Abstract
Right-handed people show an advantage in detecting a delay in visual feedback concerning an active movement of their right hand when it is viewed in a natural perspective compared to when it is seen as if viewing another person’s hand (Hoover and Harris in Exp Brain Res 233:1053–1060, 2012. doi:10.1007/s00221-014-4181-9; Exp Brain Res 222:389–397, 2015a. doi:10.1007/s00221-012-3224-3). This self-advantage is unique to their dominant hand and may reflect an enhanced sense of ownership which contributes to how right-handed people relate to the world. Here we asked whether left-handers show the same pattern of performance for their dominant hand. We measured the minimum delay that could be detected by 29 left-handers when viewing either their dominant or non-dominant hand from ‘self’ or ‘other’ perspectives and compared their thresholds to an age-matched sample of 22 right-handers. Right-handers showed a significant signature self-advantage of 19 ms when viewing their dominant hand in an expected ‘self’ perspective compared to ‘other’ perspectives. Left-handers, however, showed no such advantage for either their dominant or non-dominant hand. This lack of self-advantage in detecting delayed visual feedback might indicate a less secure sense of body ownership amongst left-handers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Our hands are the main organs with which we interact with the world: they are our presence in the world and an organ of the mind (Radman 2013). Our hands are used to aid thought (e.g., in counting) and are an important social tool with which we gesture, communicate, caress, and fight. Other people’s hands are important to us too as indicators of what they might be about to do. Try not to look where someone is pointing! Learning to manipulate objects with the hands plays a critical role in cognitive development through a child’s interactions with the world (Piaget 1954; Bremner and Cowley 2013) and in the construction of the body schema (Merleau-Ponty 1945). It is through interactions with the world, largely with the hands, that a child learns to distinguish themselves from the rest of the world and to develop a distinct sense of self (Rochat 1989, 1998, 2003; Rochat and Striano 2000). The hand then can be regarded as the agent, or even the representative, of the self.
Unlike most mammals, humans generally show a strong handedness preference (Bryden et al. 2000; Chatagny et al. 2013) with approximately 90 % preferring the right (Oldfield 1971; Annett 1985). This preference is associated with the dominant hand (and arm) being stronger (Farthing et al. 2005), faster (Annett et al. 1979) and more precise in performing motor tasks (Heuer 2007; for a review see Goble and Brown 2008). Hand preference may be more than just for convenience: because of the extensive use of the preferred hand, people with dominant right hands experience and interact differently with their environment than people with dominant left hands. Handedness, therefore, frames right- and left-handers’ creation of the mental representations of their bodies and actions differently: the body-specificity hypothesis (Casasanto 2009). This hypothesis posits that ‘peri-dominant-hand space’ (left for left-handers and right for right-handers) provides a reference frame that distinguishes self from non-self body parts (Conson et al. 2010).
Although many behaviours in left-handers are simply reversed relative to right-handers along with their handedness preference, there is some indication that the difference between left- and right-handers may be more profound leading to left-handers having a more easily disrupted perception of self. Thus, left-handers may be slightly more vulnerable to the rubber hand illusion which involves accepting an artificial hand as part of one’s own body (Ocklenburg et al. 2011 but c.f., Haans et al. 2008). Left-handedness has been linked to cognitive disorders, such as schizophrenia (Lishman and McMeekan 1976; Orr et al. 1999; Webb et al. 2013), which are associated with a reduced sense of agency (Franck et al. 2001; Maeda et al. 2013), less confidence of being in control of one’s actions (Gallagher 2000) and a weaker sense of body ownership (as seen for example through an increased rubber hand illusion; Thakkar et al. 2011). Might this mean that even healthy left-handers may have a propensity for lowered body ownership relative to right-handers?
The sense that you are in control of your body’s actions—the sense of agency (Gallagher 2000)—is associated with the sense that you own your body (van den Bos and Jeannerod 2002). Body ownership, however, can be dissociated from agency (Sang et al. 2006; Balslev et al. 2007) and can be quantified by assessing the tolerance for a temporal asynchrony between visual feedback and efferent and proprioceptive information during a self-generated movement (Hoover and Harris 2012, 2015a, b). Performing this task requires the participants to identify the image as being of their own hand and is therefore a test of body ownership. Right-handers are more sensitive at detecting such an asynchrony when viewing movement of their dominant hand in the natural ‘self’ perspective than when viewing their dominant hand from another perspective or their non-dominant hand from any perspective (Hoover and Harris 2012). We interpret this characteristic self-advantage as an implicit measure of body ownership suggesting a special place for the right hand in the self-representation of right-handers. Can such a self-advantage be found for left-handers?
