Abstract
Connectivity between shallow coral reefs and adjacent deeper habitats may be crucial to reef ecosystem stability. However, deeper habitats such as mesophotic coral ecosystems (MCEs) remain understudied. We investigated the depth structuring of shore-fish assemblages in the central Philippines across shallow (10–30 m) and mesophotic (upper: 30–60 m, lower: 60–90 m) depth zones. Baited video surveys in two coastal sites ~ 30 km apart showed strong declines with depth in fish species richness and abundance including fishery target species. Corallivores, herbivores/detritivores, omnivores and planktivores showed the strongest declines. Invertivores and generalist carnivores dominated abundance at mesophotic depths. Data from the coastal sites were analysed with published data from an offshore island (Apo Island) < 25 km away to provide broader insights on spatial variability of shore-fish depth structuring. The percentages of species that overlapped shallow and mesophotic depths were much lower in coastal sites (9–13%, 9–11%) than the island (20–26%), suggesting higher potential vertical connectivity in the latter. Mean assemblage similarities (Bray–Curtis) between shallow and mesophotic depth zones were found to be low at all sites (0.3–19.6) and decreased with depth. Fish assemblages gradually differed across depth zones at coastal sites but mesophotic assemblages at the island were more similar to the shallow coastal assemblages. Strong correlations between fish assemblages and benthic habitat were detected, suggesting that higher cover of rocky substratum at mesophotic depths facilitates vertical connectivity at the island. Our findings highlight benthic habitat as a driver of spatial variation in the depth structuring and vertical connectivity of shore-fish assemblages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Shore-fish species constitute a dominant component of tropical shelf biodiversity, the most prominent subgroup of which are fishes living in close association with shallow coral reefs (Mora et al. 2003; Tittensor et al. 2010). While coral reef fishes have become a major focus of marine ecological studies (Mora 2015), shore fishes inhabiting the deeper parts of tropical shelves have received far less attention due to the logistical difficulties imposed by sampling at greater depths (> 30 m). Knowledge of the ecology of deeper shore fishes in the tropics come mainly from fishery-dependent studies or experimental fishing surveys conducted away from coral reefs (Longhurst and Pauly 1987). However, there is a growing interest in, and need to understand, deeper shelf areas closer to shallow coral reefs, including the structure of their fish assemblages (Loya et al. 2016; Rocha et al. 2018; Sih et al. 2019). This interest is largely being driven by questions about ecological connectivity across wide depth gradients, which could play a role in reef ecosystem stability amidst severe human- and climate-induced threats to shallow reefs (Baker et al. 2016; Loya et al. 2016).
The structuring of fish and benthic assemblages with depth may provide indications of ecological connectivity between shallow reefs and deeper habitats. In the past two decades, a good number of studies used video- or diver-based methods to describe fish assemblage structure from shallow reefs (< 30 m) to the lower-light environments of the mesophotic zone (30 to 150 m deep) (Turner et al. 2017; Whitmarsh et al. 2017). Much of this research focused on mesophotic coral ecosystems (MCEs)—the deeper, hard-bottom extensions of shallow reefs where some corals, algae and sponges can still thrive, forming habitats for fish assemblages (Baker et al. 2016). Total fish species richness, abundance or biomass are commonly reported to decline with increasing depth from shallow reefs to MCEs or soft-bottom mesophotic habitats (Brokovich et al. 2008; Andradi-Brown et al. 2016; Asher et al. 2017a; Abesamis et al. 2018). Contrasting trends with depth among the trophic components of fish assemblages have been documented but typically, herbivores show declines in species richness, abundance or biomass while carnivores (e.g. planktivores, piscivores, invertivores) may increase (Thresher and Colin 1986; Brokovich et al. 2010; Fukunaga et al. 2016; Lindfield et al. 2016). Fishery target species may exhibit greater abundance at mesophotic depths, suggesting that deeper habitats serve as a refuge from fishing for some species (Lindfield et al. 2016; Asher et al. 2017b). Shifts in fish assemblage composition occur with these observed trends but the depth distributions of some species can overlap shallow and mesophotic depths, suggesting vertical connectivity via dispersal of larvae, juveniles or adults (Tenggardjaja et al. 2014; Lindfield et al. 2016; Pyle et al. 2016). Multiple inter-related drivers may influence the observed trends including benthic habitat quality, food availability, relative predation risk, light, depth and temperature (Brokovich et al. 2010; Fitzpatrick et al. 2012; Bridge et al. 2016; Lindfield et al. 2016). However, studies of shore-fish assemblages that go beyond shallow reefs are still lacking in the Indo-Pacific, particularly in the Coral Triangle, despite increased research on mesophotic communities in the past decade (Turner et al. 2017; Lesser et al. 2019).
Global shore-fish diversity reaches its peak in the Philippines and the shallow coral reefs in this region are some of the most threatened by human activities and climate change within the Coral Triangle (Carpenter and Springer 2005; Nañola et al. 2011; Burke et al. 2012). Fish and benthic surveys have been conducted extensively on Philippine reefs with the majority of these limited to the upper 10 m (Nañola et al. 2011; Licuanan et al. 2019). In contrast, fish assemblages and their habitats in deeper areas adjacent to shallow reefs remain understudied, notwithstanding the fact that the first scientific collections of deeper shore fishes were conducted in the region more than a century ago (Smith and Williams 1999). Furthermore, despite high fishing pressure on reefs in this region (Lavides et al. 2010; Nañola et al. 2011), the importance to fisheries of deeper areas near shallow reefs remains poorly understood and thus largely unconsidered in management and conservation programs such as the establishment of marine reserves (Pinheiro et al. 2019). Several studies that used data from demersal trawl surveys detected significant changes in fish assemblage structure from shallow (10 m) to deeper (50–200 m) shelf areas in the Philippines (Federizon 1992; McManus 1997; Garces et al. 2006). However, these works offer few insights on potential vertical connectivity involving shallow reefs because of the sampling design and limitations of trawl surveys. A small number of recent studies described changes in fish assemblage structure from shallow reefs to the mesophotic zone in the Philippines using video- or diver-based methods (Abesamis et al. 2018; Rocha et al. 2018; Quimpo et al. 2019; Pinheiro et al. 2019). However, it is still unclear from these very few studies how the depth structuring of shore-fish assemblages vary spatially in this highly complex archipelago.
