Abstract
Peak bone mineral density (BMD) is one of the most important factors influencing the development of osteoporosis. It was predicted that a 10% increase in peak BMD will delay the onset of osteoporosis by 13 years. However, changes in peak BMD over time are unknown. This study aimed to investigate secular trends in peak BMD among young adults in the United States. Based on the National Health and Nutrition Examination Survey from 1999–2018, 3,975 males aged 19–28 years and 2370 females aged 31–40 years were our target population for estimating peak lumbar spine BMD. BMD was measured by dual-energy X-ray absorptiometry. Generalized linear models adjusted for multiple covariates were used to examine the secular trends in peak BMD in males and females, respectively. Secular trends for peak lumbar spine BMD from 1999–2000 to 2017–2018 were not statistically significant in males or females (all Plinear and Pquadratic > 0.05). Similar results were observed in race/ethnicity subgroups (all Plinear and Pquadratic > 0.05). However, in stratified analyses by obesity category, peak lumbar spine BMD in obese males and females increased from 1999–2000 to 2009–2010 and then decreased until 2017–2018, while peak lumbar spine BMD in non-obese females decreased from 1999–2000 to 2005–2006 and then increased until 2017–2018 (all Pquadratic < 0.05). Peak lumbar spine BMD was greater in obese males and females than in non-obese males and females up to 2009–2010, but not from 2011–2012 onwards. Overall, there were no significant secular trends in peak lumbar spine BMD. However, secular trends differed between obese and non-obese groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peak bone mineral density (BMD) is one of the most important factors influencing the development of osteoporosis and osteoporotic fractures [1,2,3,4]. A peak BMD increase of 10% is predicted to delay the onset of osteoporosis by 13 years, while variation in the age of menopause and age-related bone loss have much smaller effects [4]. If peak bone mass were to be maintained up to the average age of menopause, the prevalence of low femoral neck bone mass in females aged 50 years would be reduced from 41 to 15% [5].
Data from the National Health and Nutrition Examination Surveys (NHANES) showed that femoral neck BMD in people aged 30 years and older in 2013–2014 was lower than in 2005–2012 [6]. Further studies demonstrated that prediabetic adults aged 40 years and older had a declining trend in lumbar spine and femoral neck BMD from 2005 to 2014 [7]. Whether there is a similar decreasing trend in peak BMD among younger adults over time is unknown.
The prevalence of obesity in the United States has increased over time [8]. Obesity is closely related to bone health, mainly from the mechanical effects of skeletal loading and the regulatory role of hormones and cytokines. This includes the effects of estrogen, adiponectin, and proinflammatory cytokines, such as interleukin-6, interleukin-1, and tumor necrosis factor α, on bone metabolism [9, 10]. Therefore, we hypothesized that the secular trend of peak BMD may be influenced by obesity.
The primary objective of this research was to investigate secular trends in peak BMD using data from the NHANES from 1999–2000 to 2017–2018. Such information is critical for understanding bone health in young adults in the United States, given the vital contribution of peak BMD to the development of early osteoporosis.
Materials and Methods
Study Setting and Participants
The NHANES is a repeated cross-sectional study designed to assess the health and nutritional status of the non-institutionalized civilian resident population in the United States. Data were obtained using a complex, stratified, multistage probability cluster sampling design, including demographics data, dietary data, examination data, laboratory data, questionnaire data and limited access data. From 1999 onwards, the NHANES became a continuous survey; the survey data are released every two years. The NHANES was approved by the National Center for Health Statistics Research Ethics Review Board. Written informed consent was obtained from all participants.
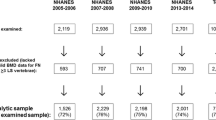
All participants with complete and valid BMD data between 1999–2000 and 2017–2018 were included in our study. We defined the ages for attainment of peak lumbar spine, femoral neck, and total hip BMD from previous studies as 24 years, 21 years, and 21 years in males, and 36 years,19 years, and 19 years in females, respectively [11, 12]. Consistent with other studies, the population between 5 years before and 5 years after the peak BMD age at each skeletal site were used to test for secular trends in peak BMD [12, 13]. After combining the above study populations, the analytic population was limited to participants aged 14–40 years (Fig. 1). Participants with a history of medication use (i.e., steroids, thyroid hormone, anti-osteoporosis medications, and anticonvulsants) or diseases (i.e., diabetes, thyroid disease, rheumatoid arthritis or osteoarthritis, cancer, chronic obstructive pulmonary disease, and congestive heart failure) affecting bone metabolism, and those with missing data on the key covariates (i.e., age, gender, BMI, race/ethnicity, and moderate or vigorous physical activity) were excluded.
