Abstract
Vision impairments such as age-related macular degeneration (AMD) and glaucoma are among the top risk factors for geriatric falls and falls-related injuries. AMD and glaucoma lead to loss of the central and peripheral visual fields, respectively. This study utilized a custom contact lens model to occlude the peripheral or central visual fields in healthy adults, offering a novel within-subject approach to improve our understanding of the etiology of balance impairments that may lead to an increased fall risk in patients with visual field loss. Two dynamic posturography tests, including an adapted version of the Sensory Organization Test and a virtual reality environment with the visual scene moving sinusoidally, were used to evaluate standing balance. Balance stability was quantified by displacement and time-normalized path length of the center of pressure. Nine young and eleven older healthy adults wore visual field occluding contact lenses during posturography assessments to compare the effects of acute central and peripheral visual field occlusion. The results found that visual field occlusion had greater impact on older adults than young adults, specifically when proprioceptive cues are unreliable. Furthermore, the results suggest that both central and peripheral visions are important in postural control; however, peripheral vision may be more sensitive to movement in the environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vision impairments are among the leading risk factors for falls and falls-related injuries. Lamoureux et al. (2008) reported a twofold increase in the risk of falling with severe vision impairment in one eye and mild/moderate visual disability in the other eye. Hip fractures are also associated with poor vision (Felson et al. 1989). Specific aspects of visual function implicated in falls include poor visual acuity (Lord and Dayhew 2001), diminished contrast sensitivity (Lord et al. 1991, 1993, 1994; Lord and Dayhew 2001), depth perception (Lord and Dayhew 2001), and visual field (VF) losses (Coleman et al. 2007; Freeman et al. 2007). In the Salisbury Eye Evaluation study, Freeman and colleagues examined associations between falls and visual impairments including visual acuity, contrast sensitivity, and VF losses. After controlling for demographic and health status, only VF losses, particularly peripheral VF losses, remained significantly associated with falls (Freeman et al. 2007). Furthermore, the impact on gait and stability is related to the amount of VF loss. Timmis et al. used a contact lens model similar to that used in the present study to occlude the central VF to varying degrees in healthy young adults. The results show that individuals exhibited gait adaptations and different obstacle avoidance techniques when 20° central VF was occluded, but not when 10° central VF was occluded (Timmis et al. 2016). This suggests that there may be a functional threshold, whereby visual field losses lead to balance and gait deficits. Consequently, in the present study, the focus is on the role of severe peripheral and central VF losses on balance.
Two of the most prominent ocular pathologies affecting visual fields in older adults are age-related macular degeneration (AMD) and primary open-angle glaucoma (Thylefors and Negrel 1994; Cruickshanks et al. 1997; Augood et al. 2006). AMD and glaucoma lead to progressive and chronic loss of the central and peripheral visual fields, respectively. Both AMD and glaucoma populations have been associated with an increased prevalence of falls; there is approximately a 2.6-fold in the risk of falling in patients with AMD compared to their age-matched controls (Szabo et al. 2008). One study conducted in Singapore in adults over 40 years old reported a fourfold increase in the risk of falls with glaucoma (Lamoureux et al. 2008). Similar trends were confirmed in other countries including Canada and USA (Guse and Porinsky 2003; Haymes et al. 2007; Bramley et al. 2008).
What remains unclear is the underlying reason as to why patients with glaucoma (i.e., with peripheral VF deficits) and with AMD (i.e., central VF deficits) experience a higher rate of falls than those with normal vision. There are two main types of mechanisms that could potentially explain the contribution of VF deficits to the risk of falling in AMD and glaucoma. The first mechanism is the inability to detect environmental hazards (Friedman et al. 2007). Patients with glaucoma have been shown to perform as well as healthy controls when crossing an obstacle course (Turano et al. 1999; Friedman et al. 2007); however, mobility performance measures in obstacle avoidance tasks have been positively linked with the size of the binocular central scotoma in AMD (Hassan et al. 2002). These findings suggest that while patients with AMD may be at an increased risk of falls due to their inability to detect environmental hazards, another mechanism is responsible for the increased risk of falls in patients with glaucoma. The second potential mechanism is linking VF with falls in the impact on balance; specifically, the impact of VF on sensory integration important for postural control. Effective integration of multisensory information (i.e., vision, proprioception, and vestibular inputs) is a process often termed “sensory re-weighting” (Peterka 2002; Mahboobin et al. 2005, 2009; Jeka et al. 2006; Asslander and Peterka 2014, 2016; Logan et al. 2014). This process involves resolving conflicting sensory inputs if two or more channels provide contradictory body state-related information and relies on accurate sensory channels to generate postural adjustments if one or more afferent inputs are inaccurate/noisy or absent (Peterka 2002; Asslander and Peterka 2014, 2016).