Here, we describe variations in shore-fish assemblage structure from shallow reefs to the mesophotic zone within a relatively small region of the central Philippines. We aimed to elucidate how depth structuring and potential vertical connectivity in fish assemblages vary spatially across this region and explored how the variations are associated with changes in benthic habitat. We examined changes in fish assemblages and the benthos across several depth zones that are commonly referred to in studies that describe ecological patterns across shallow and mesophotic depth gradients: shallow (< 30 m), upper mesophotic (30–60 m) and lower mesophotic (> 60 m) (Bongaerts et al. 2010; Loya et al. 2016; Turner et al. 2017; Lesser et al. 2019). These depth zones were sampled at two coastal sites situated on the shelf of a major island using baited remote underwater video (BRUV) systems deployed on the seafloor from 10 to 90 m deep. Trends with depth in the species richness and relative abundance of fish assemblages, their component trophic groups, species targeted by fisheries and benthic habitat were compared between the two sites. Furthermore, we investigated potential vertical connectivity at each site, fish assemblage similarity across sites and depth zones, and probable relationships between the structuring of fish assemblages and benthic habitat variables. To provide broader insights on the spatial variation of depth structuring and potential vertical connectivity in shore-fish assemblages in the study region, the data analysis included published data from Apo Island (Abesamis et al. 2018), a small offshore island located < 25 km away from the two coastal sites.
Methods
Study region
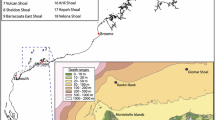
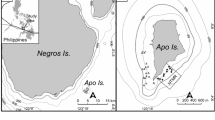
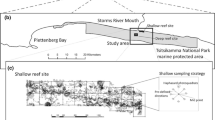
Surveys were conducted at two sites about 30 km apart on the insular shelf of south-eastern Negros, a large island in the Philippines (13,309 km2) mainly of volcanic origin (Fig. 1). The first site was situated off the coast of Dumaguete City (hereinafter Dumaguete; 9.323030°N, 123.315117°E), while the second was located off the coast of Zamboanguita municipality, near the village of Lutoban (hereinafter Lutoban; 9.068891°N, 123.158638°E). The sites were about 2 km long and 0.6 km wide. The insular shelf of south-eastern Negros is much narrower compared to continental areas in Southeast Asia (Longhurst and Pauly 1987; Garces et al. 2006), reaching depths of 100–250 m within 5–10 km from shore (Fig. 1). Coral reef development in the shallows is patchier in Dumaguete and more contiguous in Lutoban. Nearshore fishing off south-eastern Negros is done mainly by small-scale fishers using a wide variety of hook-and-lines, traps, nets and hand instruments, targeting > 150 fish species (Green et al. 2004; Abesamis and Utzurrum unpublished data). The combined impacts of fishing, pollution and siltation on marine habitats appear to be more severe in Dumaguete than in Lutoban due to much higher human population density in the former. Dumaguete has two no-take marine reserves while Lutoban has one (Fig. 1). The seaward boundaries of these reserves are confined to relatively shallow water (< 10 to 35 m deep). Dumaguete and Lutoban are about 25 and 12 km away from Apo Island, respectively (Fig. 1). Apo Island (0.7 km2) is also volcanic in origin. It is the only small island that occurs offshore from south-eastern Negros, separated by a channel > 250 m deep. The fisheries and depth structuring of fish assemblages at Apo Island have been described elsewhere (Abesamis et al. 2006, 2018). Two typhoons impacted Apo Island in 2011 and 2012, which severely affected benthic and fish assemblages at shallow and mesophotic depths within the marine reserve situated on the eastern side of the island but not the western side (Abesamis et al. 2018). These typhoons did not cause severe damage to shallow reefs in Dumaguete and Lutoban.
The study area in the central Philippines (left panel, depth contours in m) showing the locations of the two coastal study sites (right panels: a Dumaguete and b Lutoban) and Apo Island. BRUV sampling stations at each of the coastal sites are marked by letters that indicate three depth zones (S: shallow, 10–30 m; U: upper mesophotic, 30–60 m; L: lower mesophotic, 60–90 m). Broken yellow lines show the boundaries of no-take marine reserves. Map data: Google, Maxar Technologies, Becker et al. (2009)
Baited remote underwater video (BRUV)
Fishes and benthic habitat were surveyed in Dumaguete and Lutoban using single camera BRUVs. Each BRUV consists of one high-definition compact video camera (SJCAM SJ6 Legend) mounted facing forward on a horizontal base bar and protected by a steel frame (Supplementary Fig. 1). Cameras were set to 1080p and 60 fps. Bait consisted of about 0.7 kg of sardines (frozen fresh Sardinella spp. thawed prior to use) contained in a wire mesh bag suspended 1.5 m in front of each camera. BRUV is a widely used and cost-efficient method for surveying fish assemblages at depths beyond the reach of conventional diving (Whitmarsh et al. 2017; Langlois et al. 2020). The method may be biased towards sampling fish species that are attracted to the bait but it does not strongly select against non-carnivorous species (Harvey et al. 2007). Benthic habitat composition can be estimated at each BRUV sampling station, thus enabling the investigation of fish-habitat associations (Langlois et al. 2020; see below). However, the use of BRUVs to survey the benthos is subject to constraints such as the limited field of view and forward-facing orientation of the stationary cameras.
BRUV sampling stations were chosen haphazardly at each site with the aim of sampling three depth zones as evenly as possible (shallow: 10–30 m; upper mesophotic: 30–60 m; lower mesophotic: 60–90 m). Sampling was conducted between 08.00 and 17.00 h. Consecutive BRUV deployments were at least 100 m apart and fieldwork was conducted over several days at each site (Dumaguete: 16–18 September 2017 and 23–26 April 2018, Lutoban: 2–7 May 2018) to avoid bait plume interaction and reduce the likelihood of fish moving between stations (Langlois et al. 2020). The depth and geographic coordinates of each station were recorded using a GPS echosounder. BRUVs were deployed for up to 40–60 min per station but to standardise samples and further minimise the likelihood of fish moving between stations, only the first 30 min of the recorded video after the system had settled on the seafloor was analysed. A total of 54 and 57 replicate stations were successfully sampled in Dumaguete and Lutoban, respectively, with a slight under-representation of the upper mesophotic zone in Dumaguete (Fig. 1; Supplementary Table 1).
Video analysis
All visible fishes were identified to the species level whenever possible and counted using EventMeasure v.4.11 (www.seagis.com.au). MaxN, the maximum number of individuals per species observed at any given frame throughout the video sample, was used as a conservative measure of relative abundance (Cappo et al. 2004). Fish species were categorized into trophic groups (corallivore, herbivore/detritivore, omnivore, planktivore, invertivore, generalist carnivore and piscivore) based on available information on their diet (Froese and Pauly 2018) and into fishery targets or non-targets according to their importance to local commercial (for food) and subsistence fisheries (Abesamis et al. 2006; Muallil et al. 2015; Froese and Pauly 2018).