We therefore measured the thresholds for detecting delay in visual feedback for movements of the left and right hands in left-handers while they viewed either hand from a natural ‘self’ perspective and from the perspective of another and compared results with an age-matched control group of right-handers.
Methods
Participants
We tested a total of 56 (32 left- and 24 right-handed) participants, 26 of whom were recruited through the York University Research Participants Pool and were awarded class credit for their participation. The remaining 30 participants were recruited through posters and snowball technique on campus and were paid $10 per hour of participation. Handedness was determined by an adapted version of the Edinburgh Handedness Inventory (Oldfield 1971). Handedness scores could range between −100 and 100 with left-hand dominance falling between −30 and −100 and right-hand dominance falling between 30 and 100. People who scored as ambidextrous (between −30 and 30) did not participate in this study. Participants who were not able to perform the task (see “Data analysis” section) were not included in the data analysis. In total, we analysed 29 left-handed participants (15 females; mean age = 24 ± 7 years; mean inventory score = −67.6 ± 21, range = −30 to −100) and 22 right-handed participants (12 females; mean age = 26 ± 8 years; mean inventory score = 83.2 ± 16, range = 50–100). The experiment was approved by the York University office of research ethics and follows the guidelines of the Declaration of Helsinki. All participants gave informed consent.
Apparatus
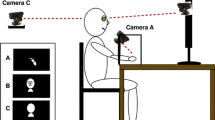
The apparatus was the same as described in previous studies (Hoover and Harris 2012, 2015a, b). Participants sat 50 cm away from an LCD screen (HP Fv583AA 20″ widescreen monitor; 1600 × 900 pixels; 5 ms refresh response time). They placed their hand on the tabletop shielded from direct view by a black cloth. A PlayStation Eye Camera (SCEI; resolution 640 × 480 pixels @ 30 Hz) was mounted on a chin rest and pointed down at their hand at an angle that captured a natural view of looking down at the hand on the table (Fig. 1).
Apparatus: participants sat on an adjustable chair at a table 50 cm from an LCD display, centred at eye level. They placed their hand on the table shielded from view by a black cloth. A PlayStation Eye Camera was mounted on the front of the chin rest and pointed down at their hand. The camera was angled to capture the view as seen from a ‘‘natural’’ egocentric perspective as if looking down at their hand. Participants used foot pedals to make responses (Hoover and Harris 2012)
Temporal and spatial manipulations of the movements
The video from the camera was fed into an iMAC (mid 2010), read by a MATLAB program (version R2009_b) and played through the LCD screen with either the minimal delay of the system (approximately 85 ms; see Hoover and Harris 2012) or with an imposed additional delay of between 33 and 231 ms. The real-time video delay was implemented with a Psychophysics Toolbox extension of MATLAB subroutine PsychVideoDelayLoop (Brainard 1997; Pelli 1997).
Participants were presented with their left or right hand viewed from four different viewing perspectives: (1) a ‘self’ perspective (unflipped: the natural, expected image of the hand), (2) vertical axis flip (flipped so that the hand appeared as the opposite hand), (3) horizontal axis flip (flipped so that the hand appeared upside down or as an other’s opposite hand) and (4) both axes flip (flipped so that the hand appeared as an other’s same hand). Visual perspectives 1 and 2 represented egocentric perspectives and visual perspectives 3 and 4 represented allocentric perspectives. Examples of these perspectives are shown as insets in Figs. 2 and 3.