Research Measurements
The data were collected through household interviews, physical examinations and laboratory tests in mobile examination centers. Age, gender, race/ethnicity, and physical activities were collected face-to-face by training interviewers using a structured questionnaire. Gender was categorized into male and female. Race/ethnicity was self-reported and categorized into Mexican American, non-Hispanic white, non-Hispanic black, and other race people (including other Hispanic and other race people). Moderate or vigorous physical activity was defined as individuals participating in moderate or vigorous leisure-time activities. Body mass index (BMI) was calculated as measured body weight (kg) divided by measured body height squared (m2) and divided into non-obese group (< 30 kg/m2) and obese group (≥ 30 kg/m2).
Serum 25(OH)D (nmol/L) for 2001–2002 to 2015–2016 was measured with radioimmunoassay (DiaSorin RIA kit, Stillwater MN)) and liquid chromatography-tandem mass spectrometry (HPLC–MS/MS). To facilitate comparisons of 25(OH)D across survey years, the 25(OH)D data for 2001–2002 to 2005–2006 were converted to HPLC–MS/MS equivalents by using regression equations [14]. Serum total calcium (mg/dl) from 2001–2002 to 2017–2018 was measured by the reaction of calcium with 5-nitro-5’methyl-BAPTA (NM-BAPTA) and EDTA or o-cresol phthalein complexes to form colored complexes or by using I.S.E. (ion selective electrode) methodology.
Details regarding the dual-energy X-ray absorptiometry (DXA) examination protocol have been published elsewhere [15, 16]. Scanning was done in fast array mode. Lumbar spine BMD was measured for survey years 1999–2018. Hip BMD was measured for survey years 2005–2010. Prior to 2010, BMD was measured with Hologic QDR-4500A fan-beam densitometers. From 2011 onwards, BMD was measured with Hologic Discovery model A densitometers. The NHANES performed cross-calibration to standardize BMD across the different Hologic DXA models, providing generally equivalent BMD values in various Hologic DXA models [17, 18]. Lumbar spine DXA scans were analyzed with Hologic Discovery software version 12.1 from 1999 to 2004, Hologic APEX software version 3.0 from 2005 to 2010, and Hologic APEX software version 4.0 from 2011 to 2018. Hip BMD scans were analyzed with Hologic Discovery software version 12.4. A previous study confirmed that there was no difference in mean BMD when analyzed using Discovery version 12.4 or APEX software version 4.0 [19]. The coefficient of variation for DXA measurements in BMD was < 0.60% [15, 16]. Anthropomorphic phantoms, quality control (QC) charts, and respondent scans were each reviewed at the mobile examination centers throughout this study to ensure the quality of the data [17]. Valid BMD data from the DXA scanning process was defined as no obstructing movable or immovable objects (including jewelry, prostheses, implants), no body parts beyond the scanning range, no excessive noise caused by obesity, and normal posture. There were 19% to 22% missing or invalid lumbar spine BMD data for the NHANES 1999–2000 to 2003–2004. According to the NHANES protocol, sequential regression multivariate imputation (SRMI) was used to impute missing BMD data to ensure a representative study sample [20].
Statistical Analysis
All analyses incorporated the survey sample weights, stratification, and clustering of the complex sampling design to ensure nationally representative estimates. All data were analyzed using the SPSS (Version 24.0, IBM SPSS, IBM Corp, Armonk, NY, USA) and the R (version: 4.1.2; R Foundation for Statistical Computing, Vienna, Austria).
