prophylactic therapy reduced the incidence or severity of PPS.
Materials and Methods
The study was approved by the Northwell Health Institutional Board Review and waiver of consent was obtained.
We retrospectively reviewed clinical and procedural data for all patients with surgical ASD repairs in our center from January 2007 to July 2017. We included patients with secundum-type ASDs, primum-type ASDs (primarily partial atrioventricular canal defects), sinus venosus (SV)-type ASDs, and coronary sinus (CS)-type ASDs. We excluded patients with other additional major CHD including complete and transitional atrioventricular canal defects, transposition of the great vessels, and ventricular septal defects. We also excluded those subjects with insufficient outpatient medical records to determine with certainty if PPS developed.
We reviewed imaging records, operative notes, discharge records, outpatient clinical data, and electrocardiograms of all subjects from prior to surgical repair until 6 months post-operatively. We recorded if ibuprofen was prescribed at hospital discharge and the dosing instructions. Subjects were deemed as having PPS if the diagnosis and typical clinical findings (i.e., fever, chest pain, rub, pericardial effusion, or pleural effusion) were documented on post-operative outpatient follow-up evaluations by the primary cardiologist within 6 weeks of surgical repair. The presence and severity of pericardial effusions were recorded as documented on official echocardiographic reports at the time of PPS diagnosis. Echocardiographic images were not interpreted again for the purpose of this study. Our institution follows published guidelines and standards for performance of pediatric echocardiograms, including grading severity of pericardial effusions criteria [6]. We recorded whether patients required pericardiocentesis as a measure of PPS severity.
Descriptive statistics were given as median and range for continuous factors, and frequency and percentage for categorical factors. The association between each categorical factor and PPS was examined using the Fisher’s exact test. The association between age (in days) and PPS was examined using the Mann–Whitney test.
Results
We reviewed the records of all isolated ASD closures (n = 245) surgically repaired in our center over the study period. Among those, 207 had adequate data and were included in the study. Median age was 2 years old (range 4 months–27 years), and 57% were females. PPS occurred in 20 subjects (10%). All PPS cases were diagnosed in the outpatient setting at a median of 8.5 days post-operatively (range 6–36 days).
Prophylactic ibuprofen had been prescribed for 89 subjects (43%) in the overall cohort. Median age was 4 years old (range 5 months–26 years). Subjects with primum-type ASD were less likely to have been prescribed ibuprofen (see Table 1). Ibuprofen was prescribed three times daily in 68% of cases, four times daily in 30% of cases, and twice daily in 2%. In total, 9 (10%) of those prescribed ibuprofen and 11 (9%) of those not prescribed were subsequently diagnosed with PPS. There was no difference in the incidence of PPS in those prescribed ibuprofen as compared to those who were not (P = 1.0).
No difference in the impact of ibuprofen on the incidence of PPS based on ASD subtype was seen (see Table 1). A total of 106 subjects with secundum-type ASDs were included in the analysis. Ibuprofen was prescribed in 59 (56%) subjects of which 7 (12%) developed PPS, as compared to 5 (11%) in those who had not been prescribed ibuprofen. No difference in the incidence of PPS was found in this subgroup analysis (P = 1.0).
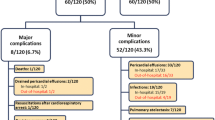
The clinical characteristics of all subjects diagnosed with PPS (n = 20) are summarized in Table 2. Pericardial (9 small, 6 moderate, 3 large) or pleural effusions (1 small, 1 moderate), greater than those seen on inpatient discharge imaging, were seen in all cases. Clinical symptoms or signs (i.e., fever, chest pain or rub) were seen in 5–25% of cases. Fever was not seen in isolation but accompanied a documented rub on exam or pericardial effusion on echocardiogram. Pericardiocentesis for a hemodynamically significant pericardial effusion was performed in 4 (20%) cases. Of the subjects with PPS who received ibuprofen, 0/9 (0%) required pericardiocentesis; of the subjects who did not receive ibuprofen, 4/11 (36%), required pericardiocentesis. This difference was not statistically significant (P = 0.94). There was no association between age of repair and diagnosis of PPS (P = 0.09). Similarly, in subgroup analysis of secundum ASDs, there was no association between age of repair and diagnosis of PPS (P = 0.09).
Discussion
PPS is a common post-operative morbidity for patients undergoing surgical ASD closure, especially those undergoing an operation for secundum-type ASDs. To our knowledge, there is no routine preventative measure in children which can help prevent PPS. Since NSAIDs like ibuprofen are the typical first-line therapy for treatment of PPS, it has been suggested that prophylactic treatment with NSAIDs might help prevent PPS.
