Abstract
Bread can be a major contributor to sodium intake, but sodium chloride reduction poses difficulties since it influences the functional properties of dough and flavor of bread. This study evaluated dough and bread properties in reduced-sodium systems containing fermented or unfermented macroalgae Saccharina latissima or Alaria esculenta. Recipes contained equal amounts of sodium (4 mg Na+/g flour), where sodium chloride contributed 3 or 3.5 mg Na+/g flour and 8.9–33.3 mg macroalgae/g flour contributed the remaining 0.5 or 1 mg Na+/g flour. A full-salt and three salt-reduced controls (6, 4, 3.5, and 3 mg Na+/g flour) were used for comparison. Empirical dough rheology, stickiness, ratios of polymeric to monomeric proteins, and bread characteristics (specific volume, crumb structure, and firmness) were measured. A trained sensory panel conducted a descriptive sensory analysis. Macroalgae addition increased water absorption and decreased dough development time, dough stability, the polymeric to monomeric protein ratio, and specific volume in a dose-dependent manner. Macroalgae addition increased the perception of saltiness, but also algae flavor and odor. Bread with fermented S. latissima received lower scores for certain undesirable sensory attributes than other bread with algae.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sodium intakes that exceed dietary recommendations are linked to increased risk for hypertension and cardiovascular diseases [1]. Bread is a major contributor to sodium intake in countries such as Norway where the average daily intake is 184 g per person [2]. Approaches to reduce sodium contents in bread include gradual reduction, sodium replacers, taste contrast, and taste enhancers [1, 3]. However, sodium chloride crucially impacts dough and bread properties, making its reduction challenging [1, 4]. Below 9 mg/g bread, dough stickiness can increase, and viscoelastic properties and loaf volumes be reduced [1, 4,5,6], causing lower product quality, operational problems, and decreased consumer acceptability [4, 5].
A novel approach to achieve sodium reduction is the incorporation of macroalgae [3]. Marine macroalgae are generally high in minerals as well as free glutamate, which evokes umami taste, thus their use may compensate for reduced-sodium chloride contents [7]. In addition, they may provide dietary fiber, proteins, trace elements and certain vitamins [8]. However, the acceptability of products with macroalgae is limited in the Western world, which is related to the ‘marine’ flavors they impart [3, 9]. In Norway, the bread category with the highest sales is breads containing at least 50% wholegrain flour [10]. The effects of salt reduction may be exacerbated when using wholemeal flour, since the presence of bran reduces gluten network formation and thereby bread volumes [11]. However, such breads also have a darker color and more intense flavor than white breads [12], which may make addition of non-wheat ingredients less noticeable.
In Norway, the brown macroalgae species Saccharina latissima (sugar kelp) and Alaria esculenta (winged kelp) are cultivated on a commercial scale [8] due to their rapid growth, high biomass yield and potential uses in food and non-food applications [13]. However, high iodine contents [8] and the adverse effects of excessive iodine intake [14] have put limitations on the amounts of unprocessed brown macroalgae that can be used in foods [8, 15]. While there is no set threshold for brown macroalgae consumption, the Norwegian Health Authority has advised consumers to be cautious of iodine in algae [16]. On the EU level, it has been recommended to monitor seaweeds for contents of heavy metals and iodine [17].
As a result of their high water content, macroalgae shelf life is limited [18] unless they are processed, e.g., using drying, lactic acid bacteria fermentation or freezing [19]. Such post-harvest treatments are crucial for establishing macroalgae supply chains. In addition, S. latissima and A. esculenta are commonly rinsed in warm tap- or saltwater, as this lowers their iodine content [20, 21]. Subsequent fermentation can further reduce the iodine content and fermented macroalgae have recently become commercially available [22]. However, strains, fermentation conditions and food use of fermented macroalgae have scarcely been reported.
The aim of this study was to investigate the use of fermented and unfermented S. latissima and A. esculenta as partial sodium chloride replacers in bread containing wholemeal wheat. Effects on dough properties were evaluated by farinograph, extensograph, dough stickiness, and protein size distribution measurements. Breads were characterized instrumentally (C-cell analysis, crumb firmness, volume) and sensory properties were assessed by a trained panel.
Materials and methods
Materials
Dough consisted of 66.6% wholewheat flour (Lantmännen Cerealia AS, Oslo, Norway) and 33.3% of a strong, refined wheat flour containing about 30 ppm of ascorbic acid (Lantmännen Cerealia). Amounts of both flours were adjusted based on flour moisture (0.139 g/g for refined and 0.127 g/g for wholemeal flour) measured with a moisture balance (Satorius Thermo Control, Sartorius, Göttingen, Germany). Flour was substituted with dry powder from fermented or non-fermented S. latissima and A. esculenta (Orkla Ocean AS, Oslo, Norway), as described below. Unfermented algae were harvested at the cultivation site of Seaweed Solutions (Frøya, Norway) and warm water-rinsed and frozen prior to drying. Fermented algae were harvested at the cultivation site of Arctic Seaweed (Misje, Norway) and fermented by the supplier using lactic acid bacteria. All batches were dried under the same conditions and milled into a fine powder with a particle size < 200 µm (Orkla Foods Norway, Arna, Norway).