We calculated means and standard errors (SEs) of the continuous covariates and percentages (%) of the categorical covariates at different skeletal sites for males and females, respectively. We conducted linear and quadratic trend tests for continuous covariates in generalized linear models with an identity link function and categorical covariates in generalized linear models with quasibinomial regression and a logit link function by treating survey years (i.e., 00, 02, 04, 06, 08, 10, 12, 14, 16, 18) as a continuous variable. Adjusted means and 95% confidence intervals for peak BMD at different skeletal sites in males and females were estimated, respectively. Generalized linear models with an identity link function were used to test for secular linear and quadratic trends in peak BMD across survey years; we adjusted for age (continuous), BMI (continuous), race/ethnicity (Mexican American, non-Hispanic white, non-Hispanic black, and other race), and moderate or vigorous physical activity (yes/no). Unadjusted models were also constructed. To test whether body weight and height impacted the results, we further adjusted for age (continuous), body weight (continuous), body height (continuous), race/ethnicity (Mexican American, non-Hispanic white, non-Hispanic black, and other race), and moderate or vigorous physical activity (yes/no) in the models. To evaluate potential differences in trends between population subgroups (BMI and race/ethnicity), subgroup analyses and interaction terms tests (i.e., survey years*BMI group, survey years*race/ethnicity, survey years2*BMI group, and survey years2*race/ethnicity) were performed for males and females, respectively. When linear or quadratic trends were statistically significant, Dunnett t-tests were performed to compare differences in peak BMD between each survey cycle and the most recent survey cycle (2017–2018) [21]. To compare differences in peak BMD among different race/ethnicity subgroups, one-way analysis of variance (ANOVA) was used separately for males and females, followed by Scheffe multiple comparisons tests. Lastly, to determine whether serum 25(OH)D and serum total calcium levels impacted the above trends, we further adjusted for serum 25(OH)D and serum total calcium in the fully adjusted models; individuals with missing serum 25(OH)D and serum total calcium data (the percentage of total missing data from 2001–2018 was 7.9% for males and 5.6% for females) were excluded from the analysis.
Results
A total of 22,439 participants aged 14–40 years were included in the analytic study population (12,773 males and 9,666 females) (Fig. 1). Males aged 19–28 years (n = 3,975) and females aged 31–40 years (n = 2,370) were our target population for estimating peak lumbar spine BMD. Lumbar spine BMD data were available from 1999–2000 to 2017–2018 (Table 1 and Supplemental Table 1). Average BMI and the percentage of obese participants in males and females increased from 1999–2000 to 2017–2018 (all Plinear < 0.05). Within BMI subgroups, average BMI for obese males, obese females, and non-obese females also showed increasing trends from 1999–2000 to 2017–2018 (all Plinear < 0.05), while average BMI of non-obese males decreased with survey years (Plinear < 0.05). Average body weight also rose among males and females from 1999–2000 to 2017–2018 (all Plinear < 0.05). Average body height in males increased from 1999–2000 to 2005–2006 and then declined until 2017–2018 (Pquadratic = 0.014), while there was no significant change in females (Plinear and Pquadratic > 0.05). The percentage of participants with moderate or vigorous physical activity in males and females showed upward trends from 1999–2000 to 2005–2006 and downward trends from 2005–2006 onwards (all Pquadratic < 0.05). There were decreasing trends from 1999–2000 to 2005–2006 and increasing trends until 2017–2018 in the percentage of other race males (Pquadratic = 0.010). In females, the percentage of Mexican American participants increased from 1999–2000 to 2017–2018 (Plinear = 0.003), while the percentage of other race participants declined before 2003–2004 and then rose from 2003–2004 to 2017–2018 (Pquadratic = 0.003).
When we analyzed participants with complete data on serum 25(OH)D and serum total calcium, we found that there was no significant change in serum 25(OH)D levels among males and females from 2001–2002 to 2017–2018 (Plinear and Pquadratic > 0.05; Supplemental Table 2). Serum total calcium levels of males and females showed decreasing trends during survey years (all Plinear < 0.001). Other characteristics, including BMI, body weight, height, moderate or vigorous physical activity, and race/ethnicity, showed identical trends over survey years as the overall population above.
We estimated peak hip BMD in males aged 16–25 years (n = 1,744) and females aged 14–23 years (n = 1,681). Hip BMD data were only available between 2005–2006 and 2009–2010 (Supplemental Table 3). The percentage of participants with moderate or vigorous physical activity decreased in females from 2005–2006 to 2009–2010 (Plinear < 0.001). The percentage of other race males increased from 2005–2006 to 2009–2010 (Plinear = 0.003).
From 1999–2000 to 2017–2018, there were no significant secular trends in peak lumbar spine BMD in males or females after adjusting for age, BMI, race/ethnicity, and moderate or vigorous physical activity (all Plinear and Pquadratic > 0.05; Fig. 2). The results remained essentially unchanged in the unadjusted models as well as in the adjusted models that included body weight and height (all Plinear and Pquadratic > 0.05; Supplemental Fig. 1). When we further adjusted for serum 25(OH)D and serum total calcium, similar results were noted (all Plinear and Pquadratic > 0.05; Supplemental Fig. 2). After adjusting for age, BMI, race/ethnicity, and moderate or vigorous physical activity, no significant secular trends were observed for peak femoral neck and total hip BMD in males and females from 2005–2006 to 2009–2010 (all Plinear and Pquadratic > 0.05; Supplemental Fig. 3).