There is some dispute in the literature as to the roles that central and peripheral visions play in maintaining balance. Three general hypotheses of VF influences on postural control have been put forth in the literature: (1) the peripheral dominance theory is that peripheral vision is more important than central vision in postural control (Amblard and Carblanc 1980); (2) the retinal invariance hypothesis suggests that central vision is just as important as peripheral vision in the control of posture (Straube et al. 1994); and (3) the functional sensitivity hypothesis suggests that the periphery of the retina is most susceptible to lamellar optic flow and the central part is most sensitive to radial optic flow, and thus, both central and peripheral have important, but functionally different, roles in maintaining posture (Stoffregen 1985; Warren and Kurtz 1992; Bardy et al. 1999). A reason why there are varying conclusions in the previous studies may be due to different experimental methods of testing the VF. Some studies do not remove part of the VF, but rather provide stimulus to only part of the field; others have used goggles to block part of the VF. These methods may not be consistently and accurately removing input to the central/peripheral visual fields; thus, our study aims to employ a novel way to induce acute VF losses using painted contact lenses in otherwise completely normal subjects to isolate the effect of VF loss alone (Nau 2012). Furthermore, studies on the influence of central and peripheral vision on balance control are difficult in patients due to the presence of systemic or ocular co-morbidities. In addition, the previous research has shown that patients with chronic central VF loss exhibit adaptations in gait kinematics and adopt more cautious strategies when obstacles are presented (Timmis and Pardhan 2012; Timmis et al. 2014). The contact lens model used in this study eliminates potential confounders of co-morbid conditions or long-term adaptations by offering a novel within-subject approach to improve our understanding of the etiology of balance impairments in individuals with VF losses at an increased risk of falls.
This study aims to determine the effect of acute peripheral and central VF losses on postural control in healthy young and older adults. Our overarching hypothesis is that both central and peripheral visions are important in the maintenance of balance; however, peripheral vision is critical in detecting motion, particularly when proprioception is inaccurate. Furthermore, we predict that these hypothesized effects of the loss of central-peripheral vision on balance will be greater in older adults than young adults. To test this hypothesis, two dynamic posturography tests will be used to assess differences in balance between (1) acute central and peripheral VF losses compared to baseline vision and (2) young and older adults wearing VF occluding contact lenses. Displacement (center of pressure RMS) and time-normalized path length (NPL) of postural sway will be measured, with a greater RMS and NPL representing a decrease in balance control. It is anticipated that both RMS and NPL will be greatest during acute peripheral VF occlusion, followed by acute central VF occlusion, with the baseline (no occlusion) producing the minimum sway measures. These differences will be amplified when sensory cues from the proprioceptive system are unreliable and when the postural system is driven by an external stimulus. In addition, it is expected that there will be a greater difference in sway during VF occlusion compared to baseline in older adults than young adults.
Methods
Nine young (aged 27.8 ± 2.1 years, 4 females) and eleven older adults (aged 72.2 ± 5.1 years, 7 females) were recruited. Eligible individuals were healthy and able to stand for at least 2 h. Exclusionary criteria included self-reported orthopedic, neurological, pulmonary, or cardiovascular abnormalities hindering normal balance and gait. In addition, individuals were excluded if they were taking any central nervous system anti-depressant drugs, including benzodiazepines or barbiturates, or taking more than five prescription drugs, as both have been associated with increased fall risk (Caramel et al. 1998; Weiner et al. 1998). Written informed consent approved by the University of Pittsburgh Institutional Review Board was obtained prior to participation.
Two visits were required to complete the study; the first visit consisted of screening procedures and the second included the experimental dynamic posturography session. Screenings excluded some potential participants from continuing to the experimental session. Subjects with cognitive impairment, defined as a score of less than 24 on the Mini-Mental Status Exam, were excluded (Folstein et al. 1975; Tombaugh and McIntyre 1992). Participants with vestibular abnormalities were excluded by rotational chair tests (ROTO) (Arriaga et al. 2005) performed by a neurologist with expertise in balance disorders. Lower extremity sensation was tested bilaterally using Semmes–Weinstein monofilaments to measure cutaneous pressure thresholds. Subjects were excluded if they could not sense the Semmes–Weinstein 5.07/10 g monofilament, which is considered to be the threshold for protective sensation (Kumar et al. 1991; Kuyk et al. 1996; Armstrong et al. 1998). Finally, participants underwent a complete ocular and vision examination by an optometrist to ensure normal vision.
At the end of their first visit, participants who met all eligibility criteria were fitted by an optometrist with custom contact lenses to be worn during the experimental session. Lenses were made with appropriate prescription for full distance correction for each participant. Two sets of lenses were custom painted for each subject: central VF occluding lenses and peripheral VF occluding lenses (Adventures in Colors, Golden, CO) (Fig. 1). Central VF occluding lenses were painted with central opacities measuring 8 mm and peripheral VF occluding lenses were painted with peripheral opacities leaving a 1 mm clear center, according to previously established parameters that have been shown to produce moderate-to-severe visual deficits (Nau 2012).