Experimental plan
All breads were prepared with a 2:1 ratio of wholemeal to refined wheat flour. Samples with added macroalgae contained equal amounts of sodium (4 mg Na+/g flour) where sodium chloride contributed 3.5 (C3A) or 3 (C3B) mg Na+/g flour and 3.3–8.9 mg macroalgae/g flour contributed the remaining 0.5 or 1 mg Na+/g flour (see Table 1 for an overview). Thus, algae amounts were based on the sodium content of the macroalgae (Supplementary Table 1) which were higher in unfermented compared to fermented powders, and higher in S. latissima than A. esculenta. A full-salt (6 mg Na+/g flour (C1)) and three salt-reduced controls (4 (C2), 3.5 (C3A), and 3 (C3B) mg Na+/g flour) were used for comparison. The amount of sodium chloride added to the full-salt control C1 was equivalent to commonly used values by bread manufacturers in Norway [23].
Farinograph analysis
A Farinograph-TS (Brabender, Duisburg, Germany) was used to analyze water absorption (WA), dough development time (DDT) and dough stability (DS) of flour mixtures without sodium chloride. Two independent analyses were carried out with a 300 g mixing bowl at 30 ℃, 63 rpm for 20 min, according to ISO 5530-1 standard.
Extensograph analysis
Viscoelastic properties of dough were measured according to AACC method 54.10, with some modifications. Doughs were prepared in a Farinograph-TS (Brabender) operated with 300 g mixing bowl at 126 rpm. Bowl temperature was adjusted to 22 ℃ to avoid dough temperature exceeding 27 ℃ at the end of mixing. Doughs were prepared with sodium chloride addition according to Table 1. Water addition was according to WA measured for Farinograph experiments minus 1.5% to reduce dough stickiness. Mixing was continued to a total energy input of 12 Wh/kg. Two independent replicates of each sample were prepared in the farinograph. The dough was further divided into two 150 ± 0.2 g pieces. Samples without macroalgae were shaped in the extensograph’s balling and rolling units while samples with macroalgae were only balled in the extensograph unit, but then hand-shaped into a cylinder (5 times back and forth rolling) since they were too sticky to be rolled in the instrument. The dough was rested for 45 and 90 min at 30 ℃ in high humidity before it was stretched by Extensograph-E (Brabender) measuring resistance to extension (BU) and extensibility (mm).
Dough Stickiness
Dough stickiness was analyzed as described by Huang and Hoseney [6], except that the contact time between the probe and sample was changed from 0.1 to 1 s to increase reproducibility. The SMS/Chen–Hoseney Dough Stickiness Rig and a 25 mm perspex cylinder probe were used to measure stickiness (g) with a TA.XTplusC Texture Analyser (Stable Micro Systems, Surrey, UK) equipped with a 500 g load cell. Dough was prepared in a farinograph with a 50 g mixing bowl, mixed as described for the extensograph but with water addition according to Farinograph WA (no subtractions as for extensograph). Dough prepared in the farinograph was first transferred to a 120 mL specimen container then to the extrusion cell of the stickiness rig, and the cell lid was screwed on. The remaining dough was placed in the cup in a warming cabinet (30 ℃) for 45 min. The dough was analyzed at 0 and after 45 min. Six replicates were performed on each independently prepared dough sample. About 1 mm of dough was extruded before each measurement, rested for 30 ± 5 s with a small lid taped with a piece of moistened cotton. A blade was used to wipe extruded dough off the surface before the next measurement. Results were processed using Exponent Connect.
Protein size distribution
Protein size distribution was analyzed by size exclusion-high-performance liquid chromatography (SE-HPLC) according to Singh, Donovan and MacRitchie [24]. A small piece of dough prepared for the extension test was incubated at 30 ℃. After 90 min, samples were frozen, freeze-dried and manually crushed to a fine powder using a mortar. Proteins from 15 mg ground samples were extracted sequentially. Sodium dodecyl sulfate (SDS)-extractable proteins were extracted with 1.5 mL 0.1 M phosphate buffer (pH 6.9) containing 1% SDS by shaking at 1500 rpm for 30 min at 75 ℃. Samples were centrifuged (13,000 rpm, 15 min) and the supernatant recovered. Remaining SDS-unextractable proteins in the pellet were extracted by adding the same phosphate buffer with sonication using Q55 Sonicator (Qsonica Sonicators, Connecticut, USA) at amplitude 100 for 30 s, and centrifuging (13,000 rpm, for 15 min). SDS-extractable and SDS-unextractable fractions were filtered through a Millex-HV PVDF 0.45 µm filter (Merck Millipore, Burlington, Massachusetts, USA). Protein extract (10 µL) was separated on a Bio-Step™ 5µm SEC-S4000 column (Phenomenex, California, USA) connected to a Dionex Ultimate 3000 (UHPLC+) (Thermo Fisher Scientific, California, USA). The eluent was 30% acetonitrile with 0.05% trifluoracetic acid (flow rate of 0.4 mL/min) and proteins monitored by UV absorption at 214 nm. The SDS-unextractable fraction resulted in one main peak consisting of large polymers, denoted F1*. The chromatogram of the SDS-extractable fraction was divided into five peaks (F1–F5). F1–F2 and F3–F4 consist of polymeric and monomeric proteins, respectively. F5 is mainly albumins and globulins. The proportion of SDS-unextractable polymeric proteins in total polymeric proteins (%UPP) was calculated as [F1*/(F1* + F1) × 100] and the ratio of polymer to monomer (Pol:Mon) as [(F1* + F1 + F2)/(F3 + F4)].
Dough pH
Three g of dough and 5 mL distilled water were first stirred by Multi OSU-20 and then by hand until dispersed. The dough suspensions were prepared from two replicates of each sample. The pH was measured with a PHM210 MeterLab pH meter (Radiometer analytic, Lyon, France).