Secular trends in peak lumbar spine bone mineral density (BMD), stratified by gender, the NHANES 1999–2018. The values plotted in each year are the means. The vertical bars represent 95% confidence intervals. Plinear and Pquadratic were adjusted for age (continuous), BMI (continuous), race/ethnicity (Mexican American, non-Hispanic white, non-Hispanic black, and other race), and moderate or vigorous physical activity (yes/no)
In subgroup analyses by obesity category, after adjusting for age, BMI, race/ethnicity, and moderate or vigorous physical activity, we noted significant interactions between obese and non-obese groups in males and females for secular trends in peak lumbar spine BMD (all Pinteraction < 0.05; Fig. 3). Peak lumbar spine BMD in obese males and females increased from 1999–2000 to 2009–2010 and decreased until 2017–2018 (all Pquadratic < 0.05). From 2009–2010 to 2011–2012, the mean peak lumbar spine BMD declined from 1.108 to 1.062 g/cm2 in obese males and from 1.120 to 1.033 g/cm2 in obese females. However, peak lumbar spine BMD in non-obese males remained stable across survey years, 1999–2018 (Plinear and Pquadratic > 0.05). Peak lumbar spine BMD in non-obese females decreased from 1999–2000 to 2005–2006 and then increased until 2017–2018 (Pquadratic < 0.05). From 1999–2000 to 2009–2010, peak lumbar spine BMD in obese males and females was higher than that in non-obese participants, while such differences were no longer seen from 2011–2012 onwards. Similarly, identical results were found when we conducted unadjusted or adjusted models that included body weight and height (Supplemental Fig. 4). Further adjustment for serum 25(OH)D and serum total calcium did not alter the observed trends (Supplemental Fig. 5). From 2005–2006 to 2009–2010, the secular trends for peak hip BMD were not significant after adjusting for age, BMI, race/ethnicity, and moderate or vigorous physical activity in obese and non-obese males and females (all Plinear and Pquadratic > 0.05; Supplemental Fig. 6).
Secular trends in peak lumbar spine bone mineral density (BMD), stratified by gender and obesity, the NHANES 1999–2018. The values plotted in each year are the means. The vertical bars represent 95% confidence intervals. Plinear, Pquadratic, Plinear interaction and Pquadratic interaction were adjusted for age (continuous), BMI (continuous), race/ethnicity (Mexican American, non-Hispanic white, non-Hispanic black, and other race), and moderate or vigorous physical activity (yes/no). * and ** indicate statistically significant difference compared with peak bone mineral density (BMD) in 2017–2018 at alpha = 0.05 and alpha = 0.01, respectively
In subgroup analyses by race/ethnicity, after adjusting for age, BMI, and moderate or vigorous physical activity, secular trends for peak lumbar spine BMD were not significant in any race/ethnicity subgroup for males and females, though peak BMD between race/ethnicity subgroups differed (all Plinear and Pquadratic > 0.05; Supplemental Fig. 7). No significant secular trends were observed for peak hip BMD in most male and female race/ethnicity subgroups between 2005–2006 and 2009–2010 when we adjusted for the above covariates (all Plinear and Pquadratic > 0.05; Supplemental Fig. 8). Overall, non-Hispanic Black males and females had higher peak lumbar spine and hip BMD than other race/ethnicity subgroups (all P < 0.05; Supplemental Figs. 7, 8). When we compared peak lumbar spine BMD between non-Hispanic Black and non-Hispanic White females, differences from 1999 to 2018 ranged from 0.011 to 0.130 g/cm2, with an overall significant mean peak BMD difference of 0.053 g/cm2 (P < 0.001).
Discussion
In this study, we found no statistically significant secular trends in peak lumbar spine BMD from 1999–2000 to 2017–2018 or peak hip BMD from 2005–2006 to 2009–2010 in males or females overall. However, in subgroup analyses, secular trends in peak lumbar spine BMD were modified by obesity; BMD was greater in obese than non-obese males and females up to 2009–2010, but not from 2011–2012 onwards.