The experimental session consisted of two dynamic posturography assessments. Prior to both tests, participant’s eyes were dilated to eliminate variability from iris movement and ensure a constant VF during data collection. The first assessment was a dynamic posturography test adapted from the Sensory Organization Test (SOT) using an Equitest posture platform (Neurocom, Inc) located in the Jordan Balance Disorders Laboratory within the Eye and Ear Institute of Pittsburgh. The SOT is a well-established test used to evaluate sensory integration abilities relevant to postural control (Nashner 1982; Bronte-Stewart et al. 2002). The platform provides sway-referenced rotations of the floor and/or the visual scene. Sway-referencing is accomplished by rotation in direct proportion to an individual’s sway, thus reducing correct sensory information from the modality being sway-referenced (vision if the visual scene is sway-referenced or proprioception if the floor is sway-referenced) (Nashner 1982). Sway-referencing was provided through the method of low-pass filtering of the COP data collected by the Equitest platform. There are six conditions in the SOT test, including combinations of fixed/sway-referenced visual scene and floor conditions and eyes closed/open conditions (Table 1). Prior to beginning the test, all participants donned a safety harness that would catch them in the event of a loss of balance. Participants were instructed to cross their arms across the chest and to stand as still as possible without locking their knees. The six conditions were presented in order and repeated three times, once for each lens condition: central VF occluding lenses, peripheral VF occluding lenses, and no occlusion. The presentation order of the peripheral VF occluding lenses and central VF occluding lenses was randomized, and the no occlusion condition was always presented last. One trial of each condition, including the eye-closed conditions (SOT conditions 2 and 5), was collected per lens condition and trial length was set at 60 s. Eye-closed conditions were repeated to act as a control as there should not be any VF occlusion effects on these conditions. Postural sway was assessed using the center of pressure (COP) data collected at a sampling frequency of 100 Hz.
The second dynamic posturography test, which examined postural response to a moving visual environment, took place in the Medical Virtual Reality Center in the Eye and Ear Institute of Pittsburgh. The Medical Virtual Reality Center contains a custom built virtual environment, termed the BNAVE (Balance Near Automatic Virtual Environment), that creates an immersive visual surround by projecting computer generated images onto three adjoining screens positioned to the left, front, and right of the participant (Jacobson et al. 2001; Musolino et al. 2006). A contiguous image is projected onto the screens and provides a full (180°) horizontal field of view. The image presented to participants consisted of a “bulls-eye” pattern of six alternating black-and-white concentric rings surrounded by a checkerboard of black-and-white squares (Fig. 2). The visual scene (central bulls-eye + peripheral checkerboard) moved radially in an anterior–posterior sinusoidal pattern with an amplitude of 20 cm and a frequency of 0.25 Hz (Jasko et al. 2003). Each trial lasted 100 s, with 20 s of fixed visual stimuli at the beginning and the end and 60 s of sinusoidal perturbed stimuli in the middle. Four trials were collected for each of the three lens conditions: two with fixed floor and two with sway-referenced floor. The presentation of lens conditions was the same as in the first posturography test. Again, a harness system was used to prevent injury if participants fell. COP data were collected at 100 Hz from a NeuroTest™ platform (Neurocom, Inc). Technical difficulties were experienced when collecting two older participants, and thus, they were excluded from further analysis in the BNAVE test only.
Sway data, as measured by the COP, from both the adapted SOT test and the BNAVE test were first down-sampled to 20 Hz and then filtered using a low-pass Butterworth filter with a cut-off frequency of 2.5 Hz. For the adapted SOT test, the first and last 5 s of each condition were removed to eliminate any transient effects. For each trial of the BNAVE test, only the 60-s portion with the sinusoidal visual perturbation were analyzed, and the first and last 5 s of this portion were removed. Due to technical difficulties, the second trial of the sway-referenced flooring condition was missing for three participants. Therefore, only the first trial of each flooring condition in the BNAVE test was used in analysis, which is also consistent with the single trial collected in the adapted SOT test.
For both the adapted SOT test and BNAVE test, two sway variables were calculated: (1) anterior–posterior displacement was quantified by the root mean square (RMS) of anterior–posterior COP and (2) the time-normalized path length (NPL) of the anterior–posterior COP. NPL was calculated by taking the absolute value of the derivative of the COP and then averaging over time (Maurer and Peterka 2005). Only outcome measures in the anterior–posterior direction were considered in the analyses, as all perturbations were in the anterior–posterior direction in both posturography tests.
A series of mixed linear models was fitted for each condition of the adapted SOT test with either RMS or NPL as the dependent variable; age group, lens condition, and their interaction as fixed effects of interest; and a participant-within-age group as a random effect to account for multiple measurements from the same participant. Upon observing distributional deviations of residuals from normality in probability–probability plots, the dependent variables were natural log transformed and the models were re-fitted. Appropriately constructed means contrasts were used to make various comparisons of interest between lens conditions and age groups. Specifically, the overall VF occlusion effect was tested within each age group and the overall age effect was tested within each VF occlusion condition. A similar analytic strategy was employed in the BNAVE test, with floor condition instead of the SOT condition in the adapted SOT test. To mitigate effects of multiple comparisons on type I errors, we made between-lens post hoc pairwise comparisons only if the overall lens effect was significant within age group. SAS® version 9.3 (SAS Institute, Inc., Cary, North Carolina) was used for all statistical analyses with α = 0.05.