Baking
A small-scale straight dough baking experiment was carried out with 10 mg dry yeast and 15 mg rapeseed oil/g flour. Water addition was according to farinograph WA. Doughs were prepared in duplicate and randomized order and mixed in a DoughLab (Perten, Stockholm, Sweden) using a 300 g mixing bowl at 126 rpm to a total energy input of 12 Wh/kg. Bowl temperature was set to 23 °C, to achieve a final dough temperature of 27 + 0.5 °C. Dough was hand-shaped and rested at 32 °C and RH 75% for 30 min after which it was divided into three pieces of 150 g and shaped by two rounds in a Dough rounder R10 (FriulCo, Maniago, Italy), then proofed at 32 °C and RH 75% for 45 min. Breads were baked for 20 min at 210 °C (oven was preheated to 240 °C) in a rotating oven (Revent type 626 GEL IAC, Revent international, Upplands Väsby, Sweden) with 10 s of steam injection. After baking, loaves were rested for a minimum of 60 min before analyses. Volume was measured using a TexVol BVM-6630 Series Analyser (Perten, Stockholm, Sweden). Further, a 2.5 cm slice was cut from the middle of each bread with a spacer and knife. A picture of each bread was taken with C-cell Color (Caliber, Warrington, UK) and crumb firmness was measured using the same Texture Analyser as for stickiness, equipped with a 5 kg load cell according to AACC Method 74–09.
Sensory analysis
The breads for descriptive analysis were prepared by upscaling the recipes from the small-scale baking with a factor of 10 to a flour weight of 3000 g. Doughs were prepared in randomized order in one replicate per sample using a Diosna SP 12 spiral mixer (Diosna, Osnabrück, Germany) for 240 s at 30 HZ followed by mixing at 40 HZ to a final dough temperature of 27 °C. Doughs were rested, proved, and baked as described for small-scale baking with a dough piece weight of 550 g and a baking time of 30 min. After cooling the breads were stored in sealed plastic bags over night. The samples for sensory analysis were C1, C2, C3A and sodium chloride reduction level C3A with all four macroalgae, see Table 1.
The sensory laboratory has been designed according to guidelines in ISO 8589:2007(E) with separate booths and electronic registration of data (Eye Question, v. 3.8.6, Logic 8, Netherlands), standardized light and a separate ventilation system.
A panel selected and trained according to ISO 8586:2012(E) performed a generic quantitative descriptive analysis based on QDA® as described by Lawless and Heymann [25]. Nofima’s highly trained and stable panel consists of 10 assessors solely hired as tasters, with an average of 15 years of experience using descriptive analysis on various kinds of foods and beverages, including bread. Panel performance is checked for every project, based on discrimination, repeatability, and panel agreement.
The panel was calibrated and trained with samples C3A and C3A + FAE, selected in informal tasting by the researchers and panel leader for being extreme examples stretching the sensory space. The descriptive terminology of the products was created in a pre-trial session, generating 23 attributes (listed in Table 2 and defined in Supplementary Table 2). Assessors evaluated the samples in duplicate, rating attribute intensity on 15-cm unstructured scales. Samples were served in plastic containers coded with 3-digit random numbers and in a sequential monadic manner following a balanced presentation order. The software used for data analysis was EyeQuestion (Logic8 BV, Utrecht, Holland) and EyeOpenR (Logic8 BV, Utrecht, Holland).
Statistical analysis
Results were analyzed using one-way analysis of variance (ANOVA), followed by Tukey’s Honestly Significant Difference test. General linear models were used to study the effect of macroalgae species, fermentation of macroalgae and sodium chloride reduction level, and interactions between the factors. Statistical analysis was carried out using Minitab Statistical Software, version 21.1 (Minitab, Inc. State College, PA, USA) or R. A significance level of α < 0.05 was used.
Results and discussion
Dough properties
Water absorption and mixing properties
Significantly higher DS and lower WA were found in control dough without macroalgae (C1) compared to all other samples (Table 3). A decrease in DDT and DS upon algae addition was also observed previously with Laminaria ochroleuca [26]. WA increased in a dose-dependent manner with macroalgae incorporation, which agrees with other studies [27,28,29]. Correspondingly, the dough with the highest amounts of algae, C3B + FAE, had significantly higher WA, as well as significantly lower DS, than all other samples. Comparing dough with the same algae type (i.e., dough samples of the C3A vs C3B series containing the same algae), higher algae levels resulted in significantly higher WA but lower DS. The increase in WA presumably involves competition for water between algae hydrocolloids and dough constituents [27, 29], as has been reported for wheat dough containing alginate [30], the main polysaccharide in brown macroalgae. The general linear model indicated that samples with S. latissima had significantly higher DDT (P < 0.01) and DS (P < 0.001) than dough with A. esculenta. However, if this is due to different properties of the two algae species or simply an effect of the higher addition levels of A. esculenta compared to S. latissima is unclear. C3A-doughs with algae had significantly higher DS (P < 0.001) and DDT (P < 0.05) than C3B-doughs. Fermented macroalgae led to significantly lower DDT (P < 0.05) and DS (P < 0.001) than unfermented macroalgae, but addition levels were slightly higher for fermented macroalgae (Table 1). In contrast to our study, an increase in DDT and DS was observed with increased amounts (1–10%) of the red macroalgae Kappaphycus alvarezii and Eucheuma denticulatum [27, 28]. These discrepancies may be due to compositional differences between macroalgae, such as the polysaccharide profile. Kappaphycus alvarezzi and Eucheuma denticulatum contain carrageenans as main polysaccharide. The addition of alginate has been shown to exert different effects on dough than the addition of carrageenan [30]. However, alginates require acid extraction to enfold their water-binding and gelling properties [31]. Further studies are needed to clarify the effect of individual constituents of brown algae in salt-reduced dough.