Previous studies on BMD trends in the United States showed no significant linear or quadratic trends in lumbar spine BMD from 2005–2006 to 2013–2014 [19]. Although partially consistent with our findings, it is important to note that their study focused on people aged 50 years or older and had a relatively short survey period. Another study found that hip BMD in people aged 30 years and older remained unchanged from 2005–2006 to 2009–2010 but significantly decreased in 2013–2014 [6]. In our study, the non-significant trends in peak hip BMD could also be a function of the short study interval.
In subgroup analyses by obesity category, we found that peak lumbar spine BMD in obese males and females increased from 1999–2000 to 2009–2010 and was higher than in non-obese participants. However, from 2009–2010 onwards, peak lumbar spine BMD in obese participants decreased and was no longer higher. Research has shown that the impact of obesity on bone health is complex. Although two recent meta-analyses have found a positive correlation between obesity and higher BMD, there was increasing evidence that the impact of obesity on BMD varied depending on its fat distribution and other related influencing factors [10, 22, 23]. The higher BMD in obese individuals might be due to the mechanical effect of body weight on bones, which was mainly determined by body lean mass [10, 24]. In our study, the percentage of individuals with moderate or vigorous physical activity was higher from 1999–2000 to 2005–2006, contributing to increased mechanical load [24]. We found that the percentage of individuals with moderate or vigorous physical activity decreased from 2007 to 2018. Previous studies found that there was an increase in sedentary time from 2007 to 2016, and an increase in total fat and high saturated fat intake from 1999 to 2016 and processed food intake from 1999 to 2018 among adults in the United States [25,26,27]. These changes led to excessive fat accumulation and abdominal obesity, which was detrimental to BMD in obese individuals, probably mainly through oxidative stress and chronic inflammation [9, 28]. Therefore, changes in the relative ratio of body lean mass to body fat mass and fat distribution in obese participants over the study period are potential explanations for our findings. Another possible explanation is that osteoblasts in obese individuals decrease as the number and metabolic rate of adipose tissue increase since both osteoblasts and adipose tissue originate from mesenchymal stem cells [29]. Obesity can also lead to the transformation of mesenchymal stem cells into bone marrow fat by promoting the inflammatory microenvironment in the bone marrow, both of which have adverse effects on BMD, especially in the lumbar vertebrae rich in trabecular bone [30,31,32]. Lastly, we found that both average BMI and body weight increased from 1999–2000 to 2017–2018, suggesting that obese participants were more obese than before; the deleterious effects of obesity (especially among obese young adults) on bone health may be worse over time.
The main changes in the NHANES from 2011 to 2018 were an oversampling of non-Hispanic Asian participants and a decrease in sample size for non-low-income non-Hispanic white participants, which might affect observed trends. Because Asian participants had lower BMD and obesity prevalence than non-Asian participants, and individuals with lower incomes were more likely to be underweight and accumulate visceral fat, this might have a negative impact on bone metabolism [33,34,35]. However, in this study, non-Hispanic Asian participants accounted for less than 7% of the total male and female population, which is unlikely to alter the BMD distribution of the total population. In addition, the results were not affected before and after adjusting for covariates including race/ethnicity, suggesting that changes in sampling strategy do not explain the findings.
In subgroup analyses by race/ethnicity, although mean peak lumbar spine BMD differences between non-Hispanic black and non-Hispanic white females varied from 1999 to 2018 (ranging from 0.011 to 0.130 g/cm2), the overall mean peak BMD difference remained statistically significant. Therefore, we believed that non-Hispanic black females had statistically higher peak lumbar spine BMD other racial groups overall, despite variability for individual survey years. Other studies have also suggested that BMD differences between races/ethnicities varied over time [6].
Osteoporotic fracture is a severe complication of osteoporosis, which can lead to reduced quality of life, disability, and even death, with significant economic burden [36]. The accrual of bone mass during childhood and adolescence is essential to reducing the risk of osteoporosis and osteoporotic fractures later in life [24]. For the overall population, secular trends in peak lumbar spine and hip BMD across survey years were not statistically significant. Therefore, from a population perspective, the role of peak BMD on later osteoporosis risk is likely to remain stable. However, it should be noted that mean peak lumbar spine BMD among obese males and females decreased by approximately 4–8% from 2009–2010 to 2011–2012. If such differences are sustained until later life, obese individuals might experience a clinically meaningful 5–10 years earlier onset of osteoporosis. Whether this is actually observed will require future long-term prospective studies.