Results
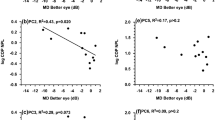
In the adapted SOT test, as expected, VF occlusion did not impact balance measures in either age group during the eye-closed conditions (SOT conditions 2 and 5). Therefore, data under these conditions will not be further discussed. VF occlusion had a significant effect on NPL, but only in older adults and during SOT condition 4 (p = 0.02, F = 4.59, Fig. 3) and SOT condition 6 (p = 0.01, F = 4.59, Fig. 3), i.e., when the floor is sway-referenced. Thus, further post hoc tests comparing VF occlusion conditions were conducted in older adults for these SOT conditions. These tests showed that under SOT condition 4, older adults exhibited on average 23% greater NPL in both central and peripheral VF occlusion conditions compared than no occlusion (p = 0.02 and p = 0.01, respectively). Central and peripheral VF occlusion yielded similar NPL values in older adults under SOT condition 4 (p > 0.1). When both vision and floor were sway-referenced in SOT condition 6, older adults exhibited on average a 23% increase in NPL when central vision was available (both in the no VF occlusion and peripheral VF occlusion conditions) compared to central VF occlusion condition (p = 0.01 and p = 0.02, respectively). In addition, older adults displayed overall significantly greater NPLs than young adults across VF occlusion and SOT conditions (p < 0.05) (Fig. 3). A final result in the adapted SOT test is that VF occlusion did not have an impact on COP displacement, i.e., RMS (p > 0.05, Table 2). This finding holds true in young and older adults. In addition, COP displacement was similar between young and older adults under all three VF occlusion and SOT conditions (p > 0.05, Table 2).
Mean and standard error of NPL (cm/s) values from the adapted SOT test. VF occlusion conditions labeled with different letters are statistically different (p < 0.05); for conditions not labeled, there were no effects of VF occlusion on NPL. Significant results are: (SOT4) NPL during central occlusion was 21% greater than no occlusion (p = 0.02) and was 25% greater during peripheral occlusion than no occlusion (p = 0.01) in older adults only; (SOT6) NPL during no occlusion was 24% greater than central occlusion (p = 0.02) and was 23% greater during peripheral occlusion than central occlusion (p = 0.01) in older adults only. In general, NPL was greater in older adults than in their young counterparts for all VF occlusion and SOT conditions (p < 0.05)
In contrast to findings in the adapted SOT test, in the BNAVE test, VF occlusion had a significant effect on COP displacement, but only in older adults. This finding holds true both when the floor is fixed (p = 0.03, F = 3.89, Table 3) and sway-referenced (p = 0.05, F = 3.39, Table 3). Thus, further post hoc tests comparing VF occlusion conditions were conducted in older adults. These tests demonstrated that in older adults, RMS increased during central VF occlusion compared to peripheral VF occlusion when the floor was fixed (45% difference, p = 0.01). Similarly, when the floor was sway-referenced, RMS of older adults was greatest when the central VF was occluded (30% greater than peripheral VF occlusion, p = 0.04; 25% greater than no occlusion, p = 0.02) (Table 3). Peripheral VF occlusion yielded similar COP displacement behavior than no occlusion conditions in older adults, both under fixed and sway-referenced floor conditions.
VF occlusion impacted NPL of older adults when the floor was fixed (p = 0.01, F = 5.98, Fig. 4). Further post hoc pairwise comparison tests in older adults when the floor was fixed revealed that NPL was significantly greater during central VF occlusion than both peripheral VF occlusion (27% difference, p = 0.01) and no occlusion (46% difference, p = 0.003). However, when the floor was sway-referenced, only young adults exhibited significant changes in NPL during different occlusion conditions (p = 0.03, F = 3.79). More specifically, NPL of young adults under sway-referenced floor conditions was 27% greater during central VF occlusion (p = 0.01) and 22% greater during peripheral VF occlusion (p = 0.04) than no occlusion. In general, NPL differences between young and older adults were minimal across VF occlusion and floor conditions (Fig. 4).
Mean and standard error of NPL (cm/s) values from the BNAVE test. VF occlusion conditions labeled with different letters are statistically different (p < 0.05); for conditions not labeled, there were no effects of VF occlusion on NPL. Significant results are: (Fixed floor) NPL during central occlusion was 27% greater than no occlusion (p = 0.01) and 46% greater than peripheral occlusion (p = 0.01) in older adults only; (Sway-referenced floor) NPL was 27% greater during central occlusion than no occlusion (p = 0.01) and 22% greater during peripheral occlusion than no occlusion (p = 0.04) in young adults only. In older adults, NPL was significantly greater than young adults only during central occlusion when the floor was fixed (p < 0.05)
Discussion
In this study, young and older healthy participants with normal vision completed balance assessments using three VF occlusion conditions: (1) acute occlusion of central vision, (2) acute occlusion of peripheral vision, and (3) no occlusion. Thus, we were able to determine the effect of acute VF loss on balance, a first step toward understanding the underlying mechanisms of falls in patients with VF defects. Comparing patient populations to understand the impact of VF deficits alone on balance is challenging due to multiple confounding factors associated with clinical conditions. Thus, prior clinical studies have not been able to fully elucidate underlying mechanisms explaining how specific VF defects result in postural instability and ultimately falls. Our contact lens model was used to create either central or peripheral VF losses in healthy populations and thus allowed the use of a within-subject approach to improve our understanding of the etiology of VF loss-related balance/gait impairments. Furthermore, the results of this study show how older adults respond differently to VF loss affects than young adults.