Benthic characteristics were measured by overlaying a 5 × 4 grid on a screen-captured image of each station and determining the most dominant benthic component within each grid cell that fully or partially overlays the benthos. Grid cells overlaying open water were excluded. Classification of the benthos followed the CATAMI scheme (Althaus et al. 2013) simplified into the following categories: hard (scleractinian) corals, soft corals, seagrass, macroalgae, sponges, other sessile invertebrates (ascidians, bryozoans, crinoids, etc.), rock, rubble and sand. The percent cover of each benthic component was then estimated per station by summing the number of grid cells dominated by each benthic category, dividing the sum by the total number of grid cells then multiplying by 100%.
Data from Apo Island
The data from Apo Island consisted of MaxN values of 253 fish species in 39 families/subfamilies (Supplementary Table 2). These were recorded by BRUV surveys in December 2014 at 26 stations situated in areas that were damaged and not damaged by typhoons (full methods in Abesamis et al. 2018). Sampling depths ranged from 10 to 80 m with the three depth zones sampled almost equally (Supplementary Table 1). Video analysis followed the same protocols as the present study, except that rubble and sand were combined into a single benthic category. In summary, the data showed that regardless of typhoon damage, fish species richness and MaxN declined with depth in most trophic groups (full results in Abesamis et al. 2018). Mean total species richness declined steadily from the shallow zone to the lower mesophotic zone while mean total MaxN remained high in the shallow and upper mesophotic zones before declining in the lower mesophotic zone (Supplementary Fig. 2). On the eastern side of the island damaged by typhoons, rock dominated benthic cover (58–62% mean cover) from the shallow to the lower mesophotic zone and hard corals were recorded only in the shallow and upper mesophotic zones at very low levels of cover (2–4%). On the opposite side not damaged by typhoons, hard corals were limited to and dominated benthic cover in the shallow zone (56% mean cover), rubble/sand dominated benthic cover in the lower mesophotic zone (59%) and rock cover was high to moderate from the upper mesophotic (64%) to the lower mesophotic (32%) zone (full results in Abesamis et al. 2018).
Data analysis
Mean species richness and MaxN of the overall fish assemblages, component trophic groups, fishery target and non-target species in Dumaguete and Lutoban were calculated per depth zone to evaluate general trends in taxonomic composition, abundance and importance to fisheries with depth. Variations in benthic habitat with depth in the two coastal sites were evaluated by calculating the mean percent cover of each benthic category per depth zone.
Variation in potential vertical connectivity across Dumaguete, Lutoban and Apo Island was investigated by calculating the proportion of the fish assemblage at each site that overlapped shallow and mesophotic depths. An approach similar to Rocha et al. (2018) was employed, which estimated relative abundance at shallow versus mesophotic depths to identify species that occurred exclusively in the shallow zone (100% of total MaxN at 10–30 m) and those that tend to be more abundant in the mesophotic zone. The percentage of species in the fish assemblage that were not classified into either of these two groups was considered indicative of vertical connectivity. A range for this percentage was calculated using a minimum of 60% and 75% of total MaxN at 30–90 m to identify species that tend to be more abundant in the mesophotic zone. Results were checked by repeating the calculations using data that excluded records with a total MaxN of 1 and those that could not be identified to the species level.
Fish assemblage similarity across all sites and depth zones was quantified by generating a Bray–Curtis similarity matrix of BRUV stations using species MaxN values that were 4th root-transformed to down-weight the influence of highly abundant species. Mean similarity within and between site-depth zone groupings was calculated from this resemblance matrix to evaluate general patterns of similarity. Non-metric multi-dimensional scaling (nMDS) plots were generated from the matrix to visualise patterns of similarity. Two-way crossed permutational analysis of variance (PERMANOVA) was used to test for statistically-significant differentiation in fish assemblages across sites (random factor) and depth zones (fixed factor). Canonical Analysis of Principal Coordinates (CAP) was used to explore potential associations between variations in fish assemblages and percent cover of benthic habitat variables, with the latter log (x + 1) transformed to improve symmetry before analysis. CAP is designed to find axes through the fish assemblage data that have the strongest correlations (δ) with the benthic data. It performs canonical analysis on principal coordinates (PCO) derived from the resemblance matrix but chooses only a subset of PCO axes (m) to avoid overparameterisation that may produce spurious correlations (Anderson and Willis 2003; Anderson et al. 2008). Analyses using nMDS, PERMANOVA and CAP were conducted separately for data that excluded or included Apo Island to examine variations in the coastal sites more closely. Similarity percentage (SIMPER) analysis was used to identify the top five fish species that contributed most significantly to the mean similarity within site-depth zone groupings. All multivariate data analyses were conducted in Primer-E v.7 with PERMANOVA+ (Anderson et al. 2008; Clarke et al. 2014).
Results
A total of 3,954 individual fish representing 326 taxa (‘species’) in 48 families/subfamilies were recorded in Dumaguete and Lutoban (Supplementary Table 2). About 6% of MaxN records were of fish that could be identified to the family or genus level only. The two coastal sites had similar species richness, with 219 and 217 species respectively. However, only about 50% of the fish species in one site also occurred in the other (110/219 species in Dumaguete and 110/217 species in Lutoban). Inclusion of Apo Island increased the total number of fish species of the entire data set to 448. Thirty-two percent (71/219) of the species in Dumaguete and 52% (113/217) of the species in Lutoban were also recorded at Apo Island.
Species richness and abundance of the overall fish assemblage declined strongly from the shallow zone to the mesophotic zone in Dumaguete and Lutoban (Fig. 2a, 3a). The declines were most pronounced in the corallivores (mostly Family Chaetodontidae), herbivores/detritivores (mostly Labridae/Scarinae and Acanthuridae), omnivores (mostly Pomacentridae) and planktivores (mostly Pomacentridae) (Fig. 2b–e, 3b–e). These trophic groups were generally more species-rich and abundant in the shallow zone in Lutoban. Species richness of invertivores (mostly Labridae and Mullidae) and generalist carnivores (mostly Nemipteridae, Lethrinidae and Carangidae) declined more gradually with depth at both sites (Fig. 2f, g). Abundance of invertivores also declined with depth at both sites but was generally higher in Dumaguete in all depth zones (Fig. 3f). Trends in the abundance of generalist carnivores were less consistent between sites with Dumaguete showing a peak in the upper mesophotic zone (Fig. 3g). Invertivores and generalist carnivores accounted for 77–90% and 90–93% of mean total MaxN in the upper and lower mesophotic zone, respectively, depending on the site (Supplementary Table 3). Piscivores (Carangidae and Serranidae/Epinephelinae) occurred in low numbers at both sites and exhibited no clear trends with depth (Figs. 2h, 3h).