There is no clear consensus in the literature regarding the diagnostic definition of PPS. The European Society of Cardiology defines PPS as having two of the following five criteria after cardiac injury: (1) fever without an alternative cause, (2) pericarditic or pleuritic chest pain, (3) pericardial of pleural rubs, (4) evidence of pericardial effusion, and/or (5) pleural effusion with elevated CRP [7]. We used these criteria along with the assessment of the primary cardiologist when reviewing outpatient records in assigning the subjects the diagnosis of PPS.
Only a single clinical trial of adults undergoing general cardiac surgery evaluated the efficacy of NSAIDs for the treatment of PPS and determined that both ibuprofen and indomethacin are more effective, as compared to placebo, in treating PPS [8]. According to the most recent recommendations by the European Society of Cardiology (Class IB) [7], ibuprofen is favored as a first-line therapy over other alternatives due to its limited side effect profile, and was chosen for this study for the same reason.
Past studies to evaluate the efficacy of NSAIDs in preventing PPS have had conflicting results. Two pediatric studies by Beland et al. [9] and Gill et al. [10] described a series of 72 (0–21 years; controlled trial) and 177 children (0–18 years; retrospective cohort) following congenital heart surgery where the incidence of PPS was not significantly reduced by the use of Aspirin. In adults following general cardiac surgery, prophylaxis with indomethacin and diclofenac has been trialed and appears to possibly have favorable results [11]. A more comprehensive systematic review combining 894 patients following both congenital heart and general cardiac surgery included three comparison groups—colchicine vs placebo (two RCTs, 471 adult patients), methylprednisolone vs placebo (one RCT 246 pediatric patients), aspirin vs historical control (one non-randomized study, 177 pediatric patients)—and concluded that only colchicine was associated with decreased risk of PPS (odds ratio 0.38) [12]. Further evidence for the use of colchicine in the prevention of PPS in adults undergoing general cardiac surgery was strengthened in the “colchicine for prevention of the PPS and post-operative atrial fibrillation (COPPS-2)” trial [13].
The etiology behind PPS and the reason it is seen at a higher incidence following surgical ASD repairs in particular, remain unclear despite being a well-accepted assertion. Most theories explaining the etiology are similar to those explaining the pericarditis seen post-myocardial infarctions (the so-called Dressler’s Syndrome [14]). These theories mainly postulate that an autoimmune-mediated response occurs targeting cardiac antigens. Some have suggested that the autoimmune response is triggered by a cross reactivity to viral antibodies. Others have focused on an inflammatory response to direct injury to pericardial cells [15,16,17]. None of these address the higher incidence seen following ASD repairs. Perhaps, it is possible that the generally healthier patient population undergoing surgical ASD repairs, typically not in active congestive heart failure, are more able to mount a potent immune or inflammatory response following cardiac surgery increasing, thereby the risk of PPS. That being said, PPS has been reported in immunosuppressed transplant recipients [18].
We sought to decrease the rate of PPS following surgical ASD repairs in our center by prescribing ibuprofen prophylactically at the time of hospital discharge. This practice began in 2010 and became more routine in 2014. Ibuprofen was prescribed at 10 mg/kg dosing or adult dosing (max 600 mg/dose) and typically prescribed at 8-h intervals. The decision to prescribe ibuprofen and the dosing interval was practitioner dependent. The decision to stop ibuprofen as an outpatient was at the discretion of the primary cardiologist who typically discontinued, or in some cases weaned ibuprofen, during the first outpatient follow-up visit (typically 1–2 weeks from discharge). Of the nine subjects diagnosed with PPS while having been prescribed ibuprofen, the median time for diagnosis was 10 days post-operatively (range 6–18 days), four of these subjects were diagnosed at 14 days or later suggesting that it is possible that ibuprofen was discontinued prior to the development of PPS.
In this study, the prophylactic treatment with ibuprofen did not reduce the incidence of PPS. The overall incidence remained approximately 10% across all subjects despite the use of ibuprofen. There was no reduction in incidence of PPS within the secundum-type ASD subgroup, classically associated with PPS in the pediatric population. There was no association demonstrated between the diagnosis of PPS and age. Interestingly, no patient who was prescribed ibuprofen required a pericardiocentesis, which suggests the possibility that the severity of the PPS was lessened by the prophylaxis. While it was not statistically significant, this study was not powered to investigate that association. A future study should be undertaken to investigate the effects of ibuprofen on need for pericardiocentesis in patients with PPS.
Limitations of this study include its retrospective nature and lack of prospective randomization to treatment and non-treatment arms. However, the two groups were relatively similar. The nature of PPS makes it necessary that we rely on outpatient, subjective, documentation for the diagnosis of PPS. Thus, differences in the clinical approach of practicing cardiologists in diagnosing PPS might have impacted the overall incidence of PPS in our cohort. Ibuprofen was assessed as prescribed, and true subject compliance, or the effect of dosing intervals, cannot be assessed. Lastly, this study cannot account for confounding changes occurring at the same time period such as modifications in surgical or cardiopulmonary bypass perfusion techniques.