The two balance tests used in this experiment allowed us to investigate the impact of acute central/peripheral VF loss on balance when visual cues are reliable (adapted SOT test, conditions 1 and 4), reduced (adapted SOT test, Conditions 3 and 6) and moving (sinusoidal visual scene movement at 0.25 Hz in the BNAVE test) as well as when balance-related proprioceptive cues are altered. When balance-related visual cues are reliable, acute VF occlusion has a little effect unless proprioceptive cues are altered via sway-referencing (SOT condition 4). Sway NPL increased in both central and peripheral VF occlusion compared to baseline in older adults, but not young adults. This suggests that older adults are more sensitive to any occlusion of the VF when they cannot rely more heavily on proprioceptive inputs.
There were no VF occlusion differences seen in standing balance when visual cues were unreliable and vestibular and proprioceptive inputs were normal (SOT condition 3). However, when both visual and proprioceptive cues were altered via sway-referencing (SOT condition 6), in older adults only, having central vision as in the no occlusion and peripheral occlusion produced a greater balance control challenge (greater NPL values) compared to occluding the central VF (i.e., having peripheral VF only). This finding suggests that having peripheral vision alone increases the sensitivity of the postural control system to motion, which is important when proprioceptive information is unreliable. Thus, peripheral vision may be influential for maintaining balance in ways that are different from central vision, particularly in older adults.
When the postural control system was externally driven by the sinusoidal movement of the visual scene in the BNAVE, older adults show an overall increase in displacement and velocity when central VF was occluded, i.e., only peripheral vision is available. These findings confirm our previous findings from the adapted SOT6 condition that the postural control system is most sensitive to motion-related visual cues from the peripheral VF. Thus, in the BNAVE test, sensitivity of the postural control system to these cues from the peripheral VF is the greatest when they are erroneous. Young adults showed an overall effect of VF occlusion (both central and peripheral) compared to baseline, but only when the floor was sway-referenced. This suggests that the postural control system in young adults is only impacted by VF loss during the most challenging balance conditions when both visual and proprioceptive sensory inputs are altered.
This study found that only NPL was sensitive to differences in age groups within occlusion condition, specifically in the adapted SOT test. NPL, similar to mean velocity, in particular, has been shown to be a sensitive measure of balance control in elucidating postural instabilities including Parkinson’s disease (Rocchi et al. 2002; Maurer et al. 2003; Maurer and Peterka 2005) and older adults (Maki et al. 1990; Prieto et al. 1996; Maurer and Peterka 2005). Similarly, NPL was more sensitive to differences in age groups, as well as VF occlusion, than RMS in this study. There was no consistent age difference within occlusion condition in NPL for the BNAVE test, suggesting that older adults were generally able to adapt to the externally driven visual input to the same extent as young adults. It should be noted that the older participants in this study were overall healthy and exclusionary criteria for this study were very rigorous and individuals with vestibular, proprioceptive, neurological, or musculoskeletal conditions prevalent in older populations were not considered for this study. Thus, greater age effects would be expected if older participants were representative of the general population; however, for the purpose of teasing out the effects of VF occlusion alone, strict exclusionary criteria were necessary for this study.
A potential limitation to this study is the use of sway-referencing to minimize proprioceptive inputs, as it may impact the visual input. It is well established that sway-referencing the floor produces decreased balance stability (Nashner 1982; Peterka 2002); the results of this study, as expected, show an increase in both displacement and velocity during sway-referenced floor conditions compared to baseline. Because sway-referencing increases sway and head motion, sway-referencing the floor also increased the amplitude of visual motion experienced by participants. Therefore, visual input may have had a greater impact on balance and the differences seen between VF occlusion conditions may not have been due to sensory re-weighting but to a change in visual information available to the central versus peripheral sensory systems. However, this effect is minimized in SOT condition 6, when both the floor and vision are sway-referenced and visual motion is synched with body sway.
As discussed previously, the data from both tests used in the present study suggest that both central and peripheral visions are important to maintaining balance, but peripheral vision in particular may be more sensitive to motion. The results of the present study lend support to the functional sensitivity hypothesis that postulates central and peripheral play different but necessary roles in postural control. The previous literature is mixed regarding which theory is most accurate, with some supporting the peripheral dominance theory (Lestienne et al. 1977; Amblard and Carblanc 1980) and others refuting it in support of the functional sensitivity hypothesis (Stoffregen 1985; Warren and Kurtz 1992; Bardy et al. 1999).
The central neural processing of visual information is consistent with the specific influences of peripheral vision on balance seen in this study. More specifically, neuroimaging studies have demonstrated that the representation of the far periphery of the retina sends inputs to area MT/MST (middle temporal area/medial superior temporal area) (Palmer and Rosa 2006). MST in particular has been linked to hierarchical vestibular processing (Chen et al. 2011). Therefore, interruption of this pathway through elimination of peripheral visual input should have a negative effect on postural control, as seen by the increased postural NPL when peripheral vision was occluded and proprioception was minimized. The tracing studies above provide the structural and mechanistic evidences for the clinical findings that interruption of peripheral vision (for example in glaucoma) more negatively impacts a patient’s ability to maintain postural control than patients with central vision loss caused by macular degeneration (Ramulu 2009; Popescu et al. 2011). In addition, visual information is divided into two streams in the brain: parvocellular ganglion cells in the fovea of the retina send information to the ventral stream, while magnocellular ganglion cells in the periphery send information to the dorsal stream (Livingstone and Hubel 1987). The dorsal stream contains a fast cortical pathway and has a more direct connection to and from V1/V2 than the ventral stream (Stephen et al. 2002). Therefore, because visual cues from the peripheral field have a more direct and faster pathway through the brain, when the peripheral VF is blocked, these fast-moving signals are no longer available to the postural control system.