The current study has several strengths. We studied nationally representative BMD data from the NHANES, ensuring the generalizability of our results to the general population in the United States. In addition, our study investigated secular trends in peak lumbar spine BMD across 20 years, which was a considerable period, and the sample size was large enough for subgroup analyses.
Several limitations of this study are acknowledged. First, due to the cross-sectional data in the NHANES, we were unable to determine the longitudinal effects of factors on peak BMD trends. Second, although the NHANES performed strict quality control for BMD measurements, it remains unclear whether there was any impact on measurements in obese people. In particular, obese individuals have greater tissue thickness than non-obese individuals, which can negatively affect BMD measurements due to scanning artifacts [37]. Although we adjusted for BMI in our study, which partially captures tissue thickness, we cannot fully exclude the adverse effects of tissue thickness on peak BMD trend results across survey years. Finally, we only studied the secular trends in peak hip BMD from 2005–2006 to 2009–2010. Whether a longer study duration would change our findings warrants further research.
In summary, there were no significant secular trends in peak lumbar spine and hip BMD in males and females. However, secular trends for peak lumbar spine BMD in obese participants decreased from 2009–2010 to 2017–2018, and peak lumbar spine BMD in obese participants was no longer higher than that in non-obese participants from 2011–2012 onwards. Additional prospective studies are needed to confirm this apparent decline among obese individuals and understand the reason(s).
References
Heaney RP, Abrams S, Dawson-Hughes B, Looker A, Marcus R, Matkovic V, Weaver C (2000) Peak bone mass. Osteoporos Int 11:985–1009. https://doi.org/10.1007/s001980070020
Tonnesen R, Schwarz P, Hovind PH, Jensen LT (2016) Physical exercise associated with improved BMD independently of sex and vitamin D levels in young adults. Eur J Appl Physiol 116:1297–1304. https://doi.org/10.1007/s00421-016-3383-1
Lorenzo JA, Canalis E, Raisz LG (2007) Metabolic bone disease. In: Kroenberg HM (ed) Williams textbook of endocrinology, 11th edn. Saunders, Philadelphia, pp 1373–1410
Hernandez CJ, Beaupre GS, Carter DR (2003) A theoretical analysis of the relative influences of peak BMD, age-related bone loss and menopause on the development of osteoporosis. Osteoporos Int 14:843–847. https://doi.org/10.1007/s00198-003-1454-8
Kanis JA, Johnell O, Oden A, Jonsson B, De Laet C, Dawson A (2000) Risk of hip fracture according to the World Health Organization criteria for osteopenia and osteoporosis. Bone 27:585–590. https://doi.org/10.1016/s8756-3282(00)00381-1
Xu Y, Wu Q (2018) Decreasing trend of bone mineral density in US multiethnic population: analysis of continuous NHANES 2005–2014. Osteoporos Int 29:2437–2446. https://doi.org/10.1007/s00198-018-4648-9
Chen C, Chen Q, Nie B, Zhang H, Zhai H, Zhao L, Xia P, Lu Y, Wang N (2020) Trends in bone mineral density, osteoporosis, and osteopenia among U.S. adults with prediabetes, 2005–2014. Diabetes Care 43:1008–1015. https://doi.org/10.2337/dc19-1807
Fan K, Lv F, Li H, Meng F, Wang T, Zhou Y (2023) Trends in obesity and severe obesity prevalence in the United States from 1999 to 2018. Am J Hum Biol 35:e23855. https://doi.org/10.1002/ajhb.23855
Cao JJ (2011) Effects of obesity on bone metabolism. J Orthop Surg Res 6:30. https://doi.org/10.1186/1749-799x-6-30
Rinonapoli G, Pace V, Ruggiero C, Ceccarini P, Bisaccia M, Meccariello L, Caraffa A (2021) Obesity and bone: a complex relationship. Int J Mol Sci. https://doi.org/10.3390/ijms222413662
Xue S, Kemal O, Lu M, Lix LM, Leslie WD, Yang S (2020) Age at attainment of peak bone mineral density and its associated factors: The National Health and Nutrition Examination Survey 2005–2014. Bone 131:115163. https://doi.org/10.1016/j.bone.2019.115163