Viscoelastic properties
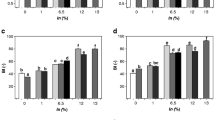
Compared to other alga types, brown macroalgae are generally lower in protein but high in non-starch polysaccharides [26, 32] which may affect elongational properties of dough systems [32]. Extensibility in control doughs decreased with salt reduction in a dose-dependent manner (C1 vs C2, C3A and C3B) after 45 min dough rest (Fig. 1a). Addition of macroalgae to salt-reduced control doughs (C3A and C3B) resulted in a further reduction of extensibility. This reduction was more pronounced for doughs containing A. esculenta compared to doughs with S. latissima, which may be due to the higher addition levels of A. esculenta (Table 1). However, C3B-FSL doughs (18.2 mg algae/g flour) had a significantly higher extensibility than C3A-FAE doughs (17.8 mg algae/g flour), which indicates an independent role of algae type. Differences in extensibility were however not apparent after 90 min rest (Fig. 1a).
Physical and chemical properties of sodium-reduced and macroalgae-enriched dough. a Extensograph extensibility and b maximum resistance to extension after 45 (Open square) or 90 (grey square) min of resting; c Stickiness after 0 (Open square) or 45 (grey square) min of resting; d pH, e the proportion of unextractable polymeric proteins in total polymeric proteins f the ratio of polymeric (Pol) to monomeric (Mon) proteins. Bars and error bars represent the average and half the range of two independently prepared doughs. Different letters denote significant differences among means (P < 0.05). AE, Alaria esculenta; SL Saccharina latissima; F, fermented. For sample name abbreviations, see Table 1
For maximum resistance to extension (Rmax), larger differences were seen at 90 than 45 min rest (Fig. 1b). Controls with reduced-sodium chloride had a significantly lower Rmax than the full-salt control (C1) at 90 min rest (P < 0.01). Most algae-containing doughs did not differ from any of the controls; however, values were closest to the full-salt control. Thus, addition of macroalgae somewhat counteracted the decrease in Rmax with sodium chloride reduction. There were no significant differences in Rmax among macroalgae-containing samples.
Viscoelastic properties of dough systems are strong determinants of bread properties (Table 4). Our extensograph results are in the range of previous studies on algae addition to bread dough with regards to extensibility, whereas Rmax was lower in our study than previously observed [28, 29]. This is most likely related to the use of 66% wholemeal wheat instead of 100% refined wheat flour in the current study and differences in algae addition levels. Systems with weaker gluten networks are more impacted by sodium chloride reduction than strong dough made with refined flour [33]. This could indicate that dough with wholemeal as used in our study may also be more affected as the network is already diluted and hindered in its formation by the bran [11]. Indeed, a sodium chloride reduction from 15 to 7.5 mg/g flour significantly weakened the dough structure in the current study, while the effects of sodium chloride reduction in refined flour systems often only manifest when difference in salt levels are relatively large, for example 0 vs 12 [34] or 15 mg sodium chloride/g flour [35].
Stickiness
Significant differences in stickiness were only present before (i.e., at 0 min) but not after resting (Fig. 1c). Significant differences were only observed between C3A + FAE and C3B + AE and C1 and C2 (P < 0.01), with C3A + FAE and C3B + AE being less sticky. These instrumental results were however not in line with subjective observations while handling the dough. In fact, macroalgae-containing dough could not be prepared in the extensograph unit. Qazi et al. [29] reported enhanced stickiness due to microalgae addition. However, to the best of our knowledge, the effects of macroalgae addition on dough stickiness have scarcely been evaluated quantitatively.
Stickiness is a complex phenomenon and poorly understood [36]. It is impacted by protein composition, hydration level, enzymatic activities, water-soluble carbohydrates, and processing [36, 37]. Stickiness has been associated with increased water mobility within a dough matrix [36] and predominantly occurs in systems with low cohesive forces (as there are few interactions between dough constituents), which is characteristic for weak gluten networks. While there were some significant differences in stickiness, they were of low magnitude and all dough samples would be categorized as non-sticky following the classification from Chen and Hoseney [38]. This may however have been influenced by sample preparation. It was difficult to create an even dough surface, presumably because the presence of whole wheat flour (and macroalgae) resulted in incomplete contact between the probe and sample. Moreover, dough dried out quickly during measurements. Beck et al. [35] reported lower instrumental stickiness in dough without sodium chloride compared to dough that contained it, contrary to expectations. In the authors’ view, data obtained with a stickiness probe did not properly describe salt-reduced dough samples. The authors proposed that gluten strands are more extensively hydrated in the absence of sodium chloride (as interactions with water are promoted over interactions among proteins if there is electrostatic repulsion). In the presence of sodium chloride, less water is bound by proteins because of more protein–protein interactions since sodium chloride shields the charges and thus reduces repulsion. The excess water leads to a higher contact force between the probe and the dough and thereby causes higher stickiness readings which are opposite to subjective evaluations [1, 26, 39]. In our study, the samples with the highest amounts of algae tended to exhibit lower instrumental stickiness than the other samples. As shown in Table 5, there were significant correlations between the stickiness after 45 min and the extensibility after 45 min, the Pol:Mon ratio and specific volume of breads (see corresponding sections below). The stickiness of dough rested for 45 min significantly correlated with its extensibility, but not with Rmax. These results may be a consequence of algae restricting water mobility, potentially due to strong interactions of algae constituents with water molecules, algae particles restricting gluten network formation and thus allowing for more protein–water interactions, or a combination of these phenomena.