While this study allowed us to examine the impact of acute short-term peripheral vision and central vision loss on balance in healthy subjects using a within-subject experimental design, future studies should compare the results from this study of healthy adults to those with chronic visual impairments such as glaucoma and AMD. One reason for the need of such comparison is that this study did not account for the long-term adaptation effects associated with these conditions. For example, recent literature suggests that structural and functional brain changes occur with glaucoma (Li et al. 2012; Zikou et al. 2012; Dai et al. 2013; Williams et al. 2013) and perhaps to a lesser extent, macular degeneration (Dilks et al. 2014; Hernowo et al. 2014; Burge et al. 2016; Prins et al. 2016). Thus, comparing results of acute VF occlusion to chronic impairments due to glaucoma and AMD will help to tease out the effects of these brain changes versus peripheral vision loss alone on balance.
Conclusions
This study examined within-subject effects of acute peripheral and central VF occlusion on standing balance. The results suggest that while both peripheral and central visions play important but different roles in maintaining balance; peripheral vision may be more sensitive to movement in the visual environment. VF occlusion had the greatest impact when balance-related sensory inputs from proprioception were unreliable, suggesting that VF loss may affect sensory integration of the postural control system. Furthermore, older adults were more sensitive to VF occlusion than young adults. Future work should compare acute VF occlusion examined in this study to chronic VF loss diseases such as glaucoma and macular degeneration.
References
Amblard B, Carblanc A (1980) Role of foveal and peripheral visual information in maintenance of postural equilibrium in man. Percept Mot Skills 51(3):903–912
Armstrong DG, Lavery LA, Vela SA, Quebedeaux TL, Fleischli JG (1998) Choosing a practical screening instrument to identify patients at risk for diabetic foot ulceration. Arch Intern Med 158(3):289–292
Arriaga MA, Chen DA, Cenci KA (2005) Rotational chair (ROTO) instead of electronystagmography (ENG) as the primary vestibular test. Otolaryngol Head Neck Surg 133(3):329–333
Asslander L, Peterka RJ (2014) Sensory reweighting dynamics in human postural control. J Neurophysiol 111(9):1852–1864
Asslander L, Peterka RJ (2016) Sensory reweighting dynamics following removal and addition of visual and proprioceptive cues. J Neurophysiol 116(2):272–285
Augood CA, Vingerling JR, de Jong PT, Chakravarthy U, Seland J, Soubrane G, Tomazzoli L, Topouzis F, Bentham G, Rahu M, Vioque J, Young IS, Fletcher AE (2006) Prevalence of age-related maculopathy in older Europeans: the European Eye Study (EUREYE). Arch Ophthalmol 124(4):529–535
Bardy BG, Warren WH Jr, Kay BA (1999) The role of central and peripheral vision in postural control during walking. Percept Psychophys 61(7):1356–1368
Bramley T, Peeples P, Walt JG, Juhasz M, Hansen JE (2008) Impact of vision loss on costs and outcomes in medicare beneficiaries with glaucoma. Arch Ophthalmol 126(6):849–856
Bronte-Stewart HM, Minn AY, Rodrigues K, Buckley EL, Nashner LM (2002) Postural instability in idiopathic Parkinson’s disease: the role of medication and unilateral pallidotomy. Brain 125(9):2100–2114
Burge WK, Griffis JC, Nenert R, Elkhetali A, DeCarlo DK, ver Hoef LW, Ross LA, Visscher KM (2016) Cortical thickness in human V1 associated with central vision loss. Sci Rep 6:23268
Caramel VM, Remarque EJ, Knook DL, Lagaay AM, Van den Brande KJ (1998) Benzodiazepine users aged 85 and older fall more often. J Am Geriatr Soc 46(9):1178–1179
Chen A, DeAngelis GC, Angelaki DE (2011) A comparison of vestibular spatiotemporal tuning in macaque parietoinsular vestibular cortex, ventral intraparietal area, and medial superior temporal area. J Neurosci 31(8):3082–3094
Coleman AL, Cummings SR, Yu F, Kodjebacheva G, Ensrud KE, Gutierrez P, Stone KL, Cauley JA, Pedula KL, Hochberg MC, Mangione CM (2007) Binocular visual-field loss increases the risk of future falls in older white women. J Am Geriatr Soc 55(3):357–364
Cruickshanks KJ, Hamman RF, Klein R, Nondahl DM, Shetterly SM (1997) The prevalence of age-related maculopathy by geographic region and ethnicity. The Colorado-Wisconsin Study of Age-Related Maculopathy. Arch Ophthalmol 115(2):242–250
Dai H, Morelli JN, Ai F, Yin D, Hu C, Xu D, Li Y (2013) Resting-state functional MRI: functional connectivity analysis of the visual cortex in primary open-angle glaucoma patients. Hum Brain Mapp 34(10):2455–2463