Mean proportion correct in detecting a delay plotted as a function of the imposed delay for left-handers using their dominant (left) (a) and non-dominant (right) (b) hands. The sigmoidal curves plotted through data are for the ‘self’ (unflipped) perspective (solid dark lines and inverted triangles), with the video flipped around the vertical axis (solid light lines and filled circles), flipped around the horizontal axis (dashed dark lines and filled squares) and flipped around both axes (dashed light lines and filled triangles). The vertical dashed line represents the 75 % threshold for the ‘self’ (unflipped) perspective, and the horizontal line represents the 75 % criterion. Average 75 % thresholds are plotted as histograms for each perspective for the dominant (c) and non-dominant (d) hands. All error bars represent SEM
Mean proportion correct in detecting a delay plotted as a function of the imposed delay for right-handers. Format as for Fig. 2
Movement
Participants made a single flexion of approximately 2 cm with their index finger of either their left or right hand. A set of 15 practice trials were completed prior to the experiment to ensure that all participants performed the appropriate movement and did not touch the table with their finger or their other fingers so as to not introduce any additional tactile cues.
Procedure
Participant sat at the table and positioned their hand in view of the camera. We used a two-interval forced choice (2IFC) discrimination paradigm to assess the thresholds for detecting temporal asynchrony. For each trial, there were two 1 s periods separated by an inter-stimulus interval of 100 ms: a minimal-delay presentation of the movement and an imposed-delay presentation of the movement. Which period had which delay was randomly chosen by MATLAB. There were eight possible differences in delays between the two periods: 0, 33, 66, 99, 132, 165, 198 and 231 ms. Participants pressed down on foot pedals throughout the experiment and raised their left or right foot to indicate which period had the imposed delay (left = first period; right = second period).
The experiment used a counterbalanced block design where the movements of the left and right hands were tested in two blocks. The eight differences of delay were each presented eight times for the four viewing conditions in a random order for a total of 256 trials per hand. After 128 trials, in any given block participants were given a break. Each session of 128 trials lasts approximately 15 min. The entire experiment consisted of 512 trials and took approximately 1 h to complete.
Data analysis
The minimum delay that could be detected was determined by fitting a logistic function to the proportion of times participants correctly reported which period contained the delayed presentation of their movement as a function of the imposed visual delay using:
where x is the imposed delay, x 0 is the 75 % detection threshold and b is the reciprocal of the slope of the function. The goodness of fit (r 2) of this function to the data varied between 0.6 and 0.7.
We used repeated measures analysis of variances (ANOVAs) on the individual thresholds for the statistical analysis. We corrected for violations of sphericity by using the Greenhouse–Geisser correction. Alpha was set at p < 0.05 and multiple comparisons were made with the false discovery rate p value correction (Benjamini and Hochberg 1995). Of the 56 participants tested, five participants’ (three left- and two right-handers) data were removed from the analysis because they were not able to perform the task (i.e., did not reach 75 % correct for more than half of the conditions).
Results
Figures 2 and 3 show the proportion of times participants correctly identified the period with the delay plotted as a function of the delay (system plus imposed delays) averaged across the participants for both the left-hand (Fig. 2) and right-hand (Fig. 3) groups for the four viewing perspectives. Psychometric functions are plotted through the average data. Mean thresholds and reciprocals of the slope of the logistic fits through each participant’s data are given in Table 1.
Discriminating visual delays for left-handers
A 2 (hands) × 4 (perspectives) repeated measures ANOVA revealed no effect of hand (F (1,28) = 0.60, 0.45, ηp 2 = 0.02) or of perspective (F (3,84) = 0.25, p = 0.86, ηp 2 = 0.01). There were no differences amongst the visual perspectives for the left-handed participants for either their dominant left (Fig. 2a, c) or non-dominant right (Fig. 2b, d) hands. Analysis of the slopes (see “Methods”) showed no effect of hand (F (1,28) = 0.22, p = 0.64, ηp 2 = 0.01) or perspective (F (3,84) = 1.32, p = 0.27, ηp 2 = 0.05) for the left-handers.