Xue S, Zhang Y, Qiao W, Zhao Q, Guo D, Li B, Shen X, Feng L, Huang F, Wang N et al (2021) An updated reference for calculating bone mineral density T-scores. J Clin Endocrinol Metab 106:e2613–e2621. https://doi.org/10.1210/clinem/dgab180
Jung KJ, Chung CY, Park MS, Kwon SS, Moon SY, Lee IH, Kim KH, Lee KM (2016) Different reference BMDs affect the prevalence of osteoporosis. J Bone Miner Metab 34:347–353. https://doi.org/10.1007/s00774-015-0676-0
Yetley EA, Pfeiffer CM, Schleicher RL, Phinney KW, Lacher DA, Christakos S, Eckfeldt JH, Fleet JC, Howard G, Hoofnagle AN et al (2010) NHANES monitoring of serum 25-hydroxyvitamin D: a roundtable summary. J Nutr 140:2030s–2045s. https://doi.org/10.3945/jn.110.121483
Centers for Disease Control and Prevention (2007) National Health and Nutrition Examination Survey (NHANES) dual energy X-ray absorptiometry (DXA) procedures manual. https://wwwn.cdc.gov/nchs/data/nhanes/2007-2008/manuals/manual_dexa.pdf. Accessed 10 September 2021
Centers for Disease Control and Prevention (2018) National Health and Nutrition Examination Survey (NHANES) Body composition procedures manual. https://wwwn.cdc.gov/nchs/data/nhanes/2017-2018/manuals/Body_Composition_Procedures_Manual_2018.pdf. Accessed 10 September 2021
Wahner HW, Looker A, Dunn WL, Walters LC, Hauser MF, Novak C (1994) Quality control of bone densitometry in a national health survey (NHANES III) using three mobile examination centers. J Bone Miner Res 9:951–960. https://doi.org/10.1002/jbmr.5650090621
Hologic Incoporated Company (2011) Practical considerations when replacing a DXA system. https://hologiced.com/wp-content/uploads/2018/06/Wilson-KE.-Practical-Considerations-When-Replacing-a-DXA-System.pdf. Accessed 19 January 2024
Looker AC, Sarafrazi Isfahani N, Fan B, Shepherd JA (2017) Trends in osteoporosis and low bone mass in older US adults, 2005–2006 through 2013–2014. Osteoporos Int 28:1979–1988. https://doi.org/10.1007/s00198-017-3996-1
Centers for Disease Control and Prevention (2008) National center for health statistics: technical documentation for the 1999-2004 dual-energy X-ray absorptiometry (DXA) multiple imputation data files. https://wwwn.cdc.gov/Nchs/data/nhanes/dxa/dxa_techdoc.pdf. Accessed 10 September 2021
Dunnett CW (1955) A multiple comparison procedure for comparing several treatments with a control. J Am Stat Assoc 50:1096–1121. https://doi.org/10.1080/01621459.1955.10501294
Turcotte AF, O’Connor S, Morin SN, Gibbs JC, Willie BM, Jean S, Gagnon C (2021) Association between obesity and risk of fracture, bone mineral density and bone quality in adults: a systematic review and meta-analysis. PLoS ONE 16:e0252487. https://doi.org/10.1371/journal.pone.0252487
Qiao D, Li Y, Liu X, Zhang X, Qian X, Zhang H, Zhang G, Wang C (2020) Association of obesity with bone mineral density and osteoporosis in adults: a systematic review and meta-analysis. Public Health 180:22–28. https://doi.org/10.1016/j.puhe.2019.11.001
Weaver CM, Gordon CM, Janz KF, Kalkwarf HJ, Lappe JM, Lewis R, O’Karma M, Wallace TC, Zemel BS (2016) The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: a systematic review and implementation recommendations. Osteoporos Int 27:1281–1386. https://doi.org/10.1007/s00198-015-3440-3
Yang L, Cao C, Kantor ED, Nguyen LH, Zheng X, Park Y, Giovannucci EL, Matthews CE, Colditz GA, Cao Y (2019) Trends in sedentary behavior among the US population, 2001–2016. JAMA 321:1587–1597. https://doi.org/10.1001/jama.2019.3636
Shan Z, Rehm CD, Rogers G, Ruan M, Wang DD, Hu FB, Mozaffarian D, Zhang FF, Bhupathiraju SN (2019) Trends in dietary carbohydrate, protein, and fat intake and diet quality among US adults, 1999–2016. JAMA 322:1178–1187. https://doi.org/10.1001/jama.2019.13771
Juul F, Parekh N, Martinez-Steele E, Monteiro CA, Chang VW (2022) Ultra-processed food consumption among US adults from 2001 to 2018. Am J Clin Nutr 115:211–221. https://doi.org/10.1093/ajcn/nqab305
Krishnan C, Choksi P, Peterson MD (2017) Abdominal adiposity and low physical activity are independently and inversely associated with bone mineral density. Obes Res Clin Pract 11:740–746. https://doi.org/10.1016/j.orcp.2017.04.002