Almost 60% (191/326) of the fish species recorded in Dumaguete and Lutoban are known to be important to local commercial and/or subsistence fisheries (Supplementary Table 2). Target species declined strongly in species richness and abundance with depth at both sites but were more species-rich and abundant than non-target fishes in all depth zones especially in the upper and lower mesophotic zones (Fig. 4). Target species comprised 64–84% of mean total species richness and 74–87% of mean total MaxN in the mesophotic zone depending on the site. Most of the target fishes in the mesophotic zone at both sites were invertivores and generalist carnivores (Supplementary Table 4).
Mean species richness and MaxN per BRUV station (upper and lower panels, respectively) of target and non-target fishes (shaded and open bars, respectively) across depth zones (shallow: 10–30 m; upper mesophotic: 30–60 m; lower mesophotic: 60–90 m) in Dumaguete and Lutoban (column a and b, respectively). Error bars are SE
Most biotic components of the benthos appeared to be limited to the shallow and upper mesophotic zones in Dumaguete and Lutoban (Supplementary Fig. 3). Hard corals were recorded only in the shallow zone in Dumaguete (maximum 17.4 m) with low mean cover (10%). All records of hard corals in Lutoban were within the shallow zone (26% mean cover) except for one record in the upper mesophotic zone at 58.8 m (<< 1% mean cover). Soft corals extended to the upper mesophotic zone in Lutoban and lower mesophotic zone in Dumaguete with highest mean cover in the upper mesophotic zone in the latter (22%). Seagrasses were recorded only in the shallow zone in Dumaguete (18% mean cover). Macroalgae were recorded in the shallow and upper mesophotic zones at both sites with low mean cover (5–9%). Mean cover of rock was very low at both sites (4–5%) and appeared to be limited to the shallow zone in Dumaguete but extended to the upper mesophotic zone in Lutoban. Rubble extended to the lower mesophotic zone in Dumaguete (5% mean cover) and the upper mesophotic zone in Lutoban (13% mean cover). Sand clearly dominated benthic cover in the upper and lower mesophotic zones at both sites (63–100% mean cover).
The percentages of the fish assemblages in Dumaguete and Lutoban that overlapped shallow and mesophotic zones (9–13% and 9–11%, respectively) were much lower than at Apo Island (20–26%), indicating higher potential vertical connectivity in the latter (Table 1; Supplementary Fig. 4). Recalculating these percentages without records that had a total MaxN of 1 and records that cannot be identified to the species level suggested a similar pattern (Dumaguete: 16–20%; Lutoban: 13–16%; Apo Island: 30–38%). Closer inspection showed that the familial composition of the overlapping species were quite different across sites, with families that are common on shallow reefs (e.g. Labridae, Acanthuridae, Chaetodontidae, Pomacentridae, Labridae/Scarinae) better represented at Apo Island (Fig. 5). Apart from the lower percentage of overlapping species at the coastal sites, there was a high percentage of species that occurred exclusively in the shallow zone, and a low percentage of species that tend to be more abundant in the mesophotic zone, at these sites. This pattern was reversed at Apo Island (Table 1). To further investigate how these opposing trends may be associated with higher potential vertical connectivity at Apo Island, we examined the depth distribution at Apo Island of the shallow species in Dumaguete and Lutoban, 54 and 83 of which, respectively, also occurred at the island (Supplementary Table 2). We found that only 30–38% of these species occurred exclusively in the shallow zone at Apo Island, depending on the site. The remaining species overlapped shallow and mesophotic zones (23–26%) or were more abundant in the mesophotic zone (39–42%), suggesting that 36 and 52 of the shallow species in Dumaguete and Lutoban, respectively, had wider or deeper depth distributions at Apo Island (calculations based on a threshold of at least 60% of total MaxN in the mesophotic zone).
Familial composition of the species that overlapped shallow and mesophotic zones in Dumaguete (a), Lutoban (b) and Apo Island (c). Values shown are the percent of the total overlapping species in each site accounted for by different families. The total number of overlapping species per site is shown in the lower right corner of each graph, which was calculated using a threshold of at least 60% of total MaxN in the mesophotic zone to differentiate species that tend to be more abundant at mesophotic depths (see “Methods” section)
Mean fish assemblage similarities between shallow and mesophotic depth zones at each site were low (range: 0.3–19.6) and decreased with depth, thus fish assemblages were least similar between shallow and lower mesophotic zones (Table 2). However, mean fish assemblage similarities within depth zones were also low (range: 10.1–34.7) but almost always higher than similarities between depth zones at each site (Table 2), indicating some degree of cohesiveness of the fish assemblages within depth zones. An nMDS plot that only included Dumaguete and Lutoban showed gradual differentiation of fish assemblages from the shallow zone to the lower mesophotic zone, with one outlier (a lower mesophotic station in Lutoban) (Fig. 6a). Differentiation between sites was more apparent in the shallow zone than in the mesophotic zone. There was no indication of an effect of sampling at different years (2017 versus 2018) or times of day (Supplementary Fig. 5). An nMDS plot that included all three sites showed that fish assemblages in the upper and lower mesophotic zones at Apo Island were more similar to most stations in the shallow zone than most stations in the mesophotic zone in the two coastal sites (Fig. 6b; also Table 2). PERMANOVA detected a significant interaction between the factors site and depth zone regardless of whether or not Apo Island was included in the analysis (Supplementary Table 5). This significant interaction was probably due to the changing magnitude and direction of the effect of depth zone on assemblage similarity across sites (Fig. 6a, b). Subsequent pair-wise comparisons indicated statistically significant differentiation in fish assemblages between all depth zones within each site (Table 3). SIMPER indicated contrasting sets of the top five fish species that contributed most significantly to assemblage similarity within depth zones at each site (Supplementary Table 6).