Discriminating visual delays for right-handers
A 2 (hands) × 4 (perspectives) repeated measures ANOVA revealed a significant interaction between which hand was used (dominant right hand or non-dominant left hand) and perspective (F (3,63) = 3.67, p = 0.02, ηp 2 = 0.15). To break down the interaction, we looked at each hand separately. For the dominant hand (Fig. 3a, c), we found a significant effect of perspective (F (2.11,44.338) = 7.62, p = 0.001, ηp 2 = 0.266). Thresholds were lower for the ‘self’ (unflipped) perspective (M ± SE = 135 ± 7 ms) compared with the ‘other’ perspectives (vertical axis flip M ± SE = 150 ± 5 ms, M ± SE difference = 14 ± 6 ms, p = 0.03; horizontal axis flip M ± SE = 160 ± 7 ms, M ± SE difference = 25 ± 7 ms, p = 0.02; and both axes flip M ± SE = 153 ± 7 ms, M ± SE difference = 18 ± 5 ms, p = 0.02). For the non-dominant hand (Fig. 3b, d), there was no effect of perspective (F (3,63) = 2.09, p = 0.11, ηp 2 = 0.09). In other words, the ability to detect the delay was best when participants were moving their dominant hand and when the images were presented in the ‘self’ perspective. When participants were moving their non-dominant hand, there were no differences across the visual perspectives. These results are similar to those found in previous studies and confirm the signature self-advantage for the right hand viewed in a ‘self’ perspective (Hoover and Harris 2012, 2015a, b). Analysis of the slopes revealed no effect of hand (F (1,21) = 0.18, p = 0.68, ηp 2 = 0.01) or perspective (F (3,63) = 1.97, p = 0.13, ηp 2 = 0.086).
Difference in thresholds between left- and right-handers
There was a between-groups effect when comparing the left- and right-handers (F (1,49) = 5.26, p = 0.03, ηp 2 = 0.10). When looking at the dominant hands, right-hander thresholds were significantly lower than left-hander thresholds in the ‘self’ (t (49) = 2.817, p = 0.01; M ± SE difference = 28 ± 9 ms) and vertical axis flip (t (45.5) = 2.11, p = 0.06, p = 0.04; M ± SE difference = 21 ± 10 ms) perspectives. For the non-dominant hand, we found the same pattern: right-handers had significantly lower thresholds than left-handers for the self (t (49) = 2.12, p = 0.04; M ± SE difference = 19 ± 9 ms) and vertical axis flip (t (49) = 2.44, p = 0.04; M ± SE difference = 26 ± 11 ms) perspectives. In other words, right-handers were able to detect smaller differences in delays than the left-handers when the hands were presented in the upright configurations (egocentric perspectives). They did, however, show similar abilities to detect the delay for the inverted configurations (allocentric perspectives) using either hand. Figure 4 plots the difference between the minimum delay detected in the ‘self’ and ‘other’ perspectives as a function of the laterality index for left- and right-handers (negative and positive laterality scores respectively). There was no correlation of the size of the effect with laterality index for left r (29) = 0.211, p = 0.27, r 2 = 0.04 and right r (22) = −0.206, p = 0.36, r 2 = 0.04. The distribution of these values is shown as Gaussians to the right of the graph.
Self-advantage (difference between the minimal delay detected when viewing from the ‘self’ and ‘other’ perspective in ms) plotted as a function of laterality index where negative indicates left-handedness, for each participant in the study. Left-handers are shown as closed symbols and right-handers as open symbols. The distribution of values is shown as a Gaussian on the right side of the graph for left- (grey shaded curve) and right-handers (open curve)
Analysis of the slopes showed no significant between-group differences (F (1,49) = 2.42, p = 0.13, ηp 2 = 0.05).
Discussion
In this study, left- and right-handers compared movement of their left or right hand with a video image, viewed in either a ‘self’ or ‘other’ perspective, to estimate whether there was a delay in the visual feedback. The results for right-handers replicated the pattern shown in our previous studies which reported lower thresholds while viewing movements of the dominant right hand (but not the left hand) when viewing in the ‘self’ (unflipped) perspective: the signature self-advantage (Hoover and Harris 2012, 2015a, b). Left-handers showed no such difference in detection thresholds across hands and perspectives and showed no self-advantage for detecting temporal asynchrony for their dominant hand in the ‘self’ (unflipped) perspective. Instead left-handers’ thresholds for both hands in all the conditions tested were comparable to those of right-handers viewing either hand in the inverted ‘other’ perspectives. There were no differences in the slopes of the functions across hands and perspectives for either left- or right-handers, suggesting that the task was equally difficult in all conditions for both groups.