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284:143–147. https://doi.org/10.1126/science.284.5411.143
Shen W, Scherzer R, Gantz M, Chen J, Punyanitya M, Lewis CE, Grunfeld C (2012) Relationship between MRI-measured bone marrow adipose tissue and hip and spine bone mineral density in African-American and Caucasian participants: the CARDIA study. J Clin Endocrinol Metab 97:1337–1346. https://doi.org/10.1210/jc.2011-2605
Bilgiç E, Boyacıoğlu Ö, Gizer M, Korkusuz P, Korkusuz F (2020) Chapter 6—architecture of bone tissue and its adaptation to pathological conditions. In: Angin S, Şimşek IE (eds) Comparative kinesiology of the human body. Academic Press, Amsterdam, pp 71–90
da Silva SV, Renovato-Martins M, Ribeiro-Pereira C, Citelli M, Barja-Fidalgo C (2016) Obesity modifies bone marrow microenvironment and directs bone marrow mesenchymal cells to adipogenesis. Obesity (Silver Spring) 24:2522–2532. https://doi.org/10.1002/oby.21660
Noel SE, Santos MP, Wright NC (2021) Racial and ethnic disparities in bone health and outcomes in the United States. J Bone Miner Res 36:1881–1905. https://doi.org/10.1002/jbmr.4417
Gong S, Wang K, Li Y, Zhou Z, Alamian A (2021) Ethnic group differences in obesity in Asian Americans in California, 2013–2014. BMC Public Health 21:1589. https://doi.org/10.1186/s12889-021-11612-z
Zhang Y, Tan C, Tan W (2023) BMI, socioeconomic status, and bone mineral density in U.S. adults: mediation analysis in the NHANES. Front Nutr 10:1132234. https://doi.org/10.3389/fnut.2023.1132234
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res 22:465–475. https://doi.org/10.1359/jbmr.061113
Yu EW, Thomas BJ, Brown JK, Finkelstein JS (2012) Simulated increases in body fat and errors in bone mineral density measurements by DXA and QCT. J Bone Miner Res 27:119–124. https://doi.org/10.1002/jbmr.506
Acknowledgements
The authors acknowledge the data from the National Health and Nutrition Examination Survey (NHANES). All data are available at the NHANES website (https://wwwn.cdc.gov/nchs/nhanes/default.aspx).
Funding
This work is supported by a research grant from the Changchun Scientific and Technological Development Program (Grant Number: 21ZGM28). LML is supported by a Tier 1 Canada Research Chair (CRC-2017–00186). This work is also supported by a research grant from the Norman Bethune Program (Grant Number: 2023B11).
Author information
Authors and Affiliations
Contributions
Conception and design (XZ and SY); data curation, formal analysis, and validation (XZ, LY and JZ); interpretation of data (all authors); drafting the article (XZ); supervision (SY, BK, LL and WL); funding acquisition (SY and BK); critically revising the article for important intellectual content (all authors); final approval of the version to be published (all authors); and agreement to be accountable for all aspects of the work (all authors).
Corresponding author
Ethics declarations
Conflict of interest
Xiaohua Zhang, Linghua Yang, Juan Zhang, Lisa M. Lix, William D. Leslie, Bo Kan, Shuman Yang are declare that they have no conflicts of interest.
Ethics Approval
The data from NHANES has been approved by the National Center for Health Statistics Research Ethics Review Board; they are Protocol #98-12, Protocol #2005-06, Continuation of Protocol #2005-06, Continuation of Protocol #2005-06, Protocol #2011-17, Continuation of Protocol #2011-17, Continuation of Protocol #2011-17, Continuation of Protocol #2011-17 (Effective through October 26, 2017), and Protocol #2018-01 (Effective beginning October 26, 2017).
Consent to Participate
Written informed consent was obtained from all participants (i.e., individual participants, legal guardians, and the parents); this can be obtained from the NHANES website (https://www.cdc.gov/nchs/nhanes/biospecimens/participants.htm).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Yang, L., Zhang, J. et al. Secular Trends in Peak Bone Mineral Density: The National Health and Nutrition Examination Survey 1999–2018. Calcif Tissue Int 114, 480–489 (2024). https://doi.org/10.1007/s00223-024-01198-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-024-01198-0