Two-dimensional nMDS plots of fish assemblage similarity for a Dumaguete (Dgt) and Lutoban (Lut) only and b Dumaguete, Lutoban and Apo Island (Apo) across three depth zones (shallow, 10–30 m; upper mesophotic, 30–60 m; lower mesophotic, 60–90 m). The outlier in plot a is not shown in plot b for clarity
CAP of the two coastal sites showed strong correlations between variations in fish assemblages and benthic habitat using the first two canonical axes (δ1 = 0.85; δ2 = 0.62) with an m of 10 (out of 110 PCO axes) that explained 60% of the variability in the resemblance matrix (Fig. 7a; Supplementary Table 7). Most shallow stations in Lutoban and some shallow stations in Dumaguete were associated with higher hard coral cover (and rock cover to a lesser extent) while most stations in the lower mesophotic zone at both sites were associated with higher sand cover (Fig. 7a). The remaining stations, which were mostly shallow stations in Dumaguete and upper mesophotic stations at the two sites, showed a gradient ranging from associations with a higher cover of biotic components other than hard coral (most strongly with macroalgal cover) to associations with higher sand cover. CAP of the three sites showed very strong correlations between fish assemblages and benthic habitat (δ1 = 0.90; δ2 = 0.86; m = 35 out of 136 PCO axes that explained 90% of the resemblance matrix) and patterns of fish-benthos associations in Dumaguete and Lutoban that were broadly similar to the previous CAP (Fig. 7b; Supplementary Table 7). The shallow stations in Apo Island clearly reflected the effects of the typhoons at that site, showing a gradient of fish assemblages that were associated with the higher hard coral cover on one side (not affected by typhoons) to those associated with higher rock cover on the other (damaged by typhoons). Most fish assemblages in the upper and lower mesophotic zones in Apo Island were associated with higher rock cover (and sponge cover to a lesser extent), contrasting with mesophotic fish assemblages at the coastal sites, which were associated with higher sand cover.
CAP exploring probable associations between variations in fish assemblage structure and benthic habitat in a Dumaguete (Dgt) and Lutoban (Lut) only and b Dumaguete, Lutoban and Apo Island (Apo) across three depth zones (shallow: 10–30 m; upper mesophotic: 30–60 m; lower mesophotic: 60–90 m). Benthic habitat categories: HC hard coral, SC soft coral, SG seagrass, MA macroalgae, SP sponge, OT other sessile invertebrates (ascidians, bryozoans, crinoids, etc.); RK rock, RU rubble, SA sand
Discussion
This study is the first to describe spatial variation in the structuring of shore-fish assemblages in the Philippines from shallow reefs to adjacent mesophotic habitats within a moderate geographic scale (tens of km). Fish assemblages in two coastal sites at Negros Island showed marked changes in species richness, abundance, trophic structure and importance to local fisheries with depth, which were fairly concordant between the sites. Fish assemblage structure (species composition weighted by abundance) at both sites gradually differed with depth, showing low levels of assemblage similarity within depth zones but even less similarity among depth zones. The depth structuring of these assemblages had strong associations with variations in benthic habitat, transitioning from assemblages in areas with a higher cover of biota (hard corals, seagrass, macroalgae, soft corals) in the shallow and upper mesophotic zones to assemblages in areas dominated by sand in the upper and lower mesophotic zones. Potential vertical connectivity was estimated to be lower in the coastal sites compared to Apo Island, with fish assemblages in the upper and lower mesophotic zones of the latter showing similarities with shallow coastal assemblages. Most mesophotic assemblages at Apo Island were associated with higher cover of rocky substratum, contrasting with mesophotic coastal assemblages that were associated with sand-dominated bottoms, and suggesting that benthic habitat quality at mesophotic depths influences the extent of vertical connectivity. These results highlight variation in benthic habitat as an important driver of spatial variation in the depth structuring and potential vertical connectivity of shore-fish assemblages from shallow to mesophotic depths.
The declines in overall fish species richness and abundance with depth in the coastal sites at Negros Island were not unexpected. Previous studies have documented general declines in the same parameters across shallow to mesophotic depth gradients in other tropical localities (Thresher and Colin 1986; Fitzpatrick et al. 2012; Asher et al. 2017a), including Apo Island (Abesamis et al. 2018). In fact, declines in species richness in all trophic groups except piscivores occurred at both Negros and Apo islands, with corallivores, herbivores/detritivores and omnivores consistently exhibiting the steepest declines with depth (Abesamis et al. 2018). However, in terms of abundance, only corallivores, herbivores/detritivores, omnivores and invertivores consistently showed declines with depth at both islands while trends in planktivores, generalist carnivores and piscivores were variable, suggesting that the latter groups are less strongly limited by changing conditions with increasing depth.
Spatial variation in how the species richness or abundance of fish assemblages and their trophic groups change with depth may be strongly influenced by variations in the availability of shelter and food resources in the benthic habitat (Brokovich et al. 2010; Fitzpatrick et al. 2012). For instance, Lindfield et al. (2016) showed that the species richness and abundance of herbivores and detritivores remained largely unchanged from shallow to mesophotic depths in the Marianas due to continued availability of structurally-complex reef habitat and algal resources down to 60 m or more, which strongly contrasts with the present study. In Hawaii, Asher et al. (2017a) showed contrasting trends between more structurally-complex hard-bottom sites and less complex soft-bottom sites in the abundance of different trophic groups from shallow to mesophotic depths. Additionally, and consistent with the present study, they found that mobile invertivores and generalist macropiscivores were characteristic of deeper (53–100 m) sand-dominated areas where the prey items of these groups are presumably abundant. The only other study of spatial variation in the depth structuring of shore-fish assemblages in the Philippines also demonstrated the probable influence of benthic habitat but across a larger geographic scale and narrower depth range than the present study. Quimpo et al. (2019) showed that differentiation of fish assemblages across three sites in northern Philippines (100–300 km apart) may be largely explained by variations in the relative cover of biotic (hard corals, soft corals, algal assemblages) and abiotic (sand, silt) components of the benthos, with the site that showed the strongest declines in species richness and abundance from shallow (10–20 m) to mesophotic depths (35–40 m) having a much higher cover of hard corals in the shallows than the other sites.
The spatial variation in potential vertical connectivity documented by this study may be explained by broad differences in the availability of hard and more structurally-complex substratum in the mesophotic zone. Benthic data in the two coastal sites at Negros Island suggested that hard corals were virtually limited to the shallow zone and rock cover was minimal (4–5% mean cover) in the shallow and upper mesophotic zones (Supplementary Fig. 3). In contrast, at Apo Island, rock cover was much higher (32–62%) in the upper and lower mesophotic zones with relatively high structural complexity (Abesamis et al. 2018). Furthermore, unlike the coastal sites, hard corals were recorded at Apo Island in the upper mesophotic zone (down to 40 m on the eastern side; Abesamis et al. 2018) although the cover was very low (2%) probably due to typhoon damage. These differences between the two islands may be indicative of contrasting conditions over thousands of years that promoted or hindered the development of MCEs, such as differences in shelf geomorphology, water turbidity and siltation levels (Montaggioni 2005; Baker et al. 2016). The higher availability of more complex hard substratum in the mesophotic zone at Apo Island may have made it possible for reef fishes to have wider or deeper local depth distributions, which is consistent with the observation that a significant number (36–52) of shallow species in the coastal sites occurred in the mesophotic zone at Apo Island. For the more mobile of these species (e.g., Labridae, Chaetodontidae, Acanthuridae, Labridae/Scarinae, Caesionidae), a wider depth distribution may be due in part to greater vertical connectivity between shallow and mesophotic depths via adult movements within individual home ranges, but this can only be verified through tagging studies (e.g., acoustic telemetry).