Size distribution of proteins
Polymeric glutenins are responsible for dough elasticity and strength, monomeric gliadins for its viscosity and extensibility. At optimum ratio, they allow gas cell walls to expand without rapid gas loss [11]. The size of glutenin polymers (%UPP) impacts gluten’s elastic properties [40]. The %UPP did not differ between samples (Fig. 1e). This is probably due to the high variability among replicates. On the other hand, our results showed that the Pol:Mon ratio (Fig. 1f) decreased with an increased amount of macroalgae in dough (P < 0.001). Aamondt et al. [41] demonstrated that gluten proteins became smaller and more soluble during dough mixing but aggregated or assembled again during dough rest. Lower Pol:Mon ratios in our algae-containing dough imply that the glutenin size remained small (equivalent to monomers) even after 90 min rest. Macroalgae powders contained 10–17% protein (Supplementary Table 1). Algae proteins may thus have contributed to and increased the monomer fraction. Moreover, the lower Pol:Mon ratios are most likely due to the presence of algae restricting protein aggregation and thereby limiting the size of gluten proteins. Whether this is due to algae particles providing steric hindrance or individual algae constituents interfering with network formation would need to be determined by future research. Overall, our results suggest that the algae incorporation into dough influences the aggregation of gluten proteins negatively and in a dose-dependent manner.
pH
Except for C3A + FSL, dough with fermented algae had significantly lower pH than all control samples or dough with unfermented algae (Fig. 1d). The pH in dough with unfermented algae was 5.95–6.02, whereas dough with fermented algae ranged from 5.57 to 5.86. The pH in dough with fermented algae was presumably lower because fermentation reduced the pH of the algae raw materials (Supplementary Table 1). Species of macroalgae (P < 0.01) and sodium chloride level (P < 0.01) influenced the pH in dough with macroalgae.
Bread properties
Specific volumes decreased with increasing algae levels (Table 4), in line with the weakened dough properties as indicated by the farinograph results (Table 1) and the Pol:Mon ratio (Fig. 1f). Mamat et al. [27], Qazi et al. [29] and Graça et al. [42] also reported lower specific volumes as algae contents increased, due to disruption of the gluten structure. Rosell et al. [30] observed lower specific volumes for dough containing pure alginate. In our samples, only the two breads with the lowest algae amounts, C3A + SL and C3A + FSL, did not differ from their salt-matched controls. Macroalgae species (P < 0.001), sodium chloride level (P < 0.05) and use of fermented algae (P < 0.01) exerted significant effects on specific volume of bread. Aside from the UPP and Pol:Mon ratio, the specific volume also had a significant and positive correlation with the pH (Table 5). This aligns with Holmes and Hoseney [43] who found that bread volume increased from pH 4.65 to 6.15. Lower sodium chloride concentrations enhance gas production [4], which may contribute to higher specific volume as sodium chloride modulates yeast metabolism and thus CO2 production [1].
The lack of significant difference among bread firmness in our study may have been due to opposing influences exerted by sodium reduction and algae incorporation, and comparatively low algae addition levels. Studies that used higher algae addition levels (up to 16%) showed an increase in bread crumb firmness with increased amounts of K. alvarezii [27], Tetraselmis chuii [29], Chlorella vulgaris [42] and Fucus vesiculosus [32].
Breads differed significantly in slice brightness, which was lower in breads with more macroalgae (Table 4). There were significant positive correlations between specific volume and brightness, and both parameters negatively correlated with firmness and positively with the Pol:Mon ratio (Table 5). These results reflect the compacter, darker and denser loaves obtained from macroalgae-enriched dough (also visible in Fig. 2b), due to the macroalgae interfering with protein aggregation and imparting color. Despite these impacts of macroalgae addition on dough and bread properties, no significant differences in crumb structure were observed between the breads with c-cell analysis (Table 4, Fig. 2a). With the general linear model significantly higher (P < 0.05) cell areas were observed in samples with S. latissima compared to A. esculenta, however, differences were of low magnitude.
Crumb structure of breads. One image per recipe was chosen at random. a The images are adapted from the C-cell software and illustrate cell size and distribution. Small cells are colored dark blue, larger ones are shown in lighter shares of blue, green and yellow. Cells large enough to be classified as holes are outlined in red. b shows corresponding images on a gray scale. For sample names, see Table 1. AE Alaria esculenta, SL Saccharina latissima; F fermented
Sensory properties
Sensory properties were impacted by the sodium chloride level and algae type (Table 6). While both salt-reduced controls (C2 and C3A) were evaluated as significantly less salty than the full-salt control C1, none of the macroalgae-containing samples were perceived as significantly different in saltiness from C1. This indicates that algae addition increased the perception of saltiness in sodium chloride-reduced bread. An increase in saltiness perception in breads with brown macroalgae (6% and 8% Chondrus crispus and Ascophyllum nodosum) was also observed by Lamont and McSweeney [9]. However, salty flavor negatively affected the liking of bread with C. crispus. On the other hand, Gorman et al. [3] showed that the use of 4% brown macroalgae powder (species not reported) allowed for 20% sodium chloride reduction in bread without negatively affecting the acceptability, whereas higher reductions led to more “not salty enough” responses by consumers. Neither of these studies reported the macroalgae’s intrinsic sodium contents, moreover the sodium content in bread was not consistent between samples [3, 9].