Self-advantage as an indicator of self
Right-handers in the present study detected a visual delay that was about 19 ms shorter in the ‘self’ perspective (dark line inverted triangles in Fig. 3a) than the minimum visual delay detected in the inverted perspectives (light solid and dark and light dashed lines in Fig. 3a). These results confirm previous estimates of the self-advantage (21 ms, Hoover and Harris 2012, 2015a, b and 20 ms, Zopf et al. 2015). We interpret the self-advantage as resulting from the view of the hand in the natural perspective matching the internal representation of the hand, thus being interpreted more directly as one’s own hand. The upright configurations correspond to egocentric or ‘plausible self’ perspectives because the hand is seen in postures could be adopted naturally. The inverted configurations, however, correspond to postures that are impossible to achieve with one’s own hands and are therefore regarded as egocentric or ‘other’ perspectives.
A similar self-advantage has been reported for other behavioural measures such as in recognition of self in static images of body parts (Saxe et al. 2006; Conson et al. 2010), judging laterality of hands (Parsons 1994; Dyde et al. 2011) and enhanced body ownership illusions (e.g., the rubber hand illusion, Holmes and Spence 2007) when the respective stimuli are presented in a ‘plausible self’ posture. Left-handers mirror right-handers whereby performance on some tasks is better with their dominant left hand compared to their non-dominant right hand. For example, during an explicit self-recognition task using images of hands presented in ‘self’ (upright) and ‘other’ (inverted) perspectives Conson et al. (2010) found fastest reaction times for left-handers recognizing their dominant hand viewed in the ‘self’ perspective. When making speeded left–right hand judgments while imagining themselves as schematic human figures, left-handers responded more quickly when a target was located on the left side of the figure compared to when it was on the right side (Gardner and Potts 2010). Their results suggest that hand dominance, and subsequently the motor experiences that stem from this dominance, affects body/self-perception and predicts a self-advantage for the left hand in left-handed participants. The lack of a self-advantage in left-handers that we found using implicit rather than explicit measure of body ownership thus suggests that left-handers do not differentiate between ‘self’ and ‘other’ as clearly as right-handers.
No self-advantage for left-handers
Left-handers tend to display a weaker (and less consistent) hand preference than right-handers (Annett 1985; McManus 1995, 2003; Bryden et al. 2000). Consistent with this, our mean left-hand participants’ absolute laterality scores were about 15 points lower than those of the right-handers. This may contribute to why right-handers’ thresholds differed with the hand performing the task, whereas left-handers’ thresholds did not (c.f., Marzoli et al. 2013): they were not left-handed enough! However, the extent of left- or right-handedness does not seem to be a factor (Fig. 4). Despite their scores on a handedness inventory, left-handers tend to be more ambidextrous than right-handers (Annett 1967; Gonzales et al. 2007; Linkenauger et al. 2009). For example, varying the orientation of a tool has less effect on whether left-handers think they can reach it than it does on right-handers. Right-handers score consistently worse in this test thinking incorrectly that tools presented in an unusual orientation are out of reach (Linkenauger et al. 2009). If left-handers extend this ability to their assessment of movements of their hands and regard either as representing themselves equally, then this might contribute to them having equal delay thresholds for each hand; that is, left-hander’s tendency towards being ambidextrous might result in them having the equivalent of two right hands—both of which can be regarded as agents of the self. The fact that the minimal delay that left-handers could detect under all viewing conditions was equivalent to thresholds in the ‘other’ perspective for right-handers argues against this, however.
Marzoli et al. (2015) have shown that both left- and right-handers tend to ‘perceive’ an action being performed with the right hand when confronted with ambiguous silhouettes of people performing actions with their hand or arms. These results suggest a greater attentional bias for all people towards the right hand and may reflect the experience of left-handers in a predominantly right-handed world. There are instances where left-handers identify as left-handed, but performance on dexterity tasks shows that they are actually right-hand dominant (Chatagny et al. 2013). Although our participants were identified as left-handers using the Edinburgh Handedness Inventory (Oldfield 1971), it may be that they retained some right-hand dominance. No such self-advantage for the right hand in left-handers was observed which argues against this possibility contributing to the results of our task. However, further research investigating how lowered hand dominance contributes to the creation of the body schema and subsequently the sense of body ownership for left-handers may reveal some important differences between left- and right-handers.