For the more site-attached species (e.g. Pomacentridae), a wider or deeper depth distribution at Apo Island could be due mainly to the greater availability of complex hard substratum facilitating recruitment and establishment of territories in the mesophotic zone. More generally speaking, however, this suggests that greater availability of suitable benthic habitat in the mesophotic zone can enhance the vertical component of larval connectivity for species that can recruit and breed at mesophotic depths, allowing mesophotic populations to seed nearby or distant shallow populations, or vice-versa (Tenggardjaja et al. 2014; Vaz et al. 2016). This mode of vertical connectivity has far more potential than adult vertical connectivity in subsidising fish populations on shallow reefs provided that mesophotic habitats are largely spared by disturbances that impact shallow reefs. However, recent studies have questioned the potential of mesophotic communities to significantly provide larval recruitment on shallow reefs and the immunity of mesophotic communities from man-made and natural disturbances (Bongaerts et al. 2017; Abesamis et al. 2018; Rocha et al. 2018). If connectivity between the mesophotic and shallow zones is relatively low, and if shallow coral reef habitats are degraded by disturbances, shallow reef fish assemblages are likely to shift in species composition, relative abundance and trophic structure driven by fish species that tend to become more abundant under degraded reef conditions (Lowe et al. 2019; Russ et al. 2020). Some of these fish species may still thrive deeper (e.g., Labridae, see Fig. 5) and drive much of the relatively low levels of recruitment that could originate from the mesophotic zone.
Several caveats apply to our interpretation of the influence of benthic habitat on the depth structuring of shore-fish assemblages in the study region. First, we acknowledge that Apo Island may not be an ideal site to compare with the coastal sites due to the damage it incurred from typhoons. The relatively high cover of structurally-complex, rocky substratum at mesophotic depths at that site probably resulted from typhoon damage (Abesamis et al. 2018) and could thus be atypical of mesophotic fore-reef slopes in the region. Another concern is that Apo Island was sampled several years earlier than the two coastal sites (2014 vs. 2017/2018), thus temporal variation may have been a confounding factor to some extent. Future work in this region should aim to expand sampling to other sites that may better represent geographic variation in benthic habitat quality from shallow to mesophotic depths, taking care to control for potential inter-annual or seasonal variation. Site selection should take into account island geomorphology as this is likely to have significant associations with benthic habitat and, therefore, the depth structuring of fish assemblages.
Second, BRUV may not be the best method to describe the benthic habitat because of the way the cameras are oriented relative to the seafloor. Estimates of relative benthic cover from BRUV would inevitably be subject to parallax error, raising doubts about their accuracy and precision. However, it seems unlikely that the relatively large number of BRUV stations per site would not be able to capture the strongest trends in the benthic habitat even with forward-facing cameras (Bennett et al. 2016). Parallax would also be less of a problem in areas with low relief (e.g. sand flats), which were prevalent in the sites. But perhaps a bigger concern with BRUV is that it can only provide a limited field of view of the benthos and may, therefore, miss some habitat features. For instance, local informants have reported the occurrence of reef-like structures (potential MCEs) between 45 and 80 m deep in Lutoban, some of which have been explored by technical divers, but the BRUV surveys at that site detected such structures (scored as ‘rock’) in only one station at 45 m deep. Sonar and remotely-operated vehicles may be far better than BRUVs in describing the benthic habitat but these tools were not available to us for this study.
Third, the high density of sampling stations at each site (Fig. 1) could have inflated the estimates of relative fish abundance at some stations and in turn affected the reported fish-benthic habitat associations because of potential fish movement between stations. This high density of stations resulted from the desire to sample the seafloor as much as possible given the limited field of view of BRUVs while trying to maintain sampling independence between stations. Although the effective area of bait attraction around each BRUV is very difficult to estimate (Harvey et al. 2007), the high density of sampling stations may not be too great a concern given that about 90% of all inter-station distances at each site were relatively large (> 200 m) and the duration of video analysed per station (30 min) was relatively short (see Whitmarsh et al. 2017 and Langlois et al. 2020 for comparison with other BRUV sampling protocols).
Few studies have reported trends in the availability of fishery target species from shallow reefs to adjacent mesophotic habitats in the Philippines. We found no evidence consistent with the mesophotic zone serving as a refuge for fishery target species probably because of the lack of extensive structurally-complex habitats at mesophotic depths in the two coastal sites (Lindfield et al. 2016). Instead, we documented declines in target fish species richness and abundance with depth, reinforcing the importance of shallow reefs as sources of food fish for local fisheries in the region. However, because the local fisheries exploit such a wide range of shore-fish species, target fishes still accounted for high proportions of fish species richness and abundance at mesophotic depths. This directs attention to the need to explicitly include mesophotic habitats near shallow reefs in fisheries management interventions, which is an issue that is currently under-emphasised in local management regimes. For instance, marine reserves are regarded as a cornerstone of fisheries management throughout the Philippines but reserve establishment by local communities has traditionally focused on protecting only the shallow areas where coral reefs occur (Weeks et al. 2010). This was the case in the two coastal sites (Fig. 1) and the same situation is prevalent in the region.
This study represents a small but novel contribution to the limited basic knowledge on the ecology of shore fishes beyond shallow coral reefs in the Philippines. The ecological patterns we reported here provide a reference point for further studies that aim to resolve the nature, strength and variability of probable ecological linkages between shallow reefs and mesophotic habitats. We appeal for more research on shore-fish assemblages and their depth structuring in this region, their vulnerability to the suite of threats that imperil shallow habitats, and their variability in relation to the complex island geomorphology that underlies the rich biological heritage of the Philippine archipelago.