Although macroalgae increased the perception of saltiness in sodium chloride-reduced bread, these samples also scored higher on several undesirable properties (algae flavor and odor, cloying, bitter, rancid, and metallic flavor). Lamont and McSweeney [9] also reported moderate to strong seafood taste as well as a strong aftertaste in bread containing 6 or 8% A. nodosum and C. crispus. These negative sensory properties can lead to poor consumer acceptance as shown for muffins [44] and bread [9]. Nie et al. [45] reported that certain volatile compounds derived from algae give products an off-flavor. Moreover, there were significant differences in whiteness between bread with and without macroalgae, and bread with fermented A. esculenta had the lowest score. Mamat et al. [44] observed that the color of muffins was significantly more liked when they contained 2% as opposed to 10% algae powder, probably due to less green color.
Among texture attributes, only chewing resistance was evaluated as significantly different, between C3A and C3A + FAE (Table 6). Thus, the algae amount in our study did not affect the textural attributes to the same extent as taste, odor, and appearance. Higher amounts of algae and low sodium chloride concentrations reportedly lead to dry, hard, and dense texture in bread and poor consumer acceptance [3, 9]. However, previous studies did not optimize WA, which may have contributed to the negative characteristics in the breads with algae. By optimizing WA according to algae addition, as in this study, algae-containing bread with a comparable texture to regular bread can be obtained. Particularly noteworthy is that the level of juiciness was maintained in the samples with added algae (no significant differences to the control samples). The use of unfermented algae led to similar sensory scores for every evaluated attribute (Table 6). However, bread with fermented S. latissima exhibited a slightly different pattern than the other algae breads, with a less pronounced seaweed/kelp and rancid odor. Bruhn et al. [22] observed reduced marine flavors and saltiness in heat-treated and fermented S. latissima which is in line with our results. Given that fermentation extends the shelf-life of algae and facilitates their handling by manufacturers [18, 19], fermented S. latissima is promising for applications such as bread. In contrast, inclusion of fermented A. esculenta did not result in more favorable sensory properties compared to unfermented A. esculenta.
Macroalgae as salt replacers in bread
This study adds to the growing body of research that suggests macroalgae are suitable to partially substitute sodium chloride and was the first to evaluate the effect of fermented vs unfermented macroalgae in bread with whole wheat flour. Our results demonstrated that addition levels are crucial, and that fermentation of macroalgae may influence how they affect a food product when added as ingredient. Overall, incorporation of fermented S. latissima is recommended over fermented A. esculenta (as well as their unfermented counterparts) as it gave breads with fewer off-flavors.
Our study could not demonstrate a relationship between algae addition and instrumental dough stickiness. Future studies would be needed to investigate the relationships between algae macromolecule structure, dough stickiness and the water status, for example via thermogravimetric analysis [36].
Our results suggest the possibility of choosing an adequate algae type, pre-processing method (such as fermentation), sodium reduction level (e.g., 33%) and tailor the algae addition level to achieve acceptable bread quality. Low (10–20 mg/g) addition levels of blanched/rinsed macroalgae flour could solve many of the problems associated with macroalgae use in food. Such low amounts not only limit iodine, cadmium, and arsenic contents to acceptable levels, thereby minimizing health risks for consumers (Ballance et al. submitted) but also minimally impact dough rheology, bread quality as well as result in breads with acceptable sensory qualities. Further studies should evaluate if relatively low amounts of macroalgae, as used in our study, can contribute to sodium reduction (due to their enhancement of salty taste) as well as intake of essential minerals. For industrial applications, consumer acceptability studies could also give valuable input during product development.
Conclusion
Both the incorporation of macroalgae as well as sodium chloride reduction weakened dough matrices and reduced specific volumes. As our samples’ sodium contents were matched, the amount of macroalgae seems to be responsible for these phenomena. Because fermented A. esculenta had the lowest sodium content, dough with this algae type contained the highest amount of macroalgae and showed the most pronounced differences to other samples. Macroalgae addition gave a distinct algae flavor which may negatively influence consumer acceptance. The lowest addition levels of S. latissima (8.9 and 9.7 mg/g for SL and FSL, respectively) were sufficiently low to avoid adverse effects on dough functionality and bread volume. Moreover, addition of FSL resulted in fewer undesirable sensory characteristics and a more favorable sensory profile than the other macroalgae. Hence, tailoring the processing method to a particular species in order to achieve desired properties of algae-containing products is possible.
Data availability
The data from analyses of the current study are available within the article. Raw data are available based on reasonable request.