A more likely correlate with the lack of self-advantage in left-handers is the lack of asymmetry of body space shown in left-handers in general (Gentilucci et al. 1998; Hach and Schütz-Bosbach 2010; Takeda et al. 2010), probably related to lesser lateralization in body and action-specific regions of the brain (such as parietal and extrastriate areas; Willems et al. 2009, 2010a, b; Willems and Hagoort 2009; Vingerhoets et al. 2012). Hach and Schütz-Bosbach (2010) found that a difference in handedness led to differences in how people represent their body space. When participants were asked to point to parts of their body or to remove circular patches from their body while blindfolded, right-handers had a tendency to bias the locations of body parts and the circular patches more to the right than they actually were, whereas left-handers showed no such spatial bias. A lack of asymmetry was also found a mental rotation task where participants identified hand images presented in varying orientations. Right-handers more quickly identified right hands compared to left hands, whereas left-handers showed no difference in performance across hands (Takeda et al. 2010). Similarly, when presented with left and right hands gripping objects in congruent and incongruent grips, right-handers more easily identified right hands, whereas left-handers again showed no bias in hand identification (Gentilucci et al. 1998). This same symmetrical performance was evident when participants were asked to imagine others’ actions: right-handers imagined a greater proportion of movements being performed with their right hand, whereas left-handers had no greater proportion of imagined actions being performed with their left hand than with their right (Marzoli et al. 2013). These observations suggest that left-handers do not identify with either hand, do not have a heightened sense of ownership of their dominant hand, and for them neither hand acts as a specialized agent of self.
Conclusion
We suggest that the focus of attention and self-expression through the right hand of right-handers is associated with the way they interact with the world. The absence of a self-advantage in left-hander suggests that they do not have such a precise internal self-representation of a hand movement. We speculate that this may be associated with a lowered sense of body ownership in general and consequently a less clear representation of self. This may in turn be related to why left-handers seem to be more vulnerable to cognitive disorders associated with disorders in the representation of self such as schizophrenia (Lishman and McMeekan 1976; Webb et al. 2013).
References
Annett M (1967) The binomial distribution of right, mixed and left handedness. Q J Exp Psychol 19:327–333. doi:10.1080/14640746708400109
Annett M (1985) Left, right, hand and brain: right shift theory. Lawrence Erlbaum Associates, Hillsdale
Annett J, Annett M, Hudson PT, Turner A (1979) The control of movement in the preferred and non-preferred hands. Q J Exp Psychol 31:641–652. doi:10.1080/14640747908400755
Balslev D, Cole J, Miall RC (2007) Proprioception contributes to the sense of agency during visual observation of hand movements: evidence from temporal judgments of action. J Cogn Neurosci 19:1535–1541. doi:10.1126/science.7569931
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57:289–300
Brainard DH (1997) The psychophysics toolbox. Spat Vis 10:433–436
Bremner AJ, Cowley D (2013) Developmental origins of the hand in the mind, and the role of the hand in the development of the mind. In: Radman Z (ed) Hand, an organ mind. MIT Press, Cambridge, pp 27–55
Bryden PJ, Pryde KM, Roy EA (2000) A performance measure of the degree of hand preference. Brain Cogn 44:402–414. doi:10.1006/brcg.1999.1201
Casasanto D (2009) Embodiment of abstract concepts: good and bad in right- and left-handers. J Exp Psychol Gen 138:351–367. doi:10.1037/a0015854