Dilks DD, Julian JB, Peli E, Kanwisher N (2014) Reorganization of visual processing in age-related macular degeneration depends on foveal loss. Optom Vis Sci 91(8):e199–206
Felson DT, Anderson JJ, Hannan MT, Milton RC, Wilson PW, Kiel DP (1989) Impaired vision and hip fracture. The Framingham Study. J Am Geriatr Soc 37(6):495–500
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198
Freeman EE, Munoz B, Rubin G, West SK (2007) Visual field loss increases the risk of falls in older adults: the Salisbury eye evaluation. Invest Ophthalmol Vis Sci 48(10):4445–4450
Friedman DS, Freeman E, Munoz B, Jampel HD, West SK (2007) Glaucoma and mobility performance: the Salisbury eye evaluation project. Ophthalmology 114(12):2232–2237
Guse CE, Porinsky R (2003) Risk factors associated with hospitalization for unintentional falls: Wisconsin hospital discharge data for patients aged 65 and over. WMJ 102(4):37–42
Hassan SE, Lovie-Kitchin JE, Woods RL (2002) Vision and mobility performance of subjects with age-related macular degeneration. Optom Vis Sci 79(11):697–707
Haymes SA, Leblanc RP, Nicolela MT, Chiasson LA, Chauhan BC (2007) Risk of falls and motor vehicle collisions in glaucoma. Invest Ophthalmol Vis Sci 48(3):1149–1155
Hernowo AT, Prins D, Baseler HA, Plank T, Gouws AD, Hooymans JM, Morland AB, Greenlee MW, Cornelissen FW (2014) Morphometric analyses of the visual pathways in macular degeneration. Cortex 56:99–110
Jacobson J, Redfern MS, Furman JM, Whitney SL, Sparto PJ, Wilson JB, Hodges LH (2001) Balance NAVE: a virtual reality facility for research and rehabilitation of balance disorders. In: Proceedings of the ACM symposium on Virtual reality software and technology, Baniff, Alberta, Canada
Jasko JG, Loughlin PJ, Redfern MS, Sparto PJ (2003) The role of central and peripheral vision in the control of upright posture during anterior-posterior optic flow. In: Proceedings of the 27th Annual Meeting of the American Society of Biomechanics, Toledo, OH
Jeka J, Allison L, Saffer M, Zhang Y, Carver S, Kiemel T (2006) Sensory reweighting with translational visual stimuli in young and elderly adults: the role of state-dependent noise. Exp Brain Res 174(3):517–527
Kumar S, Fernando DJ, Veves A, Knowles EA, Young MJ, Boulton AJ (1991) Semmes-Weinstein monofilaments: a simple, effective and inexpensive screening device for identifying diabetic patients at risk of foot ulceration. Diabetes Res Clin Pract 13(1–2):63–67
Kuyk T, Elliott JL, Biehl J, Fuhr PS (1996) Environmental variables and mobility performance in adults with low vision. J Am Optom Assoc 67(7):403–409
Lamoureux EL, Chong E, Wang JJ, Saw SM, Aung T, Mitchell P, Wong TY (2008) Visual impairment, causes of vision loss, and falls: the Singapore Malay Eye Study. Invest Ophthalmol Vis Sci 49(2):528–533
Lestienne F, Soechting J, Berthoz A (1977) Postural readjustments induced by linear motion of visual scenes. Exp Brain Res 28(3–4):363–384
Li C, Cai P, Shi L, Lin Y, Zhang J, Liu S, Xie B, Shi Y, Yang H, Li S, Du H, Wang J (2012) Voxel-based morphometry of the visual-related cortex in primary open angle glaucoma. Curr Eye Res 37(9):794–802
Livingstone M, Hubel D (1987) Psychophysical evidence for separate channels for the perception of form, color, movement, and depth. J Neurosci 7(11):3416–3468
Logan D, Kiemel T, Jeka JJ (2014) Asymmetric sensory reweighting in human upright stance. PLoS ONE 9(6):e100418
Lord SR, Dayhew J (2001) Visual risk factors for falls in older people. J Am Geriatr Soc 49(5):508–515
Lord SR, Clark RD, Webster IW (1991) Visual acuity and contrast sensitivity in relation to falls in an elderly population. Age Ageing 20(3):175–181
Lord SR, Ward JA, Williams P, Anstey KJ (1993) An epidemiological study of falls in older community-dwelling women: the Randwick falls and fractures study. Aust J Public Health 17(3):240–245
Lord SR, Ward JA, Williams P, Anstey KJ (1994) Physiological factors associated with falls in older community-dwelling women. J Am Geriatr Soc 42(10):1110–1117
Mahboobin A, Loughlin PJ, Redfern MS, Sparto PJ (2005) Sensory re-weighting in human postural control during moving-scene perturbations. Exp Brain Res 167(2):260–267
Mahboobin A, Loughlin P, Atkeson C, Redfern M (2009) A mechanism for sensory re-weighting in postural control. Med Biol Eng Comput 47(9):921–929
Maki BE, Holliday PJ, Fernie GR (1990) Aging and postural control. A comparison of spontaneous- and induced-sway balance tests. J Am Geriatr Soc 38(1):1–9
Maurer C, Peterka RJ (2005) A new interpretation of spontaneous sway measures based on a simple model of human postural control. J Neurophysiol 93(1):189–200