Data availability
The datasets collected and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abesamis RA, Alcala AC, Russ GR (2006) How much does the fishery at Apo Island benefit from spillover of adult fish from the adjacent marine reserve? Fish Bull 104:360–375
Abesamis RA, Langlois T, Birt M, Thillainath E, Bucol AA, Arceo HO, Russ GR (2018) Benthic habitat and fish assemblage structure from shallow to mesophotic depths in a storm-impacted marine protected area. Coral Reefs 37:81–97
Althaus F, Hill N, Ferrari R, Edwards L, Przeslawski R, Schönberg CHL, Stuart-Smith R, Barrett N, Edgar G, Colquhoun J, Tran M, Jordan A, Gowlett-Holmes K (2013) A standardised vocabulary for identifying benthic biota and substrata from underwater imagery: the CATAMI classification scheme. PLoS ONE 10:e0141039
Anderson MJ, Willis TJ (2003) Canonical analysis of principal coordinates: a useful method of constrained ordination for ecology. Ecology 84:511–525
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER. Guide to Software and Statistical Methods. PRIMER-E: Plymouth
Andradi-Brown DA, Macaya-Solis C, Exton DA, Gress E, Wright G, Rogers AD (2016) Assessing Caribbean shallow and mesophotic reef fish communities using baited-remote underwater video (BRUV) and diver-operated video (DOV) survey techniques. PLoS ONE 11:e0168235
Asher J, Williams ID, Harvey ES (2017a) Mesophotic depth gradients impact reef fish assemblage composition and functional group partitioning in the Main Hawaiian Islands. Front Mar Sci. https://doi.org/10.3389/fmars.2017.00098
Asher J, Williams ID, Harvey ES (2017b) An assessment of mobile predator populations along shallow and mesophotic depth gradients in the Hawaiian Archipelago. Sci Rep 7:1–18
Baker EK, Puglise KA, Harris PT (2016) Mesophotic coral ecosystems: A lifeboat for coral reefs? UNEP and GRID-Arendal, Arendal
Becker JJ, Sandwell DT, Smith WHF, Braud J, Binder B, Depner J, Fabre D, Factor J, Ingalls S, Kim S-H, Ladner R, Marks K, Nelson S, Pharaoh A, Trimmer R, Von Rosenberg J, Wallace G, Weatherall P (2009) Global Bathymetry and Elevation Data at 30 Arc Seconds Resolution: SRTM30_PLUS. Mar Geodesy 32:355–371
Bennett K, Wilson SK, Shedrawi G, McLean DL, Langlois TJ (2016) Can diver operated stereo-video surveys for fish be used to collect meaningful data on benthic coral reef communities? Limnol. Oceanogr. Meth. 14:874–885
Bongaerts P, Ridgway T, Sampayo EM, Hoegh-Guldberg O (2010) Assessing the ‘deep reef refugia’ hypothesis: focus on Caribbean reefs. Coral Reefs 29:309–327
Bongaerts P, Riginos C, Brunner R, Englebert N, Smith SR, Hoegh-Guldberg O (2017) Deep reefs are not universal refuges: reseeding potential varies among coral species. Sci Adv 3:e1602373
Bridge TC, Luiz OJ, Coleman RR, Kane CN, Kosaki RK (2016) Ecological and morphological traits predict depth-generalist fishes on coral reefs. Proc R Soc B 283:20152332
Brokovich E, Einbinder S, Shashar N, Kiflawi M, Kark S (2008) Descending to the twilight zone: changes in coral reef fish assemblages along a depth gradient down to 65 m. Mar Ecol Prog Ser 371:253–262
Brokovich E, Ayalon I, Einbinder S, Segev N, Shaked Y, Genin A, Kark S, Kiflawi M (2010) Grazing pressure on coral reefs decreases across a wide depth gradient in the Gulf of Aqaba, Red Sea. Mar Ecol Prog Ser 399:69–80
Burke L, Reytar K, Spalding M, Perry A (2012) Reefs at risk revisited in the Coral Triangle. World Resources Institute
Cappo M, Speare P, De’ath G, (2004) Comparison of baited remote underwater video stations (BRUVS) and prawn (shrimp) trawls for assessments of fish biodiversity in inter-reefal areas of the Great Barrier Reef Marine Park. J Exp Mar Biol Ecol 302:123–152
Carpenter KE, Springer VG (2005) The center of the center of marine shorefish distribution: the Philippine Islands. Environ Biol Fishes 72:467–480
Clarke KR, Gorley RN, Somerfield PJ, Warwick RM (2014) Change in marine communities: an approach to statistical analysis and interpretation. Primer-E Ltd.
Federizon RR (1992) Description of the subareas of Ragay Gulf, Philippines, and their fish assemblages by exploratory data analysis. Mar Freshw Res 43:379–391
Fitzpatrick BM, Harvey ES, Heyward AJ, Twiggs EJ, Colquhoun J (2012) Habitat specialization in tropical continental shelf demersal fish assemblages. PLoS ONE 7(6):e39634
Froese R, Pauly D (2018) Fishbase. www.fishbase.org
Fukunaga A, Kosaki RK, Wagner D, Kane C (2016) Structure of mesophotic reef fish assemblages in the Northwestern Hawaiian Islands. PLoS ONE 11:e0157861
Garces LR, Stobutzki I, Alias M, Campos W, Koongchai N, Lachica-Alino L, Mustafa G, Nurhakim S, Srinath M, Silvestre G (2006) Spatial structure of demersal fish assemblages in South and Southeast Asia and implications for fisheries management. Fish Res 78:143–157
Green SJ, Flores JO, Dizon-Corrales JQ, Martinez RT, Nuñal DRM, Armada NB, White AT (2004) The fisheries of Central Visayas, Philippines: status and trends. Coastal Resource Management Project of the Department of Environment and Natural Resources and the Bureau of Fisheries and Aquatic Resources of the Department of Agriculture. Cebu City, Philippines
Harvey ES, Cappo M, Butler JJ, Hall N, Kendrick GA (2007) Bait attraction affects the performance of remote underwater video stations in assessment of demersal fish community structure. Mar Ecol Prog Ser 350:245–254
Lavides MN, Polunin NVC, Stead SM, Tabaranza DG, Comeros MT, Dongallo JR (2010) Finfish disappearances around Bohol, Philippines inferred from traditional ecological knowledge. Environ Conserv 36:235–244
Langlois T, Goetze J, Bond T, Monk J, Abesamis RA et al (2020) A field and video annotation guide for baited remote underwater stereo-video surveys of demersal fish assemblages. Methods Ecol Evol. https://doi.org/10.1111/2041-210X.13470
Lesser MP, Slattery M, Laverick JH, Macartney KJ, Bridge TC (2019) Global community breaks at 60 m on mesophotic coral reefs. Glob Ecol Biogeogr 28:1403–1416