Chatagny P, Badoud S, Kaeser M et al (2013) Distinction between hand dominance and hand preference in primates: a behavioral investigation of manual dexterity in nonhuman primates (macaques) and human subjects. Brain Behav 3:575–595. doi:10.1002/brb3.160
Conson M, Aromino AR, Trojano L (2010) Whose hand is this? Handedness and visual perspective modulate self/other discrimination. Exp Brain Res 206:449–453. doi:10.1007/s00221-010-2418-9
Dyde RT, MacKenzie K, Harris LR (2011) How well do you know the back of your hand? Reaction time to identify a rotated hand silhouette depends on whether it is interpreted as a palm or back view. J Vis 11:868
Farthing JP, Chilibeck PD, Binsted G (2005) Cross-education of arm muscular strength is unidirectional in right-handed individuals. Med Sci Sports Exerc 37:1594–1600. doi:10.1249/01.mss.0000177588.74448.75
Franck N, Farrer C, Georgieff N et al (2001) Defective recognition of one’s own actions in patients with schizophrenia. Am J Psychiatry 158:454–459
Gallagher I (2000) Philosophical conceptions of the self: implications for cognitive science. Trends Cogn Sci 4:14–21
Gardner MR, Potts R (2010) Hand dominance influences the processing of observed bodies. Brain Cogn 73(1):35–40
Gentilucci M, Daprati E, Gangitano M (1998) Right-handers and left-handers have different representations of their own hand. Cogn Brain Res 6:185–192. doi:10.1016/S0926-6410(97)00034-7
Goble DJ, Brown SH (2008) The biological and behavioral basis of upper limb asymmetries in sensorimotor performance. Neurosci Biobehav Rev 32:598–610. doi:10.1016/j.neubiorev.2007.10.006
Gonzales C, Whitwell R, Morrissey B, Ganel T, Goodale M (2007) Left handedness does not extend to visually guided precision grasping. Exp Brain Res 182:275–279
Haans A, Ijsselsteijn WA, de Kort YA (2008) The effect of similarities in skin texture and hand shape on perceived ownership of a fake limb. Body Image 5:389–394
Hach S, Schütz-Bosbach S (2010) Sinistrals’ upper hand: evidence for handedness differences in the representation of body space. Brain Cogn 72:408–418
Heuer H (2007) Control of the dominant and nondominant hand: exploitation and taming of nonmuscular forces. Exp Brain Res 178:363–373. doi:10.1007/s00221-006-0747-5
Holmes NP, Spence C (2007) Dissociating body image and body schema with rubber hands. Behav Brain Sci 30:211. doi:10.1017/S0140525X07001501
Hoover AEN, Harris LR (2012) Detecting delay in visual feedback of an action as a monitor of self recognition. Exp Brain Res 222:389–397. doi:10.1007/s00221-012-3224-3
Hoover AEN, Harris LR (2015a) The role of the viewpoint on body ownership. Exp Brain Res 233:1053–1060. doi:10.1007/s00221-014-4181-9
Hoover AEN, Harris LR (2015b) Disrupting vestibular activity disrupts body ownership. Multisens Res 28:581–590. doi:10.1163/22134808-00002472
Linkenauger SA, Witt JK, Stefanucci JK et al (2009) The effects of handedness and reachability on perceived distance. J Exp Psychol Hum Percept Perform 35:1649–1660. doi:10.1037/a0016875
Lishman WA, McMeekan ERL (1976) Hand preference patterns in psychiatric patients. Br J Psychiatry 129:158–166. doi:10.1192/bjp.129.2.158
Maeda T, Takahata K, Muramatsu T et al (2013) Reduced sense of agency in chronic schizophrenia with predominant negative symptoms. Psychiatry Res 209:386–392. doi:10.1016/j.psychres.2013.04.017
Marzoli D, Menditto S, Lucafò C, Tommasi L (2013) Imagining others’ handedness: visual and motor processes in the attribution of the dominant hand to an imagined agent. Exp Brain Res 229:37–46. doi:10.1007/s00221-013-3587-0
Marzoli D, Lucafò C, Pagliara A, Cappuccio R, Brancucci A, Tommasi L (2015) Both right- and left-handers show a bias to attend others’ right arm. Exp Brain Res 233:415–424. doi:10.1007/s00221-014-4124-5
McManus IC (1995) Familial sinistrality: the utility of calculating exact genotype probabilities for individuals. Cortex 31:3–24
McManus C (2003) Right hand, left hand. Weidenfeld & Nicolson, London
Merleau-Ponty M (1945) Phenomenology of Perception. Gallimard, Paris
Ocklenburg S, Ruther N, Peterburs J et al (2011) Laterality in the rubber hand illusion. Laterality 16:174–187