Maurer C, Mergner T, Xie J, Faist M, Pollak P, Lucking CH (2003) Effect of chronic bilateral subthalamic nucleus (STN) stimulation on postural control in Parkinson’s disease. Brain 126(Pt 5):1146–1163
Musolino MC, Loughlin PJ, Sparto PJ, Redfern MS (2006) Spectrally similar periodic and non-periodic optic flows evoke different postural sway responses. Gait Posture 23(2):180–188
Nashner LM (1982) Adaptation to altered support and visual conditions during stance: patients with vestibular deficits. J Neurosci 5:117–124
Nau AC (2012) A contact lens model to produce reversible visual field loss in healthy subjects. Optometry 83(6):278–284
Palmer SM, Rosa MG (2006) A distinct anatomical network of cortical areas for analysis of motion in far peripheral vision. Eur J Neurosci 24(8):2389–2405
Peterka RJ (2002) Sensorimotor integration in human postural control. J Neurophysiol 88(3):1097–1118
Popescu ML, Boisjoly H, Schmaltz H, Kergoat MJ, Rousseau J, Moghadaszadeh S, Djafari F, Freeman EE (2011) Age-related eye disease and mobility limitations in older adults. Invest Ophthalmol Vis Sci 52(10):7168–7174
Prieto TE, Myklebust JB, Hoffmann RG, Lovett EG, Myklebust BM (1996) Measures of postural steadiness: differences between healthy young and elderly adults. IEEE Trans Biomed Eng 43(9):956–966
Prins D, Plank T, Baseler HA, Gouws AD, Beer A, Morland AB, Greenlee MW, Cornelissen FW (2016) Surface-based analyses of anatomical properties of the visual cortex in macular degeneration. PLoS ONE 11(1):e0146684
Ramulu P (2009) Glaucoma and disability: which tasks are affected, and at what stage of disease? Curr Opin Ophthalmol 20(2):92–98
Rocchi L, Chiari L, Horak FB (2002) Effects of deep brain stimulation and levodopa on postural sway in Parkinson’s disease. J Neurol Neurosurg Psychiatry 73(3):267–274
Stephen JM, Aine CJ, Christner RF, Ranken D, Huang M, Best E (2002) Central versus peripheral visual field stimulation results in timing differences in dorsal stream sources as measured with MEG. Vision Res 42(28):3059–3074
Stoffregen TA (1985) Flow structure versus retinal location in the optical control of stance. J Exp Psychol Hum Percept Perform 11(5):554–565
Straube A, Krafczyk S, Paulus W, Brandt T (1994) Dependence of visual stabilization of postural sway on the cortical magnification factor of restricted visual fields. Exp Brain Res 99(3):501–506
Szabo SM, Janssen PA, Khan K, Potter MJ, Lord SR (2008) Older women with age-related macular degeneration have a greater risk of falls: a physiological profile assessment study. J Am Geriatr Soc 56(5):800–807
Thylefors B, Negrel AD (1994) The global impact of glaucoma. Bull World Health Organ 72(3):323–326
Timmis MA, Pardhan S (2012) Patients with central visual field loss adopt a cautious gait strategy during tasks that present a high risk of falling. Invest Ophthalmol Vis Sci 53(7):4120–4129
Timmis MA, Scarfe AC, Tabrett DR, Pardhan S (2014) Kinematic analysis of step ascent among patients with central visual field loss. Gait Posture 39(1):252–257
Timmis MA, Scarfe AC, Pardhan S (2016) How does the extent of central visual field loss affect adaptive gait? Gait Posture 44:55–60
Tombaugh TN, McIntyre NJ (1992) The mini-mental state examination: a comprehensive review. J Am Geriatr Soc 40(9):922–935
Turano KA, Rubin GS, Quigley HA (1999) Mobility performance in glaucoma. Invest Ophthalmol Vis Sci 40(12):2803–2809
Warren WH, Kurtz KJ (1992) The role of central and peripheral vision in perceiving the direction of self-motion. Percept Psychophys 51(5):443–454
Weiner DK, Hanlon JT, Studenski SA (1998) Effects of central nervous system polypharmacy on falls liability in community-dwelling elderly. Gerontology 44(4):217–221
Williams AL, Lackey J, Wizov SS, Chia TM, Gatla S, Moster ML, Sergott R, Spaeth GL, Lai S (2013) Evidence for widespread structural brain changes in glaucoma: a preliminary voxel-based MRI study. Invest Ophthalmol Vis Sci 54(8):5880–5887
Zikou AK, Kitsos G, Tzarouchi LC, Astrakas L, Alexiou GA, Argyropoulou MI (2012) Voxel-based morphometry and diffusion tensor imaging of the optic pathway in primary open-angle glaucoma: a preliminary study. AJNR Am J Neuroradiol 33(1):128–134
Acknowledgements
Funding provided by the National Institutes on Aging [R03 AG04374]. We would like to thank Dr. Joseph Furman and his team for screening participants with balance disorders.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
O’Connell, C., Mahboobin, A., Drexler, S. et al. Effects of acute peripheral/central visual field loss on standing balance. Exp Brain Res 235, 3261–3270 (2017). https://doi.org/10.1007/s00221-017-5045-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-5045-x