Licuanan WY, Robles R, Reyes M (2019) Status and recent trends in coral reefs of the Philippines. Mar Pollut Bull 142:544–550
Lindfield SJ, Harvey ES, Halford AR, McIlwain JL (2016) Mesophotic depths as refuge areas for fishery-targeted species on coral reefs. Coral Reefs 35:125–137
Longhurst AR, Pauly D (1987) Ecology of tropical oceans. Academic Press, San Diego
Lowe JR, Williamson DH, Ceccarelli DM, Evans RD, Russ GR (2019) Responses of coral reef wrasse assemblages to disturbances and marine reserve protection on the Great Barrier Reef. Mar Biol 166:119
Loya Y, Eyal G, Treibitz T, Lesser MP, Appeldoorn R (2016) Theme section on mesophotic coral ecosystems: advances in knowledge and future perspectives. Coral Reefs 35:1–9
McManus JW (1997) Ecological community structure analysis: applications in fisheries management. In: Silvestre G, Pauly D (eds) Status and management of tropical coastal fisheries in Asia. ICLARM Conference Proceedings 53, p 133–142
Montaggioni LF (2005) History of Indo-Pacific coral reef systems since the last glaciations: development patterns and controlling factors. Earth Sci Rev 71:1–75
Mora C (2015) Ecology of fishes on coral reefs. Cambridge University Press, Cambridge
Mora C, Chittaro PM, Sale PF, Kritzer JP, Ludsin SA (2003) Patterns and processes in reef fish diversity. Nature 421:933–936
Muallil RN, Deocadez MR, Martinez RJS, Mamauag SS, Nañola CL Jr, Aliño PM (2015) Community assemblages of commercially important coral reef fishes inside and outside marine protected areas in the Philippines. Reg Stud Mar Sci 1:47–54
Nañola CL Jr, Aliño PM, Carpenter KC (2011) Exploitation-related reef fish species richness depletion in the epicenter of marine biodiversity. Environ Biol Fishes 90:405–420
Pinheiro HT, Shepherd B, Castillo C, Abesamis RA, Copus JM, Pyle RL, Greene BD, Coleman RR, Whitton RK, Thillainath E, Bucol AA, Birt M, Catania D, Bell MV, Rocha LA (2019) Deep reef fishes in the world’s epicentre of marine biodiversity. Coral Reefs 38:985–995. https://doi.org/10.1007/s00338-019-01825-5
Pyle RL, Boland R, Bolick H, Bowen BW, Bradley CJ, Kane C, Kosaki RK, Langston R, Longenecker K, Montgomery A, Parrish FA, Popp BN, Rooney J, Smith CM, Wagner D, Spalding HL (2016) A comprehensive investigation of mesophotic coral ecosystems in the Hawaiian Archipelago. PeerJ 4:e2475
Quimpo TJR, Cabaitan PC, Olavides RDD, Dumalagan EE Jr, Munar J, Siringan FP (2019) Spatial variability in reef-fish assemblages in shallow and upper mesophotic coral ecosystems in the Philippines. J Fish Biol 94:17–28
Rocha LA, Pinheiro HT, Shepherd B, Papastamatiou YP, Luiz OJ, Pyle RL, Bongaerts P (2018) Mesophotic coral ecosystems are threatened and ecologically distinct from shallow water reefs. Science 361:281–284
Russ GR, Rizzari JR, Abesamis RA, Alcala AC (2020) Coral cover a stronger driver of reef fish trophic biomass than fishing. Ecol Appl. https://doi.org/10.1002/eap.2224
Sih TL, Daniell JJ, Bridge TC, Beaman RJ, Cappo M, Kingsford MJ (2019) Deep-reef fish communities of the Great Barrier Reef shelf-break: trophic structure and habitat associations. Diversity 11:26
Smith DG, Williams JT (1999) The great Albatross Philippine expedition and its fishes. Mar Fish Rev 61:31–41
Tenggardjaja KA, Bowen BW, Bernardi G (2014) Vertical and horizontal genetic connectivity in Chromis verater, an endemic damselfish found on shallow and mesophotic reefs in the Hawaiian Archipelago and Adjacent Johnston Atoll. PLoS ONE 9:e115493
Thresher RE, Colin PL (1986) Trophic structure, diversity and abundance of fishes of the deep reef (30–300 m) at Enewetak, Marshall Islands. Bull Mar Sci 38:253–272
Turner JA, Babcock RC, Hovey R, Kendrick GA (2017) Deep thinking: a systematic review of mesophotic coral ecosystems. ICES J Mar Sci 74:2309–2320
Tittensor DP, Mora C, Jetz W, Lotze HK, Ricard D, Berghe EV, Worm B (2010) Global patterns and predictors of marine biodiversity across taxa. Nature 466:1098–1103
Vaz AC, Paris CB, Olascoaga MJ, Kourafalou VH, Kang H, Reed JK (2016) The perfect storm: match-mismatch of bio-physical events drives larval reef fish connectivity between Pulley Ridge mesophotic reef and the Florida Keys. Cont Shelf Res 125:136–146
Whitmarsh SK, Fairweather PG, Huveneers C (2017) What is Big BRUVver up to? Methods and uses of baited underwater video. Rev Fish Biol Fisheries 27:53–73
Weeks R, Russ GR, Alcala AC, White AT (2010) Effectiveness of marine protected areas in the Philippines for biodiversity conservation. Conserv Biol 24:531–540
Acknowledgements
This work was supported by the Philippine Commission on Higher Education, the British Council and the Australian Research Council. We thank AC Alcala and HP Calumpong for facilitating support at Silliman University. T Langlois and M Birt introduced us to BRUV methodology. D Inocencio, G Duran, F Quinilitan, A Quinilitan, AJ Paer, CK Paer, D Calumpong, J Maypa, A Yucor, S Knudsen, Marine Conservation Philippines and Large Marine Vertebrates Research Institute provided support during fieldwork. K Carpenter, B Russell and H Pinheiro were consulted for fish identification. We are grateful to four anonymous reviewers for their insightful comments which greatly improved the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
Research was approved by the Silliman University Research Ethics Committee. The local governments of Dumaguete City and the Municipality of Zamboanguita granted permission to conduct this study within their jurisdictions.
Additional information
Responsible Editor: D. Goulet.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by J. A. Anticamara and undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Abesamis, R.A., Utzurrum, J.A.T., Raterta, L.J.J. et al. Shore-fish assemblage structure in the central Philippines from shallow coral reefs to the mesophotic zone. Mar Biol 167, 185 (2020). https://doi.org/10.1007/s00227-020-03797-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-020-03797-5