Conclusion
Prophylactic ibuprofen prescription following surgical ASD repair did not affect the rate of PPS diagnosis. Its effect on need for pericardiocentesis in patients with PPS may deserve further investigation.
Abbreviations
- PPS:
-
Post-pericardiotomy syndrome
- ASD:
-
Atrial septal defect
- CHD:
-
Congenital heart disease
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- SV:
-
Sinus venosus
- CS:
-
Coronary sinus
References
Hoffman JI, Kaplan S (2002) The incidence of congenital heart disease. J Am Coll Cardiol 39(12):1890–1900
Campbell M (1970) Natural history of atrial septal defect. Br Heart J 32(6):820–826
Clapp SK, Garson A Jr, Gutgesell HP, Cooley DA, McNamara DG (1980) Postoperative pericardial effusion and its relation to postpericardiotomy syndrome. Pediatrics 66(4):585–588
Heching HJ, Bacha EA, Liberman L (2015) Post-pericardiotomy syndrome in pediatric patients following surgical closure of secundum atrial septal defects: incidence and risk factors. Pediatr Cardiol 36(3):498–502
Gouriet F, Levy PY, Casalta JP et al (2015) Etiology of pericarditis in a prospective cohort of 1162 cases. Am J Med 128(7):784-e1
Weitzman LB, Tinker WP, Kronzon I, Cohen ML, Glassman E, Spencer FC (1984) The incidence and natural history of pericardial effusion after cardiac surgery—an echocardiographic study. Circulation 69(3):506–511
Adler Y, Charron P, Imazio M et al (2015) 2015 ESC guidelines for the diagnosis and management of pericardial diseases: the task force for the diagnosis and management of pericardial diseases of the European Society of Cardiology (ESC) endorsed by: the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 36(42):2921–2964
Horneffer PJ, Miller RH, Pearson TA, Rykiel MF, Reitz BA, Gardner TJ (1990) The effective treatment of postpericardiotomy syndrome after cardiac operations. A randomized placebo-controlled trial. J Thorac Cardiovasc Surg 100(2):292–296
Beland MJ, Paquet M, Gibbons JE, Tchervenkov CI, Dobell AR (1990) Pericardial effusion after cardiac surgery in children and effects of aspirin for prevention. Am J Cardiol 65(18):1238–1241
Gill PJ, Forbes K, Coe JY (2009) The effect of short-term prophylactic acetylsalicylic acid on the incidence of postpericardiotomy syndrome after surgical closure of atrial septal defects. Pediatr Cardiol 30(8):1061–1067
Cantinotti M, Spadoni I, Assanta N et al (2014) Controversies in the prophylaxis and treatment of postsurgical pericardial syndromes: a critical review with a special emphasis on paediatric age. J Cardiovasc Med 15(12):847–854
Imazio M, Brucato A, Markel G et al (2011) Meta-analysis of randomized trials focusing on prevention of the postpericardiotomy syndrome. Am J Cardiol 108(4):575–579
Imazio M, Brucato A, Ferrazzi P et al (2014) Colchicine for prevention of postpericardiotomy syndrome and postoperative atrial fibrillation: the COPPS-2 randomized clinical trial. JAMA 312(10):1016–1023
Dressler W (1956) A post-myocardial infarction syndrome; preliminary report of a complication resembling idiopathic, recurrent, benign pericarditis. J Am Med Assoc 160(16):1379–1383
Imazio M, Hoit BD (2013) Post-cardiac injury syndromes. An emerging cause of pericardial diseases. Int J Cardiol 168(2):648–652
Khan AH (1992) The postcardiac injury syndromes. Clin Cardiol 15(2):67–72
Jones DA, Radford DJ, Pohlner PG (2001) Outcome following surgical closure of secundum atrial septal defect. J Paediatr Child Health 37(3):274–277
Cabalka AK, Rosenblatt HM, Towbin JA et al (1995) Postpericardiotomy syndrome in pediatric heart transplant recipients. Immunologic characteristics. Tex Heart Inst J 22(2):170–176
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest relevant to this article to disclose.
Research Involving Human and Animal Participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
A waiver of informed consent was obtained from the Northwell Health institutional review board.
Rights and permissions
About this article
Cite this article
Rabinowitz, E.J., Meyer, D.B., Kholwadwala, P. et al. Does Prophylactic Ibuprofen After Surgical Atrial Septal Defect Repair Decrease the Rate of Post-Pericardiotomy Syndrome?. Pediatr Cardiol 39, 1535–1539 (2018). https://doi.org/10.1007/s00246-018-1926-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-018-1926-4