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Orr KGD, Cannon M, Gilvarry CM et al (1999) Schizophrenic patients and their first-degree relatives show an excess of mixed-handedness. Schizophr Res 39:167–176. doi:10.1016/S0920-9964(99)00071-7
Parsons LM (1994) Temporal and kinematic properties of motor behavior reflected in mentally simulated action. J Exp Psychol Hum Percept Perform 20:709–730. doi:10.1037/0096-1523.20.4.709
Pelli DG (1997) The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat Vis 10:437–442
Piaget J (1954) The construction of reality in the child. Routledge & Kegan Paul, London
Radman Z (2013) The hand, an organ of the mind. The MIT Press, Cambridge
Rochat P (1989) Object manipulation and exploration in 2- to 5-month-old infants. Dev Psychol 25:871–884. doi:10.1037/0012-1649.25.6.871
Rochat P (1998) Self-perception and action in infancy. Exp Brain Res 123:102–109
Rochat P (2003) Five levels of self-awareness as they unfold early in life. Conscious Cogn 12:717–731
Rochat P, Striano T (2000) Perceived self in infancy. Infant Behav Dev 23:513–530. doi:10.1016/S0163-6383(01)00055-8
Sang FYP, Jáuregui-Renaud K, Green DA, Bronstein AM, Gresty MA (2006) Depersonalisation/derealisation symptoms in vestibular disease. J Neurol Neurosurg Psychiatry 77:760–766. doi:10.1136/jnnp.2005.075473
Saxe R, Jamal N, Powell L (2006) My body or yours? The effect of visual perspective on cortical body representations. Cereb Cortex 16:178–182. doi:10.1093/cercor/bhi095
Takeda K, Shimoda N, Sato Y et al (2010) Reaction time differences between left- and right-handers during mental rotation of hand pictures. Laterality 15:415–425. doi:10.1080/13576500902938105
Thakkar KN, Nichols HS, McIntosh LG, Park S (2011) Disturbances in body ownership in schizophrenia: evidence from the rubber hand illusion and case study of a spontaneous out-of-body experience. PLoS ONE 6(10):e27089. doi:10.1371/journal.pone.0027089
van den Bos E, Jeannerod M (2002) Sense of body and sense of action both contribute to self-recognition. Cognition 85:177–187
Vingerhoets G, Acke F, Alderweireldt AS, Nys J, Vandemaele P, Achten E (2012) Cerebral lateralization of praxis in right- and left-handedness: same pattern, different strength. Hum Brain Mapp 33:763–777. doi:10.1002/hbm.21247
Webb JR, Schroeder MI, Chee C et al (2013) Left-handedness among a community sample of psychiatric outpatients suffering from mood and psychotic disorders. SAGE Open 3:2158244013503166. doi:10.1177/2158244013503166
Willems RM, Hagoort P (2009) Hand preference influences neural correlates of action observation. Brain Res 1269:90–104. doi:10.1016/j.brainres.2009.02.057
Willems RM, Toni I, Hagoort P, Casasanto D (2009) Body-specific motor imagery of hand actions: neural evidence from right- and left-handers. Front Hum Neurosci 3:39. doi:10.3389/neuro.09.039.2009
Willems RM, Hagoort P, Casasanto D (2010a) Body-specific representations of action verbs: neural evidence from right- and left-handers. Psychol Sci 21:67–74. doi:10.1177/0956797609354072
Willems RM, Peelen MV, Hagoort P (2010b) Cerebral lateralization of face-selective and body-selective visual areas depends on handedness. Cereb Cortex 20:1719–1725
Zopf R, Friedman J, Williams MA (2015) The plausibility of visual information for hand ownership modulates multisensory synchrony perception. Exp Brain Res 233:2311–2321 doi:10.1007/s00221-015-4300-2
Acknowledgments
This research was supported by a Discovery Grant 46271-2015 from the Natural Sciences and Engineering Research Council of Canada to LRH. AENH was supported by a York University Provost Dissertation scholarship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoover, A.E.N., Elzein, Y. & Harris, L.R. Left-handers show no self-advantage in detecting a delay in visual feedback concerning an active movement. Exp Brain Res 234, 1915–1923 (2016). https://doi.org/10.1007/s00221-016-4595-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4595-7