References
Silow C, Axel C, Zannini E, Arendt EK (2016) Current status of salt reduction in bread and bakery products—a review. J Cer Sci 72:135–145. https://doi.org/10.1016/j.jcs.2016.10.010
Melnæs BK, Lundberg-Hallén N, Helland-Kigen, KM, Lund-Blix NA, Myhre JB, Johansen AMW, Andersen LF (2012) Norkost 3 - En landsomfattende kostholdsundersøkelse blant menn og kvinner i Norge i alderen 18–70 år, 2010–11. Utgitt: 06/2012 Bestillingsnummer: IS-2000
Gorman M, Moss R, Barker S, Falkeisen A, Knowles S, McSweeney MB (2023) Consumer perception of salt-reduced bread with the addition of brown seaweed evaluated under blinded and informed conditions. J Sci Food Agr 103:2337–2346. https://doi.org/10.1002/jsfa.12473
Belz M, Ryan L, Arendt E (2012) The impact of salt reduction in bread: a review. Crit Rev Food Sci 52:514–524. https://doi.org/10.1080/10408398.2010.502265
Adhikari B, Howes T, Bhandari BR, Truong V (2001) Stickiness in foods: a review of mechanisms and test methods. Int J Food Prop 4:1–33. https://doi.org/10.1081/JFP-100002186
Huang WN, Hoseney RC (1999) Isolation and identification of a wheat flour compound causing sticky dough. Cereal Chem 76:276–281. https://doi.org/10.1094/CCHEM.1999.76.2.276
Nguyen L, Salanță L, Socaci S, Tofana M, Fărcaş A, Pop C (2020) A mini review about monosodium glutamate. B UASVM Food Sci TE 77:1–12
Biancarosa I, Belghit I, Bruckner CG, Liland NS, Waagbø R, Amlund H et al (2018) Chemical characterization of 21 species of marine macroalgae common in Norwegian waters: benefits of and limitations to their potential use in food and feed. J Sci Food Agr 98:2035–2042. https://doi.org/10.1002/jsfa.8798
Lamont T, McSweeney M (2021) Consumer acceptability and chemical composition of whole-wheat breads incorporated with brown seaweed (Ascophyllum nodosum) or red seaweed (Chondrus crispus). J Sci Food Agr 101:1507–1514. https://doi.org/10.1002/jsfa.10765
Lekhal S, Slapø H, Karevold KI (2016) Grove kornprodukter: Utvikling, vekst og nye produkter 2014–2016 NorgesGruppens Sunnhetsrapport
Hemdane S, Jacobs PJ, Dornez E, Verspreet J, Delcour JA, Courtin CM (2016) Wheat (Triticum aestivum L.) bran in bread making: a critical review. CRFSFS 15:28–42. https://doi.org/10.1111/1541-4337.12176
Heiniö RL, Noort M, Katina K, Alam SA, Sozer N, De Kock HL et al (2016) Sensory characteristics of wholegrain and bran-rich cereal foods–a review. Trends Food Sci Technol 47:25–38. https://doi.org/10.1016/j.tifs.2015.11.002
Stévant P, Rebours C, Chapman A (2017) Seaweed aquaculture in Norway: recent industrial developments and future perspectives. Aquacult Int 25:1373–1390. https://doi.org/10.1007/s10499-017-0120-7
EFSA (2014) Scientific opinion on dietary reference values for iodine. EFSA J 12(5):3660
Mæhre HK, Malde MK, Eilertsen KE, Elvevol EO (2014) Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J Sci Food Agr 94:3281–3290. https://doi.org/10.1002/jsfa.6681
Hogstad S, Licht Cederberg D, Eriksen H, Kollander B, Olafsson G, Mikkelsen B (2023) A Nordic approach to food safety risk management for use as food: current status and basis for future work. Publication number 2022: 564. www.norden.org/publications. Accessed 30 Jan 2024
Commission recommendation (EU) 2018/464 of 19 March 2018 on the monitoring of metals and iodine in seaweed, halophytes and products based on seaweed. Official Journal of the European Union
Blikra MJ, Altintzoglou T, Løvdal T, Rognså G, Skipnes D, Skåra T et al (2021) Seaweed products for the future: using current tools to develop a sustainable food industry. Trends Food Sci Technol 118:765–776. https://doi.org/10.1016/j.tifs.2021.11.002
FAO & WHO (2022) Report of the expert meeting on food safety for seaweed–Current status and future perspectives. Rome 28–29 October 2021. Food Saf Qual Series No.13
Krook JL, Duinker A, Larssen WE, Birkeland IM, Skeie S, Horn SJ, Stévant P (2023) Approaches for reducing the iodine content of the brown seaweed Saccharina latissima—effects on sensory properties. J Appl Phycol. https://doi.org/10.1007/s10811-023-02974-5
Nielsen CW, Holdt SL, Sloth JJ, Marinho GS, Sæther M, Funderud J Rustad T (2020) Reducing the high iodine content of Saccharina latissima and improving the profile of other valuable compounds by water blanching. Foods 9:569. https://doi.org/10.3390/foods9050569
Bruhn A, Brynning G, Johansen A, Lindegaard MS, Sveigaard HH, Aarup B et al (2019) Fermentation of sugar kelp (Saccharina latissima)—effects on sensory properties, and content of minerals and metals. J Appl Phycol 31:3175–3187. https://doi.org/10.1007/s10811-019-01827-4
Hatløy A, Bråthen K (2022) The Salt Partnership 2019–2021. Progress and achievements. Fafo Report 2022: 32. Retrieved from https://www.fafo.no/images/pub/2022/20835.pdf
Singh N, Donovan R, MacRitchie F (1990) Use of sonication and size-exlusion high-performance liquid chromatography in the study of wheat flour proteins. II. Relative quantity of glutenin as a measure of breadmaking quality. Cereal Chem 67:161–170
Lawless HT, Heymann H (2010) Sensory evaluation of food: principles and practices (Vol 2). Springer, New York
Amoriello T, Mellara F, Amoriello M, Ceccarelli D, Ciccoritti R (2021) Powdered seaweeds as a valuable ingredient for functional breads. Eur Food Res Technol 247:2431–2443. https://doi.org/10.1007/s00217-021-03804-z
Mamat H, Matanjun P, Ibrahim S, Md. Amin SF, Abdul Hamid M, Rameli AS, (2014) The effect of seaweed composite flour on the textural properties of dough and bread. J Appl Phycol 26:1057–1062. https://doi.org/10.1007/s10811-013-0082-8
Onyango C, Luvitaa SK, Lagat K, K’osambo L (2021) Impact of carrageenan copolymers from two red seaweed varieties on dough and bread quality. J Appl Phycol 33:3347–3356. https://doi.org/10.1007/s10811-021-02524-x
Qazi WM, Ballance S, Uhlen AK, Kousoulaki K, Haugen JE, Rieder A (2021) Protein enrichment of wheat bread with the marine green microalgae Tetraselmis chuii–Impact on dough rheology and bread quality. LWT Food Sci Tech 143:111115. https://doi.org/10.1016/j.lwt.2021.111115
Rosell CM, Rojas JA, de Barber CB (2001) Influence of hydrocolloids on dough rheology and bread quality. Food Hydrocoll 15:75–81
Helgerud T, Gåserød O, Fjæreide T, Andersen PO, Larsen CK (2010). In: Imeson A (ed) Food stabilisers, thickeners and gelling agents. Wiley-Blackwell
Arufe S, Della Valle G, Chiron H, Chenlo F, Sineiro J, Moreira R (2018) Effect of brown seaweed powder on physical and textural properties of wheat bread. Eur Food Res Technol 244:1–10. https://doi.org/10.1007/s00217-017-2929-8
Avramenko NA, Hopkins EJ, Hucl P, Scanlon MG, Nickerson MT (2020) Effect of salts from the lyotropic series on the handling properties of dough prepared from two hard red spring wheat cultivars of differing quality. Food Chem 320:126615. https://doi.org/10.1016/j.foodchem.2020.126615
Lynch EJ, Dal Bello F, Sheehan EM, Cashman KD, Arendt EK (2009) Fundamental studies on the reduction of salt on dough and bread characteristics. Food Res Int 42:885–891. https://doi.org/10.1016/j.foodres.2009.03.014
Beck M, Jekle M, Becker T (2012) Impact of sodium chloride on wheat flour dough for yeast-leavened products. I Rheological attributes. J Sci Food Agric 92:585–592. https://doi.org/10.1002/jsfa.4612
Avramenko N, Tyler R, Scanlon M, Hucl P, Nickerson M (2018) The chemistry of bread making: the role of salt to ensure optimal functionality of its constituents. Food Rev Int 34:204–225. https://doi.org/10.1080/87559129.2016.1261296
Grausgruber H, Hatzenbichler E, Ruckenbauer P (2003) Analysis of repeated stickiness measures of wheat dough using a texture analyzer. J Texture Stud 34:69–82. https://doi.org/10.1111/j.1745-4603.2003.tb01056.x
Chen WZ, Hoseney RC (1995) Development of an objective method for dough stickiness. LWT 28:467–473. https://doi.org/10.1006/fstl.1995.0079
Diler G, Le-Bail A, Chevallier S (2016) Salt reduction in sheeted dough: a successful technological approach. Food Res Int 88:10–15. https://doi.org/10.1016/j.foodres.2016.03.013
Gupta R, Khan K, Macritchie F (1993) Biochemical basis of flour properties in bread wheats. I. Effects of variation in the quantity and size distribution of polymeric protein. J Cer Sci 18:23–41. https://doi.org/10.1006/jcrs.1993.1031
Aamodt A, Magnus EM, Hollung K, Uhlen AK, Færgestad EM (2006) Dough and hearth bread characteristics influenced by protein composition, protein content, DATEM, and their interactions. J Food Sci 70:C214–C221. https://doi.org/10.1111/j.1365-2621.2005.tb07128.x
Graça C, Fradinho P, Sousa I, Raymundo A (2018) Impact of Chlorella vulgaris on the rheology of wheat flour dough and bread texture. LWT 89:466–474. https://doi.org/10.1016/j.lwt.2017.11.024
Holmes J, Hoseney R (1987) Chemical leavening: effect of pH and certain ions on breadmaking properties. Cereal Chem 64:343–348
Mamat H, Akanda JMH, Zainol MK, Ling YA (2018) The influence of seaweed composite flour on the physicochemical properties of muffin. J Aquat 27:635–642. https://doi.org/10.1080/10498850.2018.1468841
Nie J, Fu X, Wang L, Xu J, Gao X (2022) A systematic review of fermented Saccharina japonica: fermentation conditions, metabolites, potential health benefits and mechanisms. Trends Food sci Technol 123:15–27. https://doi.org/10.1016/j.tifs.2022.03.001
Acknowledgements
The authors would like to thank the Norwegian Research Council (Project 294946; the Norwegian Seaweed Biorefinery Platform) for funding and the Norwegian Fund for Research Fees for Agricultural Products (FFL) for supporting the study through the project “Food Pilot Plant Norway” (Project Number 296083) and “Food For Future” (Project Number 314318).
Funding
Open access funding provided by Norwegian University of Life Sciences. Norges Forskningsråd, Project 294946, Norwegian Fund for Research Fees for Agricultural Products, 296083, 314318.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Johanna Liberg Krook is employed by Orkla Ocean, a supplier of seaweed products. The other authors declare that they have no conflicts of interest.
Ethical approval
Nofima’s sensory panelists are employees hired for the sole task of sensory testing, and as such, protocols are standard and covered by contractual agreement. As such, we do not seek ethical approvement for every project. However, all research related activities performed at Nofima are regulated under the Research Ethics Act 2017, and shall be carried out in accordance with relevant guidelines from national and international advisory bodies, conventions and agreements (cf. National Committee for Research Ethics in Science and Technology (NENT), National Committee for Research Ethics in Social Sciences and the Humanities (NESH), the European Group on Ethics in Science and New Technologies (EGE)/European Commission, the International Committee of Medical Journal Editors, and ICMJE (the Vancouver Convention). Also, there is compliance with ethical principles and Applicable international, EU and national law (in particular, EU Directive 95/46/EC). Johanna Liberg Krook is employed by Orkla Ocean AS, a supplier of seaweed products.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tyl, C., Koga, S., Krook, J.L. et al. Fermented and unfermented brown macroalgae as partial salt replacers in sodium-reduced dough and bread. Eur Food Res Technol 250, 1573–1585 (2024). https://doi.org/10.1007/s00217-024-04498-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-024-04498-9